Irritable Bowel Syndrome
Deep-Dive Articles
Rome IV Subtypes (IBS-C/D/M/U)
Rome IV diagnostic criteria, Bristol Stool Chart, IBS-C vs IBS-D vs IBS-M vs IBS-U distinctions, and why subtype drives treatment.
Low-FODMAP Diet
The Monash 3-phase protocol — elimination, reintroduction, personalization. Why it works, why it's temporary, and the food lists you actually need.
Visceral Hypersensitivity & Brain-Gut Axis
Why your gut feels normal sensations as painful — central sensitization in the gut, vagal tone, HPA axis, and the overlap with fibromyalgia and anxiety.
IBS & SIBO Overlap
How often IBS is really SIBO in disguise, breath testing, when to treat SIBO instead, and why rifaximin helps both.
Post-Infectious IBS
Gastroenteritis-triggered IBS, anti-CdtB/anti-vinculin antibodies, the IBSmart test, and why a Bali stomach bug 2 years ago may still be running your gut.
IBS Medications Guide
Rifaximin, linaclotide, plecanatide, lubiprostone, tenapanor, eluxadoline, alosetron, antispasmodics — what to ask your GI about, by subtype.
Gut-Directed Hypnotherapy & CBT
70-80% response rates in major trials rival any medication. Nerva, Regulora, Mahana IBS apps; CBT-for-IBS; tricyclic neuromodulators at low doses.
Peppermint, Probiotics & Herbals
Enteric-coated peppermint oil (IBgard), Bifidobacterium infantis 35624, triphala, iberogast, artichoke leaf — the evidence and what actually works.
Bile Acid Malabsorption in IBS-D
The missed cause of IBS-D — SeHCAT, C4 testing, and bile acid sequestrants.
Table of Contents
- Deep-Dive Articles
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- References
- Featured Videos
1. Overview
Irritable bowel syndrome (IBS) is a chronic, relapsing-remitting functional gastrointestinal disorder characterized by recurrent abdominal pain associated with defecation or a change in bowel habits, in the absence of identifiable structural, biochemical, or infectious etiology. IBS is classified within the broader framework of disorders of gut-brain interaction (DGBI), reflecting its pathophysiological basis in dysregulated bidirectional communication between the central nervous system (CNS) and the enteric nervous system (ENS).
IBS represents the most commonly diagnosed functional gastrointestinal disorder in gastroenterology practice and has substantial economic impact through direct healthcare costs and reduced work productivity. Four subtypes are recognized based on predominant bowel habit: IBS with predominant constipation (IBS-C), IBS with predominant diarrhea (IBS-D), IBS with mixed bowel habits (IBS-M), and IBS unclassified (IBS-U).
2. Epidemiology
IBS affects an estimated 10–15% of the global population, with prevalence varying by diagnostic criteria and geographic region. Using Rome IV criteria, the global prevalence is approximately 4–5%, lower than the 10–15% reported with Rome III criteria due to stricter frequency thresholds. The condition is more prevalent in South America and Southeast Asia when applying broader criteria.
Women are 1.5–2 times more likely than men to receive an IBS diagnosis in Western countries, though this sex difference is less marked in non-Western populations, suggesting sociocultural factors in health-seeking behavior. IBS can occur at any age but is most frequently diagnosed between the ages of 18 and 50. Prevalence declines with advancing age. Socioeconomic status, anxiety, and prior traumatic life events are independent risk factors.
IBS accounts for 12% of all primary care visits and 28% of gastroenterology referrals. Annual direct and indirect healthcare costs in the United States exceed $20 billion, driven by physician visits, diagnostic testing, pharmaceutical expenditures, and occupational absenteeism.
3. Pathophysiology
IBS pathophysiology is multifactorial and heterogeneous, encompassing alterations in gut motility, visceral hypersensitivity, gut-brain axis dysregulation, immune activation, microbiome composition, and epithelial barrier function:
- Visceral hypersensitivity: A defining feature of IBS; patients exhibit lower pain thresholds to colorectal balloon distension (allodynia) and exaggerated pain responses to normal stimuli (hyperalgesia). Central sensitization at the level of the spinal cord and brain, including altered descending pain modulation, amplifies afferent nociceptive signals from the gut.
- Altered gut motility: IBS-D is characterized by accelerated colonic and small intestinal transit, reduced rectosigmoid compliance, and exaggerated postprandial contractility (gastrocolic reflex). IBS-C shows delayed colonic transit, increased retrograde contractions, and prolonged proximal colonic segmentation. High-amplitude propagating contractions (HAPCs) may occur in both subtypes.
- Gut-brain axis dysregulation: The bidirectional vagal and spinal afferent pathways show heightened signaling in IBS. Functional neuroimaging (fMRI) demonstrates altered activation in the anterior cingulate cortex, insula, and prefrontal cortex in response to rectal stimulation. Serotonin (5-HT) signaling is central: enterochromaffin cells release 5-HT in response to mechanical and chemical stimuli; IBS-D patients exhibit elevated postprandial 5-HT levels, while IBS-C patients have reduced 5-HT availability. SERT (serotonin reuptake transporter) expression is altered in the colonic mucosa.
- Microbiome dysbiosis: Metagenomics studies show consistent alterations in the gut microbiome in IBS, including reduced microbial diversity, altered Firmicutes-to-Bacteroidetes ratio, and specific changes in Lactobacillus, Bifidobacterium, and Faecalibacterium prausnitzii abundance. Hydrogen and methane gas production from fermentation correlates with bloating and altered transit in IBS subtypes.
- Post-infectious IBS (PI-IBS): Develops in 10–30% of patients following acute gastroenteritis; persistent mucosal immune activation with increased mast cells, enterochromaffin cells, and T-lymphocyte infiltration; risk factors include severity of acute illness, female sex, anxiety, and prior psychological stress.
- Increased intestinal permeability: Demonstrated by elevated urine lactulose-to-mannitol ratios and reduced expression of tight junction proteins (occludin, claudin-1, ZO-1) in IBS-D; facilitates antigen translocation and low-grade mucosal inflammation.
- Low-grade mucosal inflammation: Increased mast cell density and mast cell proximity to mucosal nerve fibers; mast cell mediators (histamine, tryptase, prostaglandins) activate nociceptive afferents. Elevated colonic mucosal IL-1β, IL-6, and TNF-α in subsets of IBS patients.
- Food and dietary factors: Fermentable oligosaccharides, disaccharides, monosaccharides, and polyols (FODMAPs) are poorly absorbed in the small intestine, rapidly fermented by colonic bacteria, and osmotically active — leading to luminal distension, gas production, and symptom provocation in susceptible individuals.
4. Etiology and Risk Factors
Risk factors for IBS development:
- Acute gastroenteritis (bacterial, viral, or parasitic): 3–4-fold increased risk of PI-IBS; causative organisms include Campylobacter jejuni, Salmonella, Escherichia coli O157:H7, and norovirus
- Female sex (in Western populations)
- Age 18–50 years
- Psychological comorbidities: anxiety (OR ~3.0), depression (OR ~2.7), somatization disorder, post-traumatic stress disorder, and a history of physical or sexual abuse
- Family history (heritability estimated at 20–57% in twin studies)
- Antibiotic use, particularly broad-spectrum agents (disrupts microbiome)
- Food hypersensitivity (not IgE-mediated; likely immune-mediated mucosal sensitization)
- Small intestinal bacterial overgrowth (SIBO) — positive lactulose or glucose hydrogen breath test in 30–85% of IBS-D patients in some series, though prevalence varies widely
5. Clinical Presentation
Core symptoms:
- Recurrent abdominal pain: typically cramping, lower abdominal predominance; relieved by defecation in some but not all patients; often triggered by meals, stress, menstruation
- Altered bowel habits: diarrhea, constipation, or alternating; urgency and incomplete evacuation are common
- Abdominal bloating and visible distension: particularly postprandial; more prominent in IBS-C and IBS-M
- Mucus in stool (without blood)
IBS subtypes by predominant stool form (Bristol Stool Form Scale):
- IBS-C: >25% stools Bristol type 1–2 and <25% type 6–7
- IBS-D: >25% stools Bristol type 6–7 and <25% type 1–2
- IBS-M: >25% stools both type 1–2 and type 6–7
- IBS-U: Meets Rome IV criteria but bowel habit pattern cannot be classified into above subtypes
Extraintestinal and associated symptoms:
- Fatigue (50–80% of patients)
- Sleep disturbances and non-restorative sleep
- Fibromyalgia (comorbid in 30–65% of IBS patients)
- Chronic pelvic pain, dyspareunia, dysmenorrhea
- Interstitial cystitis / bladder pain syndrome
- Temporomandibular joint disorder
- Anxiety and depressive disorders
- Headache and migraine
Alarm features requiring further investigation (red flags):
- Rectal bleeding or hematochezia
- Unintentional weight loss (>10% body weight)
- Nocturnal symptoms awakening from sleep
- New onset in patients over 50 years
- Family history of colorectal cancer, IBD, or celiac disease
- Fever, elevated CRP, or elevated fecal calprotectin (>150–200 µg/g)
- Progressive dysphagia or odynophagia
6. Diagnosis
Rome IV Diagnostic Criteria (2016)
Recurrent abdominal pain, on average at least 1 day per week in the last 3 months, associated with two or more of the following criteria, with symptom onset at least 6 months before diagnosis:
- Related to defecation (may be relieved or worsened)
- Associated with a change in frequency of stool
- Associated with a change in form (appearance) of stool
Note: Rome IV replaced "improvement with defecation" (Rome III) with "related to defecation" to encompass patients in whom pain worsens with defecation. The minimum frequency threshold was increased from 3 days/month (Rome III) to 1 day/week (Rome IV), resulting in a lower diagnosed prevalence.
Diagnostic Approach — Positive Diagnosis Strategy
IBS should be diagnosed based on symptom criteria without extensive investigation in patients without alarm features, to avoid diagnostic delay and unnecessary procedures. Recommended baseline investigations:
- Full blood count (FBC) and CRP/ESR — to exclude IBD and anemia
- Thyroid function tests — to exclude hypothyroidism (IBS-C) or hyperthyroidism (IBS-D)
- Celiac serology (anti-tTG IgA + total IgA) — 3–5-fold higher celiac prevalence in IBS-D
- Fecal calprotectin — highly sensitive marker to differentiate IBS from IBD; values <50 µg/g support IBS diagnosis (NPV >97% for IBD); values >150–200 µg/g warrant colonoscopy
- Stool culture and C. difficile toxin — in IBS-D with recent antibiotic use or travel history
- Colonoscopy: indicated in patients ≥45–50 years (colorectal cancer screening), those with alarm features, or calprotectin elevation; random mucosal biopsies to exclude microscopic colitis in IBS-D
Additional Investigations in Selected Cases
- Hydrogen/methane breath testing: glucose or lactulose challenge for SIBO (IBS-D) and lactulose for intestinal transit; methane-dominant breath test profile associated with IBS-C
- Gastric emptying scintigraphy: when gastroparesis is suspected
- Anorectal manometry: for patients with severe constipation to exclude dyssynergic defecation
- Wireless motility capsule (SmartPill): whole gut transit measurement in refractory IBS-C
7. Treatment
Treatment is individualized, stepwise, and guided by predominant bowel habit subtype. A strong therapeutic alliance and patient education are foundational.
Dietary Interventions
- Low-FODMAP diet: The most evidence-based dietary intervention; reduces intraluminal osmotic load and fermentation; 50–76% of IBS patients respond with significant symptom reduction; should be implemented in 3 phases: restriction (6–8 weeks), reintroduction, and personalization. Requires dietitian guidance to ensure nutritional adequacy.
- General dietary advice: Regular meals, reduced fat and spicy foods, limited caffeine and alcohol, adequate fluid intake, soluble fiber supplementation for IBS-C (psyllium; insoluble fiber such as bran may worsen symptoms)
- Gluten-free diet: Benefits reported in IBS-D patients without celiac disease; may reflect FODMAP reduction (fructans in wheat) rather than gluten itself
Pharmacological Treatment — IBS-D
- Loperamide: µ-opioid receptor agonist; reduces intestinal transit and rectal urgency; effective for diarrhea and urgency but not abdominal pain; 2–4 mg as needed or before meals
- Rifaximin (Xifaxan): Non-absorbable antibiotic targeting gut dysbiosis and SIBO; FDA-approved for IBS-D (550 mg TID × 14 days); provides symptom relief for 10–12 weeks; re-treatment is effective
- Eluxadoline (Viberzi): Mixed µ/κ-opioid receptor agonist and δ-receptor antagonist; FDA-approved for IBS-D; reduces pain and diarrhea; contraindicated in patients without a gallbladder or with heavy alcohol use due to sphincter of Oddi spasm risk
- Alosetron (Lotronex): 5-HT3 receptor antagonist; FDA-approved for severe IBS-D in women who have failed conventional therapy; reduces pain and diarrhea; requires REMS program due to risk of ischemic colitis and severe constipation
- Low-dose tricyclic antidepressants (TCAs): Amitriptyline 10–75 mg at bedtime; neuromodulatory effects through norepinephrine and 5-HT reuptake inhibition and anticholinergic slowing of transit; most evidence-based neuromodulator for global IBS symptoms
Pharmacological Treatment — IBS-C
- Linaclotide (Linzess): Guanylate cyclase-C (GC-C) receptor agonist; increases intracellular cGMP → activates CFTR chloride channel → intestinal secretion and accelerated transit; reduces visceral pain by suppressing colonic afferent firing; FDA-approved for IBS-C (290 µg daily)
- Plecanatide (Trulance): GC-C agonist structurally analogous to uroguanylin; FDA-approved for IBS-C (3 mg daily); similar mechanism to linaclotide with pH-dependent activation
- Lubiprostone (Amitiza): Type 2 chloride channel (ClC-2) activator; increases intestinal fluid secretion; FDA-approved for IBS-C in women ≥18 years (8 µg BID)
- Tenapanor (Ibsrela): NHE3 (sodium/hydrogen exchanger 3) inhibitor; reduces sodium absorption in the intestinal epithelium, increasing luminal water and reducing visceral pain; FDA-approved for IBS-C (50 mg BID)
- Selective serotonin reuptake inhibitors (SSRIs): Accelerate colonic transit; useful in IBS-C patients with comorbid anxiety or depression; fluoxetine 20 mg daily
- Osmotic laxatives: Polyethylene glycol (PEG) for constipation relief; does not address pain component
Centrally Acting Neuromodulators (All Subtypes)
- Low-dose TCAs: First-line neuromodulator; amitriptyline, nortriptyline, imipramine
- SNRIs: Duloxetine, venlafaxine; useful particularly in IBS with comorbid depression, fibromyalgia, or neuropathic pain
- Gabapentinoids: Pregabalin, gabapentin; evidence for visceral pain reduction; particularly useful in post-infectious IBS with central sensitization
Psychological and Behavioral Therapies
- Cognitive behavioral therapy (CBT): Most robust evidence; shown to reduce IBS symptom severity scores, pain, and disability; telephone-delivered and web-based CBT are effective alternatives
- Gut-directed hypnotherapy: 70–80% response rates in controlled trials; durable effects up to 5 years; effective even without formal psychological comorbidity
- Mindfulness-based stress reduction (MBSR): Reduces symptom severity and psychological distress; particularly useful in patients with anxiety and catastrophizing
Probiotics
Several species and strains have demonstrated modest benefit in randomized trials, though results are inconsistent across studies. Bifidobacterium infantis 35624 and multi-species formulations including Lactobacillus and Bifidobacterium species have the most supporting evidence for global symptom reduction. Strain specificity is important — results are not generalizable across products.
8. Complications
IBS does not cause structural bowel disease, cancer, or shortened life expectancy. However, its chronic nature is associated with significant morbidity:
- Severe quality-of-life impairment comparable to chronic obstructive pulmonary disease and congestive heart failure on validated quality-of-life instruments
- Occupational disability and reduced work productivity (presenteeism and absenteeism)
- High rates of anxiety and depressive disorders, which may be both predisposing and consequential
- Healthcare overutilization: excessive colonoscopies, imaging, and unnecessary procedures due to diagnostic uncertainty
- Nutritional deficiency from overly restrictive diets, particularly poorly supervised low-FODMAP or elimination diets
- Social isolation and impaired interpersonal relationships
- Adverse effects from long-term pharmacotherapy (constipation with loperamide, sedation with TCAs, antibiotic resistance with rifaximin overuse)
9. Prognosis
IBS is a chronic condition with a fluctuating course; spontaneous remission occurs in approximately 30–40% of patients over 5 years. Symptom severity may fluctuate with stress, diet changes, intercurrent illness, hormonal changes, and life events. Subtype conversion is common: 30–40% of IBS patients transition between subtypes over time.
Predictors of poor prognosis include comorbid anxiety, depression and somatization; severe baseline symptom scores; high levels of healthcare seeking; abuse history; and post-infectious onset with severe acute illness. Early intervention with psychological therapies in high-risk patients appears to modify the long-term course favorably.
IBS does not increase the risk of colorectal cancer, IBD, or other serious GI disorders when properly diagnosed. The primary burden is related to quality-of-life impairment and economic costs.
10. Prevention
- Post-infectious IBS prevention: Prompt treatment of acute gastroenteritis and judicious antibiotic use to reduce PI-IBS risk; probiotic supplementation during and after acute gastroenteritis may reduce the risk (limited evidence)
- Antibiotic stewardship: Reducing unnecessary broad-spectrum antibiotic courses to preserve microbiome diversity
- Psychological intervention: Early CBT or stress management in high-risk individuals (anxiety, prior trauma, severe acute illness) may reduce IBS incidence or severity
- Diet and lifestyle: Mediterranean dietary pattern, adequate physical activity, and adequate sleep are associated with lower IBS prevalence in observational studies
- No pharmacological agent has been validated for IBS prevention in high-risk populations
11. Recent Research and Advances
- Rome IV criteria revision: The 2016 Rome IV criteria refined diagnostic thresholds to improve specificity; ongoing discussion about whether biomarkers (fecal calprotectin, plasma cytokines, serum anti-CdtB and anti-vinculin antibodies) should be incorporated into future diagnostic algorithms.
- Biomarker development: Anti-CdtB (cytolethal distending toxin B) and anti-vinculin antibodies, generated after Campylobacter infection causing molecular mimicry in enteric neurons, are elevated in IBS-D; a commercially available serum panel (ibs-smart) has FDA clearance for differentiating IBS-D from IBD, with a positive predictive value above 90%.
- Fecal microbiota transplantation (FMT): Multiple RCTs have shown conflicting results; a large Norwegian RCT (Johnsen et al., 2018) demonstrated superiority of FMT over placebo at 3 months but waning effects at 1 year; optimal protocols (donor selection, delivery route, number of infusions) remain under investigation.
- Tenapanor mechanism elucidation: Post-hoc analyses of the IBSRELA Phase 3 trials confirmed that NHE3 inhibition reduces visceral hypersensitivity through phosphate-mediated mechanisms, expanding understanding beyond fluid secretion.
- Psychedelic-assisted therapy: Early-phase trials of psilocybin and MDMA-assisted psychotherapy in refractory IBS are ongoing, based on their neuroplasticity effects in the anterior cingulate cortex and insula.
- Dietary precision medicine: Microbiome-informed dietary recommendations using machine learning to predict individual response to low-FODMAP diet versus Mediterranean diet versus specific carbohydrate diet are under development.
- Mast cell targeted therapy: Phase 2 trials of cromoglicate (mast cell stabilizer), ketotifen (H1 antihistamine), and ebastine in IBS patients with mast cell-nerve fiber proximity have shown promising visceral pain reduction.
12. References
- Lacy BE, Mearin F, Chang L, et al. Bowel Disorders. Gastroenterology. 2016;150(6):1393–1407. doi:10.1053/j.gastro.2016.02.031
- Ford AC, Lacy BE, Talley NJ. Irritable Bowel Syndrome. N Engl J Med. 2017;376(26):2566–2578. doi:10.1056/NEJMra1607547
- Simrén M, Törnblom H, Palsson OS, et al. Visceral hypersensitivity is associated with GI symptom severity in functional GI disorders: consistent findings from five different patient cohorts. Gut. 2018;67(2):255–262. doi:10.1136/gutjnl-2016-312361
- Halmos EP, Power VA, Shepherd SJ, Gibson PR, Muir JG. A diet low in FODMAPs reduces symptoms of irritable bowel syndrome. Gastroenterology. 2014;146(1):67–75. doi:10.1053/j.gastro.2013.09.046
- Pimentel M, Lembo A, Chey WD, et al. Rifaximin Therapy for Patients with Irritable Bowel Syndrome without Constipation. N Engl J Med. 2011;364(1):22–32. doi:10.1056/NEJMoa1004409
- Lembo AJ, Lacy BE, Zuckerman MJ, et al. Eluxadoline for Irritable Bowel Syndrome with Diarrhea. N Engl J Med. 2016;374(3):242–253. doi:10.1056/NEJMoa1505180
- Chey WD, Lembo AJ, Lavins BJ, et al. Linaclotide for Irritable Bowel Syndrome with Constipation. N Engl J Med. 2012;366(17):1624–1633. doi:10.1056/NEJMoa1200553
- Pittayanon R, Lau JT, Yuan Y, et al. Gut Microbiota in Patients With Irritable Bowel Syndrome — A Systematic Review. Gastroenterology. 2019;157(1):97–108. doi:10.1053/j.gastro.2019.03.049
- Ford AC, Quigley EM, Lacy BE, et al. Effect of Antidepressants and Psychological Therapies, Including Hypnotherapy, in IBS. Am J Gastroenterol. 2014;109(9):1350–1365. doi:10.1038/ajg.2014.148
- Pimentel M, Morales W, Rezaie A, et al. Development and validation of a biomarker for diarrhea-predominant irritable bowel syndrome in human subjects. PLoS ONE. 2015;10(5):e0126438. doi:10.1371/journal.pone.0126438
- Johnsen PH, Hilpüsch F, Cavanagh JP, et al. Faecal microbiota transplantation versus placebo for moderate-to-severe irritable bowel syndrome: a double-blind, randomised, placebo-controlled, parallel-group, single-centre trial. Lancet Gastroenterol Hepatol. 2018;3(1):17–24. doi:10.1016/S2468-1253(17)30338-2
- Mearin F, Lacy BE, Chang L, et al. Bowel Disorders. Gastroenterology. 2016;150(6):1393–1407.e5. doi:10.1053/j.gastro.2016.02.031
- Barbara G, Stanghellini V, Brandi G, et al. Interactions between commensal bacteria and gut sensorimotor function in health and disease. Am J Gastroenterol. 2005;100(11):2560–2568. doi:10.1111/j.1572-0241.2005.00230.x
- Drossman DA, Chang L, Chey WD, et al. Rome IV Multidimensional Clinical Profile for Functional Gastrointestinal Disorders. Rome Foundation, 2016. doi:10.1053/j.gastro.2016.02.015
- Whorwell PJ, Prior A, Faragher EB. Controlled trial of hypnotherapy in the treatment of severe refractory irritable bowel syndrome. Lancet. 1984;2(8414):1232–1234. doi:10.1016/S0140-6736(84)92793-4
- Farmer AD, Randall HA, Aziz Q. It's a gut feeling: how the gut microbiome affects the state of mind. J Physiol. 2014;592(14):2981–2988. doi:10.1113/jphysiol.2013.270389
Research Papers
Curated PubMed topic searches of peer-reviewed literature on Irritable Bowel Syndrome (IBS). Each link opens a live PubMed query so you always see the most current studies.
- PubMed: Rome IV IBS criteria
- PubMed: Low-FODMAP diet for IBS
- PubMed: IBS microbiome
- PubMed: Rifaximin in IBS
- PubMed: Post-infectious IBS
- PubMed: Visceral hypersensitivity
- PubMed: Gut-brain axis in IBS
- PubMed: Probiotics in IBS
- PubMed: Peppermint oil for IBS
- PubMed: Eluxadoline and linaclotide
- PubMed: CBT for IBS
- PubMed: Bile acid diarrhea and IBS
Connections
- Deep Dives: Rome IV Subtypes · Low-FODMAP · Brain-Gut Axis · IBS/SIBO Overlap · Post-Infectious IBS · Medications Guide · Hypnotherapy & CBT · Peppermint & Probiotics · Bile Acid Malabsorption
- Gastroenterology
- SIBO (Small Intestinal Bacterial Overgrowth) — deep dives: Breath Test Guide, Low-FODMAP Diet, Rifaximin Protocols, Herbal Antimicrobials, Root Causes
- Inflammatory Bowel Disease
- Celiac Disease
- Symptoms: Bloating · Constipation · Chronic Diarrhea — see also all Symptoms
- Anxiety
- Depression
- Gut Health
- Elimination Diet
- Peppermint
- Ginger
Featured Videos

Irritable Bowel Syndrome (IBS): Symptoms, Causes & Treatment | Dr. Srikant Mohta

Irritable Bowel Syndrome (IBS): Causes, Symptoms, Bristol Stool Chart, Types and Treatment
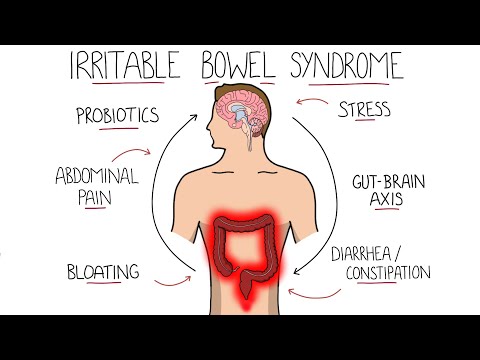
Irritable Bowel Syndrome (IBS) - Including Symptoms, Criteria & Treatment!

Do I have IBS ? | Symptoms & Diagnosis | Irritable Bowel Syndrome

How to CURE Irritable Bowel Syndrome in 5 Simple Steps

Irritable bowel syndrome: Mayo Clinic Radio

Inflammatory Bowel Disease (IBD): Symptoms, Treatment, and Prevention | Mass General Brigham

Heal Irritable Bowel Syndrome (IBS) with Natural Treatments by Dr. Rajsree

Irritable Bowel Syndrome Treatments | GI Society