Post-Infectious IBS
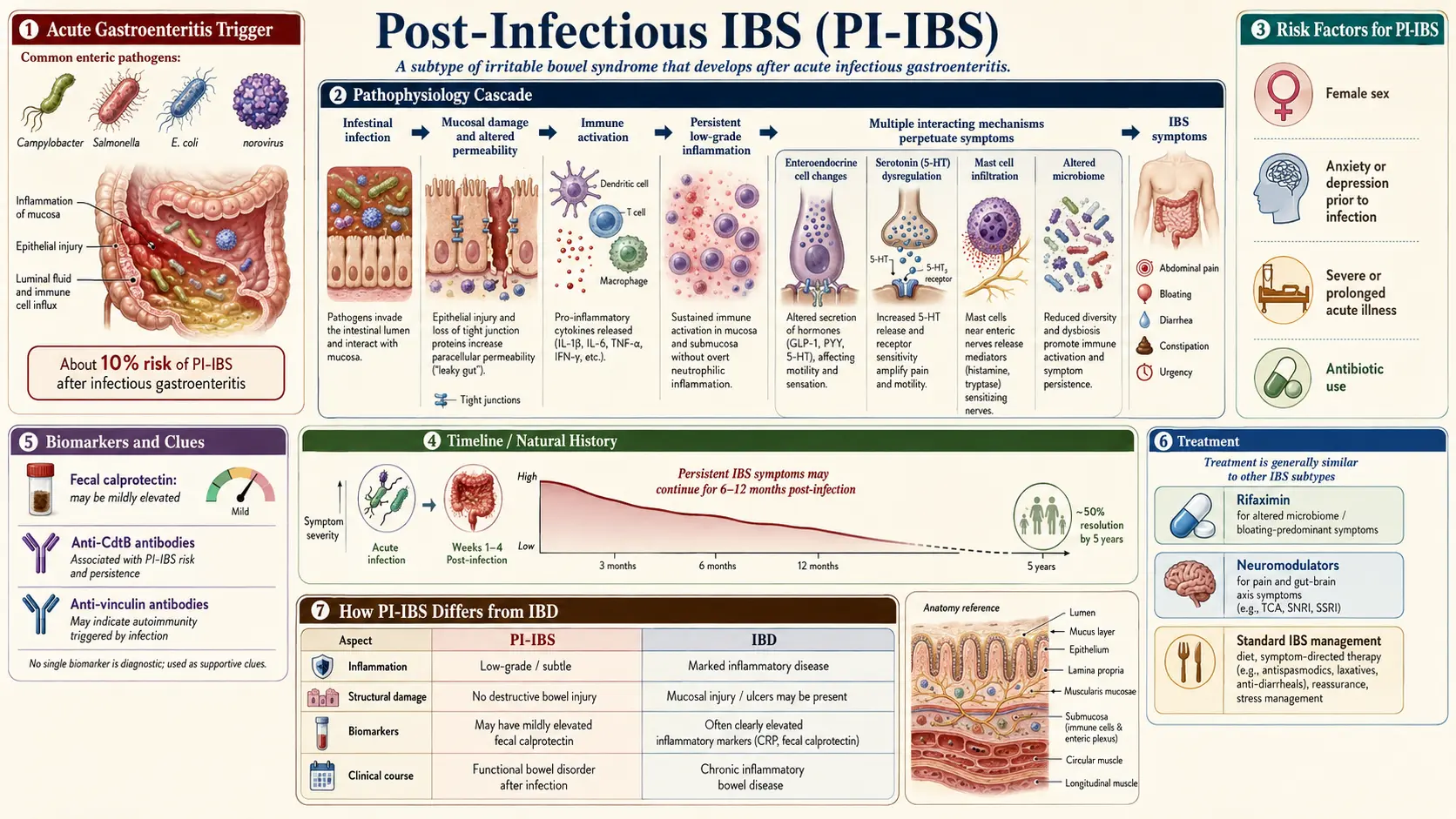
Table of Contents
- What Post-Infectious IBS Is
- The Numbers — How Common and Who Gets It
- The Walkerton Outbreak — The Definitive Study
- Risk Factors — Who Is Most Likely to Develop PI-IBS
- Which Pathogens Cause It
- Symptom Pattern — How PI-IBS Looks and Feels
- The CdtB – Vinculin Mechanism
- The ibs-smart Blood Test
- Travel History — The Missing Clue
- Post-Giardia IBS and the Bergen Outbreak
- Overlaps — ME/CFS, Lyme, Alpha-Gal
- Treatment — What Actually Works
- Time Course — How Long Does It Last?
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
What Post-Infectious IBS Is
Post-infectious IBS (PI-IBS) is irritable bowel syndrome that starts, to the day, after an episode of acute gastroenteritis — food poisoning, traveler's diarrhea, a bout with a parasite, or a waterborne outbreak. Before the infection you digested food normally. You had the "stomach flu" or that three-day nightmare on a Mexico beach, and the acute illness resolved — but the bathroom urgency, bloating, cramping, and unpredictable stools never quite went away. Six months later, a year later, you still do not trust a long car ride or a restaurant meal.
That is not a coincidence, and it is not "in your head." PI-IBS is now recognized as the best-understood subtype of irritable bowel syndrome, with a concrete triggering event, a measurable biological mechanism, and a blood test that can confirm it. About one in ten people who get acute infectious gastroenteritis go on to develop chronic IBS-type symptoms that meet Rome criteria at six to twelve months. For many of them, the infection was the single hinge between a normal gut and a decade of misery.
This article walks through what the condition is, who gets it, how it damages the gut, how to test for it, and what treatments compress the otherwise long natural history. If your IBS began after a specific illness you can still remember the date of, this is the page for you.
The Numbers — How Common and Who Gets It
The headline statistic, replicated across dozens of cohorts and summarized in the Klem et al. 2017 meta-analysis of more than 21,000 patients: roughly 10% of people who suffer acute infectious gastroenteritis develop IBS within 12 months, compared with a background rate of about 1–2% in matched controls who did not get sick. That is a four-to-seven-fold increase in IBS risk, persisting for years after the infection clears.
Put differently: walk into any gastroenterology clinic and ask new IBS patients when their symptoms started. Between 6% and 17% will name a specific episode of gastroenteritis as the onset — often a memorable one like a food-poisoning outbreak, a cruise-ship norovirus, or a foreign trip. PI-IBS is not a rare curiosity. It is one of the most common mechanisms by which people enter chronic IBS.
The Walkerton Outbreak — The Definitive Study
In May 2000, the municipal water supply of Walkerton, Ontario — a town of about 5,000 people — was contaminated with E. coli O157:H7 and Campylobacter jejuni after heavy rains washed livestock runoff into a shallow well. Roughly 2,300 residents became acutely ill; seven died. Because the exposed population was an entire town with known medical records, the outbreak became a natural experiment in what happens after acute bacterial gastroenteritis.
John Marshall and colleagues at McMaster University followed the Walkerton cohort for a decade. Their landmark 2010 paper, published in Gastroenterology, reported that 36.2% of Walkerton residents with acute gastroenteritis went on to develop IBS within two to three years — three times the rate in unexposed controls. Eight years later, about 15% still met IBS criteria, and many more had subclinical symptoms. Severity of the acute illness — bloody diarrhea, weight loss, fever — was the strongest predictor of long-term PI-IBS.
Walkerton, together with Robin Spiller's UK bacterial-gastroenteritis cohort (published in Gut 2009), established beyond reasonable doubt that acute enteric infection is a causal trigger for a substantial fraction of chronic IBS. These are not "stressed people who happened to have food poisoning." The gut itself was changed by the infection.
Risk Factors — Who Is Most Likely to Develop PI-IBS
Not everyone who gets gastroenteritis ends up with PI-IBS. The risk varies roughly tenfold depending on who you are and what happened to you during the acute illness. Consistent predictors across multiple cohorts:
- Female sex. Women are roughly twice as likely as men to develop PI-IBS after the same infection. Hormonal modulation of pain processing and gut motility is the leading suspect.
- Younger age at infection. People under 60 develop PI-IBS at much higher rates than older adults, possibly because aging gut mucosa mounts a less vigorous antibody response.
- Severity of the acute illness. Longer diarrhea (greater than seven days), fever, bloody stools, and weight loss during the acute infection each roughly double the PI-IBS risk.
- Psychological distress at the time of infection. Anxiety, depression, or high life stress in the month around the acute illness is a replicated risk factor. This does not mean IBS is "psychological" — it means a stressed nervous system permits central sensitization to lock in after the gut injury.
- Antibiotic use during or after the infection. Paradoxically, antibiotics given for the gastroenteritis (or any antibiotic given in the surrounding months) increase PI-IBS risk, likely by flattening the protective microbiome and allowing dysbiosis to take hold.
- Bacterial rather than viral pathogens. Bacterial infections carry the highest PI-IBS risk. Viral gastroenteritis (norovirus, rotavirus) produces PI-IBS at meaningfully lower rates, though not zero. Protozoal infections (Giardia) are among the strongest triggers of all.
If you are a woman in your twenties or thirties who got food poisoning on vacation, was put on ciprofloxacin, and had bloody diarrhea for ten days while also working a high-stress job, the odds you developed PI-IBS are not 10% — they are closer to 40%.
Which Pathogens Cause It
The culprits are the usual enteric infection suspects, ranked roughly by PI-IBS risk in the literature:
- Campylobacter jejuni — the single most-studied trigger. Causes about 10–15% PI-IBS at one year.
- Shigella — high PI-IBS rates, especially in travel-associated infections.
- Salmonella — the canonical "food poisoning" bacterium; solid PI-IBS association.
- Enterotoxigenic E. coli (ETEC) and Shiga-toxin-producing E. coli (STEC) — the Walkerton pathogens; the organisms behind most traveler's diarrhea.
- Clostridioides difficile — after C. diff colitis, up to 25% of patients develop persistent IBS-type symptoms.
- Giardia lamblia — protozoal, not bacterial; produces some of the highest PI-IBS rates on record (see the Bergen outbreak below).
- Norovirus and viral gastroenteritis — lower risk, roughly 4–5% PI-IBS, but not negligible given how common noro is.
Symptom Pattern — How PI-IBS Looks and Feels
PI-IBS tends to look different from the constipation-predominant IBS that runs in families or appears gradually in adolescence. The typical PI-IBS patient describes:
- Diarrhea-predominant (IBS-D) or mixed (IBS-M) pattern — rarely pure constipation.
- Urgency. The sudden, drop-everything need to find a bathroom, often within minutes of eating. Many patients map every gas station between home and work.
- Bloating and visible distension. A flat morning belly that balloons through the day.
- Post-prandial cramping — pain or griping within 30–60 minutes of a meal, partially relieved by a bowel movement.
- Food intolerances that were not there before. Dairy, gluten, onions, and high-FODMAP foods suddenly become triggers. This is usually SIBO-overlay physiology (see the sibling IBS & SIBO overlap and low-FODMAP diet articles).
- A clearly remembered start date. Patients with PI-IBS can usually say "before the Mexico trip in 2019 I was fine" with certainty. Non-PI IBS patients usually cannot.
The CdtB – Vinculin Mechanism
For thirty years, the stumbling block in PI-IBS research was the mechanism. The infection is long gone. Stool cultures are negative. Biopsies look almost normal. So what, precisely, is broken?
In a series of papers culminating in 2015, Mark Pimentel and colleagues at Cedars-Sinai identified the mechanism. Most invasive enteric bacteria (Campylobacter, Salmonella, Shigella, E. coli, several others) secrete a toxin called cytolethal distending toxin, subunit B (CdtB). CdtB damages host DNA and, crucially, triggers a durable antibody response. Your immune system makes anti-CdtB antibodies to clear the infection — and those antibodies often stick around for years after the bacteria are gone.
Unfortunately, a portion of CdtB is a molecular mimic for a human protein called vinculin. Vinculin is a cell-adhesion protein expressed densely in the gut's neural infrastructure — specifically in the interstitial cells of Cajal (ICC), the pacemaker cells that generate the small intestine's migrating motor complex (MMC). The MMC is the "housekeeping wave" that sweeps bacteria and debris from the small bowel into the colon between meals, roughly every 90 to 120 minutes, and it is what keeps the small intestine normally near-sterile.
When anti-CdtB antibodies cross-react with vinculin, they attack the interstitial cells of Cajal. ICC density drops. The MMC weakens or disappears. Small-intestinal contents stop being swept downstream. Bacteria migrate up from the colon and colonize the small bowel — the textbook setup for small intestinal bacterial overgrowth (SIBO). Fermentation of food in the small bowel produces the bloating, urgency, and post-prandial distress that defines PI-IBS.
This is why PI-IBS, SIBO, and IBS-D so often overlap: they are the same disease at different stages.
The ibs-smart Blood Test
The CdtB – vinculin discovery produced the first IBS-specific blood test. Marketed as ibs-smart (previously IBSchek), developed at Cedars-Sinai and now offered by Gemelli Biotech, it measures two IgG antibody levels:
- Anti-CdtB — evidence of prior invasive bacterial gastroenteritis.
- Anti-vinculin — evidence that the cross-reactive autoimmunity has begun damaging your own gut neural tissue.
A result is considered strongly positive when both antibodies are elevated. Specificity for PI-IBS is reported at around 90% or higher in the validation cohorts, meaning a double-positive result essentially rules in the post-infectious mechanism and rules out inflammatory bowel disease, celiac, and functional constipation. Sensitivity is lower — a negative does not rule PI-IBS out entirely, particularly for older infections where antibody titers have waned.
Clinically, the test does three things. It gives patients a concrete, biological diagnosis (huge psychologically, after years of being told "it's just IBS"). It predicts SIBO susceptibility, because vinculin damage is what permits overgrowth to recur. And it predicts response to rifaximin, the non-absorbed antibiotic that treats SIBO — double-positive patients tend to respond well and to relapse predictably every few months, which is exactly the pattern expected from permanent ICC damage.
The test costs around $150–$250 and is rarely covered by insurance. It is worth the money if your IBS started after a specific infection, if you respond to rifaximin but relapse, or if you simply want to know whether your symptoms have a measurable biological basis.
Travel History — The Missing Clue
The single most undervalued question in an IBS workup is: where were you, and what happened, in the weeks before your symptoms began?
Patients who spent a week in Bali, Mexico, India, or Southeast Asia in 2018, 2019, or 2021 and came home with Bali belly, Montezuma's revenge, or Delhi belly are often still carrying the symptoms years later without connecting the dots. The acute illness felt like a bad week and then resolved. The chronic bathroom problem that started right afterward felt like a separate issue. In PI-IBS it is not.
Travel-associated ETEC, Campylobacter, and Shigella are among the most common triggers worldwide. If you can place the start of your IBS within a month of foreign travel, an ibs-smart blood test is likely to come back positive, and the diagnostic and treatment pathway (rifaximin, prokinetics, low-FODMAP, see rifaximin protocol) is well-worn.
Post-Giardia IBS and the Bergen Outbreak
In autumn 2004, the municipal water supply of Bergen, Norway — a city of about 250,000 — was contaminated with Giardia lamblia. Roughly 1,300 confirmed cases (and thousands of likely unreported ones) developed acute giardiasis. Norwegian researchers, led by Kurt Hanevik and colleagues, followed the cohort for more than a decade.
The results were striking. At three years, 46% of the Giardia-exposed group met criteria for IBS, compared with 14% of controls. At six years the IBS rate was still 40%. At ten years a substantial fraction remained symptomatic. Post-Giardia IBS also carries a high rate of co-occurring chronic fatigue — a pattern that sits at the boundary of PI-IBS and ME/CFS.
The practical implication: if your IBS started after drinking from a stream, using untreated well water, traveling in areas where Giardia is endemic, or eating contaminated food, a stool PCR panel looking specifically for Giardia, Cryptosporidium, and Blastocystis is worth doing even years out. Treatment with tinidazole or nitazoxanide occasionally clears a low-grade persistent infection and resolves symptoms.
Overlaps — ME/CFS, Lyme, Alpha-Gal
PI-IBS sits inside a broader family of post-infectious chronic illnesses. The patterns overlap, and many patients have more than one label.
ME/CFS (myalgic encephalomyelitis / chronic fatigue syndrome). A substantial fraction of ME/CFS patients can date their illness to a specific infection — Epstein-Barr, enterovirus, Giardia, or another gut pathogen. Gut symptoms are common in ME/CFS and often meet IBS criteria. The Bergen post-Giardia cohort was notable for exactly this overlap. See the Chronic Fatigue Syndrome overview and the Post-Viral Triggers deep dive.
Lyme disease. Tick-borne Borrelia infection produces gastrointestinal symptoms in roughly a quarter of acute cases, and chronic or post-treatment Lyme syndrome frequently includes IBS-pattern bowel complaints. If you live in an endemic region and your IBS began after a summer of tick exposure or an unexplained febrile illness, see Lyme Disease.
Alpha-Gal syndrome. A tick-induced IgE antibody to the sugar galactose-alpha-1,3-galactose produces delayed reactions to mammalian meat — often with urgent diarrhea, cramping, and urgency three to six hours after eating. It mimics IBS-D perfectly and is routinely missed. If your symptoms cluster after red-meat meals, see Alpha-Gal Syndrome.
Treatment — What Actually Works
PI-IBS is one of the few IBS subtypes with a mechanistically targeted treatment plan. In rough order of evidence:
Rifaximin. A non-absorbed antibiotic that sits in the small bowel and reduces bacterial overgrowth. The TARGET 3 trials showed that two weeks of rifaximin produces durable symptom improvement in a substantial fraction of IBS-D patients, with retreatment effective at relapse. Double-positive ibs-smart patients respond especially well. See the sibling rifaximin protocol for full dosing.
Prokinetics. Because the core injury in PI-IBS is impaired MMC function, drugs that stimulate the MMC between meals reduce relapse. Low-dose naltrexone (LDN, 2.5–4.5 mg at bedtime), prucalopride (Motegrity), and off-label low-dose erythromycin are used by SIBO-focused clinicians to stretch the interval between rifaximin courses.
Low-FODMAP diet. A short-term elimination of fermentable carbohydrates lowers gas production and symptom severity while the underlying biology is addressed. It is not a forever diet — long-term FODMAP restriction starves beneficial microbes. See the sibling low-FODMAP diet article for the three-phase protocol.
Low-dose naltrexone (LDN). Beyond its prokinetic role, LDN has emerging evidence for modulating the low-grade gut inflammation and visceral hypersensitivity that persist after enteric infection. The dose is trivial, the cost low, side effects minimal. Several case series and small trials in IBS and Crohn's support it.
Gut-directed hypnotherapy. PI-IBS installs a layer of central sensitization — the spinal cord and brain amplify pain signals from a gut that has already healed. Gut-directed hypnotherapy (Manchester protocol, Nerva app, or in-person) is one of the few interventions with randomized-trial evidence for reducing this component. Roughly 70% of patients respond, and the effect is durable. It is not "it's all in your head" — it is rewiring the pain-processing circuitry that the infection helped recruit.
Treat what else is lurking. A stool PCR panel for Giardia, Blastocystis, and Dientamoeba, plus bile-acid testing in IBS-D, plus (when indicated) a glucose or lactulose breath test for SIBO, completes the workup. Do not settle for a bare "IBS" label when the biology is knowable.
Time Course — How Long Does It Last?
Untreated, the natural history of PI-IBS is slow. Median time to symptom resolution in the untreated Walkerton and Spiller cohorts was in the range of two to five years, with a meaningful minority — perhaps 15–20% — symptomatic at a decade. The Bergen post-Giardia cohort has an even longer tail.
The good news: treatment compresses this timeline substantially. Patients who combine rifaximin, a prokinetic, a temporary low-FODMAP phase, and gut-directed hypnotherapy often report major improvement within three to six months. Relapses happen — the underlying vinculin damage may be permanent — but the relapse-and-retreat cycle can keep you functional indefinitely.
The single most important thing a PI-IBS patient can do is recognize the pattern. An IBS workup that stops at "we think it's IBS, try fiber" is a decade of unnecessary suffering. An IBS workup that asks "when did this start, and what happened?" opens the door to a treatable disease.
Key Research Papers
- Marshall JK, et al. Eight-year prognosis of postinfectious irritable bowel syndrome following waterborne bacterial dysentery (Walkerton). Gastroenterology. 2010.
- Spiller R, Garsed K. Postinfectious irritable bowel syndrome. Gut. 2009.
- Pimentel M, et al. Development and validation of a biomarker for diarrhea-predominant IBS (anti-CdtB and anti-vinculin antibodies). PLoS ONE. 2015.
- Klem F, et al. Prevalence, risk factors, and outcomes of irritable bowel syndrome after infectious enteritis: a systematic review and meta-analysis. Gastroenterology. 2017.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on post-infectious IBS biology, biomarkers, and management:
- Post-infectious irritable bowel syndrome
- Walkerton outbreak and long-term IBS
- Anti-CdtB and anti-vinculin antibodies in IBS
- Campylobacter and post-infectious IBS
- Post-Giardia IBS and the Bergen cohort
- Interstitial cells of Cajal, MMC, and SIBO
- Rifaximin for IBS-D (TARGET 3)
- Low-dose naltrexone in functional bowel disease
- Gut-directed hypnotherapy for IBS
Connections
- Irritable Bowel Syndrome
- Post-Viral Triggers
- Alpha-Gal Syndrome
- Lyme Disease
- Chronic Fatigue Syndrome
- IBS and SIBO Overlap
- SIBO
- Low-FODMAP Diet for IBS
- Visceral Hypersensitivity and Brain-Gut Axis
- Gut-Directed Hypnotherapy and CBT
- IBS Medications Guide
- Peppermint, Probiotics, and Herbals
- Rome IV Subtypes
- Anxiety
- Gut-Brain Axis
- Bloating
- Inflammatory Bowel Disease
- Breath Test Guide