Endoscopy, Biopsy, and Marsh Classification in Celiac Disease
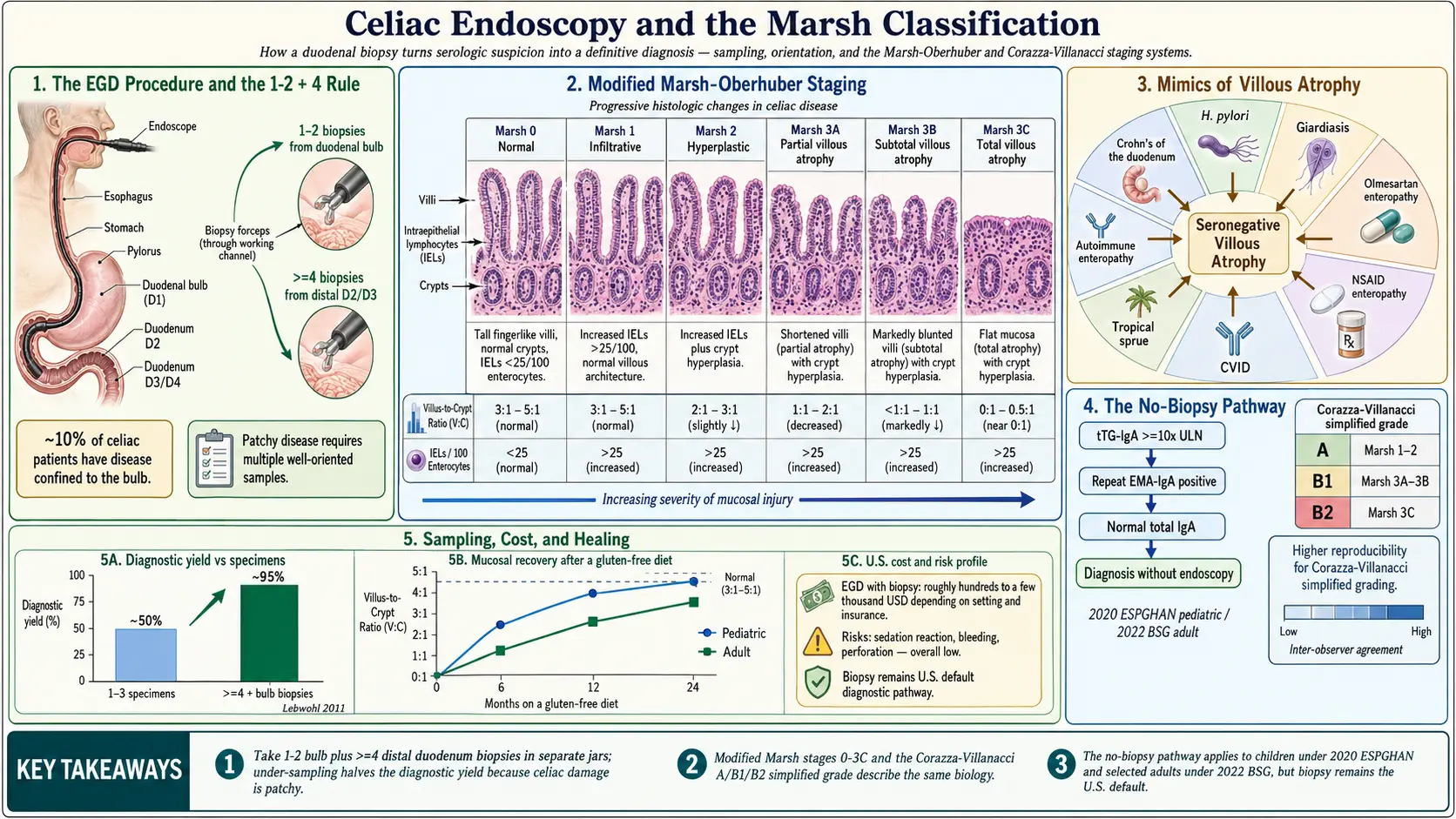
Table of Contents
- Why a Biopsy Still Matters
- The EGD Procedure, Step by Step
- Sedation Choices — MAC, Propofol, and Moderate
- Proper Biopsy Technique — The 1-2 + 4 Rule
- Orientation and Why It Changes the Answer
- The Marsh-Oberhübner Classification
- Corazza-Villanacci A/B/C — The Simplified Grade
- When Marsh 1 Is Enough to Diagnose
- Other Causes of Villous Atrophy
- Capsule Endoscopy and MR Enterography
- The No-Biopsy Pathway
- Follow-Up Biopsy and Mucosal Healing
- Cost, Prep, and Risks
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
Why a Biopsy Still Matters
For decades, the small-bowel biopsy has been called the "gold standard" for celiac disease. That phrase is worn down from overuse, but the reason it stuck is simple: celiac disease is defined, at its core, by a specific pattern of damage to the lining of the small intestine, and the only way to see that pattern is to look at it under a microscope. Blood tests tell you the immune system is reacting to gluten. Genetics tell you the reaction is biologically possible. The biopsy tells you whether the damage has actually happened yet, how bad it is, and whether something else might be mimicking it.
In the last ten years, that picture has shifted. European and British guidelines now allow selected adults and children to skip the biopsy entirely if their blood work is extreme enough. That change is real and legitimate, but it applies to a minority of patients. For most people evaluated for celiac disease in the United States, an upper endoscopy with duodenal biopsies remains the step that turns a suspicion into a diagnosis.
This article walks through what happens the day of the procedure, how the tissue is interpreted, and why the quality of the biopsy — how many samples, from where, and how they are handled — matters just as much as the technology doing the looking.
The EGD Procedure, Step by Step
An upper endoscopy is called an EGD, short for esophagogastroduodenoscopy — esophagus, stomach, and duodenum. That last word is the important one for celiac evaluation. The duodenum is the first foot or so of small intestine, and it is where gluten-driven damage tends to hit hardest.
You will be asked to fast for at least eight hours before the procedure. No food, no milk, no gum, no coffee. Most centers allow small sips of water with morning medications up to two hours beforehand. An empty stomach is not a formality; anything in there obscures the view, irritates the scope passage, and raises the aspiration risk during sedation.
On arrival, you change into a gown, an IV is placed, and baseline vitals are recorded. In the procedure room you lie on your left side. A soft plastic bite block goes between your teeth to protect them and keep your mouth open. Sedation is pushed through the IV, and within a minute or two you are asleep or deeply relaxed.
The gastroenterologist passes a flexible scope — about the thickness of an index finger — through your mouth, down the esophagus, through the stomach, past the pylorus (the valve at the stomach's exit), and into the duodenum. The scope has a camera, a light, and a working channel through which tiny biopsy forceps can be extended. The doctor inspects the lining visually, then pushes the forceps out, takes small tissue samples (each about the size of a pinhead), and pulls them back through the channel into a jar of formalin fixative.
Start to finish, the examination itself takes ten to twenty minutes. You wake up in recovery over the next thirty to sixty minutes. Somebody must drive you home — sedation impairs reflexes and judgment for the rest of the day.
Sedation Choices — MAC, Propofol, and Moderate
Two main sedation approaches are used. Knowing the difference helps you ask the right questions when your procedure is scheduled, because your experience and your bill will both depend on it.
Moderate ("conscious") sedation uses a combination of a benzodiazepine (usually midazolam) and a short-acting opioid (usually fentanyl), pushed by the endoscopy nurse under the gastroenterologist's supervision. You are drowsy, comfortable, and usually amnestic, but you can respond to voice. This is cheaper and does not require a dedicated anesthesiologist. Recovery is a little slower and a small fraction of patients remember parts of the procedure.
Monitored anesthesia care (MAC) with propofol is delivered by an anesthesiologist or certified nurse anesthetist who stays in the room. Propofol works in seconds, wears off in minutes, and produces a deeper, more predictable sleep. Most patients remember nothing. It is the default in many U.S. endoscopy centers today because throughput is faster and patient satisfaction is higher. The downside is cost: a separate anesthesia bill, often several hundred to over a thousand dollars on top of the procedure itself, and coverage varies by insurer.
Neither approach is medically required for a routine celiac EGD. If cost is a concern, ask whether moderate sedation is an option. If prior procedures have been unpleasant or you have high anxiety, request MAC/propofol and confirm in advance that your insurance covers it.
Proper Biopsy Technique — The 1-2 + 4 Rule
Here is where many celiac diagnoses are made or missed. Celiac damage is famously patchy. The immune attack does not wallpaper the duodenum evenly — it hits some villi hard, spares others a centimeter away, and leaves a mosaic. A single biopsy or even two in one spot can land in normal-looking tissue and return a falsely reassuring result.
Current guidelines from the American College of Gastroenterology, the British Society of Gastroenterology, and ESPGHAN all converge on the same minimum:
- 1 to 2 biopsies from the duodenal bulb (the first segment, immediately past the pylorus).
- At least 4 biopsies from the distal duodenum — the second and third parts, often called D2 and D3.
That is a total of five to six samples, each dropped into a separate labeled jar so the pathologist knows where it came from. Fewer than four samples from D2 miss a meaningful fraction of celiac cases. A large U.S. study by Lebwohl and colleagues (2011) showed that when endoscopists submitted only one to three specimens, the diagnostic yield for celiac disease dropped by roughly half compared with four or more.
The bulb biopsies were added to guidelines in the 2010s after studies found that roughly 10% of celiac patients have disease confined to the bulb — the only part of the duodenum where villi look abnormal. Skipping the bulb in those patients hands them a false negative. Historically the bulb was avoided because its normal architecture is slightly irregular (Brunner's glands create small distortions), and pathologists can mistake normal bulb tissue for mild disease. The solution is not to skip it but to label it clearly so the pathologist interprets it in context.
Before your procedure, it is entirely reasonable to ask your gastroenterologist: "Will you be taking at least one or two samples from the bulb and four from the distal duodenum, in separate jars?" A competent celiac workup will say yes without needing to think about it.
Orientation and Why It Changes the Answer
Pathology is a craft as much as a science. A duodenal biopsy is a tiny curl of mucosa, and when it is embedded in paraffin and sliced, the angle of the cut decides what the pathologist sees. A well-oriented biopsy is sliced perpendicular to the surface, so each villus appears as a tall finger and each crypt as a deep well. A poorly oriented biopsy is sliced obliquely, and normal villi can look flattened or missing — an artifact, not disease.
Good endoscopy labs float each biopsy onto a small piece of filter paper or cucumber slice before dropping it into formalin, which helps the tissue orient. Good pathology labs have a technician who embeds the sample with the villi pointing upward. Neither is guaranteed. If your biopsy report says "orientation suboptimal" or "cannot be fully assessed," that is a reason to repeat, not to diagnose. If your story strongly suggests celiac and your biopsy is equivocal, ask whether orientation was adequate.
The Marsh-Oberhübner Classification
In 1992, the British pathologist Michael Marsh proposed a staging system for gluten-related small-bowel damage. In 1999, Oberhuber refined it, and the result — called the Marsh-Oberhübner or simply modified Marsh classification — remains the language pathologists use today. Understanding it lets you read your own biopsy report.
- Marsh 0 — Normal. Villi are tall and fingerlike, crypts are the expected depth, and the number of lymphocytes inside the surface lining (intraepithelial lymphocytes, or IELs) is under 25–30 per 100 enterocytes. A Marsh 0 biopsy in a person on a normal gluten-containing diet with negative serology effectively rules out active celiac disease.
- Marsh 1 — Infiltrative. Villi and crypts still look normal, but IELs are elevated above 25–30 per 100 enterocytes. This is the earliest detectable sign of immune activation. Marsh 1 is not specific — many things cause it — but combined with positive celiac antibodies, it can be diagnostic.
- Marsh 2 — Hyperplastic. Elevated IELs plus crypt hyperplasia (the crypts become deeper and more active as the mucosa tries to replace dying enterocytes faster). Villi are still standing but stressed.
- Marsh 3A — Partial villous atrophy. Villi are visibly shorter than normal, IELs are elevated, crypts are hyperplastic.
- Marsh 3B — Subtotal villous atrophy. Villi are markedly blunted, barely protruding above the mucosal surface.
- Marsh 3C — Total villous atrophy. Villi are essentially gone. The mucosa looks flat, like a cobblestone plaza where a forest used to stand. Classic, severe celiac.
Some older reports still use "Marsh 4" for irreversible hypoplastic atrophy, but that category has been largely retired — most "Marsh 4" cases turn out to be refractory celiac or enteropathy-associated T-cell lymphoma (EATL), which are handled separately (see the refractory celiac article).
Corazza-Villanacci A/B/C — The Simplified Grade
Because pathologists disagreed more often than anyone liked on where exactly Marsh 3A ended and 3B began, in 2007 Corazza and Villanacci proposed a simpler three-tier scheme:
- Grade A — non-atrophic (elevated IELs with or without crypt hyperplasia). Roughly corresponds to Marsh 1 and 2.
- Grade B1 — atrophic, villus-to-crypt ratio greater than 1:1. Roughly corresponds to Marsh 3A and 3B.
- Grade B2 — atrophic, villi no longer visible. Roughly corresponds to Marsh 3C.
Agreement among pathologists on the Corazza-Villanacci grade is substantially higher than on modified Marsh subtypes. Some U.S. centers now report both. Clinically, the two systems mean the same thing — they are vocabulary, not biology.
When Marsh 1 Is Enough to Diagnose
Marsh 1 — elevated lymphocytes without architectural change — is a trap in both directions. On its own it is not celiac disease; roughly half of Marsh 1 biopsies turn out to be caused by something else (see the next section). But in the right context, Marsh 1 is plenty.
The combination that supports a celiac diagnosis at Marsh 1 is:
- Classic symptoms (chronic diarrhea, weight loss, iron-deficiency anemia, dermatitis herpetiformis, or a family history).
- Positive celiac serology — elevated tissue transglutaminase IgA (tTG-IgA) or endomysial antibody (EMA), or positive deamidated gliadin peptide antibodies. See the serology article.
- Positive HLA-DQ2 or DQ8 genetics — see the HLA genetics article.
- Clear improvement on a strict gluten-free diet.
A Marsh 1 biopsy alone, with negative antibodies and no symptoms, is not celiac. But when the serology, genetics, and symptoms all line up, Marsh 1 is considered early celiac disease and treated as such.
Other Causes of Villous Atrophy
Villous atrophy is celiac disease's signature, but it is not unique to celiac. A pathologist who sees blunted villi is trained to keep a short list of alternatives in mind, and so should you — particularly if your serology is negative.
- Helicobacter pylori infection — typically causes increased IELs in the duodenum (Marsh 1 pattern), rarely frank atrophy.
- Giardiasis — Giardia trophozoites adhere to the duodenal lining and can cause partial villous atrophy that resolves with antibiotic treatment.
- Intestinal tuberculosis — rare in the U.S. but important in endemic regions; usually shows granulomas.
- Olmesartan enteropathy — the blood-pressure drug olmesartan (Benicar) can cause a sprue-like villous atrophy that is indistinguishable from celiac except by history and resolution off the drug.
- NSAID enteropathy — chronic ibuprofen, naproxen, or diclofenac use injures the small bowel.
- Mycophenolate mofetil (MMF) — an immunosuppressant used after transplant; well-documented cause of villous atrophy.
- Common variable immunodeficiency (CVID) — produces a celiac-like picture, usually with a striking absence of plasma cells in the lamina propria.
- Tropical sprue — a post-infectious enteropathy seen in travelers returning from the tropics.
- Autoimmune enteropathy — rare; often associated with anti-enterocyte antibodies.
- Crohn's disease with duodenal involvement can look similar; usually other features point to Crohn's (see the Crohn's article).
This list is why "villous atrophy + negative celiac antibodies" always triggers a second thought. It is called seronegative villous atrophy, and the workup shifts away from gluten and toward drugs, infections, and rarer immunopathologies.
Capsule Endoscopy and MR Enterography
The EGD only reaches the duodenum. Occasionally the question is bigger: Is there disease further downstream? Is something else, like small-bowel lymphoma or ulceration, masquerading? Two additional tools help.
Video capsule endoscopy (VCE) is a pill-sized camera you swallow. It transmits images of the entire small bowel as it travels through. For celiac, VCE is not a diagnostic tool — it cannot take biopsies — but it is useful in refractory celiac disease, to look for ulcerative jejunitis, strictures, or mass lesions suggestive of lymphoma.
MR enterography (MRE) is an MRI of the small bowel after drinking a large volume of contrast fluid. It shows wall thickening, lymph node enlargement, and enhancement patterns. Like VCE, it is reserved for atypical or refractory cases.
Neither test replaces the EGD with biopsies for routine celiac diagnosis. They are adjuncts for when the standard workup does not add up.
The No-Biopsy Pathway
Europe moved first. ESPGHAN (European pediatric gastroenterology society) published a no-biopsy pediatric pathway in 2012 and updated it in 2020. A child can be diagnosed with celiac disease without endoscopy if all of the following are true:
- tTG-IgA is at least 10 times the upper limit of normal.
- Endomysial antibody (EMA-IgA) is positive on a separate blood draw.
- Total IgA is normal (to rule out IgA deficiency, which would invalidate the tTG-IgA result).
Symptoms and HLA typing are no longer required under the 2020 update. In adults, the British Society of Gastroenterology adopted a similar pathway in 2022: adults with tTG-IgA ≥ 10× ULN plus a positive EMA on a second sample can be diagnosed without biopsy in selected cases, particularly when endoscopy access is limited.
The American College of Gastroenterology remains more cautious. In the U.S., biopsy is still recommended for almost all adults, with no-biopsy diagnosis reserved for children meeting ESPGHAN criteria or adults for whom endoscopy is genuinely unsafe. Part of the reason is practical — U.S. labs report tTG values in different units and different reference ranges, making "10× ULN" less uniform than in Europe.
The bottom line for patients: if your tTG is mildly or moderately elevated, you will be offered endoscopy. If it is very high (above ten times the upper limit of normal) and you are in a country or a system using the no-biopsy pathway, you may be diagnosed on blood work alone. Ask your gastroenterologist which pathway applies.
Follow-Up Biopsy and Mucosal Healing
For years, a repeat endoscopy at one to two years on a gluten-free diet was standard, to confirm that the villi had regrown. That practice has softened. Current guidelines recommend a follow-up biopsy mainly when:
- Symptoms persist despite an apparently strict gluten-free diet.
- Serology remains elevated beyond twelve months.
- New symptoms appear after a period of stability, raising concern for refractory celiac or lymphoma.
- The original diagnosis was uncertain.
In uncomplicated celiac disease with resolved symptoms and normalizing serology, many gastroenterologists now skip the repeat scope. Healing is rarely complete at twelve months anyway — adults often need two years or more for histologic recovery, and a substantial minority never fully heal even on a strict diet. The decision is individualized.
Cost, Prep, and Risks
An EGD in the United States is one of the more variable-priced procedures in medicine. A cash-pay ambulatory surgery center may bill $1,500 to $2,500 all-in. A hospital outpatient department may bill $3,000 to $5,000 or more for the same procedure, and the MAC anesthesia adds another $500 to $1,500 on top. Insurance negotiated rates are lower, but copays and deductibles can still leave patients with a four-figure bill. Before scheduling, call the facility and ask for an itemized estimate that includes facility fee, physician fee, pathology fee, and anesthesia fee — those four line items are billed separately and surprise patients who were only quoted one of them.
Prep is simple compared with a colonoscopy. Nothing to eat or drink after midnight the night before (or for eight hours before the procedure if it is in the afternoon). Small sips of water with essential morning medications are usually fine — confirm with the scheduling nurse. Bring a driver. Plan to rest the remainder of the day.
Risks of a diagnostic EGD are low but not zero:
- Perforation of the esophagus, stomach, or duodenum — approximately 1 in 10,000 diagnostic EGDs. A hole in the gut wall is a surgical emergency.
- Bleeding from a biopsy site — usually self-limiting, more concerning in patients on blood thinners. Rates around 1 in 500 to 1 in 1,000.
- Aspiration during sedation — minimized by proper fasting.
- Adverse reactions to sedation — usually mild (nausea, low blood pressure), very rarely serious.
- Sore throat and mild bloating afterward — common, self-limiting within 24 hours.
The overall serious-complication rate for diagnostic upper endoscopy is roughly 1 in 500 to 1 in 1,000. For someone with a reasonable pretest probability of celiac disease, the benefit of a confirmed diagnosis — and the lifetime of gluten-free commitment it justifies — far outweighs that risk.
Key Research Papers
- Marsh MN. Gluten, major histocompatibility complex, and the small intestine. A molecular and immunobiologic approach to the spectrum of gluten sensitivity ('celiac sprue'). Gastroenterology. 1992.
- Oberhuber G, Granditsch G, Vogelsang H. The histopathology of coeliac disease: time for a standardized report scheme for pathologists. Eur J Gastroenterol Hepatol. 1999.
- Corazza GR, Villanacci V, et al. Comparison of the interobserver reproducibility with different histologic criteria used in celiac disease. Clin Gastroenterol Hepatol. 2007.
- Lebwohl B, Kapel RC, Neugut AI, Green PH, Genta RM. Adherence to biopsy guidelines increases celiac disease diagnosis. Gastrointest Endosc. 2011.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on celiac biopsy technique, Marsh grading, and the no-biopsy pathway:
- Celiac disease duodenal biopsy technique
- Duodenal bulb biopsy in celiac disease
- Marsh classification in celiac disease
- Corazza-Villanacci grading system
- No-biopsy pathway for celiac diagnosis in adults
- ESPGHAN pediatric celiac guidelines
- Seronegative villous atrophy differential diagnosis
- Olmesartan-associated enteropathy
- Mucosal healing and follow-up biopsy in celiac disease
- Video capsule endoscopy in refractory celiac disease
Connections
- Celiac Disease Overview
- Gluten-Free Diet: A Practical Guide
- Dermatitis Herpetiformis
- Nutritional Deficiencies and Bone Health
- IBD Diagnostic Workup: Calprotectin, Colonoscopy, MRE
- Crohn's Disease
- Irritable Bowel Syndrome
- SIBO
- Anemia
- Full-Body MRI Screening
- Celiac Serology Testing tTG DGP EMA
- Genetic Testing HLA DQ2 and DQ8
- Non Celiac Gluten Sensitivity
- Refractory Celiac and EATL