Calcium – Essential Mineral for Human Health
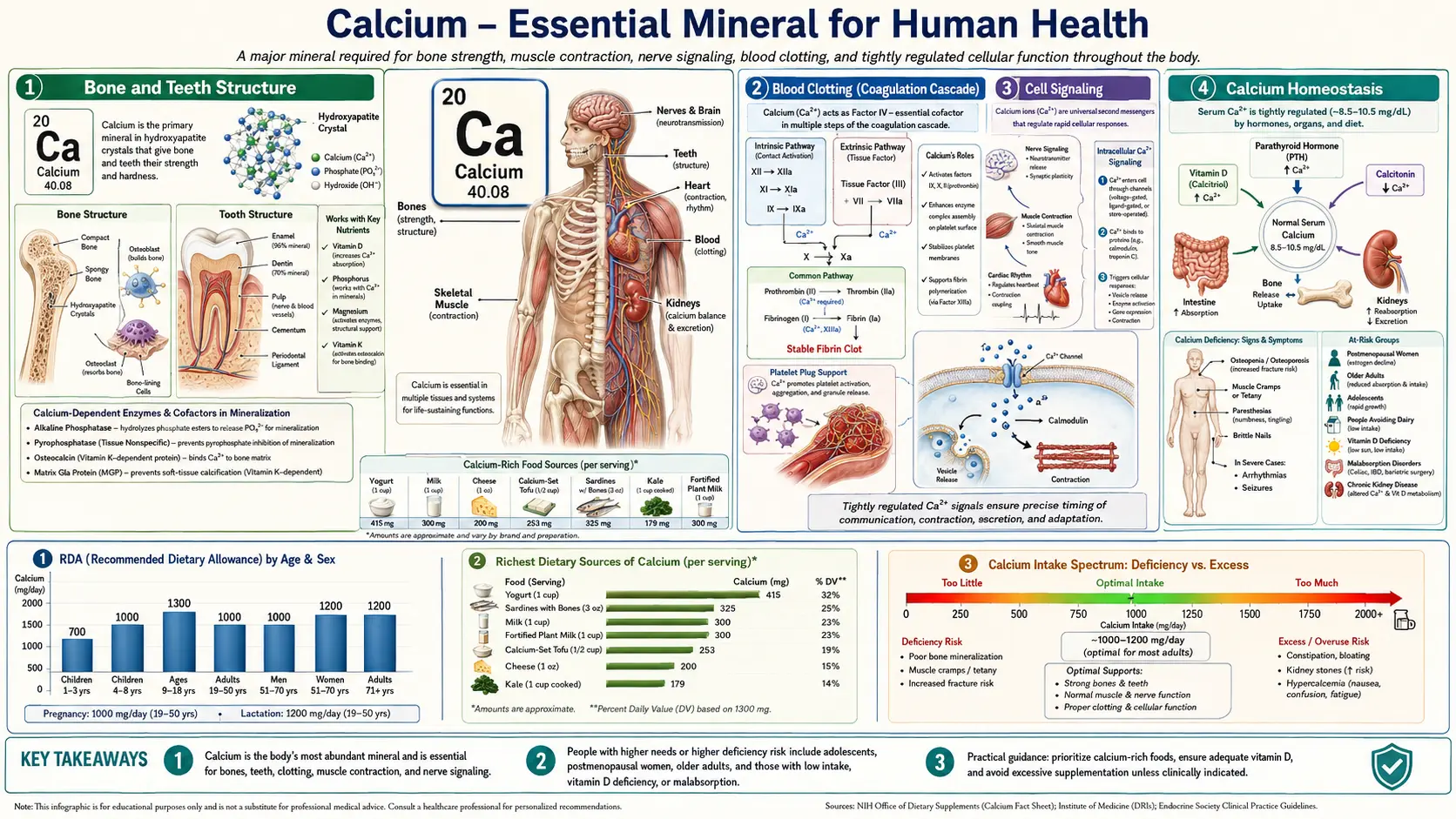
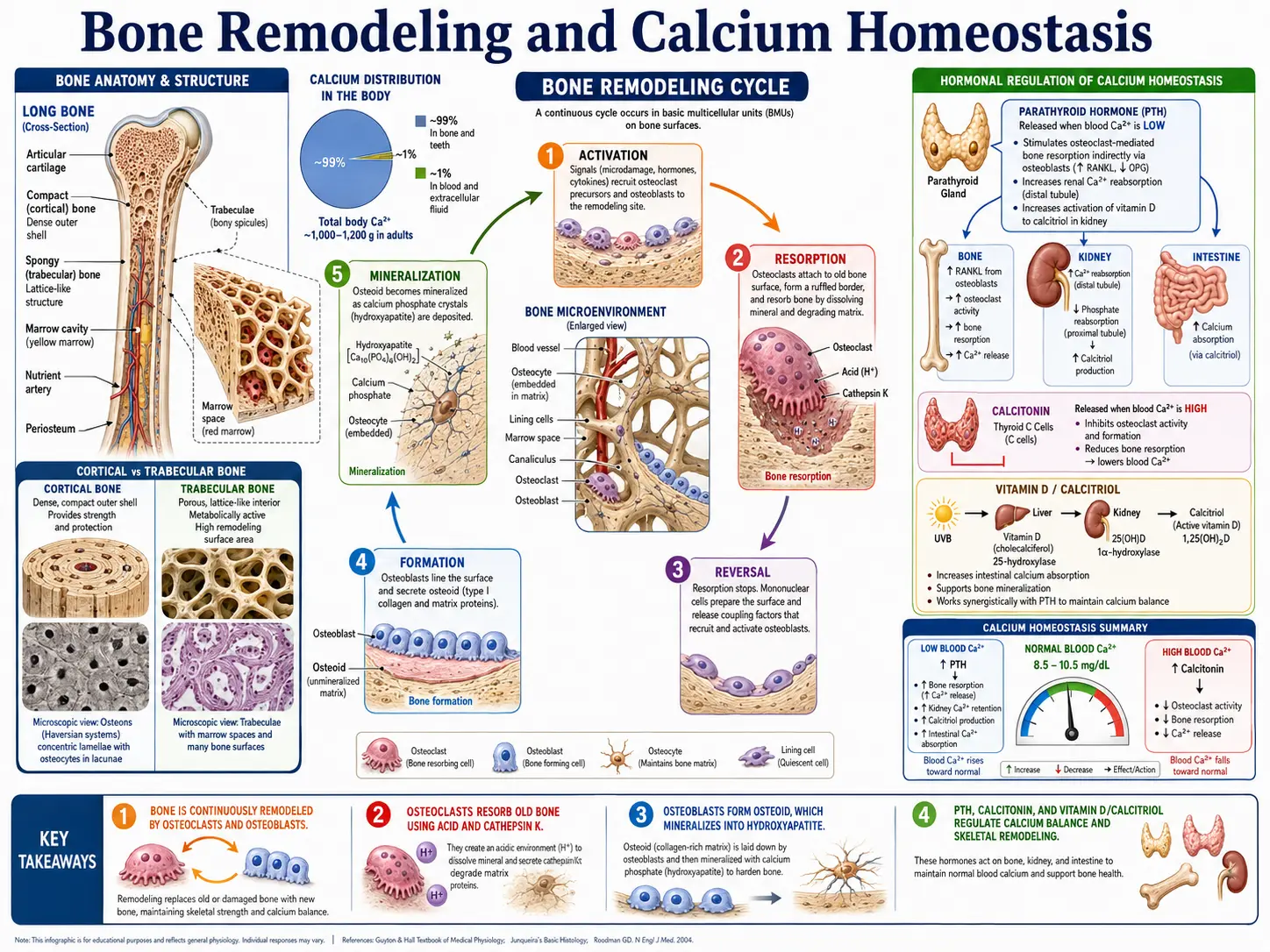
Calcium is the most abundant mineral in the human body, accounting for approximately 1,000 to 1,200 grams in the average adult. Roughly 99% of the body's calcium is stored in bones and teeth, where it provides structural integrity. The remaining 1% circulates in the blood, resides in extracellular fluid, and is distributed within cells, where it performs a remarkably diverse array of physiological functions. Maintaining precise calcium concentrations in these compartments is critical to survival, and the body employs a sophisticated hormonal regulatory system to achieve this balance.
Table of Contents
- Bone and Teeth Structure
- Muscle Contraction
- Nerve Transmission
- Blood Clotting (Coagulation Cascade)
- Enzyme Activation
- Hormone Secretion
- Cell Signaling
- Cardiovascular Function
- Acid-Base Balance
- Calcium Homeostasis
- Connections
- Connections
- Featured Videos
Bone and Teeth Structure
Calcium serves as the principal structural component of the skeletal system and dentition. Within bone and tooth enamel, calcium combines with phosphate to form crystalline hydroxyapatite (Ca10(PO4)6(OH)2), a mineral lattice that confers rigidity and compressive strength.
- Hydroxyapatite – This calcium-phosphate crystal accounts for approximately 65% of bone mass by weight. The hydroxyapatite lattice is deposited within a collagen matrix, creating a composite material that is both hard and somewhat flexible, allowing bones to resist fracture under mechanical stress.
- Bone as a calcium reservoir – Beyond its structural role, the skeleton functions as a dynamic reservoir from which calcium can be mobilized into the bloodstream when dietary intake is insufficient. This process is tightly regulated to prevent excessive bone resorption.
- Dental enamel – Tooth enamel is the hardest substance in the body, composed of approximately 96% hydroxyapatite by weight. Adequate calcium availability during tooth development is essential for proper enamel mineralization and long-term resistance to dental caries.
- Dentin and cementum – Beneath enamel, dentin contains roughly 70% hydroxyapatite, while cementum, which covers the tooth root, is similarly mineralized. Both structures depend on calcium for their formation and maintenance.
Muscle Contraction
Calcium ions (Ca2+) are indispensable for the contraction of all three types of muscle tissue: skeletal, cardiac, and smooth muscle. The mechanism by which calcium triggers contraction is known as excitation-contraction coupling.
- Skeletal muscle – When a motor neuron fires an action potential, calcium is released from the sarcoplasmic reticulum into the cytoplasm. Calcium binds to troponin C on the thin filament, causing a conformational change that moves tropomyosin and exposes myosin-binding sites on actin. Cross-bridge cycling then produces force and shortening.
- Cardiac muscle – Calcium enters cardiac myocytes through L-type voltage-gated calcium channels during the plateau phase of the action potential. This extracellular calcium triggers a much larger release from the sarcoplasmic reticulum via ryanodine receptors, a process called calcium-induced calcium release (CICR).
- Smooth muscle – In smooth muscle, calcium binds to calmodulin rather than troponin. The calcium-calmodulin complex activates myosin light-chain kinase (MLCK), which phosphorylates the myosin regulatory light chain and permits cross-bridge cycling.
Nerve Transmission
Calcium plays a pivotal role in synaptic transmission, the process by which neurons communicate with one another and with effector cells such as muscle fibers and glandular cells.
- Neurotransmitter release – When an action potential arrives at the presynaptic terminal, voltage-gated calcium channels open, allowing Ca2+ to flow into the terminal. The rise in intracellular calcium triggers the fusion of synaptic vesicles with the presynaptic membrane, releasing neurotransmitters such as acetylcholine, norepinephrine, and glutamate into the synaptic cleft.
- Synaptotagmin – Calcium binds to synaptotagmin, a sensor protein on synaptic vesicles, which then interacts with SNARE proteins to catalyze membrane fusion and exocytosis.
- Neural excitability – Extracellular calcium concentration influences the threshold for neuronal firing. Low calcium levels (hypocalcemia) increase neural excitability, which can manifest as tingling, numbness, and in severe cases, tetany or seizures.
Blood Clotting (Coagulation Cascade)
Calcium is designated as Factor IV in the coagulation cascade and is required at multiple steps in the clotting process. Without adequate ionized calcium, blood cannot coagulate properly.
- Gamma-carboxyglutamate (Gla) domains – Several clotting factors (II, VII, IX, and X) contain Gla domains that require calcium ions to bind to phospholipid surfaces on activated platelets. This membrane binding is essential for the assembly of the tenase and prothrombinase complexes.
- Intrinsic and extrinsic pathways – Calcium participates in both the intrinsic pathway (involving factors XII, XI, IX, and VIII) and the extrinsic pathway (involving tissue factor and factor VII). It is also required for the common pathway, where prothrombin is converted to thrombin.
- Fibrin stabilization – Factor XIII, a transglutaminase activated by thrombin in the presence of calcium, cross-links fibrin monomers to form a stable, insoluble clot.
- Clinical relevance – Chelating agents such as EDTA and citrate prevent coagulation in blood collection tubes by binding calcium, underscoring its essential role in the clotting cascade.
Enzyme Activation
Calcium ions serve as cofactors or regulators for numerous enzymes throughout the body, influencing metabolic pathways in virtually every organ system.
- Protein kinase C (PKC) – Calcium, along with diacylglycerol (DAG), activates PKC, a family of enzymes that phosphorylate serine and threonine residues on target proteins. PKC is involved in cell proliferation, differentiation, and immune responses.
- Phospholipase A2 – This enzyme, which releases arachidonic acid from membrane phospholipids (a precursor for prostaglandins and leukotrienes), requires calcium for its activity.
- Calpains – These calcium-dependent cysteine proteases participate in cytoskeletal remodeling, cell migration, apoptosis, and signal transduction.
- Calcineurin – A calcium- and calmodulin-dependent serine/threonine phosphatase that plays a critical role in T-cell activation and immune function. It is the target of immunosuppressive drugs cyclosporine and tacrolimus.
Hormone Secretion
Calcium regulates the release of hormones from endocrine glands through mechanisms similar to neurotransmitter exocytosis.
- Insulin secretion – In pancreatic beta cells, glucose metabolism raises intracellular ATP, which closes ATP-sensitive potassium channels, depolarizes the cell membrane, and opens voltage-gated calcium channels. The resulting calcium influx triggers insulin granule exocytosis.
- Parathyroid hormone (PTH) – The calcium-sensing receptor (CaSR) on parathyroid cells detects extracellular calcium levels. When calcium falls, CaSR signaling decreases, permitting increased PTH secretion. When calcium is high, CaSR activation suppresses PTH release.
- Aldosterone – Angiotensin II stimulates aldosterone secretion from the adrenal zona glomerulosa partly through calcium signaling pathways.
- Catecholamines – Calcium influx into adrenal chromaffin cells is required for the exocytotic release of epinephrine and norepinephrine.
Cell Signaling
Calcium functions as one of the most versatile and ubiquitous second messengers in cellular signaling. Intracellular calcium concentration is maintained at extremely low levels (approximately 100 nanomolar) compared to extracellular fluid (approximately 1.2 millimolar), creating a 10,000-fold gradient that allows rapid and dramatic signaling events when calcium channels open.
- IP3-mediated calcium release – Many G-protein-coupled receptors activate phospholipase C, which cleaves PIP2 into IP3 and DAG. IP3 binds to receptors on the endoplasmic reticulum, releasing stored calcium into the cytoplasm.
- Calmodulin – This ubiquitous calcium-binding protein undergoes a conformational change upon binding four Ca2+ ions, enabling it to activate a wide array of downstream targets, including kinases, phosphatases, and ion channels.
- CaMKII (calcium/calmodulin-dependent protein kinase II) – A multifunctional kinase activated by calcium-calmodulin that plays key roles in synaptic plasticity, learning, memory, and cardiac function.
- Store-operated calcium entry (SOCE) – When intracellular calcium stores are depleted, the ER sensor STIM1 activates Orai1 channels in the plasma membrane, allowing sustained calcium entry from the extracellular space.
- Calcium oscillations – Many cell types exhibit rhythmic fluctuations in cytoplasmic calcium concentration. The frequency and amplitude of these oscillations encode information that determines specific cellular responses such as gene transcription, secretion, or contraction.
Cardiovascular Function
Beyond its role in cardiac muscle contraction, calcium influences multiple aspects of cardiovascular physiology.
- Cardiac pacemaker activity – Calcium currents contribute to the spontaneous depolarization of sinoatrial node cells, helping to establish and maintain heart rhythm. The "calcium clock" mechanism, involving rhythmic calcium release from the sarcoplasmic reticulum, works in concert with the membrane voltage clock.
- Vascular smooth muscle tone – Calcium influx through L-type calcium channels in vascular smooth muscle cells is a primary determinant of vessel tone and blood pressure. Calcium channel blockers (e.g., amlodipine, nifedipine) lower blood pressure by reducing this influx.
- Endothelial function – Calcium signaling in endothelial cells stimulates nitric oxide synthase (eNOS), promoting nitric oxide production and vasodilation.
- Cardiac action potential – The plateau phase (phase 2) of the ventricular action potential depends on inward calcium current through L-type channels, which sustains depolarization and allows sufficient time for ventricular contraction before repolarization.
Acid-Base Balance
Calcium metabolism is closely linked to the body's acid-base status, with changes in blood pH directly affecting calcium availability and function.
- pH and protein binding – Approximately 40% of serum calcium is bound to albumin. Alkalosis increases albumin's affinity for calcium, reducing the ionized (physiologically active) fraction and potentially causing symptoms of hypocalcemia even when total calcium is normal. Acidosis has the opposite effect, increasing ionized calcium.
- Bone buffering – Chronic metabolic acidosis stimulates calcium release from bone as the skeleton's alkaline mineral salts help buffer excess acid. Prolonged acidosis can therefore contribute to bone demineralization.
- Renal handling – The kidneys adjust calcium reabsorption in response to acid-base status. Acidosis tends to increase urinary calcium excretion, while alkalosis promotes calcium retention.
Calcium Homeostasis
The body maintains serum ionized calcium within a narrow range of approximately 1.1 to 1.3 mmol/L through a tightly coordinated hormonal system involving three primary regulators: parathyroid hormone, calcitonin, and active vitamin D (calcitriol).
Parathyroid Hormone (PTH)
- Stimulus – PTH is secreted by the four parathyroid glands in response to a fall in serum ionized calcium, detected by the calcium-sensing receptor (CaSR).
- Bone effects – PTH stimulates osteoclast-mediated bone resorption, releasing calcium and phosphate from the skeleton into the bloodstream.
- Renal effects – PTH increases calcium reabsorption in the distal convoluted tubule and thick ascending limb of Henle, reduces phosphate reabsorption in the proximal tubule (promoting phosphaturia), and stimulates the enzyme 1-alpha-hydroxylase, which converts 25-hydroxyvitamin D to its active form, 1,25-dihydroxyvitamin D (calcitriol).
- Net effect – PTH raises serum calcium and lowers serum phosphate.
Calcitonin
- Stimulus – Calcitonin is released by the parafollicular C cells of the thyroid gland in response to elevated serum calcium.
- Mechanism – Calcitonin inhibits osteoclast activity, reducing bone resorption and thereby lowering serum calcium. It also promotes renal calcium excretion.
- Physiological significance – The role of calcitonin in adult human calcium homeostasis is considered modest compared to PTH and vitamin D. However, it may be more important during periods of calcium stress such as pregnancy, lactation, and skeletal growth.
Vitamin D (Calcitriol)
- Activation pathway – Vitamin D3 (cholecalciferol) is synthesized in the skin upon UVB exposure or obtained from the diet. It is hydroxylated in the liver to 25-hydroxyvitamin D (calcidiol) and then in the kidney to the active hormone 1,25-dihydroxyvitamin D (calcitriol) by 1-alpha-hydroxylase, an enzyme stimulated by PTH.
- Intestinal absorption – Calcitriol's most important action is to increase intestinal absorption of calcium (and phosphate). It induces the expression of calbindin, a calcium-binding protein in enterocytes, and upregulates calcium transport channels (TRPV6) and basolateral calcium pumps.
- Bone effects – Calcitriol supports bone mineralization by maintaining adequate serum calcium and phosphate concentrations. At high levels, it can also stimulate osteoclast differentiation and bone resorption through the RANK/RANKL pathway.
- Renal effects – Calcitriol enhances calcium reabsorption in the distal nephron, working in concert with PTH.
Integrated Homeostatic Response
When serum calcium falls, PTH secretion increases within seconds to minutes. PTH rapidly mobilizes calcium from bone, conserves calcium at the kidney, and stimulates calcitriol production. Calcitriol then enhances intestinal calcium absorption over hours to days. When serum calcium rises above the set point, PTH secretion is suppressed, calcitonin secretion increases modestly, renal calcium excretion rises, and the system returns to equilibrium. This multi-organ, multi-hormone feedback loop ensures that calcium levels remain within the physiological range necessary for cellular function and survival.
Connections
- Calcium Benefits Deep Dive
- Calcium for Bone Health
- Calcium for Muscle Function
- Calcium for Cardiovascular Health
- Calcium for Nerve Transmission
- Calcium Chloride — the IV emergency salt, and why the "acidify the body" theory is a myth
- Calcium and Muscle Function — excitation-contraction coupling, cramps, tetany, and the calcium-magnesium balance for cardiac and skeletal muscle.
- Osteoporosis
- Vitamin D3
- Vitamin K
- Magnesium
- Potassium
- Phosphorus
- Boron
- Silicon
- Manganese and Bone Formation
- Collagen
- Milk
- Cramp Prevention
- Hypertension
- Kidney Disease
- Vitamin C
- Celiac Disease