Celiac Disease
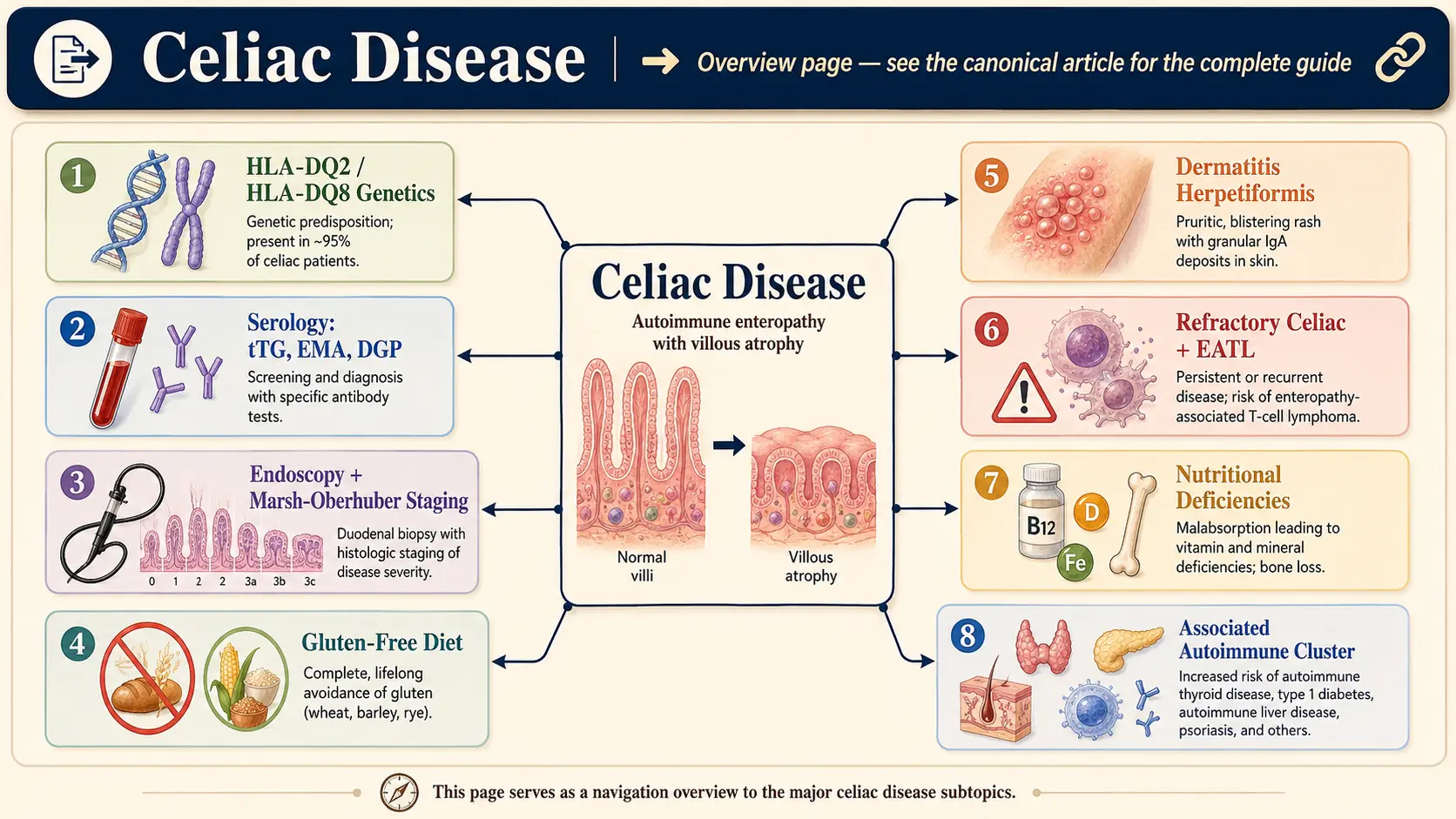
Deep-Dive Articles
Serology: tTG, DGP, EMA
Which antibody tests, in which order, and the trap of seronegative celiac. How to test correctly while still eating gluten.
HLA-DQ2 / DQ8 Genetic Testing
When genetic testing helps, when it's misleading, and why a negative result is more informative than a positive one.
Endoscopy, Biopsy & Marsh Staging
Marsh 0-III villous atrophy grading, proper biopsy technique (4-6 samples from duodenum including bulb), patchy disease, and when to skip the biopsy.
Gluten-Free Diet: Practical Guide
The 20-ppm threshold, cross-contamination in shared kitchens, restaurant strategy, FDA/Codex labeling, hidden-gluten sources, and grocery brands that actually stay safe.
Non-Celiac Gluten Sensitivity
How NCGS is diagnosed by exclusion, why most "gluten sensitivity" is actually fructan sensitivity, and how to do a proper N-of-1 trial.
Refractory Celiac & EATL
Type 1 vs Type 2 refractory celiac disease, aberrant intraepithelial lymphocytes, the lymphoma progression risk, and modern treatments (budesonide, azathioprine, cladribine).
Nutritional Deficiencies & Bone Health
Iron, B12, folate, D, copper, zinc, selenium — the post-diagnosis workup, repletion protocols, and when to get a DEXA scan.
Associated Autoimmune Conditions
Hashimoto's, Type 1 diabetes, primary biliary cholangitis, autoimmune hepatitis, Sjögren's, dermatitis herpetiformis — what to screen for and when.
Dermatitis Herpetiformis: Celiac of the Skin
The itchy blistering rash of celiac — dapsone, diet, and perilesional biopsy.
Table of Contents
- Deep-Dive Articles
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- References
- Featured Videos
1. Overview
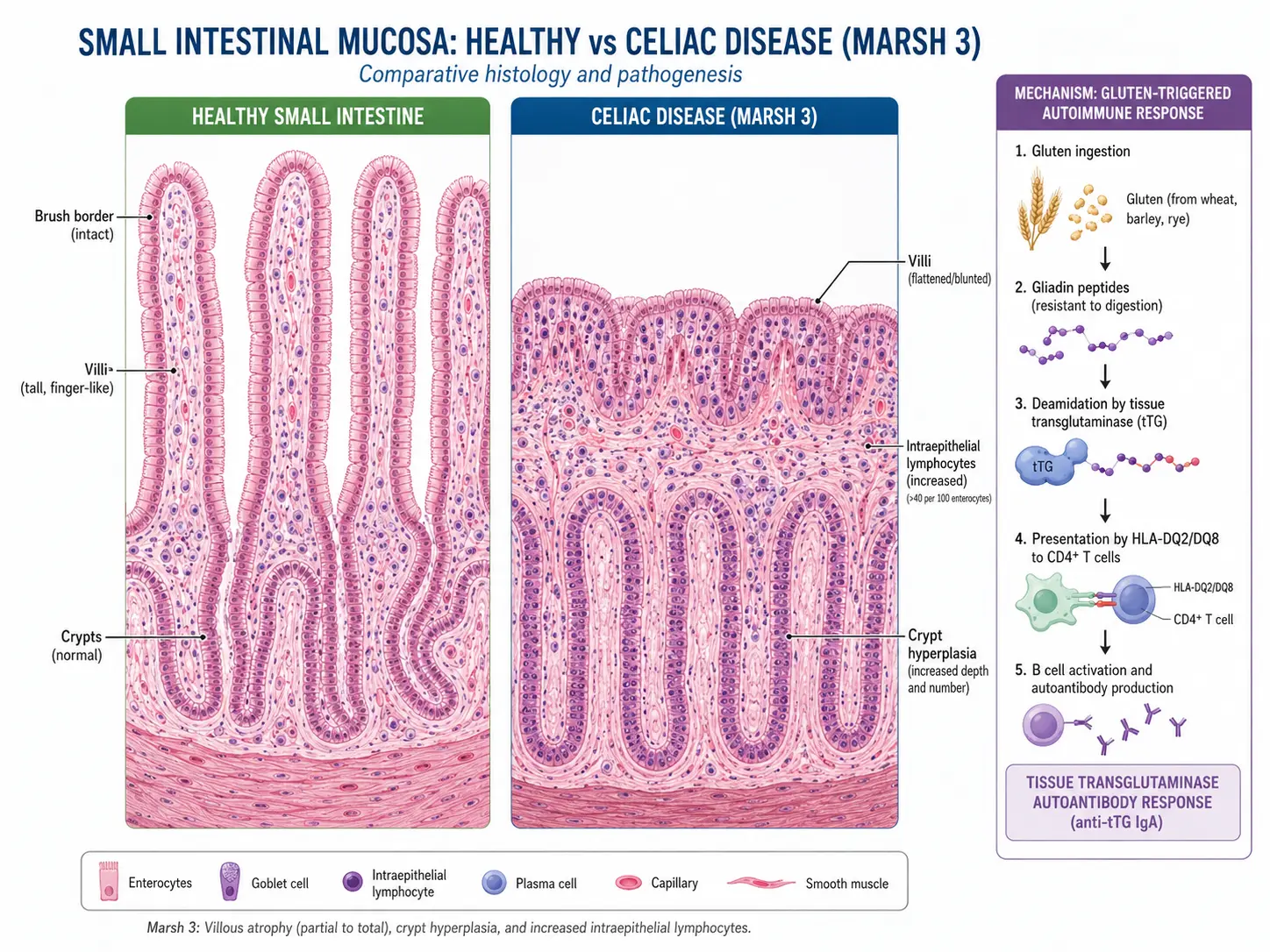
Celiac disease (CD) is a chronic, immune-mediated enteropathy triggered by the ingestion of gluten — a storage protein complex found in wheat (Triticum aestivum), barley (Hordeum vulgare), and rye (Secale cereale) — in genetically susceptible individuals. Upon gluten exposure, an aberrant adaptive immune response targets the small intestinal mucosa, resulting in villous atrophy, crypt hyperplasia, and intraepithelial lymphocytosis. The ensuing malabsorption can affect virtually every organ system.
Celiac disease is distinct from non-celiac gluten sensitivity (NCGS) and wheat allergy in that it involves both innate and adaptive immune mechanisms, characteristic serological markers, and specific histopathological lesions. It is classified as an autoimmune condition because the enzyme tissue transglutaminase 2 (tTG2) serves as both an antigen modifier and a primary autoantigen.
2. Epidemiology
Celiac disease affects approximately 1% of the global population, though the majority of cases remain undiagnosed. Large population-based serological studies estimate a worldwide prevalence of 0.7–1.4%, with significant geographic variation. Prevalence is highest in Northern Europe (up to 2–3% in Finland and Sweden) and among populations of European ancestry in North America and Australia.
The female-to-male ratio is approximately 2:1 for symptomatic disease; subclinical and silent forms show a more equal sex distribution. Diagnosis can occur at any age, with two incidence peaks: early childhood (12–24 months after gluten introduction) and the fourth to sixth decades of life. An "iceberg" model is used to describe the large proportion of undiagnosed individuals — only an estimated 20–30% of celiac disease cases in Western countries have been formally diagnosed.
Prevalence appears to be rising, independent of improved detection rates, suggesting environmental contributions such as alterations in microbiome composition, neonatal feeding practices, and changes in wheat processing.
3. Pathophysiology
The pathogenesis of celiac disease involves a complex interplay between genetic susceptibility, environmental triggers, and dysregulated immune activation:
- Gluten processing: Dietary gluten is incompletely digested by luminal proteases, yielding immunostimulatory peptides — most notably the 33-mer gliadin peptide. These peptides cross the intestinal epithelial barrier via transcellular or paracellular routes, partly facilitated by increased intestinal permeability mediated by zonulin.
- Tissue transglutaminase 2 (tTG2) deamidation: Within the lamina propria, tTG2 deamidates glutamine residues to glutamate in gliadin peptides, significantly increasing their affinity for the HLA-DQ2 and HLA-DQ8 molecules on antigen-presenting cells (APCs).
- Adaptive immune response: Deamidated gliadin peptides presented by HLA-DQ2/DQ8 on dendritic cells and macrophages activate gluten-specific CD4+ T helper cells. These Th1-polarized cells secrete pro-inflammatory cytokines — predominantly IFN-γ and TNF-α — that drive epithelial damage and upregulate matrix metalloproteinases responsible for villous destruction.
- Autoimmune component: CD4+ T cells also provide B-cell help, driving the production of IgA anti-tTG2 and anti-endomysial antibodies (EMA). Anti-tTG2 antibodies can directly inhibit tTG2 enzymatic activity and affect angiogenesis, contributing to extraintestinal manifestations.
- Innate immune activation: IL-15 produced by stressed enterocytes activates intraepithelial lymphocytes (IELs) expressing the NK receptor NKG2D. These cytotoxic IELs kill epithelial cells expressing the stress ligand MICA, amplifying mucosal damage.
- Resultant histology: Progressive mucosal injury follows the Marsh-Oberhuber classification, culminating in complete villous atrophy (Marsh 3c), crypt hyperplasia, and a dense IEL infiltrate exceeding 25 IELs per 100 enterocytes.
4. Etiology and Risk Factors
Genetic factors: HLA class II molecules are the primary genetic determinants. Approximately 90–95% of celiac disease patients carry HLA-DQ2.5 (encoded by DQA1*05 and DQB1*02), and most of the remainder carry HLA-DQ8 (DQA1*03/DQB1*0302) or HLA-DQ2.2. HLA typing has high negative predictive value (>99%) but low positive predictive value, as 30–40% of the general population carries susceptibility alleles. Non-HLA genes — including IL2, IL21, CTLA4, CCR3, and RGS1 — contribute an additional estimated 50% of genetic risk.
Environmental triggers and risk factors:
- Gluten ingestion — necessary but not sufficient
- First-degree relative with celiac disease (10–15% lifetime risk)
- Associated autoimmune conditions: type 1 diabetes mellitus (5–10% prevalence), autoimmune thyroid disease (3–5%), IgA deficiency (10-fold increased risk), Down syndrome, Turner syndrome, Williams syndrome
- Infant feeding practices: early or late introduction of gluten, possibly breastfeeding cessation before gluten introduction
- Gastrointestinal infections: rotavirus and adenovirus 12 infection in early childhood may trigger disease in susceptible individuals
- Microbiome dysbiosis: alterations in gut microbial composition, including reduced Lactobacillus and Bifidobacterium species
- Increased wheat protein content in modern grain varieties and altered food processing
5. Clinical Presentation
Celiac disease presents across a broad spectrum, ranging from classic malabsorptive symptoms to subclinical or silent disease incidentally detected through screening.
Classical presentation (predominantly in young children):
- Chronic diarrhea — often voluminous, pale, malodorous, and floating (steatorrhea)
- Abdominal distension and bloating
- Failure to thrive, growth retardation, delayed puberty
- Muscle wasting, particularly of the buttocks and proximal limbs
- Irritability and behavioral changes
Non-classical presentation (more common in older children and adults):
- Iron-deficiency anemia refractory to oral supplementation
- Fatigue and malaise
- Bone pain, osteopenia, osteoporosis with fragility fractures
- Peripheral neuropathy, ataxia (gluten ataxia)
- Dermatitis herpetiformis — intensely pruritic, symmetrical vesiculobullous rash on extensor surfaces; IgA deposits at the dermal-epidermal junction are pathognomonic
- Recurrent oral aphthous ulcers
- Elevated transaminases (celiac hepatitis)
- Infertility, recurrent miscarriage, menstrual irregularities
- Dental enamel hypoplasia (permanent teeth)
- Hyposplenia and increased infection susceptibility
Silent celiac disease: Positive serology and characteristic biopsy findings without symptoms; detected through targeted screening of at-risk groups.
Potential celiac disease: Positive serology with normal duodenal histology (Marsh 0–1); some patients may develop overt disease over time.
6. Diagnosis
Diagnosis requires a combination of serology, histopathology, HLA typing, and clinical response to a gluten-free diet (GFD). Testing must be performed while the patient is consuming a gluten-containing diet.
Serological Tests
- IgA anti-tissue transglutaminase (anti-tTG IgA): First-line test; sensitivity 95–98%, specificity 94–97%. Measure simultaneously with total serum IgA to exclude IgA deficiency.
- IgA anti-endomysial antibodies (EMA-IgA): Specificity approaching 100%; sensitivity 85–98%; observer-dependent immunofluorescence assay used for confirmation.
- Deamidated gliadin peptide antibodies (anti-DGP IgG and IgA): Useful in IgA-deficient patients; higher sensitivity than native anti-gliadin antibodies.
- IgA deficiency: Present in ~2–3% of celiac patients; use IgG-based assays (anti-tTG IgG, anti-DGP IgG) in these individuals.
Histopathology — Marsh-Oberhuber Classification
Multiple biopsies (at least 4 from the second/third part of the duodenum and at least 1 from the duodenal bulb) are required for diagnosis in adults:
- Marsh 0: Normal mucosa
- Marsh 1: Increased IELs (>25 per 100 enterocytes) — infiltrative lesion; compatible with but not diagnostic of celiac disease
- Marsh 2: IEL infiltration + crypt hyperplasia
- Marsh 3a: Partial villous atrophy
- Marsh 3b: Subtotal villous atrophy
- Marsh 3c: Total villous atrophy — villous-to-crypt ratio <1:1
HLA Typing
HLA-DQ2 and HLA-DQ8 typing is primarily used to exclude celiac disease (negative predictive value >99%). It is particularly useful in equivocal cases, patients already on a GFD, or those unable to undergo gluten challenge.
Pediatric Non-Biopsy Diagnosis (ESPGHAN 2020 Guidelines)
In symptomatic children with anti-tTG IgA >10 times the upper limit of normal, confirmed by positive EMA-IgA on a separate blood sample, and HLA-DQ2/DQ8 positivity, duodenal biopsy can be omitted — provided the child responds clinically to a GFD.
Differential Diagnosis
- Common variable immunodeficiency (CVID) with enteropathy
- Autoimmune enteropathy
- Tropical sprue
- Giardiasis
- Microscopic colitis
- Non-celiac gluten sensitivity (NCGS) — diagnosis of exclusion; seronegative, normal histology
7. Treatment
Strict Lifelong Gluten-Free Diet (GFD)
The GFD remains the only proven, effective treatment for celiac disease. Elimination of all wheat, barley, and rye from the diet leads to symptom resolution, serological normalization, and mucosal healing. Oats are tolerated by most celiac patients but must be certified gluten-free to avoid cross-contamination; a small subset (<5%) reacts to avenin in oats.
- The threshold for gluten toxicity is <10 mg/day; most guidelines recommend <20 ppm gluten in GFD-labeled foods
- Nutritional supplementation is often required at diagnosis: iron, folate, vitamin D, calcium, vitamin B12, zinc, magnesium
- Referral to a registered dietitian with celiac disease expertise is strongly recommended
- Regular follow-up: repeat serology at 6 and 12 months, then annually; repeat biopsy at 1–2 years to confirm mucosal healing in adults
Pharmacological Therapies
Currently, no approved pharmacological therapies exist beyond the GFD. However, investigational agents include:
- Latiglutenase (ALV003): Oral glutenase enzyme combination (prolyl endopeptidase + cysteine endoprotease) that degrades gluten peptides in the stomach — completed Phase 2 trials with mixed results
- Larazotide acetate (AT-1001): Tight junction regulator that reduces intestinal permeability by blocking the zonulin pathway; Phase 3 trial results demonstrated symptom reduction in accidental gluten exposure
- ZED1227: First-in-class tTG2 inhibitor; Phase 2 CZAR trial (2021) showed significantly reduced villous atrophy during gluten challenge compared with placebo
- Nexvax2: Antigen-specific immunotherapy targeting HLA-DQ2.5 — Phase 2 trials discontinued due to lack of efficacy on symptom scores
Refractory Celiac Disease (RCD)
Defined as persistent or recurrent symptoms and villous atrophy despite strict GFD for >12 months, after exclusion of intentional or inadvertent gluten ingestion and other causes:
- RCD Type I: Normal IEL phenotype; treated with nutritional support, budesonide (9 mg/day), or open-capsule budesonide; azathioprine as second-line
- RCD Type II: Clonal aberrant IEL population (CD3+CD8-CD103+); carries risk of enteropathy-associated T-cell lymphoma (EATL); treated with cladribine, stem cell transplantation in selected patients
8. Complications
- Malnutrition and micronutrient deficiencies: Iron, calcium, vitamin D, folate, vitamin B12, zinc, fat-soluble vitamins (A, E, K)
- Osteoporosis and fragility fractures: Elevated fracture risk up to 40% compared to the general population, particularly at the hip and spine
- Malignancy: Enteropathy-associated T-cell lymphoma (EATL) — rare but life-threatening; risk increased 4–6-fold compared to general population; small bowel adenocarcinoma; hepatocellular carcinoma; esophageal and oropharyngeal cancers
- Reproductive complications: Infertility, recurrent pregnancy loss, intrauterine growth restriction, low birth weight
- Neurological complications: Gluten ataxia, peripheral neuropathy, epilepsy with occipital calcifications, cognitive impairment
- Hyposplenia: Functional splenic atrophy increases susceptibility to encapsulated organisms such as Streptococcus pneumoniae, Haemophilus influenzae type b, and Neisseria meningitidis
- Associated autoimmune diseases: Type 1 diabetes, autoimmune thyroiditis, primary biliary cholangitis, Sjogren syndrome
- Refractory celiac disease (RCD): Affects 1–2% of celiac patients; Type II carries 30–50% 5-year mortality
9. Prognosis
The overall prognosis for celiac disease is excellent with strict adherence to a GFD. Most patients experience complete symptom resolution within weeks to months. Serological normalization typically occurs within 6–12 months, while mucosal healing in adults may require 2 years or longer, and is often incomplete in older adults. Studies suggest that 60–70% of adults achieve complete histological recovery after 2 years on a strict GFD.
Mortality in celiac disease is marginally elevated compared to the general population, primarily due to malignancy risk and cardiovascular complications from malnutrition. Patients who are diagnosed early, adhere strictly to the GFD, and receive appropriate nutritional support have mortality rates comparable to the unaffected population. RCD Type II carries significantly elevated mortality (30–50% at 5 years) due to risk of EATL transformation.
10. Prevention
There is currently no established strategy to prevent the development of celiac disease in genetically susceptible individuals. Evidence-based recommendations include:
- Timing of gluten introduction: Current European and American guidelines recommend introducing gluten between 4 and 12 months of age; the ESPGHAN consensus no longer supports a specific age window as definitively protective
- Breastfeeding: Earlier studies suggested a protective role; current evidence does not confirm breastfeeding prevents celiac disease, though it may delay onset
- PreventCD trial: A landmark multicenter RCT demonstrating that neither small-dose gluten introduction at 4 months nor breastfeeding at time of introduction prevented celiac disease in high-risk (HLA-DQ2 positive) infants at 3-year follow-up
- Screening in first-degree relatives: Recommended due to 10–15% lifetime prevalence; serological screening at age 3 (after at least 1 year of gluten ingestion) or in symptomatic individuals at any age
- Microbiome interventions: Under investigation; no current recommendations for probiotic use in prevention
11. Recent Research and Advances
- tTG2 inhibition (ZED1227): The Phase 2 CZAR trial published in NEJM (2021) demonstrated that ZED1227, an irreversible tTG2 inhibitor, significantly protected against gluten-induced villous atrophy during a 6-week gluten challenge, validating tTG2 as a therapeutic target and representing the most promising pharmacological advance to date.
- Tight junction modulation: Larazotide acetate has demonstrated consistent reduction of symptoms during inadvertent gluten exposure in Phase 3 trials, positioning it as a potential adjunct therapy for non-responsive symptoms.
- Microbiome research: Metagenomic studies have identified consistent dysbiosis in untreated celiac disease, with depletion of butyrate-producing bacteria and enrichment of pro-inflammatory species. Fecal microbiota transplantation (FMT) studies are ongoing.
- Non-biopsy diagnosis expansion: The ESPGHAN 2020 revised guidelines extended non-biopsy diagnosis criteria and are being evaluated for adaptation to adult populations in select cases.
- Gluten immunotherapy: Nexvax2, a peptide immunotherapy, failed Phase 2 trials but refined understanding of HLA-DQ2 immunodominant epitopes; next-generation tolerogenic nanoparticle therapies are in preclinical development.
- Wheat bioengineering: Development of low-immunogenic wheat strains with silenced α-gliadin genes through RNAi and CRISPR-Cas9 technology; clinical studies ongoing.
- Digital health monitoring: Wearable biosensors and AI-driven dietary tracking apps are being validated for real-time gluten exposure detection and dietary adherence monitoring.
12. References
- Lebwohl B, Sanders DS, Green PHR. Coeliac disease. Lancet. 2018;391(10115):70–81.
- Al-Toma A, Volta U, Auricchio R, et al. European Society for the Study of Coeliac Disease (ESsCD) guideline for coeliac disease and other gluten-related disorders. United European Gastroenterol J. 2019;7(5):583–613.
- Husby S, Koletzko S, Korponay-Szabó I, et al. European Society Paediatric Gastroenterology, Hepatology and Nutrition Guidelines for Diagnosing Coeliac Disease 2020. J Pediatr Gastroenterol Nutr. 2020;70(1):141–156.
- Rubio-Tapia A, Hill ID, Semrad C, et al. American College of Gastroenterology Guidelines Update: Diagnosis and Management of Celiac Disease. Am J Gastroenterol. 2023;118(1):59–76.
- Schuppan D, Mäki M, Lundin KEA, et al. A Randomized Trial of a Transglutaminase 2 Inhibitor for Celiac Disease. N Engl J Med. 2021;385(1):35–45.
- Fasano A. All disease begins in the (leaky) gut: role of zonulin-mediated gut permeability in the pathogenesis of some chronic inflammatory diseases. F1000Res. 2020;9:F1000 Faculty Rev-69.
- Lionetti E, Castellaneta S, Francavilla R, et al. Introduction of gluten, HLA status, and the risk of celiac disease in children. N Engl J Med. 2014;371(14):1295–1303.
- Vriezinga SL, Auricchio R, Bravi E, et al. Randomized feeding intervention in infants at high risk for celiac disease. N Engl J Med. 2014;371(14):1304–1315.
- Meresse B, Malamut G, Cerf-Bensussan N. Celiac disease: an immunological jigsaw. Immunity. 2012;36(6):907–919.
- Catassi C, Fabiani E, Iacono G, et al. A prospective, double-blind, placebo-controlled trial to establish a safe gluten threshold for patients with celiac disease. Am J Clin Nutr. 2007;85(1):160–166.
- Volta U, Caio G, De Giorgio R, et al. Non-celiac gluten sensitivity: a work-in-progress entity in the spectrum of wheat-related disorders. Best Pract Res Clin Gastroenterol. 2015;29(3):477–491.
- Ilus T, Kaukinen K, Virta LJ, Pukkala E, Collin P. Incidence of malignancies in diagnosed celiac patients: a population-based estimate. Am J Gastroenterol. 2014;109(9):1471–1477.
- Leffler DA, Kelly CP, Abdallah HZ, et al. A randomized, double-blind study of larazotide acetate to prevent the activation of celiac disease during gluten challenge. Am J Gastroenterol. 2012;107(10):1554–1562.
- Trynka G, Hunt KA, Bockett NA, et al. Dense genotyping identifies and localizes multiple common and rare variant association signals in celiac disease. Nat Genet. 2011;43(12):1193–1201.
- Marsh MN. Gluten, major histocompatibility complex, and the small intestine. A molecular and immunobiologic approach to the spectrum of gluten sensitivity ('celiac sprue'). Gastroenterology. 1992;102(1):330–354.
- Malamut G, Cellier C. Refractory celiac disease. Gastroenterol Clin North Am. 2019;48(1):137–144.
- Schiepatti A, Savioli J, Vernero M, et al. Pitfalls in the Diagnosis of Coeliac Disease: A Review of Literature. Nutrients. 2020;12(6):1711.
Research Papers
Curated PubMed topic searches of peer-reviewed literature on Celiac Disease. Each link opens a live PubMed query so you always see the most current studies.
- PubMed: Celiac disease diagnosis guidelines
- PubMed: Gluten-free diet in celiac disease
- PubMed: Tissue transglutaminase antibodies
- PubMed: HLA-DQ2/DQ8 susceptibility
- PubMed: Refractory celiac disease
- PubMed: Dermatitis herpetiformis
- PubMed: Epidemiology of celiac disease
- PubMed: Celiac disease and bone density
- PubMed: Enteropathy-associated T-cell lymphoma
- PubMed: Larazotide acetate trials
- PubMed: ZED1227 TG2 inhibitor
- PubMed: Non-celiac gluten sensitivity
Connections
- Gluten-Free Diet: A Practical Guide
- Dermatitis Herpetiformis
- Nutritional Deficiencies and Bone Health
- Endoscopy, Biopsy, and Marsh Classification
- Inflammatory Bowel Disease
- Irritable Bowel Syndrome
- Small Intestinal Bacterial Overgrowth (SIBO)
- Crohn's Disease
- Ulcerative Colitis
- Elimination Diet
- Vitamin D3
- Iron
- Calcium
- Anemia
- Osteoporosis
- Psoriasis
- Gut Healing
- Vitamin B12