Vitamin B12 (Cobalamin): The Nerve, Blood, and Vitality Vitamin
Vitamin B12, known scientifically as cobalamin, is a water-soluble vitamin with a distinction shared by no other nutrient: it is the largest and most structurally complex vitamin in the human body, containing at its center a single atom of the trace mineral cobalt — from which its name derives. In natural medicine, B12 is revered as the "vitality vitamin" for its profound and irreplaceable roles in nervous system integrity, red blood cell formation, DNA synthesis, methylation, energy production, and mental health. It is also the nutrient most frequently deficient in the modern world, with estimates suggesting that up to 40% of the general population may have suboptimal B12 levels — a silent epidemic with devastating consequences for the brain, nerves, blood, and every cell that depends on methylation for its proper function.
Table of Contents
- Nervous System — Protection, Maintenance, and Repair
- Methylation — The Engine of Cellular Function
- Red Blood Cell Formation and Megaloblastic Anemia
- Brain Health, Cognitive Function, and Dementia Prevention
- Mental Health — Depression, Anxiety, and Psychosis
- Energy Production and Mitochondrial Function
- Cardiovascular Health
- Bone Health
- Digestive Health and the Absorption Cascade
- Immune System Function
- Fertility and Reproductive Health
- Skin, Hair, and Mucous Membranes
- Detoxification and Cellular Protection
- Natural Food Sources
- Recommended Daily Intake
- Supplemental Forms
- Assessment of B12 Status — Beyond Serum B12
- Synergistic Nutrients
- Populations at Higher Risk of Deficiency
- Signs of Deficiency
- Special Therapeutic Applications
- Connections
- Featured Videos
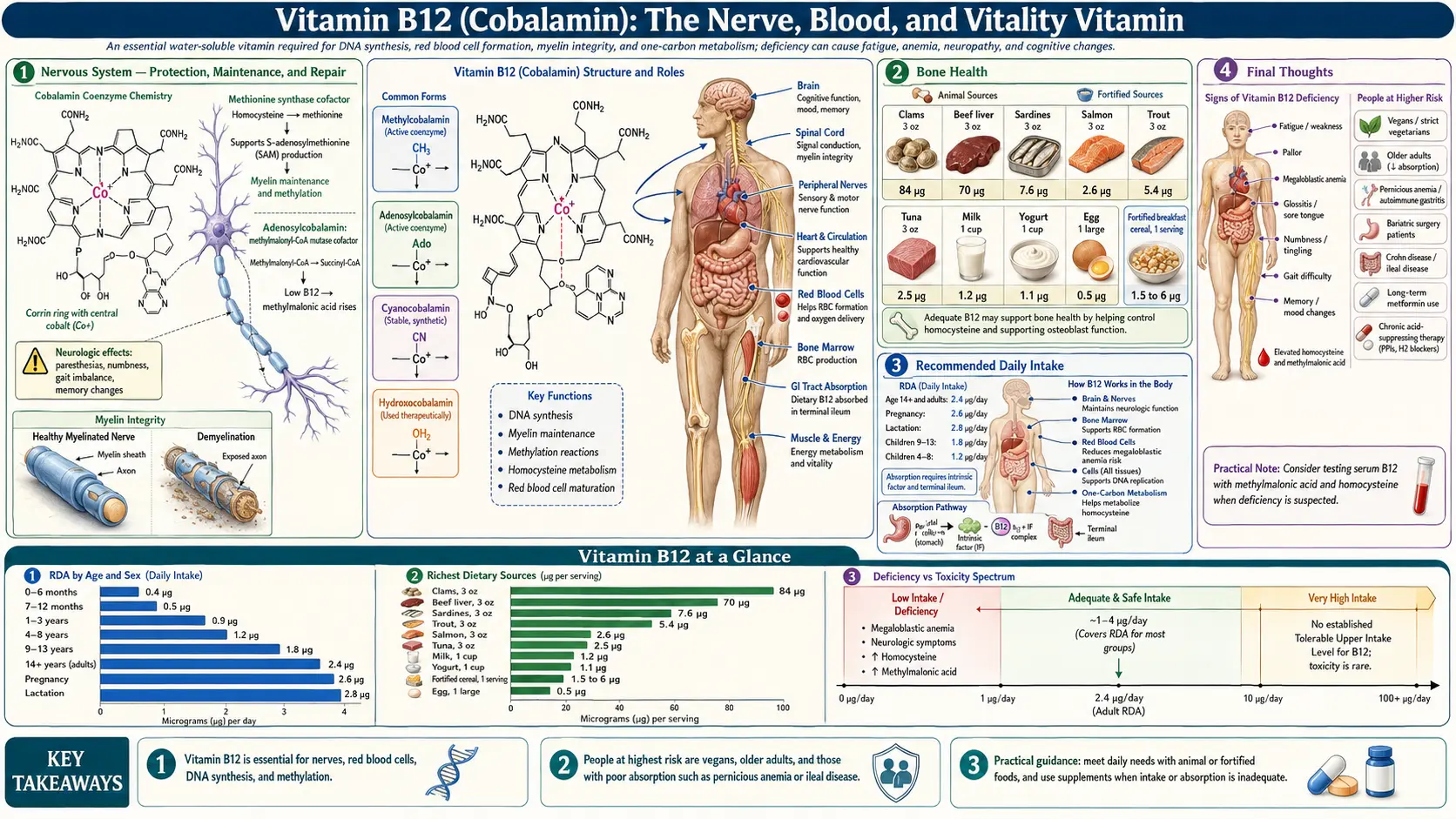
1. Nervous System — Protection, Maintenance, and Repair
If there is one role for which Vitamin B12 is most essential and most feared when deficient, it is its irreplaceable function in maintaining the integrity of the nervous system.
- Myelin Sheath Synthesis: B12 is absolutely required for the synthesis and maintenance of myelin — the fatty, insulating sheath that wraps around nerve fibers throughout the brain, spinal cord, and peripheral nervous system. Myelin enables rapid, accurate electrical signal transmission between neurons. Without B12, myelin progressively degenerates, leading to nerve dysfunction and, eventually, irreversible neurological damage.
- S-Adenosylmethionine (SAMe) and Myelin: B12 drives the methylation cycle that produces SAMe, which in turn donates methyl groups required for the synthesis of phosphatidylcholine and sphingomyelin — the primary lipid components of myelin. B12 deficiency therefore starves the nervous system of the raw materials it needs to maintain its own insulation.
- Subacute Combined Degeneration: Prolonged, severe B12 deficiency causes subacute combined degeneration of the spinal cord — a progressive demyelination of the dorsal and lateral columns that produces numbness, tingling, loss of proprioception (position sense), gait instability, weakness, and eventually paralysis. This is one of the most feared complications of B12 deficiency, and if not caught early, the damage can become permanent.
- Peripheral Neuropathy: Numbness, tingling, burning, and "pins and needles" sensations in the hands and feet are among the most common neurological manifestations of B12 deficiency. These symptoms can precede anemia by months or even years, making them critical early warning signs.
- Optic Neuropathy: B12 deficiency can damage the optic nerve, causing visual disturbances, reduced visual acuity, and in severe cases, optic atrophy and vision loss.
- Autonomic Neuropathy: The autonomic nervous system — governing heart rate, blood pressure, digestion, bladder function, and temperature regulation — can be affected by B12 deficiency, causing orthostatic hypotension, urinary incontinence, gastroparesis, and abnormal sweating.
- Irreversibility Warning: Unlike many nutritional deficiencies, neurological damage from prolonged B12 deficiency may be only partially reversible or entirely irreversible once demyelination has progressed beyond a certain point. Early detection and treatment are paramount.
2. Methylation — The Engine of Cellular Function
B12 is one of only two essential cofactors (alongside folate) for the methylation cycle — the biochemical process that touches virtually every aspect of cellular health.
- Methionine Synthase: B12 (as methylcobalamin) is the essential cofactor for the enzyme methionine synthase, which transfers a methyl group from 5-methyltetrahydrofolate (5-MTHF) to homocysteine, regenerating methionine. This single reaction sits at the intersection of folate metabolism, homocysteine clearance, and SAMe production — making B12 the gatekeeper of the entire methylation cycle.
- SAMe Production: Methionine is converted to S-adenosylmethionine (SAMe), the universal methyl donor that participates in over 200 methylation reactions including DNA methylation (epigenetics), neurotransmitter synthesis, phospholipid production, creatine formation, and hormone metabolism.
- The Methyl Trap: When B12 is deficient, 5-MTHF cannot donate its methyl group and becomes "trapped" in its methylated form. This creates a functional folate deficiency even when folate intake is adequate — disrupting both DNA synthesis (causing megaloblastic anemia) and methylation simultaneously. This phenomenon, called the "methyl trap," explains why B12 deficiency produces the same hematological picture as folate deficiency.
- Epigenetic Regulation: By driving SAMe production, B12 supports the DNA methylation that governs gene expression — turning genes on and off in patterns critical for normal development, cellular differentiation, and cancer prevention.
- Homocysteine Clearance: The methionine synthase reaction is the primary pathway for homocysteine remethylation. B12 deficiency causes homocysteine accumulation — an independent risk factor for cardiovascular disease, stroke, dementia, and pregnancy complications.
3. Red Blood Cell Formation and Megaloblastic Anemia
- DNA Synthesis for Erythropoiesis: Red blood cell precursors in the bone marrow are among the most rapidly dividing cells in the body. B12, through its role in releasing folate from the methyl trap, is essential for the thymidine synthesis that drives DNA replication during erythropoiesis.
- Megaloblastic Anemia: B12 deficiency produces megaloblastic (macrocytic) anemia — identically to folate deficiency. Red blood cell precursors cannot properly replicate their DNA, resulting in abnormally large, immature, oval-shaped red blood cells (macro-ovalocytes) that are inefficient at carrying oxygen. Symptoms include profound fatigue, weakness, pallor, shortness of breath, rapid heart rate, and dizziness.
- Pernicious Anemia: The most well-known cause of B12 deficiency is pernicious anemia — an autoimmune condition in which the body produces antibodies against intrinsic factor (IF) or the parietal cells of the stomach that produce IF. Without intrinsic factor, B12 cannot be absorbed in the ileum regardless of dietary intake. Pernicious anemia requires lifelong B12 supplementation, typically via injection or high-dose sublingual/oral forms.
- Pancytopenia: In severe B12 deficiency, the production of all blood cell lines can be affected — red blood cells (anemia), white blood cells (leukopenia), and platelets (thrombocytopenia) — a dangerous condition called pancytopenia.
- Hypersegmented Neutrophils: As with folate deficiency, hypersegmented neutrophils (white blood cells with 5+ nuclear lobes) on a peripheral blood smear are among the earliest and most sensitive indicators of B12-related megaloblastic changes.
- Critical Distinction: Supplementing with folate (or folic acid) can correct the anemia of B12 deficiency by bypassing the methyl trap, but it does NOT address the neurological damage, which continues to progress silently. This is why masking B12 deficiency with folic acid is considered dangerous, and why both nutrients must always be assessed together.
4. Brain Health, Cognitive Function, and Dementia Prevention
- Brain Atrophy: Low B12 status is consistently associated with accelerated brain atrophy (loss of brain volume) — a hallmark of cognitive decline and dementia. Landmark studies have demonstrated that supplementation with B12, folate, and B6 can slow the rate of brain shrinkage by up to 53% in individuals with elevated homocysteine and mild cognitive impairment.
- Alzheimer's Disease: Low B12, elevated homocysteine, and impaired methylation are all strongly associated with increased Alzheimer's disease risk. B12 deficiency contributes to the accumulation of neurotoxic compounds, oxidative stress, and the disruption of the phospholipid membranes essential for synaptic function.
- Vascular Dementia: Through its homocysteine-lowering effects, B12 protects cerebral vasculature and reduces the risk of vascular dementia — the second most common form of dementia after Alzheimer's.
- Cognitive Performance: Adequate B12 status is associated with better performance on tests of memory, processing speed, verbal fluency, and executive function across all age groups, but particularly in the elderly.
- Pseudodementia: In some cases, cognitive impairment caused by B12 deficiency can mimic true dementia so convincingly that it is misdiagnosed as Alzheimer's disease. These cases are potentially fully reversible with B12 repletion — making B12 assessment essential in every dementia workup.
- White Matter Lesions: B12 deficiency and elevated homocysteine are associated with increased white matter hyperintensities on brain MRI — areas of demyelination and vascular damage that correlate with cognitive decline.
5. Mental Health — Depression, Anxiety, and Psychosis
- Neurotransmitter Synthesis: B12, through the methylation cycle and SAMe production, supports the synthesis and metabolism of serotonin, dopamine, norepinephrine, and melatonin. Deficiency impairs the production of these mood-regulating neurotransmitters at the most fundamental biochemical level.
- Depression: Low B12 status is one of the most consistently reported nutritional findings in major depressive disorder. Studies demonstrate that adequate B12 is associated with better treatment response to antidepressants, and B12 supplementation may improve mood outcomes in depressed patients — particularly those with documented deficiency.
- Anxiety and Irritability: B12 deficiency-related neurotransmitter dysfunction and nervous system irritability can manifest as anxiety, panic, emotional lability, and agitation.
- Psychosis and Psychiatric Manifestations: Severe B12 deficiency has been documented to cause frank psychiatric symptoms including paranoia, delusions, hallucinations, violent behavior, and personality changes — sometimes called "megaloblastic madness." These psychiatric manifestations can occur even before anemia develops, making B12 status an essential investigation in any new-onset psychiatric presentation.
- Insomnia: B12 plays a role in melatonin synthesis and circadian rhythm regulation. Deficiency may contribute to sleep disturbances, delayed sleep phase syndrome, and poor sleep quality.
- Postpartum Depression: Depleted B12 stores after pregnancy and lactation may contribute to postpartum depression, and repletion is increasingly included in natural postpartum recovery protocols.
6. Energy Production and Mitochondrial Function
- Methylmalonyl-CoA Mutase: B12 (as adenosylcobalamin) is the essential cofactor for methylmalonyl-CoA mutase, the enzyme that converts methylmalonyl-CoA to succinyl-CoA — a critical step that feeds odd-chain fatty acids, branched-chain amino acids (valine, isoleucine), and certain other substrates into the Krebs cycle for ATP production.
- Methylmalonic Acid (MMA) Accumulation: When B12 is deficient, methylmalonyl-CoA cannot be converted to succinyl-CoA, and methylmalonic acid (MMA) accumulates in the blood and urine. Elevated MMA is the most specific and sensitive biomarker of functional B12 deficiency at the tissue level, and is often the first metabolic abnormality to appear — even before serum B12 levels drop below the conventional reference range.
- Mitochondrial Health: By supplying succinyl-CoA to the Krebs cycle, B12 directly supports mitochondrial energy production. Deficiency starves the mitochondria of a key substrate, contributing to fatigue and reduced cellular energy output.
- Heme Synthesis: Succinyl-CoA is also the starting substrate for heme biosynthesis — the production of the iron-containing molecule in hemoglobin. B12 deficiency therefore impairs oxygen transport through two mechanisms: megaloblastic anemia (DNA synthesis failure) and reduced heme production.
- Chronic Fatigue: One of the most universally reported symptoms of B12 deficiency is profound, unrelenting fatigue that does not improve with rest. This reflects the combined effects of impaired energy metabolism, anemia, and neurological dysfunction.
- Exercise Tolerance: Reduced aerobic capacity, premature fatigue, and poor exercise recovery can all stem from B12 deficiency-related anemia and impaired mitochondrial function.
7. Cardiovascular Health
- Homocysteine — The Primary Mechanism: B12's most important cardiovascular function is its role in converting homocysteine to methionine via methionine synthase. Elevated homocysteine directly damages vascular endothelium, promotes oxidative stress, increases platelet aggregation, impairs nitric oxide production, and accelerates atherosclerosis.
- The B12-Folate-B6 Triad: Optimal homocysteine management requires adequate levels of all three nutrients working in concert: folate and B12 for remethylation, and B6 for transsulfuration. Deficiency of any one member of this triad compromises the entire system.
- Stroke Prevention: Meta-analyses have demonstrated that B-vitamin supplementation (B12 + folate ± B6) reduces stroke risk, particularly in populations without mandatory folic acid food fortification.
- Peripheral Vascular Disease: Elevated homocysteine from B12 deficiency is a risk factor for peripheral arterial disease and deep vein thrombosis.
- Endothelial Function: B12, by reducing homocysteine and supporting nitric oxide production, helps maintain healthy vascular endothelial function — the critical interface between blood and vessel walls.
- Heart Failure: Emerging evidence suggests that B12 deficiency is common among heart failure patients and may contribute to disease severity through homocysteine-mediated vascular damage and impaired myocardial energy metabolism.
8. Bone Health
- Homocysteine and Collagen: Elevated homocysteine from B12 deficiency interferes with collagen cross-linking in bone tissue, weakening the organic bone matrix independently of bone mineral density. This means standard DEXA scans may underestimate fracture risk in B12-deficient individuals.
- Osteoblast Function: B12 appears to support osteoblast (bone-building cell) activity and the methylation reactions required for normal bone metabolism.
- Osteoporosis and Fracture Risk: Multiple large epidemiological studies have demonstrated that low B12 status is independently associated with reduced bone mineral density and significantly increased hip fracture risk, particularly in the elderly.
- Methylation and Bone Turnover: The methylation cycle supported by B12 influences the expression of genes involved in bone remodeling, potentially affecting the balance between bone formation and resorption.
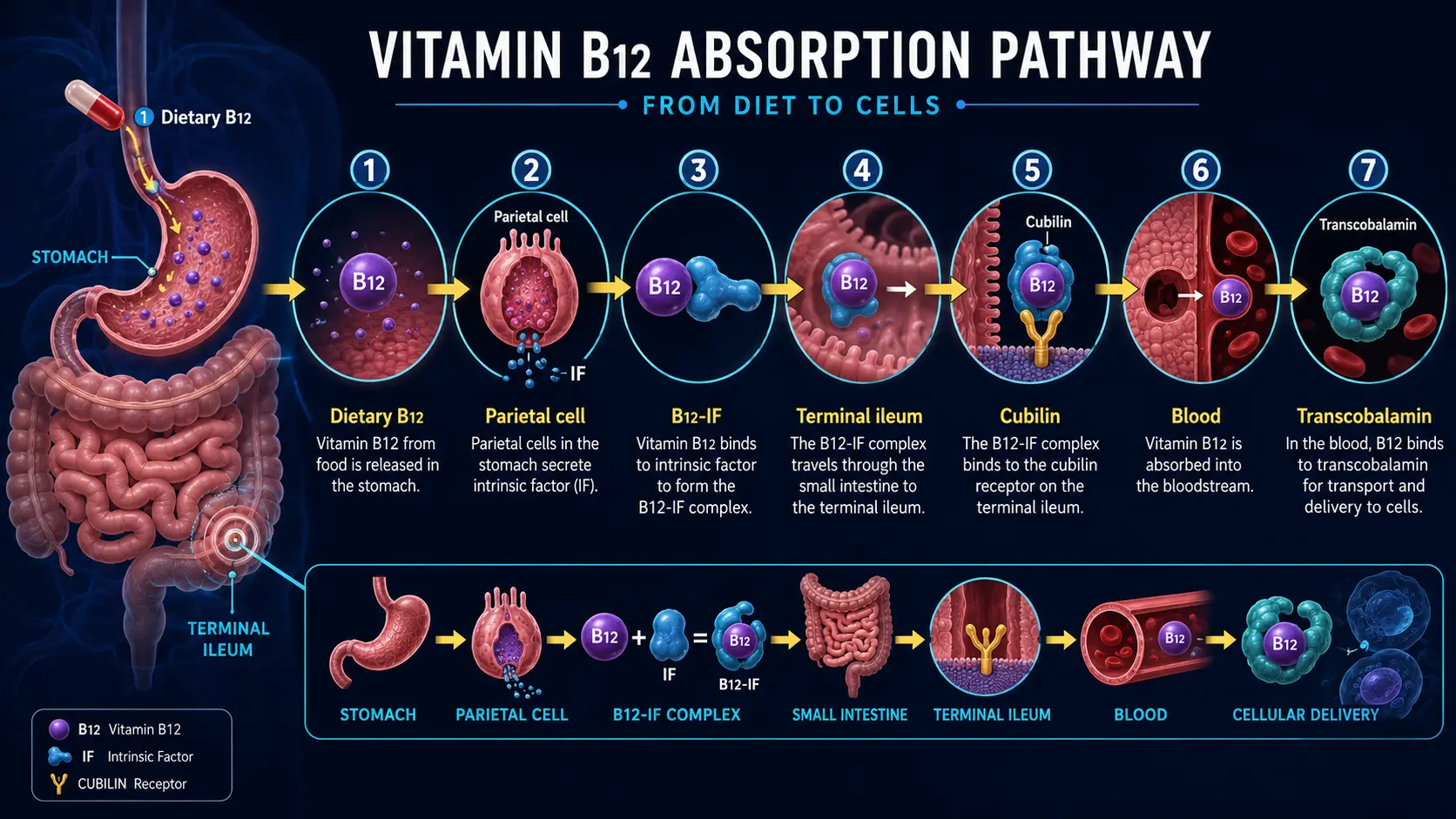
9. Digestive Health and the Absorption Cascade
B12 has the most complex absorption process of any vitamin — requiring multiple steps that are each vulnerable to disruption.
- Step 1 — Stomach Acid Release: B12 in food is bound to proteins. Hydrochloric acid (HCl) and pepsin in the stomach cleave B12 from its protein carriers. Individuals with low stomach acid (hypochlorhydria) — including those on proton pump inhibitors (PPIs) or H2 blockers — cannot efficiently release B12 from food.
- Step 2 — Haptocorrin Binding: Free B12 binds to haptocorrin (R-protein), a salivary and gastric glycoprotein, which protects B12 from acid degradation as it passes through the stomach.
- Step 3 — Intrinsic Factor: In the duodenum, pancreatic enzymes degrade haptocorrin, releasing B12 to bind with intrinsic factor (IF) — a glycoprotein produced exclusively by the parietal cells of the stomach. This B12-IF complex is essential for absorption.
- Step 4 — Ileal Absorption: The B12-IF complex travels to the terminal ileum, where it binds to specific receptors (cubilin-amnionless complex) and is absorbed via receptor-mediated endocytosis. This is the only efficient absorption site for physiological doses of B12.
- Step 5 — Transport: Once absorbed, B12 is bound to transcobalamin II (TC-II) for delivery to tissues throughout the body. The fraction bound to TC-II (called holotranscobalamin or holoTC) represents the biologically active, tissue-available B12 — and is increasingly recognized as a superior marker of B12 status compared to total serum B12.
- Enterohepatic Recirculation: B12 is secreted in bile and reabsorbed in the ileum — a recycling process that can conserve B12 for years in healthy individuals. However, any disruption to this cycle (biliary disease, ileal resection, bacterial overgrowth) accelerates depletion.
- Passive Absorption: Approximately 1–2% of oral B12 can be absorbed passively (without intrinsic factor) across the intestinal mucosa by mass action. This is the mechanism that allows high-dose oral B12 supplementation (1,000–5,000 mcg) to be effective even in individuals lacking intrinsic factor.
10. Immune System Function
- Immune Cell Proliferation: T-cells, B-cells, and NK cells all require DNA synthesis for rapid clonal expansion during immune responses. B12, through its role in releasing folate from the methyl trap, supports the DNA synthesis essential for this proliferative burst.
- NK Cell Activity: B12 deficiency is associated with reduced natural killer cell cytotoxicity — a critical frontline defense against virally infected and malignant cells.
- T-Cell Function: Low B12 status impairs T-cell proliferation, reduces CD8+ cytotoxic T-cell counts, and alters the CD4/CD8 ratio — weakening cell-mediated immunity.
- Antibody Production: Impaired B-cell function and immunoglobulin synthesis in B12 deficiency reduce the body's ability to produce antibodies in response to infections.
- Inflammatory Modulation: B12 has been shown to modulate inflammatory cytokine production, and deficiency is associated with elevated inflammatory markers that can drive chronic disease.
- Autoimmunity Connection: Pernicious anemia itself is an autoimmune condition, and individuals with one autoimmune disease are at increased risk for others. B12 deficiency is frequently comorbid with Hashimoto's thyroiditis, type 1 diabetes, vitiligo, and other autoimmune conditions.
11. Fertility and Reproductive Health
- Female Fertility: B12 is essential for proper DNA synthesis and methylation during oocyte (egg) development. Deficiency has been associated with anovulation, implantation failure, and recurrent early pregnancy loss.
- Male Fertility: B12 supports spermatogenesis — the production and maturation of sperm cells. Deficiency is associated with reduced sperm count, impaired motility, increased DNA fragmentation in sperm, and elevated homocysteine in seminal fluid.
- Pregnancy Outcomes: Low maternal B12 status is associated with increased risk of neural tube defects, preterm birth, low birth weight, preeclampsia, and recurrent miscarriage — effects mediated through impaired methylation, elevated homocysteine, and defective DNA synthesis.
- Polycystic Ovary Syndrome (PCOS): Women with PCOS, particularly those taking metformin (which depletes B12), are at increased risk of B12 deficiency with potential impacts on fertility and metabolic health.
- Epigenetic Programming: Maternal B12 status influences the epigenetic programming of the developing fetus through DNA methylation, potentially affecting the child's long-term metabolic and neurological health.
12. Skin, Hair, and Mucous Membranes
- Hyperpigmentation: B12 deficiency can cause a distinctive reversible hyperpigmentation of the skin, particularly over the knuckles, nail beds, and oral mucosa. This finding is especially common in individuals with darker skin tones and can be a valuable clinical clue.
- Vitiligo: Loss of skin pigmentation (vitiligo) has been associated with B12 deficiency and autoimmune pernicious anemia. Some case reports document repigmentation following B12 supplementation.
- Glossitis and Oral Changes: A smooth, beefy-red, painful tongue (Hunter's glossitis) is a classic sign of B12 deficiency. Angular cheilitis, mouth ulcers, and oral burning syndrome may also occur.
- Hair Health: B12 deficiency has been associated with premature hair graying and hair loss, likely related to impaired DNA synthesis and methylation in rapidly dividing hair follicle cells.
- Nail Changes: Bluish-black nail discoloration and fragile nails have been reported in B12 deficiency.
13. Detoxification and Cellular Protection
- Glutathione Support: B12 drives the methylation cycle that produces methionine, which feeds the transsulfuration pathway (B6-dependent) to produce cysteine — the rate-limiting precursor to glutathione. Adequate B12 is therefore essential for maintaining glutathione levels, the body's master antioxidant and central molecule of Phase II liver detoxification.
- Cyanide Detoxification: Hydroxocobalamin (a natural form of B12) has such a strong affinity for cyanide that it is used as an FDA-approved antidote for cyanide poisoning (Cyanokit®). It binds cyanide to form cyanocobalamin, which is then safely excreted by the kidneys.
- Nitric Oxide Scavenging: B12 can scavenge excess nitric oxide and peroxynitrite — reactive nitrogen species that cause oxidative and nitrosative stress in the brain and throughout the body. This neuroprotective function may be particularly relevant in conditions involving neuroinflammation.
- Sulfite Detoxification: Through its support of methylation and sulfur amino acid metabolism, B12 indirectly supports the body's ability to process and eliminate sulfites — relevant for individuals with sulfite sensitivity.
14. Natural Food Sources
Vitamin B12 is unique among all vitamins in that it is produced exclusively by microorganisms (bacteria and archaea). It does not occur naturally in any plant food. All dietary B12 comes from animal foods (where B12 is concentrated from microbial synthesis in the animal's gut) or from bacterial fermentation.
Richest Whole Food Sources
- Clams and Shellfish: The single richest natural food source of B12 — a 3-ounce serving of clams provides over 1,000% of the daily value
- Beef Liver and Organ Meats: Extraordinarily concentrated in B12, liver is the traditional food most valued for preventing and treating B12 deficiency
- Sardines, Mackerel, and Herring: Small, oily fish that provide B12 along with omega-3 fatty acids, vitamin D, and selenium
- Wild-Caught Salmon and Trout: Excellent sources of B12 in a nutrient-dense, omega-3-rich food matrix
- Beef, Lamb, and Venison (grass-fed): Red meat provides highly bioavailable B12 along with iron, zinc, and other B vitamins
- Eggs (pasture-raised): Provide B12 primarily in the yolk, in a convenient, affordable, and versatile food
- Dairy Products (grass-fed): Yogurt, cheese, and milk contain meaningful amounts of B12
- Chicken and Turkey: Moderate sources of B12, with dark meat providing slightly more than white meat
- Nutritional Yeast (fortified): The primary B12 source for many vegans — note that only fortified nutritional yeast contains B12; unfortified nutritional yeast does not
- Nori Seaweed: Some studies suggest that certain nori varieties contain bioactive B12, though the amount and bioavailability remain debated. It should not be relied upon as a primary B12 source
Critical Note for Vegans and Vegetarians
- No Reliable Plant Sources: There are no proven, reliable, unfortified plant sources of bioactive Vitamin B12. Claims about spirulina, chlorella, tempeh, and fermented foods containing B12 are largely based on the presence of B12 analogues (pseudovitamin B12) — inactive forms that cannot fulfill B12's metabolic functions and may actually interfere with true B12 absorption and assays.
- Mandatory Supplementation: All vegans and most vegetarians should supplement with B12 without exception. This is not a matter of debate in nutritional science — it is a biological necessity. Natural practitioners recommend methylcobalamin or hydroxocobalamin supplementation for all individuals following plant-based diets.
- Storage and Depletion Timeline: The liver stores 2–5 mg of B12 — enough to last 3–5 years in a healthy individual with intact enterohepatic circulation. However, once stores are depleted, deficiency develops rapidly, and neurological damage can occur before anemia appears.
15. Recommended Daily Intake
The following are general guidelines measured in micrograms (mcg):
- Adult Men and Women: 2.4 mcg per day
- Pregnant Women: 2.6 mcg per day
- Breastfeeding Women: 2.8 mcg per day
- Children (4–8 years): 1.2 mcg per day
- Children (9–13 years): 1.8 mcg per day
- Adults over 50: The Institute of Medicine recommends that all adults over 50 obtain B12 from supplements or fortified foods, as the ability to extract B12 from food protein declines significantly with age due to reduced stomach acid production
Therapeutic Doses: In natural medicine practice, supplemental doses commonly range from 500 mcg to 5,000 mcg daily for maintenance and repletion. For pernicious anemia and severe deficiency, intramuscular injections of 1,000 mcg are administered daily or weekly until repletion, followed by monthly maintenance injections or daily high-dose oral/sublingual supplementation. B12 has no established upper toxicity limit — it is considered safe even at very high doses, as excess is excreted in the urine.
16. Supplemental Forms
The form of B12 matters significantly in natural medicine, as each has distinct metabolic properties and clinical applications.
- Methylcobalamin: The methylated, coenzyme form of B12 that participates directly in the methionine synthase reaction (methylation cycle). It is the preferred form for methylation support, neurological health, mood disorders, and general supplementation. It is light-sensitive and less stable than cyanocobalamin, requiring proper storage.
- Hydroxocobalamin: A naturally occurring form produced by bacteria, with the longest retention time in the body and the greatest affinity for binding and clearing cyanide and nitric oxide. It is the preferred form for intramuscular injections in many countries (especially Europe), for cyanide detoxification, and for individuals with nitric oxide-related issues. It converts to both methylcobalamin and adenosylcobalamin in the body.
- Adenosylcobalamin (Dibencozide): The coenzyme form that participates in the methylmalonyl-CoA mutase reaction (energy production and fatty acid metabolism) within the mitochondria. It is specifically relevant for addressing elevated MMA levels and supporting mitochondrial function. Less commonly available as a standalone supplement.
- Cyanocobalamin: The synthetic, most stable, and least expensive form. It contains a cyanide molecule that must be removed and the molecule converted to active forms (methyl- or adenosylcobalamin) before use. While it is the most studied form and effective at raising serum B12 levels, natural practitioners generally prefer the bioactive forms, particularly for individuals with detoxification concerns, MTHFR variants, or neurological conditions.
- Delivery Methods:
- Sublingual tablets and lozenges: Dissolve under the tongue for direct mucosal absorption, bypassing the digestive tract. Preferred for maintenance supplementation.
- Intramuscular (IM) injections: The gold standard for severe deficiency, pernicious anemia, and neurological involvement. Provides complete bioavailability regardless of absorptive capacity.
- High-dose oral: 1,000–5,000 mcg oral B12 can achieve adequate repletion even in pernicious anemia through passive absorption (1–2% of the dose is absorbed without intrinsic factor).
- Nasal sprays and transdermal patches: Alternative delivery methods with variable bioavailability.
17. Assessment of B12 Status — Beyond Serum B12
Accurately assessing B12 status is more complex than a simple blood test, and natural practitioners should understand the full spectrum of available markers.
- Serum B12: The most commonly ordered test, but also the least sensitive. Serum B12 measures total circulating B12, including inactive analogues bound to haptocorrin. The conventional "normal" range (200–900 pg/mL) is widely considered too broad — functional deficiency can exist at levels within the "normal" range, particularly between 200–450 pg/mL.
- Methylmalonic Acid (MMA): The most specific marker of functional, tissue-level B12 deficiency. MMA rises when B12 is insufficient for the methylmalonyl-CoA mutase reaction. Elevated MMA (>0.4 µmol/L) in the presence of normal or low-normal serum B12 confirms functional deficiency.
- Homocysteine: Rises when B12 (or folate) is insufficient for the methionine synthase reaction. Sensitive but not specific to B12 alone — folate, B6, and kidney function also affect levels.
- Holotranscobalamin (HoloTC): Measures the biologically active fraction of B12 bound to transcobalamin II — the portion actually available to tissues. Increasingly recognized as the earliest and most sensitive marker of B12 depletion. Low holoTC (<35 pmol/L) indicates reduced B12 delivery to cells.
- Complete Blood Count (CBC): May reveal macrocytosis (elevated MCV) and hypersegmented neutrophils in advanced deficiency, but hematological changes are often late findings — neurological damage can be well established before the CBC becomes abnormal.
- Natural Practitioner Approach: Optimal assessment uses a combination of serum B12, MMA, and homocysteine (and holoTC when available). Target serum B12 levels of >500 pg/mL (ideally 600–900 pg/mL) with normal MMA and homocysteine are preferred in functional and natural medicine practice.
18. Synergistic Nutrients
- Folate (Vitamin B9): The most critical synergistic partner. B12 and folate are metabolically inseparable — B12 is required to release folate from the methyl trap, and folate provides the methyl group that B12-dependent methionine synthase uses to clear homocysteine. Deficiency of either one functionally impairs the other.
- Vitamin B6 (Pyridoxine): The third member of the homocysteine-management triad. B6 clears homocysteine via the transsulfuration pathway, complementing the remethylation pathway driven by B12 and folate.
- Vitamin B2 (Riboflavin): FAD (from riboflavin) is the cofactor for MTHFR, the enzyme that produces 5-MTHF — the methyl donor that B12-dependent methionine synthase uses. Without adequate B2, the folate-B12 methylation axis is impaired upstream.
- Iron: Both B12 deficiency and iron deficiency cause anemia, and they frequently coexist. B12 supports heme synthesis (via succinyl-CoA), and iron provides the metal center of the heme molecule. Both must be adequate for healthy red blood cell production.
- Betaine (TMG): An alternative methyl donor that can convert homocysteine to methionine via the BHMT enzyme, providing a backup methylation pathway when B12-dependent methionine synthase is compromised.
- Intrinsic Factor: While not a nutrient, intrinsic factor is the essential binding protein required for B12 absorption. Its absence (in pernicious anemia or after gastric surgery) is the most important physiological barrier to B12 sufficiency.
- Stomach Acid (HCl): Adequate gastric acid is required to liberate B12 from food proteins — the essential first step of absorption. Hypochlorhydria dramatically impairs food-bound B12 extraction.
19. Populations at Higher Risk of Deficiency
- Vegans: The single highest-risk population. Without supplementation, B12 deficiency is virtually inevitable on a strict vegan diet. Stores may sustain adequate levels for 3–5 years before clinical deficiency appears.
- Vegetarians: Lacto-ovo vegetarians obtain some B12 from eggs and dairy but are still at significantly increased risk compared to omnivores, particularly if these foods are consumed infrequently.
- Elderly Individuals: Age-related atrophic gastritis affects up to 30% of adults over 60, reducing stomach acid production and impairing food-bound B12 absorption. This is the most common cause of B12 deficiency in the elderly — and it occurs even with adequate dietary intake.
- Individuals on Proton Pump Inhibitors (PPIs): Long-term PPI use (omeprazole, esomeprazole, etc.) suppresses stomach acid production, significantly reducing B12 extraction from food proteins. Risk increases with duration of use.
- Individuals on Metformin: Metformin, the most widely prescribed diabetes medication worldwide, reduces B12 absorption by up to 30% through mechanisms that may involve calcium-dependent ileal absorption disruption. B12 monitoring is recommended for all metformin users.
- Individuals with Pernicious Anemia: Autoimmune destruction of parietal cells or intrinsic factor antibodies prevent B12 absorption entirely, requiring lifelong supplementation.
- Individuals with Gastrointestinal Conditions: Celiac disease, Crohn's disease (especially ileal), small intestinal bacterial overgrowth (SIBO), Helicobacter pylori infection, and tropical sprue all impair B12 absorption.
- Post-Bariatric Surgery Patients: Gastric bypass and sleeve gastrectomy fundamentally alter B12 absorption anatomy, creating lifelong supplementation requirements.
- Individuals with Alcohol Use Disorders: Alcohol damages gastric mucosa, impairs intrinsic factor production, disrupts ileal absorption, and accelerates B12 depletion.
- Pregnant and Breastfeeding Women: Increased demands for fetal development, DNA synthesis, and breast milk production elevate B12 requirements. Vegan and vegetarian mothers are at particular risk of having B12-deficient infants.
- Infants Born to B12-Deficient Mothers: Infants are born with limited B12 stores and depend on breast milk for their supply. Infants of B12-deficient or vegan/vegetarian mothers can develop severe neurological damage within months of birth if not supplemented — a medical emergency that is entirely preventable.
- Individuals on H2 Receptor Blockers: Ranitidine, famotidine, and other H2 blockers reduce stomach acid and impair food-bound B12 absorption with prolonged use.
- Individuals with Nitrous Oxide Exposure: Nitrous oxide (N₂O — "laughing gas") irreversibly oxidizes and inactivates B12. Chronic or repeated exposure (recreational use, frequent dental procedures, occupational exposure) can precipitate acute, severe B12 deficiency with devastating neurological consequences, even in individuals with previously normal B12 stores.
20. Signs of Deficiency
B12 deficiency is insidious — often developing slowly over years and manifesting through a bewildering diversity of symptoms across multiple organ systems. The triad of hematological, neurological, and psychiatric manifestations is characteristic, but symptoms may appear in any combination and at any severity.
Hematological Signs
- Fatigue, weakness, and pallor — often the earliest and most universal symptoms
- Shortness of breath on exertion
- Rapid or irregular heartbeat (palpitations, tachycardia)
- Dizziness and lightheadedness
- Easy bruising or bleeding (from thrombocytopenia in severe cases)
Neurological Signs
- Numbness and tingling in hands and feet (peripheral neuropathy)
- Burning sensations in the extremities
- Difficulty walking, unsteady gait, and balance problems
- Loss of proprioception (inability to sense body position)
- Muscle weakness and stiffness
- Tremor
- Visual disturbances (optic neuropathy)
- Bladder and bowel dysfunction (autonomic neuropathy)
Psychiatric and Cognitive Signs
- Depression and persistent low mood
- Anxiety, irritability, and emotional lability
- Brain fog, poor concentration, and memory impairment
- Confusion and disorientation
- Paranoia, delusions, and hallucinations (in severe cases)
- Personality changes
- Insomnia
Other Signs
- Glossitis — smooth, beefy-red, painful tongue
- Mouth ulcers and angular cheilitis
- Skin hyperpigmentation (knuckles, nail beds, oral mucosa)
- Premature hair graying
- Infertility and recurrent miscarriage
- Loss of appetite and weight loss
- Elevated homocysteine and elevated MMA on blood work
21. Special Therapeutic Applications
- Pernicious Anemia: Lifelong B12 replacement via IM injections (typically hydroxocobalamin 1,000 mcg every 1–3 months) or daily high-dose oral/sublingual methylcobalamin (1,000–5,000 mcg). Monitoring with serum B12, MMA, and CBC.
- Neurological Rehabilitation: Aggressive B12 repletion (daily IM injections for weeks, then weekly, then monthly) for individuals presenting with neurological symptoms. Early intervention is critical to maximize recovery potential.
- Depression (Adjunctive): Methylcobalamin 1,000–5,000 mcg daily in combination with L-methylfolate and P5P as part of a comprehensive methylation and mood-support protocol.
- Cognitive Decline Prevention: B12 + folate + B6 combination therapy for slowing brain atrophy and cognitive decline in at-risk elderly individuals with elevated homocysteine.
- Homocysteine Reduction: B12, folate, and B6 triad for cardiovascular and neurological protection, targeting homocysteine levels below 8 µmol/L.
- Fertility Optimization: B12 as part of preconception nutrient protocols for both women and men, supporting DNA synthesis, methylation, and healthy gametogenesis.
- Post-Bariatric Surgery: Lifelong B12 supplementation (sublingual, IM, or high-dose oral) with regular monitoring of serum B12 and MMA.
- Metformin Users: Routine B12 monitoring and supplementation (500–1,000 mcg daily) for all patients on metformin therapy.
- Vegan and Vegetarian Support: Non-negotiable daily B12 supplementation (methylcobalamin or hydroxocobalamin, 250–1,000 mcg daily) for all individuals following plant-based diets.
- Nitrous Oxide Recovery: Aggressive B12 repletion following N₂O exposure, particularly in individuals with pre-existing marginal B12 status. Hydroxocobalamin injections are preferred.
- Cyanide Poisoning: Hydroxocobalamin (Cyanokit®) is the FDA-approved antidote for cyanide poisoning, administered intravenously at 5–10 g doses.
Final Thoughts
Vitamin B12 is a nutrient of extraordinary biological importance and alarming epidemiological deficiency. It protects the very substance of the nervous system — the myelin that insulates every nerve in the brain and body. It powers the methylation cycle that governs gene expression, neurotransmitter production, and cardiovascular protection. It builds the red blood cells that carry life-giving oxygen to every tissue. And it does all of this while being subject to one of the most complex and vulnerable absorption processes of any nutrient, in a modern world of acid-suppressing medications, plant-based diets without adequate supplementation, autoimmune conditions, and aging digestive systems. The consequences of B12 deficiency are among the most severe of any nutritional insufficiency — ranging from crushing fatigue and depression to irreversible demyelination, psychosis, and dementia. Yet they are also among the most preventable and treatable, if caught in time. In natural medicine, B12 is not merely important — it is non-negotiable.
Guard the molecule that guards your nerves, your mind, and your blood — for when Vitamin B12 speaks, every cell in your body listens.
Connections
- Active B12 (Holotranscobalamin) Testing
- B12 Deficiency Diagnosis
- B12 and the Nervous System
- Anemia
- Homocysteine
- Vitamin B12 Test
- Methionine
- Dementia
- Depression
- Fatigue
- Alzheimer's Disease
- Peripheral Neuropathy
- Vitamin B9
- Vitamin B6
- Vitamin B2
- Iron
- Complete Blood Count
- SIBO
- Celiac Disease