Rome IV Subtypes: IBS-C, IBS-D, IBS-M, IBS-U
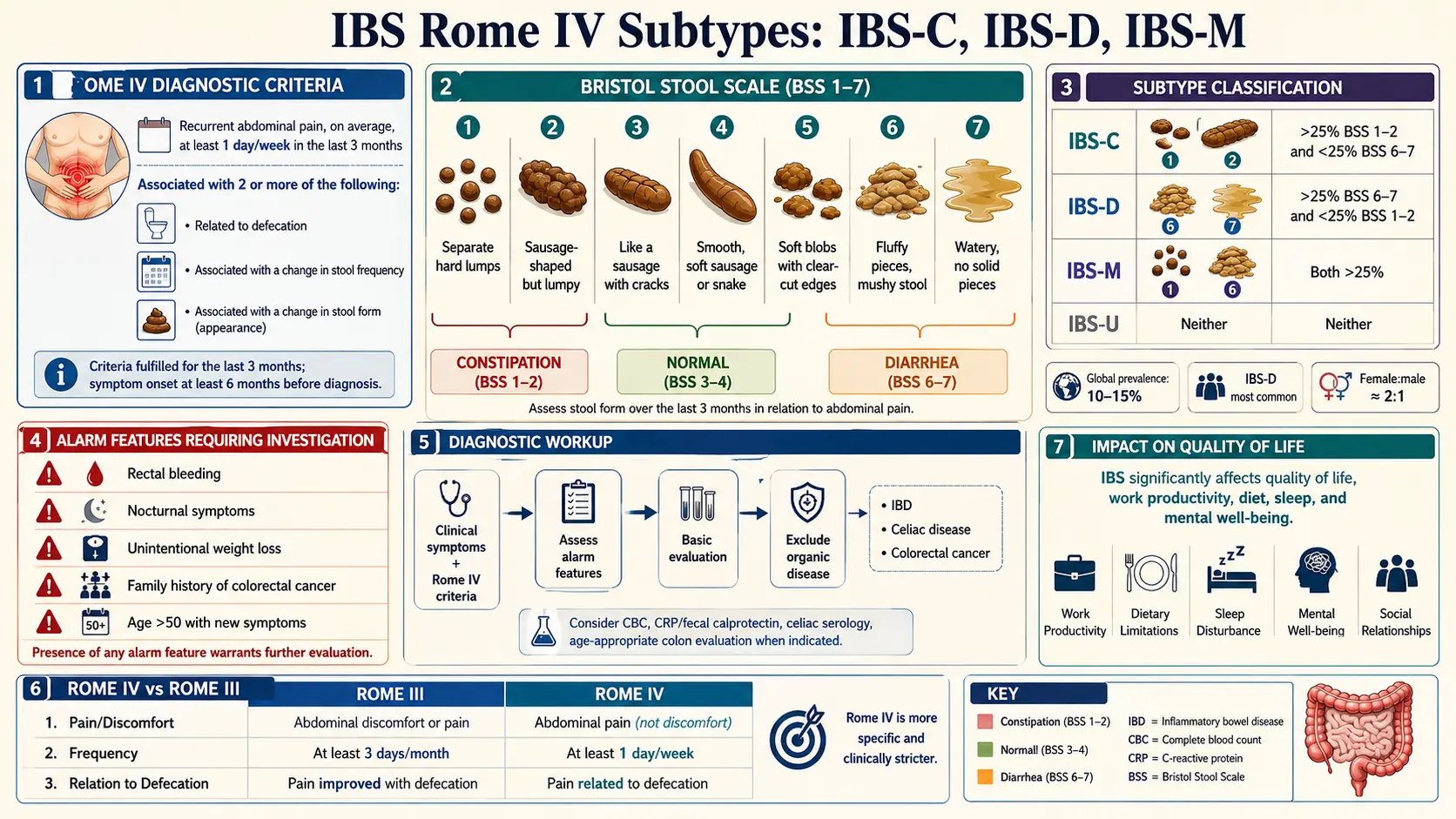
Table of Contents
- Why the Subtype Matters
- The Rome IV Criteria — What Actually Counts as IBS
- The Bristol Stool Chart — Your Measuring Stick
- How Subtypes Are Assigned
- IBS-C — Constipation-Predominant
- IBS-D — Diarrhea-Predominant
- IBS-M — Mixed
- IBS-U — Unclassified
- What Changed from Rome III to Rome IV
- Red Flags — When It Is NOT IBS
- The Minimum Workup Before Accepting the Label
- Why Your Subtype Can Shift Over Time
- IBS-C Overlap with Pelvic-Floor Dyssynergia
- IBS-D Overlap with Bile Acid Malabsorption
- Practical Tracking — Two Weeks of Data
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
Why the Subtype Matters
"IBS" by itself is not a treatment plan. Two patients walking out of the same gastroenterology clinic with the same IBS diagnosis can need almost opposite medications. One needs a laxative-like agent to pull water into the colon. The other needs a drug that slows the gut down and blocks diarrhea. Getting the subtype wrong turns a manageable condition into a miserable one — or worse, an expensive one, because most of the newer IBS medications are subtype-specific and cost hundreds of dollars per month.
The four Rome IV subtypes — IBS-C (constipation), IBS-D (diarrhea), IBS-M (mixed), and IBS-U (unclassified) — exist for exactly this reason. They are not a bureaucratic exercise. They are the practical filter that tells your clinician which drug aisle to shop in and which diet trial to try first. This article walks through how the subtypes are defined, how they are assigned from real stool data, what shifts them over time, and the two commonly missed overlap conditions (pelvic-floor dyssynergia and bile acid malabsorption) that mimic IBS-C and IBS-D.
The Rome IV Criteria — What Actually Counts as IBS
Rome IV, published in 2016 by the Rome Foundation, is the international reference standard for diagnosing IBS and the other disorders of gut-brain interaction. To meet Rome IV criteria for IBS, you need recurrent abdominal pain, on average at least 1 day per week in the last 3 months, associated with two or more of the following:
- Related to defecation (pain improves, or sometimes worsens, with a bowel movement).
- Associated with a change in stool frequency (more or fewer bowel movements than normal).
- Associated with a change in stool form or appearance (harder, looser, more pellet-like, more watery).
There is also a duration requirement: symptoms must have started at least 6 months before diagnosis, even though the "active" window for counting symptoms is the last 3 months. This two-tier clock is how Rome IV separates genuine IBS from a three-week flare of gastroenteritis or a reaction to a new medication.
Notice what is not required: no abnormal blood test, no abnormal imaging, no abnormal colonoscopy. IBS is a positive clinical diagnosis, made on symptom pattern once red flags are excluded. You do not need a negative workup to "earn" the label — you need a consistent symptom picture plus the absence of alarm features (covered below).
The Bristol Stool Chart — Your Measuring Stick
Every subtype decision rides on one tool: the Bristol Stool Form Scale, a seven-point pictorial chart developed at the Bristol Royal Infirmary in 1997 by Heaton and Lewis. It describes stool by shape and consistency rather than frequency.
- Type 1 — separate hard lumps, like nuts. Very hard to pass.
- Type 2 — sausage-shaped but lumpy.
- Type 3 — sausage-shaped with cracks on the surface.
- Type 4 — smooth, soft, sausage or snake-like. This is the "ideal" form.
- Type 5 — soft blobs with clear-cut edges.
- Type 6 — fluffy, ragged pieces, mushy.
- Type 7 — entirely liquid, no solid pieces.
For Rome IV subtyping, Types 1 and 2 are "hard stools" (a sign of slow colonic transit) and Types 6 and 7 are "loose stools" (a sign of fast transit or inflammation). Types 3, 4, and 5 are considered normal and are ignored when assigning a subtype. This is an important point often missed by patients: a day with a Type 4 bowel movement does not count for or against any subtype — it is a "normal" day.
How Subtypes Are Assigned
Rome IV defines the subtypes using only the days with abnormal stool form. You (or your clinician) count the proportion of abnormal bowel movements that are hard versus loose.
- IBS-C (constipation-predominant): more than 25% of bowel movements are Type 1 or 2, and less than 25% are Type 6 or 7.
- IBS-D (diarrhea-predominant): more than 25% are Type 6 or 7, and less than 25% are Type 1 or 2.
- IBS-M (mixed): more than 25% are Type 1 or 2 and more than 25% are Type 6 or 7. Both extremes, same patient.
- IBS-U (unclassified): meets IBS criteria but stool pattern does not fit any of the above, usually because abnormal stools are too infrequent to hit the 25% threshold.
Rome IV strongly recommends calculating these percentages from at least two weeks of daily stool logs, not from memory. Memory consistently overestimates the most unpleasant days — a patient with three dramatic diarrhea episodes a month will often describe themselves as "IBS-D" even when their real pattern is IBS-M or IBS-U. A paper log, notes app, or one of the free stool-tracking apps (Cara Care, Bowelle) settles this quickly.
IBS-C — Constipation-Predominant
IBS-C is the subtype where hard, pellet-like, or straining-heavy bowel movements dominate. The pain is classically crampy, relieved (at least partly) by finally passing stool, and often accompanied by bloating that worsens through the day. Many IBS-C patients wake up flat-bellied and look visibly distended by evening.
First-line management typically stacks in this order: soluble fiber (psyllium, not wheat bran, which can worsen bloating), adequate water, physical activity, then osmotic laxatives such as PEG 3350 (MiraLAX) — available over the counter, cheap, and well-tolerated. When that is not enough, prescription options include linaclotide (Linzess), plecanatide (Trulance), lubiprostone (Amitiza), and tenapanor (Ibsrela). These work by pulling water into the intestine and, in the case of linaclotide and plecanatide, also quiet visceral pain signaling through guanylate cyclase-C receptors on gut nerves.
Antispasmodics (hyoscyamine, dicyclomine, peppermint oil) help with the cramping component across all subtypes but do not move stool. Patients using opioids for other chronic pain should know that opioids reliably worsen IBS-C — see the chronic pain page for non-opioid alternatives.
IBS-D — Diarrhea-Predominant
IBS-D is loose, urgent, often-watery stools, frequently clustered in the morning or after meals. The hallmark is urgency — the sudden "I need a bathroom right now" feeling that patients organize their lives around, memorizing restroom locations at work, grocery stores, and along commutes. Pain is usually crampy and improves after passing stool.
First-line drug therapy is loperamide (Imodium), a cheap over-the-counter antidiarrheal that tightens the gut without entering the brain. Taken 30–60 minutes before a meal or before leaving the house, it can be life-changing for predictable triggers. Prescription options include rifaximin (Xifaxan), a poorly-absorbed antibiotic given as a two-week course that can quiet IBS-D for months — especially in patients with overlapping SIBO; eluxadoline (Viberzi), a gut-targeted opioid receptor modulator; and alosetron (Lotronex), restricted to women with severe IBS-D.
Diet-wise, a structured low-FODMAP elimination and reintroduction — covered in detail on the Low-FODMAP page — helps roughly half of IBS-D patients within four weeks. Bile acid malabsorption (see below) is the single most commonly missed IBS-D mimic and deserves a specific workup before accepting "pure" IBS-D as the final answer.
IBS-M — Mixed
IBS-M is the most complicated subtype to treat because the patient is oscillating between two opposite states. A week of constipation ends in a day or two of diarrhea, then the cycle resets. Some patients swing within a single day: hard, incomplete morning stool followed by loose urgent afternoon stools.
Two practical patterns explain most IBS-M:
- Overflow diarrhea from stool impaction. Hard stool accumulates in the rectum or sigmoid colon, and watery stool from further up leaks around it. This looks like diarrhea but the underlying problem is constipation. A rectal exam or abdominal X-ray showing a loaded colon settles this. Treatment is directed at the constipation — fighting the diarrhea with loperamide makes it worse.
- True oscillation driven by erratic transit, visceral hypersensitivity, and diet. These patients respond best to a two-track approach: low-dose osmotic laxative plus antispasmodic, with loperamide reserved only for clear-cut diarrhea days. Rigid daily use of antidiarrheals will flip them into constipation.
IBS-M patients disproportionately benefit from gut-directed hypnotherapy and CBT because the brain-gut component — anxiety about the next unpredictable episode — drives a measurable portion of the symptom load.
IBS-U — Unclassified
IBS-U is what you get when you meet the pain and abnormal-bowel-habit criteria but your abnormal days do not cluster heavily enough into either hard or loose to hit the 25% thresholds. Most commonly, these are patients with a handful of hard or loose days per month and otherwise normal Type 3–4 stools. IBS-U is a legitimate Rome IV category — it is not a "you don't really have IBS" label.
Practically, IBS-U is managed by tracking and re-evaluating. Given a few more months of symptom data, many IBS-U patients reclassify into IBS-C, IBS-D, or IBS-M. Until they do, treatment is symptom-triggered: antispasmodics for pain, peppermint oil for bloating, a short loperamide course for a bad run of loose days, and osmotic laxative for a stretch of constipation.
What Changed from Rome III to Rome IV
If you have been living with IBS for more than a decade, you may have been diagnosed under the older Rome III criteria (2006). Rome IV made two changes that matter:
- The word "discomfort" is gone. Rome III required "abdominal pain or discomfort." Rome IV dropped "discomfort" because it was vague, translated poorly across languages, and was sometimes used to diagnose IBS in patients whose real issue was functional bloating or functional dyspepsia. Rome IV insists on actual pain.
- The frequency bar went up. Rome III required symptoms at least 3 days per month. Rome IV requires at least 1 day per week on average — roughly a doubling of the minimum frequency.
The practical effect: some patients who met Rome III do not meet Rome IV. That does not mean their condition vanished. It usually means they fit better into a neighboring Rome IV category — functional abdominal bloating/distension, functional constipation, or functional diarrhea. Treatment is largely the same either way, but the label can matter for insurance coverage of specific IBS drugs.
Red Flags — When It Is NOT IBS
Rome IV and every major gastroenterology society list the same set of alarm symptoms that should stop an IBS diagnosis cold and trigger further workup. These are the features IBS does not cause. Any of them deserve prompt evaluation, not reassurance.
- Unintended weight loss — 5% or more of body weight over 6 months without trying.
- Nocturnal symptoms — pain or diarrhea that wakes you from sleep. IBS is essentially a daytime condition; waking at 3 a.m. to run to the bathroom points toward IBD, infection, or bile acid malabsorption.
- Rectal bleeding — any visible blood on the paper, in the bowl, or mixed into stool, beyond what hemorrhoids account for.
- Iron-deficiency anemia on a CBC — points to chronic occult blood loss.
- New-onset symptoms after age 50 — first-time IBS-pattern symptoms in this age group require a colonoscopy to exclude colorectal cancer.
- Family history of colorectal cancer, IBD, or celiac disease in a first-degree relative.
- Fever with abdominal symptoms — IBS does not cause fever.
- Progressive, worsening symptoms rather than a waxing/waning pattern — especially if severity climbs steadily over weeks to months.
The conditions most commonly masquerading as IBS when alarm features are present include inflammatory bowel disease (Crohn's disease and ulcerative colitis), celiac disease, microscopic colitis, colorectal cancer, ovarian cancer in women, and — for IBS-D specifically — bile acid malabsorption and hyperthyroidism.
The Minimum Workup Before Accepting the Label
Rome IV is explicit: a positive symptom-based diagnosis, plus a limited workup to exclude the most common mimics. The minimum reasonable baseline for an adult presenting with IBS-pattern symptoms:
- CBC (complete blood count) — anemia or elevated white count points away from IBS.
- CRP or ESR — inflammation markers. An elevated CRP in a patient with "IBS" should prompt IBD workup.
- Celiac serology — IgA tissue transglutaminase (tTG-IgA) plus total IgA to rule out IgA deficiency. Celiac disease can mimic either IBS-D or IBS-M precisely.
- Fecal calprotectin (especially for IBS-D or IBS-M). A value under 50 µg/g makes IBD very unlikely; values over 150–200 demand colonoscopy. This is the single most useful non-invasive test for separating IBS from IBD.
- TSH — hyperthyroidism can masquerade as IBS-D, hypothyroidism as IBS-C.
Colonoscopy is not required for every IBS patient. It becomes mandatory if any alarm symptom is present, if the patient is over 50, or if fecal calprotectin is elevated. Stool studies for pathogens and Giardia are indicated after recent travel, outbreaks, or sudden-onset post-infectious symptoms — see the post-infectious IBS page.
Why Your Subtype Can Shift Over Time
Subtype is not a life sentence. Longitudinal studies using the Rome III and IV frameworks — including the Manchester IBS registry and the Mayo Rochester Epidemiology Project cohorts — show that roughly one third to one half of IBS patients shift subtype within 1–2 years. The most common transitions are IBS-M to IBS-C, IBS-M to IBS-D, and IBS-D to IBS-M. Stable IBS-C is the most persistent subtype; IBS-U is the least stable.
Drivers of subtype shift include:
- Medication changes. Starting an opioid, an iron supplement, or a calcium-channel blocker can flip IBS-D to IBS-C overnight. Stopping them reverses it. Chronic PPI use can tilt the pattern toward looser stools.
- Diet shifts. A high-fiber or high-FODMAP diet can push a patient toward diarrhea; a low-residue diet can push toward constipation.
- Post-infectious events. Gastroenteritis frequently pushes a stable IBS-C patient into a months-long IBS-D phase.
- Hormonal cycles. Menstrual cycling shifts stool form in many women; pregnancy typically tilts toward IBS-C; menopause often unmasks IBS-D.
- Weight change and physical activity. Both independently influence colonic transit.
Practically, this means it is worth re-subtyping once a year, or any time treatment stops working. A drug that is perfect for IBS-D is actively harmful if the patient has drifted into IBS-C.
IBS-C Overlap with Pelvic-Floor Dyssynergia
Roughly one in three IBS-C patients actually has a mechanical outlet problem rather than — or in addition to — slow colonic transit. Pelvic-floor dyssynergia (also called dyssynergic defecation or anismus) is a learned pattern in which the puborectalis and external anal sphincter muscles contract when they should relax, turning defecation into a push against a closed door.
Clues that a patient's IBS-C is really pelvic-floor dyssynergia:
- Sensation of incomplete evacuation even after a long sit on the toilet.
- Need to strain, splint (press on the perineum or vaginal wall), or use digital maneuvers to pass stool.
- Normal transit studies but poor response to laxatives — or laxatives producing urgency and soiling without real evacuation.
- History of chronic straining, sexual trauma, or postpartum pelvic-floor injury.
Diagnosis is made with anorectal manometry with balloon expulsion (gold standard, available at most academic GI centers) or defecography (dynamic MRI or fluoroscopic study that films the act of defecation). Treatment is not more laxatives; it is pelvic-floor biofeedback with a trained physical therapist, typically 4–6 sessions, with success rates of 70–80% in randomized trials. Patients misdiagnosed as pure IBS-C can spend years on escalating laxative doses before anyone performs the test that would redirect them.
IBS-D Overlap with Bile Acid Malabsorption
An estimated 25–30% of patients labeled IBS-D actually have bile acid malabsorption (BAM), also called bile acid diarrhea. Normally, bile acids released into the small intestine are reabsorbed in the terminal ileum and recycled. When that reabsorption is incomplete, excess bile acids spill into the colon, where they draw in water and accelerate transit — producing urgent, watery, often pale or yellow-tinged stools, classically within an hour of eating a fatty meal and often waking the patient in the early morning.
The gold-standard test is the SeHCAT scan (selenium-75-labeled homotaurocholic acid retention test), widely used in the UK and Europe but essentially unavailable in the United States. Alternatives include serum C4 (7α-hydroxy-4-cholesten-3-one), fasting serum FGF19, or a pragmatic empirical trial of cholestyramine (a bile acid sequestrant; typical starting dose 4 g once or twice daily). A dramatic response within a week — stools reorganize, urgency resolves, bathroom maps get forgotten — is strongly suggestive of BAM and is often accepted as de facto diagnosis when SeHCAT is unavailable.
BAM is particularly common after cholecystectomy (gallbladder removal), after ileal resection (as in some Crohn's patients), and in post-infectious IBS-D. Any IBS-D patient who fails a low-FODMAP trial and loperamide should be specifically screened for BAM before cycling through more expensive therapies.
Practical Tracking — Two Weeks of Data
Before your next GI appointment, give yourself a concrete dataset to work from. For two weeks, record daily: number of bowel movements, Bristol type of each, any pain (0–10 scale, and whether it improved with the bowel movement), meals, menstrual day if applicable, medications and supplements, and sleep. A single page of graph paper or a free app will do.
At the end of two weeks, count only the abnormal days. What percent were Type 1–2? What percent were Type 6–7? Run that through the subtype rules above. Bring the log and your calculated percentages to the appointment. Clinicians work orders of magnitude faster with a stool diary than with "sometimes I'm constipated and sometimes I have diarrhea, it's complicated." The log also catches red flags you might dismiss — two nocturnal episodes in two weeks, say — that change the workup.
Key Research Papers
- Lacy BE, et al. Bowel Disorders. Gastroenterology. 2016. (Rome IV criteria for IBS and related bowel disorders)
- Mearin F, et al. Bowel Disorders (Rome IV). Gastroenterology. 2016.
- Palsson OS, et al. Development and Validation of the Rome IV Diagnostic Questionnaire for Adults. Gastroenterology. 2016.
- Drossman DA. Functional Gastrointestinal Disorders: History, Pathophysiology, Clinical Features and Rome IV. Gastroenterology. 2016.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on Rome IV subtyping, IBS diagnosis, and overlap conditions:
- Rome IV criteria for IBS
- Bristol Stool Form Scale and IBS subtyping
- IBS subtype stability and longitudinal shifts
- Fecal calprotectin in IBS versus IBD
- Pelvic-floor dyssynergia and biofeedback in IBS-C
- Bile acid malabsorption in IBS-D and SeHCAT testing
- IBS alarm symptoms and red flag features
- Linaclotide, plecanatide, and tenapanor for IBS-C
- Rifaximin and eluxadoline for IBS-D
Connections
- Irritable Bowel Syndrome
- Post-Infectious IBS
- Low-FODMAP Diet for IBS
- Gut-Directed Hypnotherapy and CBT
- IBS Medications Guide
- IBS and SIBO Overlap
- Bile Acid Malabsorption
- Peppermint, Probiotics, and Herbals
- Visceral Hypersensitivity and Brain-Gut Axis
- SIBO
- Celiac Disease
- Inflammatory Bowel Disease
- Chronic Pain
- Gallbladder Disease
- Gastroesophageal Reflux Disease
- Constipation
- Bloating
- Anxiety