HLA-DQ2 and HLA-DQ8 Genetic Testing for Celiac Disease
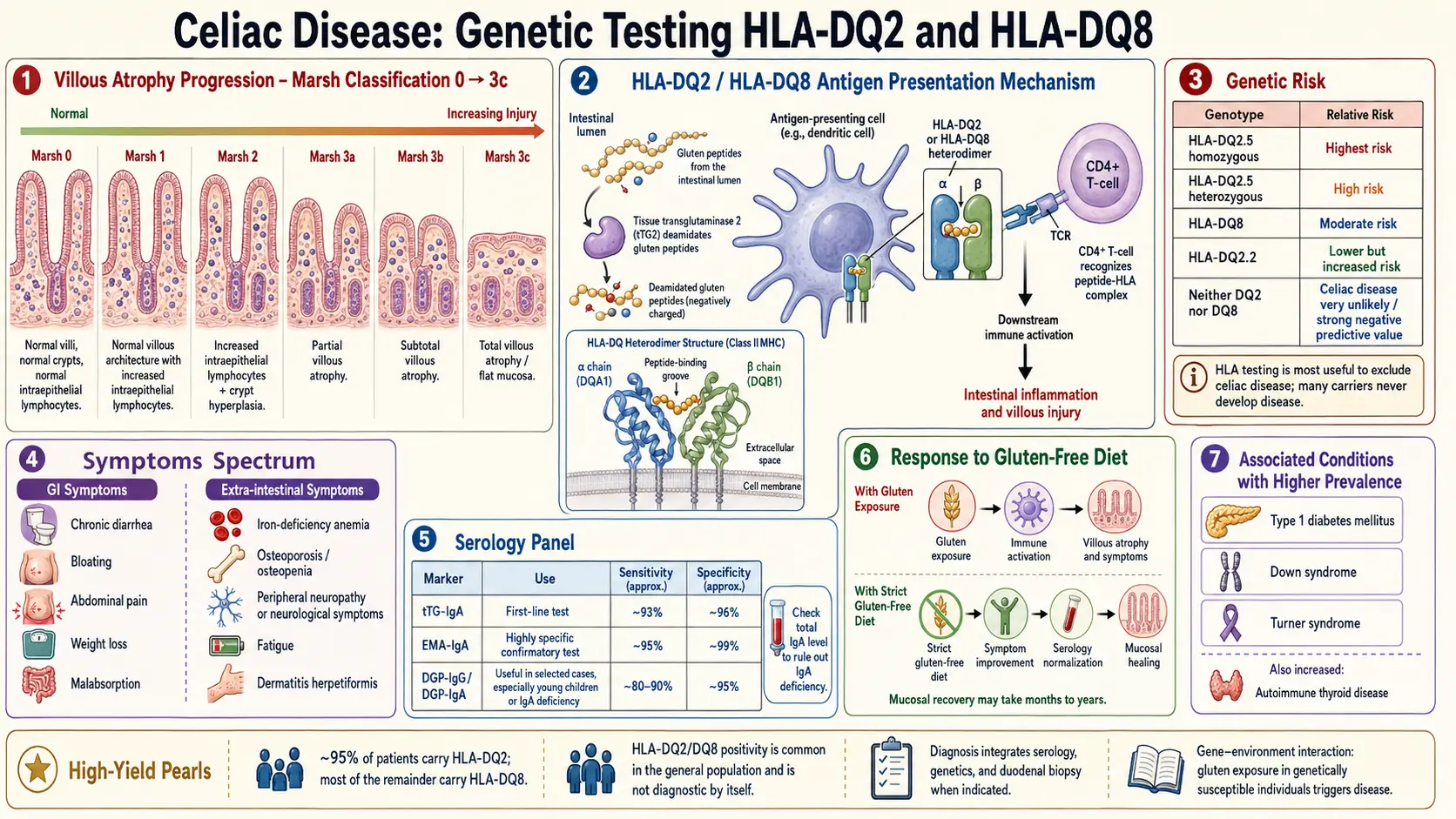
Table of Contents
- What HLA Genes Actually Are
- Meet HLA-DQ2 and HLA-DQ8
- The Numbers That Matter
- The Test's Most Useful Property: Ruling Celiac Out
- The Clinical Misinterpretation Trap
- When to Order the Test
- Dose-Response: Not All Positives Are Equal
- Labs, Methods, and Cost
- Once in a Lifetime — Genes Don't Change
- Shared Haplotypes with Type 1 Diabetes and Beyond
- How to Read Your Report
- Why 23andMe HLA Results Are Unreliable
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
What HLA Genes Actually Are
HLA stands for human leukocyte antigen. These are proteins that sit on the surface of your cells and hold up little molecular flashcards to the immune system. Every card shows a short peptide (a chopped-up piece of protein) that the cell is currently making or has swallowed. Immune cells called T cells patrol your tissues, reading the cards. If the peptide looks like something that belongs to you, the T cell moves on. If it looks foreign — a virus, a bacterium, or a tumor mutation — the T cell raises an alarm.
The genes that code for HLA proteins live on the short arm of chromosome 6, inside a dense region called the Major Histocompatibility Complex (MHC). HLA genes come in two main classes. Class I (HLA-A, -B, -C) appears on almost every nucleated cell in the body and is read by CD8+ "killer" T cells. Class II (HLA-DR, -DQ, -DP) appears mainly on specialized antigen-presenting cells — dendritic cells, macrophages, B cells — and is read by CD4+ "helper" T cells that coordinate the broader immune response.
Celiac disease is a class II story. The molecules that sit at the center of the disease — HLA-DQ2 and HLA-DQ8 — are class II receptors on antigen-presenting cells. When these receptors grab onto gluten peptides (specifically ones that have been chemically altered by an enzyme called tissue transglutaminase, or tTG) and show them to CD4+ T cells, the T cells react as if gluten were a hostile invader. That reaction drives the small-intestine damage you see on a biopsy.
Meet HLA-DQ2 and HLA-DQ8
HLA-DQ is a heterodimer: two protein chains, alpha and beta, that pair up to form one working receptor. The alpha chain is coded by the HLA-DQA1 gene and the beta chain by HLA-DQB1. Both of these live inside the MHC on chromosome 6. Which alpha and beta you inherit determines which flavor of HLA-DQ you carry.
- HLA-DQ2.5 — the classic celiac-associated heterodimer. It is made from the alpha chain DQA1*05 plus the beta chain DQB1*02. Most commonly these two alleles are inherited together on the same chromosome (in cis) as part of a shared block called the DR3-DQ2 haplotype. They can also be inherited on opposite chromosomes (in trans), which still produces a working DQ2.5 molecule but with slightly different downstream risk.
- HLA-DQ2.2 — a close cousin, made from DQA1*02 plus DQB1*02. Carries lower celiac risk than DQ2.5 because its peptide-binding groove holds gluten less tightly.
- HLA-DQ8 — made from the alpha chain DQA1*03 plus the beta chain DQB1*03:02. Usually inherited with the DR4 allele as part of the DR4-DQ8 haplotype.
When you see a report that says "HLA-DQ2 positive" or "HLA-DQ8 positive," what the lab has really done is check whether you carry the underlying DQA1 and DQB1 alleles that assemble into those receptors. A small number of labs still use serological (antibody-based) typing, but virtually all modern testing is DNA-based (PCR or sequence-based).
The Numbers That Matter
Three numbers summarize why this test exists:
- Roughly 95% of biopsy-confirmed celiac patients carry HLA-DQ2. Another 5–10% carry HLA-DQ8. Combined, almost 100% of celiac patients carry DQ2, DQ8, or both. A small number carry only half of the DQ2 heterodimer (just DQB1*02), and the rare remainder carry neither — these cases are unusual enough that some researchers question whether they are truly celiac at all.
- About 30–40% of the general European-ancestry population also carries HLA-DQ2 or HLA-DQ8. Prevalence varies by region — higher in parts of Scandinavia and Ireland, lower in East Asia.
- Only about 3% of DQ2-positive people (and a similar fraction of DQ8 carriers) ever develop celiac disease in their lifetime. The gene is necessary but nowhere near sufficient.
That third number is the one most patients — and unfortunately many clinicians — miss. Being "HLA-DQ2 positive" is far more common than being celiac, by roughly a factor of ten.
The Test's Most Useful Property: Ruling Celiac Out
Here is the single most important sentence on this page: the genuine clinical value of HLA-DQ2/DQ8 testing is its negative predictive value, not its positive predictive value.
If your test comes back negative for both HLA-DQ2 and HLA-DQ8, your chance of having or developing celiac disease is vanishingly small — well under 1%. The negative predictive value exceeds 99%. In practical terms, a truly double-negative result effectively rules out celiac.
That is surprisingly hard to do with any other celiac test. Serology (tTG-IgA, DGP, EMA) can miss cases if the patient is already gluten-free, IgA-deficient, very young, or has Marsh 1–2 disease. Biopsy can miss patchy lesions. The genetic test doesn't care whether you're eating gluten or not — your DNA is the same either way. For a family member or an ambiguous case, a clean negative on HLA is often the fastest way to close the book.
The Clinical Misinterpretation Trap
The flip side is the trap. When a patient comes back "HLA-DQ2 positive," it is extremely common for someone — a well-meaning primary care physician, a naturopath, a family member who read a blog post — to say "That means you have celiac." It does not.
It means you are one of the 30–40% of the population who carries the genetic permission for celiac. The overwhelming majority of those people never develop the disease. A positive HLA result, by itself, means nothing about whether you should eat gluten, should take the diagnosis seriously, or should start buying expensive gluten-free food.
The correct diagnostic pathway remains: symptoms plus serology plus biopsy, while still eating gluten, with HLA genetics used as a supporting or ruling-out tool — never a stand-alone answer. A positive HLA test in an asymptomatic person with negative serology and a normal biopsy simply means: you have the gene, and you do not have the disease today. Revisit testing if symptoms appear.
When to Order the Test
HLA-DQ2/DQ8 testing is not recommended for everyone. The major gastroenterology societies (AGA, ACG, ESPGHAN) reserve it for five fairly specific situations:
- Equivocal biopsy or serology-biopsy mismatch. Serology positive but biopsy normal, or biopsy suggestive but serology negative. A negative HLA in this setting steers you away from a celiac label; a positive HLA supports continued workup.
- Already on a gluten-free diet. Once you have stopped eating gluten, serology and biopsy both normalize over weeks to months, so they stop being reliable. Re-challenging with gluten for six to eight weeks is miserable for most patients and impossible for some. A negative HLA lets you skip the challenge and exit the celiac concern entirely. See the serology testing article for details on why diet confounds those tests.
- First-degree relatives of a confirmed celiac patient. Parents, siblings, and children of a celiac proband have roughly a 10% lifetime risk of developing the disease themselves. A negative HLA on a family member essentially closes the question for life. A positive HLA flags them for periodic serology screening every two to three years, or sooner if symptoms appear.
- Down syndrome, Turner syndrome, Williams syndrome, and selective IgA deficiency. These conditions carry a substantially elevated baseline celiac risk (5–15% in Down syndrome). Gene testing helps decide how aggressively to screen over the lifetime.
- Deciding whether to commit a child to long-term serology screening. A negative HLA in a young child with a celiac parent spares the family two decades of periodic blood draws.
Outside these scenarios, routine HLA testing in asymptomatic adults is not cost-effective and often creates more anxiety than clarity.
Dose-Response: Not All Positives Are Equal
Carrying HLA-DQ2 or DQ8 is not a single binary. There is a gene dosage effect, and knowing where you sit on it changes your numerical risk substantially.
From highest to lowest celiac risk:
- DQ2.5 homozygous — two full copies of the DQA1*05 / DQB1*02 haplotype, one on each chromosome. Relative celiac risk is the highest in the population: roughly 10–15 times the average. Lifetime disease risk in this group is around 15–20%.
- DQ2.5 heterozygous plus DQ2.2 — one full DQ2.5 haplotype and one DQ2.2 half-copy (DQB1*02 without DQA1*05). Also elevated; risk sits between homozygous DQ2.5 and simple DQ2.5 heterozygous.
- DQ2.5 heterozygous (one copy) — the most common celiac-associated genotype. Lifetime disease risk around 3–7%.
- DQ8 alone — lower risk than any DQ2.5 genotype, but still elevated above the general population.
- DQ2.2 alone (half-heterozygote, DQB1*02 without DQA1*05) — the lowest-risk positive result. Some labs used to report this as "celiac gene positive," which is technically true but practically misleading. Lifetime risk of celiac in this group is under 1%.
A thorough lab report will name the exact haplotype and, ideally, state whether you are homozygous or heterozygous. If your report just says "DQ2 positive" without further detail, ask your ordering physician to request the full DQA1 and DQB1 breakdown; most labs will send it on request.
Labs, Methods, and Cost
Major U.S. labs that perform clinical HLA-DQ2/DQ8 typing include LabCorp, Quest Diagnostics, Prometheus, and Mayo Clinic Laboratories. They all use PCR-based or sequence-specific oligonucleotide methods on a blood sample (some accept buccal swabs). Turnaround is typically 5–10 business days.
Cash pricing ranges from roughly $150 to $400 depending on the lab and the depth of typing (a single "DQ2/DQ8" screen is cheaper than a full DQA1/DQB1 high-resolution panel). Insurance coverage is reasonably good when the ordering diagnosis code reflects a legitimate indication — equivocal biopsy, family screening of a first-degree relative, Down syndrome, or diagnostic clarification in a patient already on a gluten-free diet. Ordered as pure curiosity in an asymptomatic adult, it will often be denied and billed back to you.
The relevant CPT codes are typically 81376 (HLA class II typing, low resolution; one locus), 81377 (one antigen equivalent), or for full high-resolution panels 81382/81383. Your ordering physician's office handles the coding; you don't need to memorize these, but having them handy is useful if you need to appeal a denial.
Once in a Lifetime — Genes Don't Change
This is a detail worth emphasizing because it saves people money. HLA genotype does not change over your lifetime. You inherited two DQA1 alleles and two DQB1 alleles at conception; they are the same in your toddler blood, your middle-aged blood, and your elderly blood. You only need the test once.
That means if you had HLA-DQ2/DQ8 testing done as a child, or twenty years ago, or at any point in the past, the old result is still valid. Retesting is a waste unless the original report is genuinely lost. Contrast this with serology (tTG-IgA), which measures your current immune activity and changes dramatically depending on whether you're eating gluten.
A related point: the test result also predates your diagnosis. If you were tested before going gluten-free, the result is identical to what a test done ten years into the diet would show. Diet does not affect HLA results.
Shared Haplotypes with Type 1 Diabetes and Beyond
The DR3-DQ2 and DR4-DQ8 haplotypes are not exclusive to celiac. They are the two highest-risk genetic backgrounds for Type 1 Diabetes (T1D) as well. Roughly 90% of people with T1D carry DR3-DQ2, DR4-DQ8, or both. That is why the two diseases cluster so strongly in families and even in the same individual — somewhere between 3% and 10% of people with T1D also develop celiac, and the overlap runs the other direction too.
If you are HLA-DQ2 or DQ8 positive and have a family history of T1D, it is worth knowing about. It does not mean you will develop T1D. It does mean your children, if they carry the same haplotypes, sit in an elevated-risk bracket for several autoimmune conditions. See the associated autoimmune conditions article for the full overlap map, including Hashimoto's thyroiditis (which shares DR3-DQ2 as a risk background) and autoimmune hepatitis.
More broadly, MHC class II risk alleles cluster across autoimmune disease. DR3 predisposes to celiac, T1D, Hashimoto's, Graves' disease, systemic lupus erythematosus, and myasthenia gravis. DR4 adds rheumatoid arthritis and T1D. On the class I side, HLA-B27 drives ankylosing spondylitis and the broader spondyloarthropathies (see HLA-B27 Explained), and HLA-B*51 is the strongest genetic marker for Behçet's disease. These are useful context: your HLA type is a background on which many possible autoimmune stories can play out, but very few of them actually will.
How to Read Your Report
A clinical HLA-DQ2/DQ8 report typically lists four alleles: two DQA1 values and two DQB1 values, with an interpretive summary at the bottom. Here is a plain-language translation of the combinations you are likely to see.
- "DQ2.5 positive (DQA1*05, DQB1*02)." You carry the highest-risk celiac gene. Your lifetime risk depends on whether one copy or two; if the report does not specify, ask. Roughly 95% of celiac patients match this pattern.
- "DQ8 positive (DQA1*03, DQB1*03:02)." The second celiac-associated pattern. Lower risk than DQ2.5 but still elevated.
- "DQ2.2 only (DQB1*02 positive, DQA1*05 negative)." Half-heterozygote. Lowest-risk positive result. Celiac is possible but uncommon.
- "DQA1*05 only, DQB1*02 negative." The other half of the DQ2.5 dimer without the partner chain. By itself this does not assemble a functioning DQ2.5 receptor and is generally considered celiac-negative for practical purposes.
- "Negative for HLA-DQ2 and HLA-DQ8." The clean-ruling-out result. Your chance of having or developing celiac is under 1%. Ongoing screening is generally not indicated.
If you are sent a report and cannot match it to one of the above, ask the ordering physician to walk through it with you. A good clinic will spend ten minutes on this — it is a high-impact conversation that can spare you years of unnecessary screening or unnecessary worry.
Why 23andMe HLA Results Are Unreliable
Direct-to-consumer genetics companies — 23andMe, AncestryDNA — do not actually sequence your HLA genes. They use SNP genotyping chips that read a sparse scatter of single-letter variants across the genome, then use statistical imputation to guess what your HLA alleles probably are based on the linkage patterns seen in a reference population.
For most genes in most ancestries, imputation works fine. For HLA, it is notoriously unreliable. The MHC region is the most polymorphic stretch of DNA in the human genome — thousands of alleles, dense recombination history, and reference panels heavily biased toward European ancestry. Imputed HLA calls for people of non-European background can be wrong a meaningful fraction of the time, and even for Europeans the subtype-level resolution (e.g. distinguishing DQ2.5 from DQ2.2) is not reliable enough for clinical decisions.
Third-party services like Promethease that interpret raw 23andMe data inherit the same limitations. If your 23andMe-derived report says you are "celiac gene positive" or "celiac gene negative," treat it as a suggestion, not a diagnosis. For any decision that actually matters — skipping a gluten challenge, clearing a family member from ongoing screening, deciding whether to pursue biopsy — order a proper clinical HLA-DQ2/DQ8 test through LabCorp, Quest, or your hospital system. The extra $200 is worth it.
Key Research Papers
- Sollid LM, et al. Evidence for a primary association of celiac disease to a particular HLA-DQ alpha/beta heterodimer. J Exp Med. 1989;169(1):345–50.
- Karell K, et al. HLA types in celiac disease patients not carrying the DQA1*05-DQB1*02 (DQ2) heterodimer: results from the European Genetics Cluster on Celiac Disease. Hum Immunol. 2003;64(4):469–77.
- Murray JA, et al. HLA DQ gene dosage and risk and severity of celiac disease. Dig Dis Sci. 2007.
- Husby S, et al. European Society Paediatric Gastroenterology, Hepatology and Nutrition Guidelines for Diagnosing Coeliac Disease 2020. J Pediatr Gastroenterol Nutr. 2020;70(1):141–56.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on HLA-DQ genetics and celiac disease:
- HLA-DQ2 and celiac disease
- HLA-DQ8 and celiac disease
- HLA-DQ2.5 homozygosity and celiac risk
- Celiac genetic testing and negative predictive value
- Celiac disease first-degree relative screening
- DR3-DQ2 haplotype and Type 1 Diabetes
- HLA imputation accuracy from SNP arrays
- Celiac disease in Down syndrome and HLA screening
Connections
- Celiac Disease Overview
- Associated Autoimmune Conditions
- Celiac Serology Testing: tTG, DGP, EMA
- Gluten-Free Diet: A Practical Guide
- Nutritional Deficiencies and Bone Health
- Non-Celiac Gluten Sensitivity
- Endoscopy, Biopsy, and Marsh Classification
- Refractory Celiac Disease and EATL
- Hashimoto's Thyroiditis
- Ankylosing Spondylitis
- HLA-B27 Explained
- Diabetes
- Dermatitis Herpetiformis