Nutritional Deficiencies and Bone Health in Celiac Disease
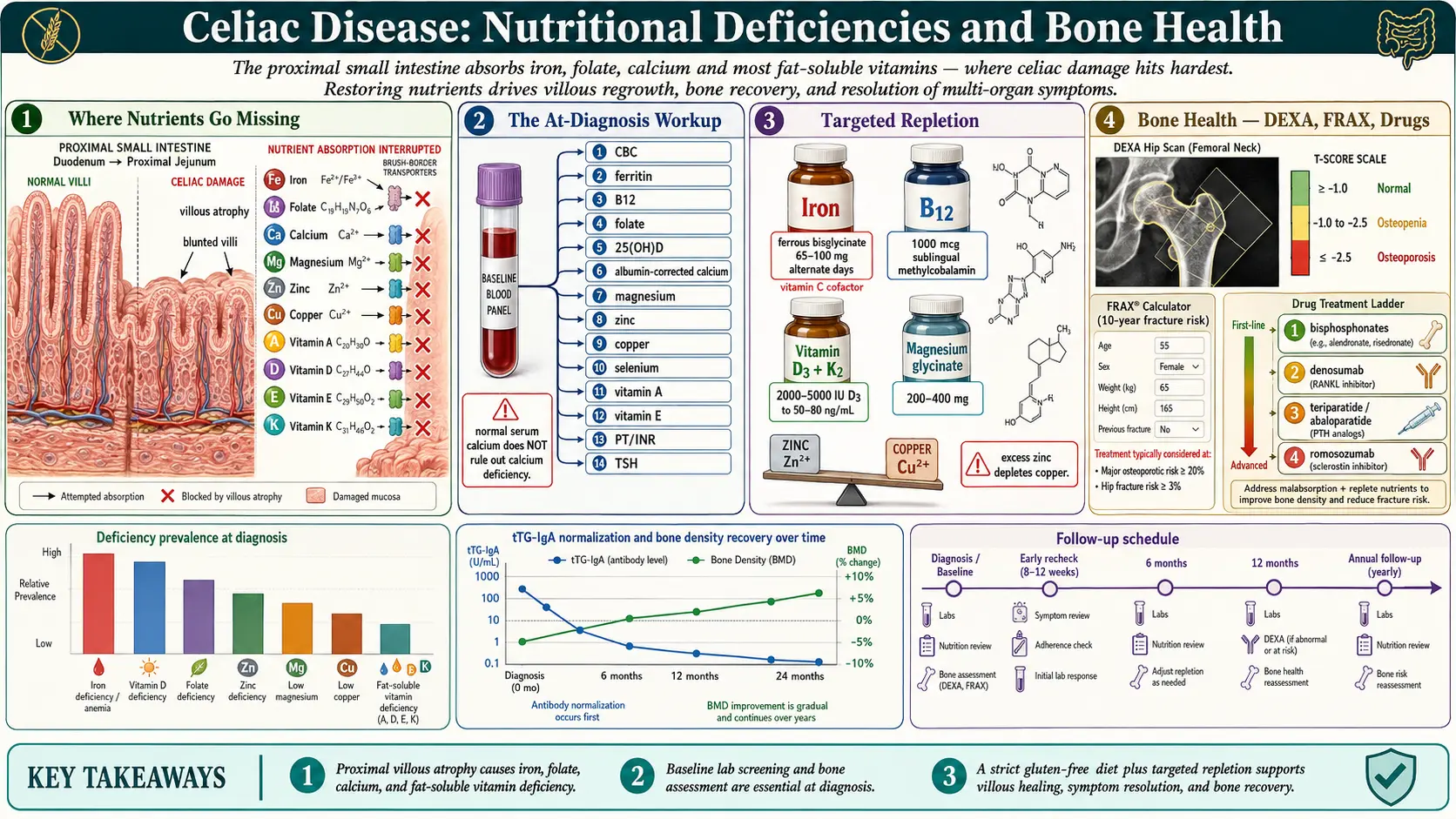
Table of Contents
- Why Nutrients Go Missing in Celiac
- The At-Diagnosis Nutritional Workup
- Iron — The Signature Celiac Deficiency
- Vitamin B12 and Folate
- Vitamin D and Calcium
- Magnesium
- Zinc and Copper — The Balancing Act
- Fat-Soluble Vitamins: A, E, K
- Protein, Albumin, and Selenium
- Bone Health: DEXA, Osteopenia, Osteoporosis
- When Bone Drugs Are Warranted
- Lactose Intolerance, Dermatitis Herpetiformis, and Pediatric Growth
- The Recheck Schedule — 3, 6, 12 Months and Beyond
- Realistic Healing Timeline
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
Why Nutrients Go Missing in Celiac
Celiac disease damages the proximal small intestine — the duodenum and upper jejunum — exactly where your body absorbs iron, folate, calcium, and most fat-soluble vitamins. The flattened villi and inflamed epithelium cannot grip and transport nutrients across the gut wall, so a person eating a seemingly adequate diet can still run low on everything. The longer undiagnosed celiac has been active, the deeper and more numerous the deficiencies tend to be.
This is not a minor footnote to your diagnosis. Nutrients drive the repair of the villi themselves, the rebuilding of bone that has been quietly thinning for years, the regeneration of red blood cells, and the normal function of every enzyme your body runs. Getting the numbers right matters as much as the gluten-free diet itself, and the first year after diagnosis is the window where it all gets sorted out.
The good news: most deficiencies correct on a strict gluten-free diet within 6 to 12 months as the villi regrow, with judicious supplementation to speed the process. Bone density takes longer — typically two years or more — but it does recover in the majority of adult patients. The work is front-loaded: one thorough workup at diagnosis, careful repletion, and two or three rechecks in the first year.
The At-Diagnosis Nutritional Workup
Ask your physician to order the following at the time of your celiac diagnosis, before you begin any supplementation. Getting a clean baseline tells you what to fix and, equally important, what not to over-supplement.
- Complete blood count (CBC) with indices. Look at MCV (mean corpuscular volume). Microcytic anemia (MCV under 80) points to iron deficiency; macrocytic anemia (MCV over 100) points to B12 or folate deficiency. A mixed picture is common in celiac because both patterns coexist — the MCV may look "normal" while hiding both.
- Ferritin, serum iron, TIBC, transferrin saturation. Ferritin is the best single marker of body iron stores and is almost always the first abnormality in celiac — iron is absorbed in the duodenum, ground zero for celiac damage.
- Vitamin B12 (serum) and ideally methylmalonic acid if B12 is low-normal.
- Folate — RBC folate is more reliable than serum folate, which fluctuates with recent meals.
- 25-hydroxyvitamin D (25(OH)D). Target 50–80 ng/mL for celiac recovery; most newly diagnosed patients sit well below 30.
- Serum calcium with albumin correction. Important caveat: blood calcium often looks normal even when total-body calcium is severely depleted, because the body will rob bone to keep serum levels in range. A normal serum calcium does not rule out calcium deficiency.
- Magnesium — RBC magnesium preferred over serum magnesium, which misses intracellular depletion.
- Zinc (plasma or serum).
- Copper (serum) and ceruloplasmin. Copper can be depleted either by celiac itself or iatrogenically later if zinc supplementation is pushed too high.
- Selenium.
- Vitamin A (retinol) and vitamin E (alpha-tocopherol).
- Vitamin K — measured functionally via prothrombin time (PT/INR). A prolonged PT in an unanticoagulated patient points to vitamin K deficiency.
- Albumin and total protein — markers of chronic malabsorption and protein-losing enteropathy.
- TSH — autoimmune thyroid disease is the single most common overlap with celiac.
This panel sounds long, but it is one blood draw and almost every line item is covered by insurance when the indication is a new celiac diagnosis. Have a copy of every result sent to you directly; you will want it when you are tracking recovery.
Iron — The Signature Celiac Deficiency
Iron deficiency is so typical of undiagnosed celiac that any unexplained iron-deficiency anemia in an adult is a reason to screen for celiac disease regardless of GI symptoms. Duodenal villi are the main site of iron absorption; when they are blunted, iron never enters the body, no matter how much is in the diet.
Oral repletion. For mild to moderate deficiency, ferrous bisglycinate 65–100 mg elemental iron is well tolerated and absorbs better than ferrous sulfate for many patients. Critically, the dosing interval matters more than the dose. Stoffel and colleagues (2017) showed that alternate-day dosing — taking iron every other day — actually produces more total absorption than daily dosing, because daily iron raises hepcidin and shuts down intestinal iron uptake for the next 24 hours. Take it on an empty stomach with vitamin C (250–500 mg) to triple absorption, and do not take it within two hours of coffee, tea, calcium, or dairy.
When IV is warranted. Oral iron fails roughly 30% of the time, either because the gut cannot tolerate it (nausea, constipation, cramping) or because absorption is still too damaged. The threshold for switching to IV is reasonable: ferritin below 20 ng/mL with oral-iron intolerance or failure after 8–12 weeks. Two modern formulations dominate the U.S. market: ferric carboxymaltose (Injectafer), typically given as two 750 mg doses a week apart, and ferric derisomaltose (Monoferric), which can deliver a full 1000 mg in a single 20-minute infusion. Both carry a small but real risk of transient hypophosphatemia, and Injectafer is more likely to cause it; Monoferric is preferable if you can get insurance coverage.
Do not ignore persistent iron deficiency after a year of strict gluten-free eating. It is one of the first clues that gluten exposure is ongoing or that refractory celiac disease is developing — see the refractory celiac article.
Vitamin B12 and Folate
These two B vitamins both cause macrocytic anemia, and celiac can drop either one. Folate is absorbed in the proximal small intestine (where celiac damage is worst), so folate deficiency is common. B12 is absorbed in the terminal ileum, which is usually spared, but concurrent autoimmune gastritis, H. pylori, or pernicious anemia (all over-represented in celiac patients) frequently takes out B12 anyway.
Folate repletion. 1 mg of folic acid (or methylfolate for patients with MTHFR variants who feel better on the methylated form) daily for 1–3 months, then drop to a maintenance multivitamin dose.
B12 repletion. For mild deficiency, 1000 mcg sublingual methylcobalamin daily works for most people. For severe deficiency, neurological symptoms, or documented pernicious anemia, start with 1000 mcg intramuscular cyanocobalamin weekly for 4–8 weeks, then monthly for maintenance. Do not wait on neurological symptoms — peripheral neuropathy and cognitive changes from B12 deficiency can become permanent if repletion is delayed more than a year.
Vitamin D and Calcium
Roughly two-thirds of newly diagnosed celiac adults are vitamin D deficient, and bone loss follows directly. Correcting vitamin D is the single most important intervention for long-term bone health in celiac disease.
Vitamin D dosing. Start with vitamin D3 2000–5000 IU daily, titrated to a 25(OH)D blood level of 50–80 ng/mL. If the starting level is under 20 ng/mL, a loading dose of 50,000 IU weekly for 8 weeks followed by daily maintenance is a reasonable alternative. Pair it with vitamin K2 (MK-7) 100–200 mcg daily to direct the extra calcium into bone rather than arteries.
Calcium. Adults need roughly 1000–1200 mg of elemental calcium per day from all sources combined (food plus supplement). Food sources are always preferred — dairy if tolerated, fortified plant milks, sardines with bones, kale, almonds. If supplementing, calcium citrate 500–600 mg elemental once or twice daily absorbs better than carbonate, especially in the low-acid stomach environment many celiac patients have. Do not co-dose calcium with iron — they compete for absorption.
Remember the caveat from the workup: serum calcium is a terrible screening test because the body holds it constant at the expense of your bones. Look at 25(OH)D, urinary calcium, and parathyroid hormone (PTH) for a truer picture. An elevated PTH with a "normal" serum calcium is a classic sign of calcium and vitamin D insufficiency.
Magnesium
Magnesium deficiency is common in celiac and chronically under-diagnosed because standard serum magnesium is insensitive — only about 1% of body magnesium lives in the blood. RBC magnesium is the better test. Symptoms of deficiency are vague but suggestive: muscle cramps (especially at night or in the calves), eyelid twitches, migraines, anxiety, poor sleep, constipation, and palpitations.
Repletion. Magnesium glycinate 200–400 mg elemental daily, taken in the evening. Glycinate is gentle on the gut and well absorbed. Magnesium citrate works too but tends to loosen stools, which may or may not be desirable depending on your GI state. Avoid magnesium oxide — it is cheap but poorly absorbed. Caution in kidney disease (reduced magnesium clearance).
Zinc and Copper — The Balancing Act
Zinc is directly required for villous regeneration, wound healing, immune function, and taste. Deficiency is common in active celiac and can actually slow gut healing. Zinc picolinate 15–30 mg elemental daily for 2–3 months is a reasonable repletion dose.
Here is the trap: zinc and copper compete for absorption in the gut. Supplementing zinc at doses above 30–40 mg daily for more than a few months reliably induces copper deficiency, which then causes its own anemia and neurological problems. If you are taking 30 mg or more of zinc beyond the first few months, add copper 1–2 mg daily on a separate schedule. Better still: check a serum copper and ceruloplasmin before and after zinc repletion, and stop supplementation once levels normalize.
Copper can also be depleted by celiac disease itself (again, duodenal absorption), so do not assume copper is fine just because you have not been supplementing zinc. Test it.
Fat-Soluble Vitamins: A, E, K
All three fat-soluble vitamins depend on intact fat absorption, which celiac disrupts. Deficiencies are less often symptomatic than iron or B12, but they show up on lab panels and matter for long-term health.
- Vitamin A. Low retinol slows villous regeneration and impairs dim-light vision. Replete with the diet first (eggs, liver, dairy) and a modest supplement (2500–5000 IU daily) if labs remain low. Do not mega-dose — vitamin A is stored in the liver and can become toxic above 10,000 IU/day sustained.
- Vitamin E. Low alpha-tocopherol can cause peripheral neuropathy and ataxia in severe cases. Mixed-tocopherol supplements (200–400 IU daily) correct mild deficiency.
- Vitamin K. Functional deficiency shows up as a prolonged PT/INR. Green leafy vegetables plus K2 (MK-7) cover most cases; persistent deficiency suggests ongoing fat malabsorption and warrants a gastroenterology workup.
Protein, Albumin, and Selenium
Chronic malabsorption pulls down albumin and total protein. An albumin below 3.5 g/dL in newly diagnosed celiac is not rare and usually corrects within 6 months of a strict gluten-free diet plus adequate protein intake. Aim for 1.0–1.2 g of protein per kilogram of body weight per day during recovery — roughly 70–100 g/day for most adults. This is higher than standard recommendations because tissue repair is running at an accelerated rate.
Selenium deficiency is under-recognized in celiac and contributes to thyroid autoimmunity. Two Brazil nuts a day (or a 100–200 mcg supplement for 2–3 months) handles most cases.
Bone Health: DEXA, Osteopenia, Osteoporosis
The 2023 American College of Gastroenterology (ACG) guidelines recommend a DEXA scan at the time of diagnosis for every adult with celiac disease. Bone loss is one of the most common silent consequences of untreated celiac, driven by years of calcium malabsorption, vitamin D deficiency, and low-grade inflammation.
A DEXA scan reports two numbers:
- T-score compares your bone density to a healthy young adult of the same sex. Used for adults over 50 or postmenopausal women.
- Z-score compares your bone density to someone your own age and sex. Used for adults under 50, premenopausal women, and children.
T-score cutoffs:
- Normal: T-score above −1.0
- Osteopenia (low bone mass): T-score between −1.0 and −2.5
- Osteoporosis: T-score at or below −2.5
- Severe osteoporosis: T-score at or below −2.5 plus one or more fragility fractures
The FRAX calculator (an online tool from the University of Sheffield) uses T-score, age, sex, weight, height, prior fracture history, parental hip fracture, smoking, alcohol, glucocorticoid use, and rheumatoid arthritis to estimate your 10-year probability of a major osteoporotic fracture. It is the standard way of deciding whether a borderline DEXA result warrants treatment.
The recovery curve. On a strict gluten-free diet with adequate calcium and vitamin D, bone mineral density improves measurably within the first year and continues to improve for 2–5 years. Nazareth and colleagues (2015) and earlier work by Larussa and colleagues (2012) both documented significant BMD gains by year two. Repeat DEXA at 2 years is the standard follow-up in adult celiac patients who were osteopenic or osteoporotic at diagnosis.
If bone density fails to improve after 2 years on a strict diet, the investigation needs to widen: persistent gluten exposure, refractory celiac, coexisting hyperparathyroidism, vitamin D malabsorption, or an endocrine cause unrelated to celiac.
When Bone Drugs Are Warranted
Most adults with celiac-associated osteopenia do not need bone drugs. The gluten-free diet plus vitamin D, calcium, protein, and weight-bearing exercise rebuilds bone on its own. Pharmacologic therapy enters the picture in a narrower set of situations:
- T-score at or below −2.5 (osteoporosis by density criteria)
- History of a fragility fracture (a fracture from a fall from standing height or less) regardless of T-score
- FRAX 10-year risk of major osteoporotic fracture above 20%, or hip fracture above 3%
The drug options, in rough order of use:
- Bisphosphonates — alendronate (Fosamax) or risedronate (Actonel). Oral weekly dosing; first-line for most patients. Must be taken on an empty stomach with a full glass of water, sitting upright for 30 minutes, to protect the esophagus. IV zoledronic acid (Reclast) once a year is an alternative for patients who cannot tolerate oral dosing.
- Denosumab (Prolia). A subcutaneous injection every 6 months. Good for patients with kidney impairment who cannot take bisphosphonates, but has a rebound bone-loss problem if stopped abruptly — treatment is effectively lifelong or must be bridged with a bisphosphonate on discontinuation.
- Teriparatide (Forteo) or abaloparatide (Tymlos). Daily subcutaneous injections for up to 2 years. Anabolic — they build new bone rather than just preventing loss. Reserved for severe osteoporosis or patients who have already fractured on antiresorptive therapy.
- Romosozumab (Evenity). Monthly subcutaneous injection for 12 months; both builds bone and reduces resorption. Severe-osteoporosis use only, with cardiovascular cautions.
Before starting any bone drug, make sure vitamin D is replete (at least 30 ng/mL, preferably 50+) and calcium intake is adequate. Bone drugs do not work well in a vitamin-D-deficient patient.
Lactose Intolerance, Dermatitis Herpetiformis, and Pediatric Growth
Temporary lactose intolerance. The enzyme that digests milk sugar — lactase — lives on the tips of intestinal villi, which are exactly what celiac destroys. Many newly diagnosed patients are secondarily lactose intolerant and feel worse when they eat dairy. This is usually temporary, resolving within 6–12 months as the villi regrow. A short dairy-free trial at diagnosis often dramatically improves symptoms; reintroduction after 6 months is worth testing before concluding you are permanently lactose intolerant.
Dermatitis herpetiformis (DH). DH is celiac disease expressed in the skin — an intensely itchy, blistering rash on the elbows, knees, buttocks, and scalp, driven by the same gluten-triggered IgA immunology. Patients with DH almost always have some degree of small-intestinal villous damage whether or not they have GI symptoms, and the nutritional workup above applies identically. The rash responds to strict gluten-free eating, though it can take months, and oral dapsone speeds relief while the diet works.
Pediatric growth failure. Short stature and delayed puberty are classic presenting signs of celiac disease in children. The encouraging news is that catch-up growth on a gluten-free diet is the rule, not the exception, when diagnosis happens before puberty closes the growth plates. Height often gains 5–10 cm over baseline projections in the first two years, and most children reach their genetic height potential. Screen any child with unexplained short stature for celiac, early.
The Recheck Schedule — 3, 6, 12 Months and Beyond
Here is a reasonable follow-up lab rhythm based on the Rubio-Tapia 2013 ACG guidelines and current practice:
- At 3 months: CBC, ferritin, 25(OH)D, tTG-IgA, basic metabolic panel. Check symptom response.
- At 6 months: Repeat the deficient items from baseline; recheck tTG-IgA (should be trending down). Evaluate adherence.
- At 12 months: Full recheck of every baseline abnormality; tTG-IgA should be normal or near-normal if strict adherence. Consider repeat upper endoscopy at 12–24 months in adults to confirm mucosal healing, particularly in those whose symptoms or antibodies have not normalized.
- At 24 months: Repeat DEXA if the baseline scan showed osteopenia or osteoporosis.
- Annually thereafter: tTG-IgA, CBC, ferritin, 25(OH)D, TSH (thyroid overlap is common), and comprehensive metabolic panel.
Realistic Healing Timeline
Expect the following, roughly:
- Weeks 1–4: Most GI symptoms begin to settle. Energy starts rising as inflammation falls.
- Months 1–3: Folate, magnesium, and zinc typically normalize quickly with repletion. Antibodies begin dropping.
- Months 3–6: Ferritin, B12, and vitamin D climb into normal range with consistent supplementation. Lactose tolerance often returns.
- Months 6–12: Villous architecture on biopsy is substantially or fully recovered in most adults. Antibodies usually negative by 12 months.
- Years 1–3: Bone mineral density climbs measurably. Weight and muscle mass return to normal. Energy and cognition continue to improve.
- Years 2–5: Peak bone recovery. Many osteopenic patients return to normal DEXA ranges without drugs.
If you are not on this curve — especially if ferritin stays low, antibodies stay high, or symptoms persist after 12 months of what you believe is strict gluten-free eating — the next step is a hard look at hidden gluten exposure (see the gluten-free diet practical guide), and if exposure is truly excluded, evaluation for refractory celiac disease (see the refractory celiac article).
Key Research Papers
- Rubio-Tapia A, Hill ID, Kelly CP, Calderwood AH, Murray JA. ACG Clinical Guidelines: Diagnosis and management of celiac disease. Am J Gastroenterol. 2013.
- Stoffel NU, et al. Iron absorption from oral iron supplements given on consecutive versus alternate days and as single morning doses versus twice-daily split dosing in iron-depleted women. Haematologica. 2019/2017 (alternate-day dosing).
- Larussa T, et al. Bone mineralization in celiac disease. Gastroenterol Res Pract. 2012.
- Nazareth S, et al. Bone health recovery on a gluten-free diet in adult celiac disease. J Clin Gastroenterol. 2015.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on nutrient deficiencies and bone health in celiac disease:
- Celiac disease and iron deficiency anemia
- Celiac disease and vitamin D deficiency
- Celiac disease, bone mineral density, and DEXA
- Celiac disease, B12 and folate deficiency
- Celiac disease, zinc, and copper
- Celiac disease, osteoporosis, and bisphosphonates
- Alternate-day oral iron dosing and hepcidin
- Celiac disease, gluten-free diet, and mucosal healing
- Dermatitis herpetiformis and celiac disease
- Pediatric celiac disease and catch-up growth
Connections
- Celiac Disease Overview
- Gluten-Free Diet: A Practical Guide
- Dermatitis Herpetiformis
- Endoscopy, Biopsy, and Marsh Classification
- Osteoporosis
- Calcium
- Vitamin D3
- Iron
- Vitamin B12
- Magnesium
- Anemia
- Homocysteine Lab Test
- IBD Fatigue, Anemia, and Nutrient Deficiencies
- Associated Autoimmune Conditions
- Genetic Testing HLA DQ2 and DQ8
- Celiac Serology Testing tTG DGP EMA
- Non Celiac Gluten Sensitivity
- Refractory Celiac and EATL