Autoimmune Conditions Associated with Celiac Disease
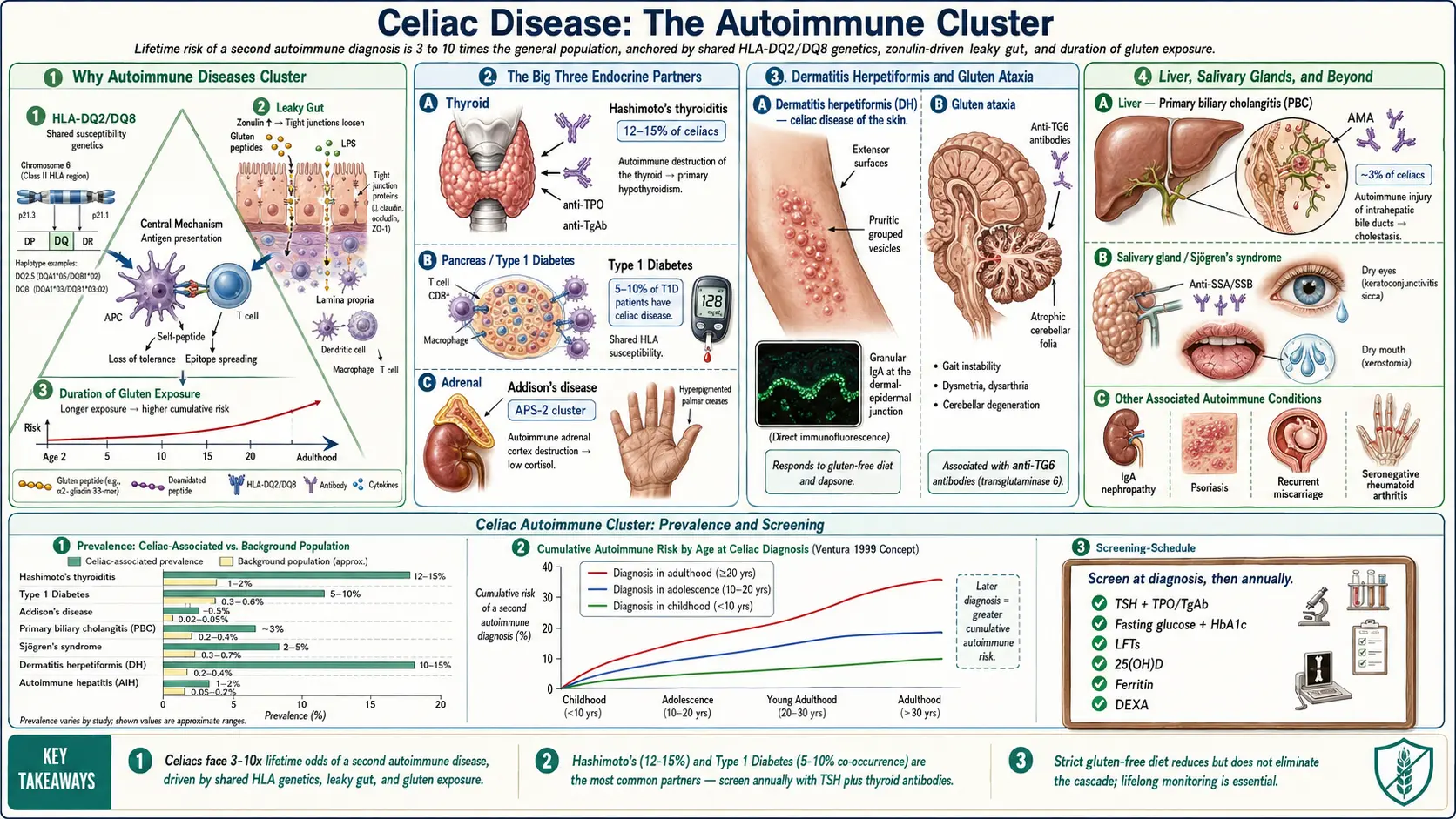
Table of Contents
- Why Autoimmune Diseases Cluster Around Celiac
- Hashimoto's Thyroiditis — The Most Common Partner
- Graves' Disease
- Type 1 Diabetes
- Addison's Disease and the APS-2 Cluster
- Sjögren's Syndrome
- Primary Biliary Cholangitis and Autoimmune Hepatitis
- Dermatitis Herpetiformis — Celiac of the Skin
- Inflammatory Bowel Disease and Autoimmune Enteropathy
- Lupus, Rheumatoid Arthritis, and Other Rheumatic Disease
- Neurological Autoimmunity — Gluten Ataxia, Neuropathy, Epilepsy
- IgA Nephropathy, Psoriasis, Cardiac, and Reproductive Issues
- Screening Recommendations — What to Check and When
- What to Do If You Develop a Second Autoimmune Disease
- Why Strict Gluten-Free Doesn't Always Stop the Cascade
- Key Research Papers
- Connections
- Featured Videos
Why Autoimmune Diseases Cluster Around Celiac
If you have celiac disease, your lifetime odds of developing a second autoimmune condition are three to ten times higher than the general population's. This is not bad luck. Celiac sits at a biological crossroads where three forces meet: a specific genetic background, a specific immune-activation pathway, and a leaky gut wall. All three are shared with a long list of other autoimmune diseases.
The genetic layer is HLA-DQ2 and HLA-DQ8. Roughly 95% of celiacs carry HLA-DQ2, and most of the rest carry HLA-DQ8. These same class-II HLA haplotypes — or their close neighbors on chromosome 6 — are overrepresented in type 1 diabetes, autoimmune thyroid disease, Addison's disease, Sjögren's syndrome, and several others. Your immune system is primed to present certain self-peptides to T cells in a way that tips toward autoreactivity. See the HLA-DQ2/DQ8 article for the genetics in detail.
The second layer is the leaky gut hypothesis, formalized by Alessio Fasano's work on zonulin. In celiacs, gluten fragments trigger release of zonulin, which loosens the tight junctions between intestinal epithelial cells. Partially digested food proteins, bacterial fragments (LPS), and other antigens leak across the gut wall into the bloodstream, where they encounter immune cells never meant to see them. Chronic low-grade translocation creates chronic systemic immune activation — a plausible background for distant-organ autoimmunity to ignite.
The third layer is time of exposure. The longer you eat gluten with untreated celiac disease, the higher your risk of a second autoimmune diagnosis climbs. Ventura and colleagues (1999) showed that children diagnosed before age 2 and placed on a gluten-free diet had autoimmune rates close to the background population, while those diagnosed as teenagers or adults had already accumulated extra risk that didn't fully reverse on the diet. The message is not to panic if you were diagnosed late; it is to take the diet seriously starting now.
Hashimoto's Thyroiditis — The Most Common Partner
Hashimoto's is the autoimmune condition you are statistically most likely to develop alongside celiac. Population prevalence of Hashimoto's is roughly 3–5%. Among celiacs, prevalence runs 12–15%, a three- to five-fold increase. The elevated risk is bidirectional: if you have Hashimoto's first, your odds of having undiagnosed celiac are roughly 2–5%, compared with about 1% in the general population.
The clinical link matters because symptoms overlap. Fatigue, brain fog, cold intolerance, constipation, weight changes, and hair thinning can all come from either untreated celiac malabsorption or hypothyroidism — or both at once. If you have celiac and still feel exhausted after a year on a strict gluten-free diet, the next lab to draw is a TSH plus thyroid antibodies.
Screening workup: TSH, free T4, anti-thyroid peroxidase (TPO) antibody, anti-thyroglobulin (TgAb) antibody. See the TPO and TgAb explainer for what each number means. A gluten-free diet modestly improves thyroid antibody levels in some patients, and some feel better combining it with an autoimmune-protocol elimination, but if your thyroid has been significantly damaged you will still need levothyroxine replacement. Diet doesn't regrow a gland.
Graves' Disease
Graves' disease is the hyperthyroid sibling of Hashimoto's — driven by TSH-receptor antibodies (TRAb or TSI) that stimulate the thyroid into overdrive. The celiac association is smaller than Hashimoto's but real: roughly a 2–4 fold increase in risk. Graves' presents with heat intolerance, weight loss despite normal eating, palpitations, tremor, anxiety, and in some cases thyroid eye disease (bulging, double vision, grittiness).
Any celiac who develops unexplained weight loss, racing heart, or heat intolerance should have a TSH checked promptly; a suppressed TSH with elevated free T4 sends you to an endocrinologist. Treatment is methimazole, radioactive iodine, or thyroidectomy — celiac diagnosis doesn't change the standard approach, but it does mean you should rule out concurrent Hashimoto's and monitor for the hashitoxicosis picture where disease swings between hyper and hypo phases.
Type 1 Diabetes
The celiac–type 1 diabetes (T1D) relationship is one of the tightest in autoimmune medicine. Roughly 5–10% of people with T1D also have celiac disease, compared with about 1% in the general population. The two conditions share HLA-DQ2/DQ8 haplotypes almost exactly; if you have T1D and carry DQ2 or DQ8, your celiac risk is particularly elevated.
In most cases, celiac is diagnosed after T1D — often picked up on routine screening rather than symptoms, because classical GI complaints may be blunted or attributed to diabetic gastroparesis. The International Society for Pediatric and Adolescent Diabetes (ISPAD) recommends screening all children with T1D for celiac at diagnosis and then annually (or at least every 2 years) for the first five years, plus any time symptoms suggest it. The American Diabetes Association makes a similar recommendation for pediatric T1D and for adults with suggestive symptoms.
Once both diagnoses are on the table, the gluten-free diet itself matters for glycemic control. Recovering intestinal villi absorb carbohydrates more efficiently, and insulin requirements often rise modestly after starting the diet — worth knowing so hypoglycemia doesn't sneak up on you. The diet does not reverse beta-cell loss, but it may slow the development of further autoimmune complications and helps prevent the nutrient deficiencies (iron, folate, B12, vitamin D) that compound an already-demanding condition.
Addison's Disease and the APS-2 Cluster
Addison's disease — autoimmune destruction of the adrenal cortex — is rare in the general population (about 1 in 10,000) but shows up in roughly 1–2% of celiacs, a dramatic relative increase. More importantly, celiac, Addison's, Hashimoto's, and type 1 diabetes tend to appear together in the same patient under the umbrella diagnosis of autoimmune polyendocrine syndrome type 2 (APS-2), also called Schmidt syndrome.
Addison's is easy to miss until it is an emergency. Classic symptoms are fatigue, darkening skin (hyperpigmentation of the palmar creases, gums, and scars), low blood pressure, salt craving, nausea, and unexplained weight loss. If you have celiac plus another autoimmune endocrine disease and you feel progressively wiped out with orthostatic symptoms, ask for a morning cortisol, ACTH level, and 21-hydroxylase antibody before an adrenal crisis announces itself. Treatment is lifelong hydrocortisone and fludrocortisone.
Sjögren's Syndrome
Sjögren's syndrome attacks exocrine glands — primarily the tear-producing lacrimal glands and the saliva-producing parotid and submandibular glands — producing dry eyes, dry mouth, joint pain, and fatigue. The celiac association is substantial: case-control studies show a 3- to 15-fold increased rate depending on the cohort. Conversely, roughly 5% of primary Sjögren's patients have celiac on serologic screening.
Both conditions are driven by class-II HLA-mediated lymphocyte infiltration, and both produce dense lymphocyte infiltrates in epithelial-lined tissues (small intestine for celiac, salivary ducts for Sjögren's). Workup in a celiac with persistent dry eyes, dry mouth, or unexplained parotid swelling: anti-SSA (Ro) and anti-SSB (La) antibodies, Schirmer test, and if suspicion is high, lip biopsy. Gluten-free diet does not treat Sjögren's itself; management is pilocarpine or cevimeline, aggressive use of preservative-free artificial tears, and monitoring for the small-but-real lymphoma risk.
Primary Biliary Cholangitis and Autoimmune Hepatitis
The liver is an underappreciated target in celiac. Two autoimmune liver diseases cluster with celiac to clinically important degrees.
Primary biliary cholangitis (PBC) — formerly primary biliary cirrhosis — is an autoimmune attack on the small intrahepatic bile ducts. Roughly 3% of celiacs have PBC, and remarkably about 6% of PBC patients have celiac on screening — one of the strongest reciprocal associations in the literature. Anyone with celiac and persistently elevated alkaline phosphatase should be checked for anti-mitochondrial antibody (AMA). Symptoms are often absent early; fatigue and itching are the classic first complaints. Treatment is ursodeoxycholic acid, with obeticholic acid as second line.
Autoimmune hepatitis (AIH) is less tightly linked than PBC but still over-represented — roughly 4–6% of celiacs show elevated transaminases from autoimmune inflammation rather than from hepatitis virus or fatty liver. Workup is ANA, anti-smooth muscle antibody (ASMA), anti-LKM-1, and IgG level; liver biopsy confirms. The practical rule: every celiac deserves a liver panel at diagnosis and annually thereafter. Mildly elevated AST and ALT in untreated celiac often normalize within 6–12 months of a strict gluten-free diet — if they don't, that's the signal to dig for autoimmune liver disease.
Dermatitis Herpetiformis — Celiac of the Skin
Dermatitis herpetiformis (DH) is not an associated condition in the usual sense — it is celiac disease, expressed primarily in the skin. Nearly every DH patient has the same small-intestinal villous changes as classical celiac, even if they have no GI symptoms. The rash is a clinical fingerprint once you've seen it: intensely itchy, grouped small vesicles and papules on the extensor elbows, knees, buttocks, lower back, and scalp, often scratched off before they can fully form into blisters. Patients describe it as the worst itch of their lives.
Diagnosis is by skin biopsy taken from normal-appearing skin adjacent to a lesion (not the lesion itself), examined with direct immunofluorescence. The diagnostic finding is granular IgA deposition at the dermal-epidermal junction, concentrated in the tips of the dermal papillae. Serology — tTG-IgA, DGP-IgA, and particularly epidermal transglutaminase (TG3) antibody — supports the diagnosis.
Treatment has two parts. The gluten-free diet is the real therapy — it treats both the skin and the underlying small-bowel disease, and over time it may allow the rash to remit entirely. But the diet takes months to bring symptoms under control, and the itch is unbearable. Dapsone is given for symptomatic relief, typically starting 25–50 mg daily and titrating to response; itch often resolves within 48–72 hours. Before starting dapsone, check G6PD status because dapsone causes hemolysis in G6PD-deficient patients. Most patients taper off dapsone within 1–2 years on a strict gluten-free diet.
Inflammatory Bowel Disease and Autoimmune Enteropathy
The overlap with Crohn's disease and ulcerative colitis is modest but real — roughly a 2- to 3-fold increased prevalence in celiacs compared with controls, and vice versa. The clinical headache is that symptom patterns overlap heavily (diarrhea, abdominal pain, weight loss, iron-deficiency anemia), and some patients have both conditions simultaneously. If your celiac serology has normalized and your diet has been strict for more than a year but symptoms persist, a repeat endoscopy with ileal biopsies and often a colonoscopy is appropriate.
Autoimmune enteropathy is rare but belongs on the differential for refractory celiac disease — villous atrophy that does not respond to a strict gluten-free diet. It is characterized by anti-enterocyte and anti-goblet-cell antibodies and requires immunosuppression rather than dietary management. See the refractory celiac article for the full workup.
Lupus, Rheumatoid Arthritis, and Other Rheumatic Disease
Systemic lupus erythematosus (SLE) shows a modest association with celiac — roughly a 2- to 3-fold increase in bidirectional screening studies, though the numbers are less robust than for thyroid or T1D. Rheumatoid arthritis (especially seronegative RA and juvenile idiopathic arthritis) overlaps with celiac at similar rates; seropositive RF-positive RA is less clearly linked.
The practical lesson for patients: unexplained joint pain, morning stiffness longer than 30 minutes, malar rash, photosensitive rash, or unexplained anemia/thrombocytopenia in a known celiac deserves a rheumatology referral and an autoimmune panel (ANA, anti-dsDNA, anti-Smith, RF, anti-CCP, complement levels). The gluten-free diet does not treat lupus or RA directly, though some patients report joint-pain improvement — probably from reducing background inflammation rather than modifying the underlying disease.
Weaker associations also exist with multiple sclerosis, myasthenia gravis, and autoimmune polymyositis/dermatomyositis. These are not strong enough to justify routine screening of celiacs, but any celiac with new unexplained neurologic weakness, muscle pain with elevated CK, or symptom patterns suggestive of MS deserves appropriate specialty workup.
Neurological Autoimmunity — Gluten Ataxia, Neuropathy, Epilepsy
Celiac is not only a gut disease. A subset of patients have neurological presentations as their dominant or even sole complaint.
Gluten ataxia is immune-mediated cerebellar injury — clumsiness, unsteady walking, slurred speech, and characteristic eye-movement abnormalities. MRI may show cerebellar atrophy. The antibody marker is anti-transglutaminase 6 (TG6), a cerebellum-enriched variant of the same enzyme family targeted in classical celiac (TG2). Strict gluten-free diet halts progression in most cases and can reverse some damage if started early; late intervention often leaves residual deficits. Not every ataxia patient with positive tTG has gluten ataxia, but in cryptogenic cerebellar ataxia, celiac serology belongs in the workup.
Peripheral neuropathy in celiac takes several forms — distal symmetric sensory ("burning feet"), small-fiber neuropathy, and less commonly mononeuritis multiplex. Some cases respond to the gluten-free diet alone. Work up B12, folate, copper, and vitamin E first; celiac malabsorption produces deficiencies that cause neuropathy through a different mechanism than direct autoimmunity.
Epilepsy with bilateral occipital calcifications — the Gobbi syndrome — is a classical but rare celiac-associated picture: seizures (often visual), calcium deposits in the occipital lobes on CT, and celiac villous atrophy. Early gluten-free diet can reduce seizure burden dramatically.
"Brain fog" — the vague cognitive slowing, word-finding difficulty, and fatigue many celiacs describe — is underdiagnosed but real, with small functional-MRI studies showing measurable attention and processing-speed changes that improve on a strict diet. If you feel sharper within weeks of tightening gluten control, you're not imagining it.
IgA Nephropathy, Psoriasis, Cardiac, and Reproductive Issues
IgA nephropathy — the most common primary glomerulonephritis worldwide — shows elevated prevalence in celiacs, probably because both diseases involve abnormal mucosal IgA responses. Microscopic hematuria or persistent proteinuria on a celiac's routine urinalysis deserves a nephrology referral.
Psoriasis has a modest but reproducible association with celiac; small trials suggest gluten-free diet can help psoriasis severity in patients who are tTG-positive at baseline, though not in tTG-negative psoriatics.
Cardiac autoimmune associations are rare but worth knowing. Idiopathic dilated cardiomyopathy has been reported at elevated rates in celiac cohorts, and case series describe partial cardiac recovery after strict gluten-free diet. Autoimmune pericarditis is a rare but documented association.
Reproductive issues are not strictly autoimmune but deserve mention because they are common and under-recognized. Unexplained infertility, recurrent miscarriage, delayed menarche, early menopause, and low-birthweight infants all occur at elevated rates in untreated celiac. The mechanisms are mixed: micronutrient malabsorption (iron, folate, zinc, selenium), chronic inflammation affecting implantation, and anti-tTG antibodies crossing the placenta. Any woman with unexplained recurrent pregnancy loss deserves a celiac workup, and many of these problems improve substantially on a strict gluten-free diet.
Screening Recommendations — What to Check and When
Here is a practical, evidence-based monitoring schedule for the celiac patient who wants to catch a second autoimmune disease early.
At diagnosis:
- CBC, ferritin, iron studies — iron deficiency is the most common deficiency and often the reason for the original referral.
- Comprehensive metabolic panel including LFTs — baseline AST, ALT, alkaline phosphatase. Mild elevation is common and often resolves on diet; persistent elevation needs liver autoantibody workup.
- TSH, free T4, anti-TPO, anti-TgAb — baseline thyroid status and antibodies.
- 25-hydroxy vitamin D, B12, folate, copper, zinc, magnesium — baseline micronutrients.
- Fasting glucose and HbA1c — establish baseline and screen for T1D (consider GAD-65 and islet-cell antibodies if family history).
- DEXA scan — baseline bone density; osteoporosis and osteopenia are substantially more common in untreated celiac (see the deficiencies and bone health article).
- Total IgA — if selective IgA deficiency is present, IgA-based antibody testing for second conditions will be falsely negative.
Within the first year after diagnosis:
- Repeat tTG to confirm response to the diet.
- Thyroid panel within 12 months — antibodies may have been negative at diagnosis but positive a year later.
- Clinical review for symptoms of other autoimmune disease.
Ongoing (every 1–2 years unless symptoms dictate sooner):
- TSH and thyroid antibodies every 1–2 years — the highest-yield second-disease screen.
- CBC, ferritin, LFTs, 25(OH)D annually.
- DEXA every 2 years if the baseline was abnormal; every 2–5 years if normal and post-menopausal or at other risk factors.
- Fasting glucose and HbA1c annually, especially with family history of T1D.
- Annual tTG to verify dietary adherence (rising tTG often signals hidden gluten exposure before symptoms return).
In type 1 diabetes, the reverse screen applies: annual tTG-IgA for the first five years after T1D diagnosis, per ISPAD, and any time suggestive symptoms appear.
What to Do If You Develop a Second Autoimmune Disease
First: it is not your fault and it does not mean you are failing at the gluten-free diet. Most second autoimmune diagnoses in celiacs occur despite excellent dietary adherence. The immune dysregulation that led to celiac in the first place does not disappear just because gluten is removed.
Second: each condition needs its own management. The gluten-free diet treats celiac and sometimes moderates the severity of others (dermatitis herpetiformis, gluten ataxia, early thyroid antibody activity), but it will not by itself treat Hashimoto's that has already destroyed the gland, T1D that has destroyed beta cells, Addison's, PBC, or Sjögren's. Expect to add disease-specific medication — levothyroxine, insulin, hydrocortisone, ursodeoxycholic acid, dapsone, pilocarpine, immunosuppressants as appropriate. None of these conflict with a strict gluten-free diet, but double-check that medications are certified gluten-free (most are, but a handful of older generics use wheat-derived excipients).
Third: coordinate your specialists. A celiac with Hashimoto's, T1D, and Addison's needs a gastroenterologist, endocrinologist, and primary care physician who talk to each other. Keep a one-page summary of your diagnoses, medications, and recent lab values and hand it to every new specialist. Polypharmacy in autoimmune poly-syndromes is where preventable errors happen.
Why Strict Gluten-Free Doesn't Always Stop the Cascade
Patients often ask, fairly, why they should bother being strict if the diet doesn't prevent a second autoimmune disease. Two answers.
First, it does reduce the cascade — just not eliminate it. Ventura's pediatric cohort study and later adult analyses both show that strict adherence reduces the probability of picking up a second autoimmune diagnosis compared with partial adherence or ongoing gluten exposure. The absolute numbers vary, but the direction is consistent.
Second, once the immune system has "tipped" — once it has generated the clonal T and B cells that will eventually produce thyroid, islet-cell, or adrenal antibodies — those cells can persist and expand even without the original gluten stimulus. The breach has happened; the army has been trained. Removing the training stimulus may slow the expansion but does not necessarily unmake it. This is why early diagnosis and early diet are so much more effective than late ones. It is also why anyone with celiac should treat monitoring for second conditions as a routine part of care, not an alarmist add-on.
None of this is a reason to eat gluten. Every crumb feeds the process. But it is a reason to have realistic expectations about what the diet can and cannot do, and to partner with a medical team that takes the whole autoimmune picture seriously rather than treating celiac as a purely dietary problem.
Key Research Papers
Peer-reviewed studies on celiac disease genetics, the autoimmune cluster, screening intervals, and how duration of gluten exposure shapes the lifetime risk of a second autoimmune diagnosis.
- Rubio-Tapia A, Hill ID, Kelly CP, Calderwood AH, Murray JA. ACG Clinical Guidelines: Diagnosis and Management of Celiac Disease. American Journal of Gastroenterology. 2013;108(5):656-676.
- Husby S, Koletzko S, Korponay-Szabó IR, et al. ESPGHAN Guidelines for the Diagnosis of Coeliac Disease (2020 update). Journal of Pediatric Gastroenterology and Nutrition. 2020;70(1):141-156.
- Lebwohl B, Sanders DS, Green PHR. Coeliac Disease. Lancet. 2018;391(10115):70-81.
- Singh P, Arora A, Strand TA, et al. Global Prevalence of Celiac Disease: Systematic Review and Meta-Analysis. Clinical Gastroenterology and Hepatology. 2018;16(6):823-836.e2.
- Lundin KEA, Wijmenga C. Coeliac Disease and Autoimmune Disease — Genetic Overlap and Screening. Nature Reviews Gastroenterology & Hepatology. 2015;12(9):507-515.
- Cosnes J, Cellier C, Viola S, et al. Incidence of Autoimmune Diseases in Celiac Disease: Protective Effect of the Gluten-Free Diet. Clinical Gastroenterology and Hepatology. 2008;6(7):753-758.
- Ventura A, Magazzù G, Greco L; SIGEP Study Group for Autoimmune Disorders in Celiac Disease. Duration of Exposure to Gluten and Risk for Autoimmune Disorders in Patients with Celiac Disease. Gastroenterology. 1999;117(2):297-303.
- Caio G, Volta U, Sapone A, et al. Celiac Disease: A Comprehensive Current Review. BMC Medicine. 2019;17(1):142.
- Reilly NR, Aguilar K, Hassid BG, et al. Celiac Disease in Normal-Weight and Overweight Children: Clinical Features and Growth Outcomes Following a Gluten-Free Diet. Journal of Pediatric Gastroenterology and Nutrition. 2011;53(5):528-531.
- Hadjivassiliou M, Sanders DS, Grünewald RA, Woodroofe N, Boscolo S, Aeschlimann D. Gluten Sensitivity: From Gut to Brain. Lancet Neurology. 2010;9(3):318-330.
- Bonamico M, Pasquino AM, Mariani P, et al. Prevalence and Clinical Picture of Celiac Disease in Turner Syndrome. Journal of Clinical Endocrinology & Metabolism. 2002;87(12):5495-5498.
- Bibbins-Domingo K, Grossman DC, Curry SJ, et al. Screening for Celiac Disease: US Preventive Services Task Force Recommendation Statement. JAMA. 2017;317(12):1252-1257.
- Reunala T, Salmi TT, Hervonen K, Kaukinen K, Collin P. Dermatitis Herpetiformis: A Common Extraintestinal Manifestation of Coeliac Disease. Nutrients. 2018;10(5):602.
Live PubMed Searches
Live PubMed queries that update as new papers are indexed.
- Celiac disease and autoimmune thyroid disease
- Celiac disease and type 1 diabetes screening
- Celiac disease and autoimmune polyendocrine syndrome
- Celiac disease and Sjögren's syndrome
- Celiac disease and primary biliary cholangitis
- Dermatitis herpetiformis and celiac disease
- Gluten ataxia and transglutaminase 6
- Celiac disease and IgA nephropathy
- Celiac disease, infertility, and recurrent pregnancy loss
Connections
- Celiac Disease Overview
- Gluten-Free Diet: A Practical Guide
- HLA-DQ2 and HLA-DQ8 Genetic Testing
- Nutritional Deficiencies and Bone Health
- Refractory Celiac Disease and EATL
- Celiac Serology Testing: tTG, DGP, EMA
- Non-Celiac Gluten Sensitivity
- Diabetes
- Lupus
- AIP and Gluten Elimination
- TPO and TgAb Antibodies Explained
- Sjogren's Syndrome
- Graves' Disease
- Hashimoto's Thyroiditis
- Irritable Bowel Syndrome
- Arthritis
- Vitiligo
- Dermatitis Herpetiformis