Refractory Celiac Disease and EATL
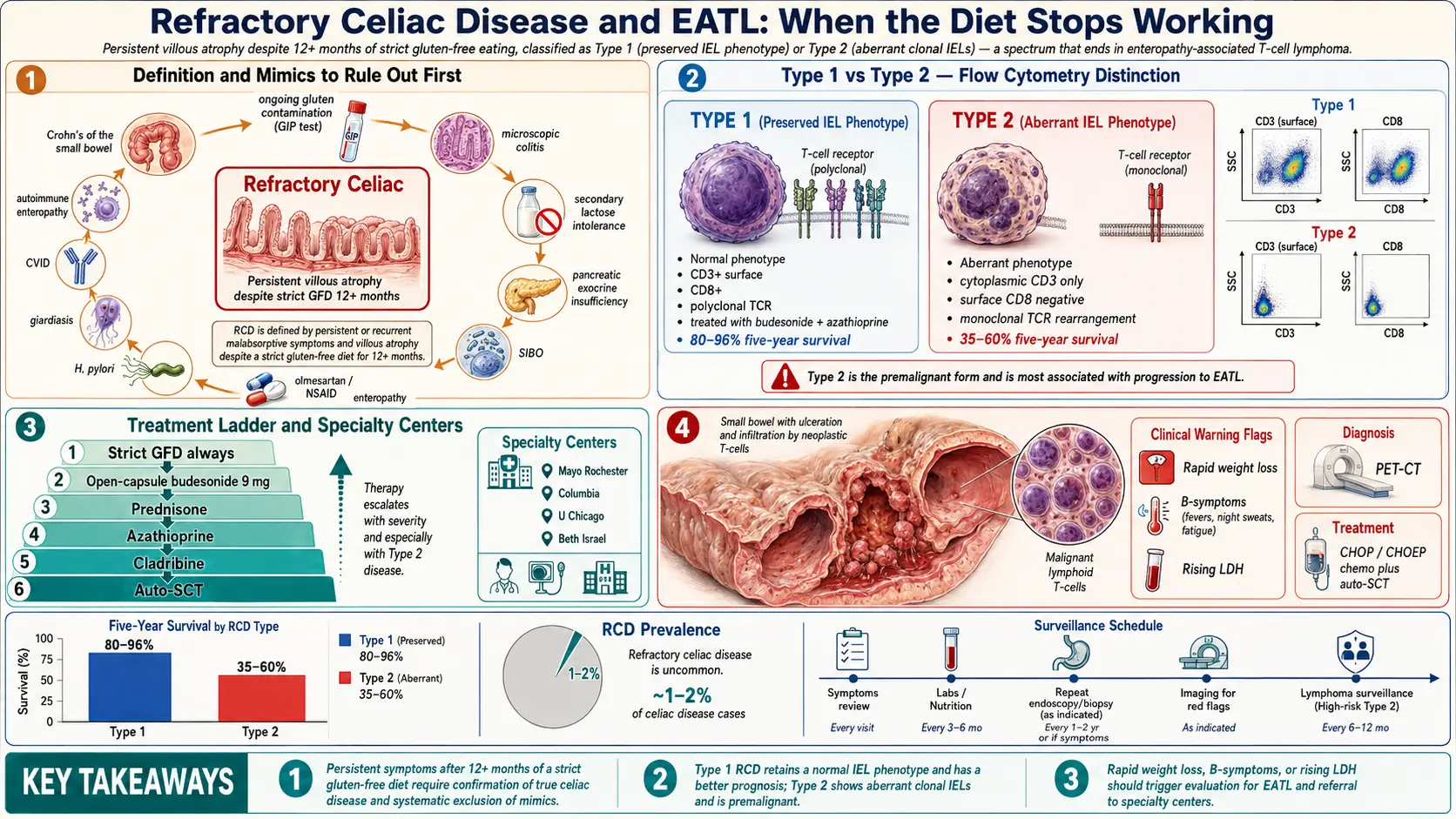
Table of Contents
- What Refractory Celiac Disease Actually Is
- How Common Is It?
- Type 1 versus Type 2 — Why the Distinction Matters
- Before You Accept the Diagnosis — What Must Be Ruled Out
- The Full Workup
- Treating Type 1 Refractory Celiac
- Treating Type 2 Refractory Celiac
- EATL — Enteropathy-Associated T-Cell Lymphoma
- Prognosis — The Honest Numbers
- When and Where to Get a Second Opinion
- Monitoring Cadence
- Living With Refractory Disease
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
What Refractory Celiac Disease Actually Is
If you have celiac disease, the standard story is simple and reassuring: cut out gluten, give your small intestine six to twenty-four months to heal, and the villi grow back, the symptoms fade, and the antibody tests turn negative. The overwhelming majority of patients follow exactly this arc.
A small minority do not. Refractory celiac disease (RCD) is the formal name for the situation in which malabsorptive symptoms — diarrhea, weight loss, fatigue, anemia, low albumin — persist or come back despite at least twelve months on a strict gluten-free diet, and a repeat biopsy still shows villous atrophy. The key word is "despite." By definition, if you are still being exposed to gluten, you do not have refractory disease — you have an unhealed gut from ongoing exposure, which is a completely different problem with a much better prognosis.
Refractory celiac is a diagnosis of exclusion. Three conditions must all be true:
- A prior, well-documented celiac diagnosis — positive tTG-IgA or EMA plus biopsy showing Marsh 3 villous atrophy, plus supportive HLA DQ2/DQ8 genetics.
- Persistent or recurrent malabsorptive symptoms plus ongoing villous atrophy on follow-up biopsy after twelve or more months of strict gluten-free eating.
- Gluten contamination ruled out as the cause, along with all the other mimics that can keep a celiac patient sick (listed in the next section).
If all three are met, you have refractory celiac disease. The next step is to figure out which type.
How Common Is It?
In large referral-center series, roughly 1–2% of celiac patients eventually meet criteria for refractory disease. That number sounds small, but celiac itself affects about 1% of the general population — so we are talking about tens of thousands of people in the United States alone.
Real-world numbers are almost certainly lower than 1–2%, because most studies come from tertiary centers that receive the hardest cases. Community-practice estimates suggest the true incidence is well under 1% of all diagnosed celiacs. The overall point stands: if you are a celiac patient doing badly, the statistical odds still favor gluten contamination, a missed mimic, or a coexisting condition — not refractory disease. Clinicians investigate those first for a reason.
Type 1 versus Type 2 — Why the Distinction Matters
Refractory celiac comes in two flavors, and they are treated as different diseases because their cell biology — and their prognosis — are genuinely different.
Type 1 (RCD-I)
The intraepithelial lymphocytes (IELs) that live between your gut lining cells look normal under the pathologist's microscope and on flow cytometry. They express the full complement of surface markers a healthy T cell should: CD3 positive, CD8 positive, TCR-αβ positive. There is no clonal rearrangement of their T-cell receptor genes — the population is diverse, the way a normal immune response should be.
Type 1 behaves a lot like a stubborn, smoldering version of ordinary celiac disease. It usually responds to immunosuppression (steroids, budesonide, sometimes azathioprine), and most patients regain a near-normal life expectancy.
Type 2 (RCD-II)
The IELs are aberrant. They have lost surface CD8, lost their surface T-cell receptor, and show cytoplasmic CD3 only — a telltale pattern on flow cytometry. Their T-cell-receptor genes show a monoclonal rearrangement, meaning a single abnormal clone has expanded to dominate the IEL population. Many of these cells express TCR-γδ rather than the usual αβ.
This is no longer quite celiac disease. It is a low-grade T-cell neoplasia confined to the intestinal epithelium — a precursor state on a continuous spectrum that ends in overt T-cell lymphoma. Type 2 is the dangerous one, and it is the form that most often progresses to EATL.
The laboratory distinction between Type 1 and Type 2 is not subtle, but it requires a pathology lab that can do flow cytometry on intraepithelial lymphocytes and TCR gene rearrangement studies. Many community hospitals cannot. This is one of the main reasons refractory celiac is referred to specialty centers — getting the subtype right changes everything that follows.
Before You Accept the Diagnosis — What Must Be Ruled Out
A gastroenterologist worth their salt will spend more time excluding mimics than confirming refractory disease. The list is long because each of these conditions is treatable, and several are far more common than true RCD.
- Ongoing gluten exposure. The single biggest cause of persistent symptoms in a diagnosed celiac. Modern testing includes gluten immunogenic peptide (GIP) assays on urine or stool — sold as GlutenDetect and similar kits. A positive GIP test on a patient who believes they are strict proves hidden exposure and reframes the entire case. A formal sit-down with a celiac-experienced registered dietitian catches contamination sources most patients miss (shared toaster, oats that are not certified gluten-free, medications with wheat starch, lip balm, communion wafers, restaurant fryer oil).
- Microscopic colitis — lymphocytic or collagenous colitis causes watery diarrhea and is more common in celiac patients than in the general population. Colonoscopy with random biopsies catches it.
- Lactose intolerance. Damaged villi cannot make lactase. Many newly diagnosed celiacs also cannot tolerate dairy for months after starting the diet, even once gluten is out.
- Pancreatic exocrine insufficiency. Celiac can impair the hormonal signals that tell the pancreas to release enzymes. Check fecal elastase — a level under 200 µg/g suggests insufficiency, under 100 is definitive. A short trial of pancreatic enzyme replacement (pancrelipase) can be diagnostic and therapeutic.
- Small intestinal bacterial overgrowth (SIBO). Damaged small-bowel motility and architecture invite bacterial colonization. A glucose or lactulose breath test clarifies. Treatment with rifaximin often unlocks months of symptom improvement.
- Lymphocytic enteritis from other causes — medications (especially olmesartan, NSAIDs, mycophenolate), Helicobacter pylori, giardiasis, common variable immunodeficiency (CVID), tropical sprue, autoimmune enteropathy.
- Other enteropathies that mimic celiac. Crohn's disease of the small bowel, eosinophilic enteritis, Whipple's disease, and autoimmune enteropathy can all look like celiac on a superficial biopsy read.
Running this exclusion list takes weeks or months. That is normal, and it is worth doing carefully — finding a treatable mimic is far better news than landing on a refractory diagnosis.
The Full Workup
Once the mimics are excluded and twelve months of documented strict gluten-free eating still leaves you with symptoms and villous atrophy, the specialty workup kicks in:
- Repeat upper endoscopy (EGD) with duodenal biopsies — at least four specimens from the distal duodenum plus one or two from the bulb. The pathologist re-confirms Marsh 3 atrophy.
- Flow cytometry on fresh IELs — this is the test that separates Type 1 (normal phenotype) from Type 2 (aberrant, CD3+ cytoplasmic, surface-CD8 negative, surface-TCR negative). It must be ordered prospectively because the sample has to reach the flow lab alive.
- T-cell receptor gene rearrangement study — a PCR-based test looking for a monoclonal TCRγ or TCRβ clone in the IEL population. A monoclonal result supports Type 2.
- HLA typing — specifically, DQ2.5 homozygosity (inheriting DQ2.5 from both parents) is a known risk factor for progressing to Type 2 and EATL.
- Small-bowel imaging — magnetic resonance enterography (MRE) or video capsule endoscopy to look for wall thickening, masses, strictures, or ulcerative jejunitis (deep ulcers in the jejunum that carry a very high risk of progression to lymphoma).
- PET-CT scan — reserved mostly for suspected Type 2 or any suspicion of EATL. Fluorodeoxyglucose-avid bowel wall or mesenteric lymph nodes should raise the alarm.
- Labs — CBC, comprehensive metabolic panel, albumin, ferritin, vitamin B12, folate, vitamin D, zinc, copper, magnesium, tTG-IgA (to track diet adherence), and LDH (a rough lymphoma marker).
Treating Type 1 Refractory Celiac
The good news first: Type 1 responds to treatment in roughly 75–80% of patients, and the goal is durable symptom remission plus mucosal healing.
Strict gluten-free eating continues — always. Even though the diet alone is not enough, stopping it makes everything worse. This is the foundation on which every other therapy is layered.
Corticosteroids are usually first-line. Two patterns:
- Oral prednisone at 20–40 mg daily, tapered over two to three months once symptoms settle. Effective but carries the usual steroid burden — bone loss, blood sugar rise, mood change, cataracts with long use.
- Budesonide 9 mg once daily, preferably the enteric-release, open-capsule formulation so the drug is delivered across the entire small bowel rather than releasing only in the terminal ileum. Budesonide has a much smaller systemic steroid footprint because 80–90% is cleared on first pass through the liver. Many celiac centers now start here rather than with prednisone.
Azathioprine (2–2.5 mg/kg/day) is the usual steroid-sparing agent for patients who relapse on taper or cannot come off budesonide. TPMT activity should be checked before starting to avoid profound myelosuppression. 6-mercaptopurine is a reasonable alternative.
With this approach, five-year survival in Type 1 is 80–96% across published series — close to, though not identical to, the general celiac population.
Treating Type 2 Refractory Celiac
Type 2 is genuinely hard. Nothing on the shelf reliably induces deep remission, and the threat of progression to lymphoma colors every decision.
The general strategy:
- Nutritional rescue first. Many patients arrive malnourished. Albumin under 3.0, severe weight loss, or refeeding risk calls for nutritional support — sometimes enteral tube feeding with an elemental formula, occasionally parenteral nutrition.
- Open-capsule budesonide 9 mg daily is still used for symptom control, though it does not clear the aberrant clone.
- Cladribine (2-chlorodeoxyadenosine) — a purine analog chemotherapy agent given in short IV cycles. Published series from the Amsterdam and Paris celiac centers show roughly half of treated Type 2 patients achieve clinical and histologic response, though the aberrant IEL clone often persists.
- Autologous hematopoietic stem-cell transplant (auto-HSCT) — reserved for Type 2 patients who fail cladribine. The Leiden group (Al-Toma and colleagues) has the longest experience. Patient-reported outcomes can be dramatic, but the treatment carries real transplant mortality risk and is offered only at a handful of centers worldwide.
- Emerging cell-targeted therapies — anti-IL-15 monoclonal antibodies (which target the cytokine driving aberrant IEL survival), JAK inhibitors, and CD30/CD52-directed agents are in clinical trials. None are standard of care yet, but enrollment in a specialty-center trial is often the best option a Type 2 patient has.
Five-year survival in Type 2 is reported between 35% and 60%, with the main driver being progression to EATL.
EATL — Enteropathy-Associated T-Cell Lymphoma
EATL is the feared endpoint. It is a rare but aggressive T-cell non-Hodgkin lymphoma that arises from the same aberrant intraepithelial lymphocyte population that defines Type 2 refractory disease.
How rare? About 1 case per 1,000 celiac patients per year in referral data — but the number is heavily concentrated in the Type 2 refractory subgroup. Within Type 2, roughly 40% progress to overt EATL within five years. For ordinary, diet-responsive celiac the lifetime risk is very low — reassurance, not alarm, is the right frame for most patients.
Presentation. Rapid weight loss, worsening abdominal pain, new palpable lymphadenopathy, a palpable abdominal mass, bowel obstruction, or acute perforation. Fevers and drenching night sweats (classic "B symptoms") may be present. LDH often rises. Any well-diagnosed celiac who takes a sudden turn for the worse — especially on a Type 2 trajectory — deserves urgent imaging and a surgical-oncology opinion.
Diagnosis is made on biopsy of the involved bowel, a surgical resection specimen, or a lymph node. PET-CT stages the disease. Bone marrow biopsy completes the workup.
Treatment combines surgical management of any obstruction or perforation, CHOP-like chemotherapy (cyclophosphamide, doxorubicin, vincristine, prednisone, sometimes with etoposide added as CHOEP), followed by consolidation autologous stem-cell transplant in patients fit enough to tolerate it. Brentuximab vedotin (anti-CD30) is used in CD30-expressing tumors.
Historically, five-year survival with EATL has hovered around 20%. Newer regimens incorporating auto-SCT in first remission have pushed that number higher in some series — the trend is genuinely encouraging but the disease remains one of the most difficult lymphomas to cure.
Prognosis — The Honest Numbers
Rounding the published literature to numbers patients can actually hold in their heads:
- Type 1 refractory celiac — 80 to 90% five-year survival. Most patients reach remission, and many live a normal lifespan. The main long-term burdens are steroid side effects and the modest background risk of progression.
- Type 2 refractory celiac — 35 to 60% five-year survival. The driver of mortality is progression to EATL, not the malabsorption itself. Catching the transition early is the single most useful thing surveillance can do.
- EATL — under 20% five-year survival historically, climbing with modern chemo-plus-auto-SCT regimens.
These numbers come from specialty-center cohorts, which are inherently enriched for the sickest patients. If you are reading this because you or someone you love just heard the words "refractory celiac," please remember — most patients fall into Type 1, most respond to treatment, and "refractory" does not mean "untreatable."
When and Where to Get a Second Opinion
Refractory celiac is a disease where experience matters enormously. The diagnostic tests (IEL flow cytometry, TCR rearrangement) are not universally available, and treatment choices (cladribine, auto-HSCT, trial enrollment) require centers that see many cases a year. In the U.S. and Europe, a short list of institutions has built dedicated celiac programs with all of these capabilities:
- Mayo Clinic (Rochester, MN) — Celiac Disease Clinic under the Division of Gastroenterology and Hepatology. The original refractory-celiac classification work by Rubio-Tapia and Murray came from this group.
- Columbia University Celiac Disease Center (New York, NY) — a major U.S. referral site for complex disease.
- University of Chicago Celiac Disease Center (Chicago, IL).
- Beth Israel Deaconess Celiac Center (Boston, MA).
- Sheffield Institute of Gluten-Related Disorders (UK).
- Amsterdam UMC celiac group (Netherlands) — the original home of cladribine and auto-HSCT protocols for Type 2.
- Hôpital Necker / Georges-Pompidou celiac center (Paris) — Malamut and Cellier's group, a foundational reference for the Type 2 phenotype.
Ask for a referral if: your local team does not offer IEL flow cytometry; a Type 2 diagnosis is being considered; you are about to start cladribine, azathioprine long-term, or auto-HSCT; or if PET-CT or small-bowel imaging shows anything worrisome. Second opinions at celiac-focused centers are routine and welcome — many accept email-based record reviews before you travel.
Monitoring Cadence
Once refractory disease is diagnosed and treatment is underway, surveillance has to be tight enough to catch trouble but not so heavy that it dominates your life.
- tTG-IgA (plus total IgA) every 3–6 months — not because it tracks the refractory disease itself, but because rising antibodies point to gluten exposure and reopen that whole question.
- Clinical visit every 3 months at a minimum during active treatment — weight, albumin, CBC, CMP, vitamin D, ferritin, B12, magnesium.
- Repeat endoscopy with biopsy + IEL flow cytometry at 6–12 month intervals while symptomatic, and whenever clinical status changes.
- Small-bowel imaging (MRE or capsule) annually in Type 2 or any patient with persistent atrophy.
- PET-CT whenever new B symptoms, a palpable mass, rising LDH, or unexplained weight loss appear — don't wait for the annual scan.
- Bone density (DEXA) every 1–2 years, especially on chronic steroids.
- Registered dietitian check-ins at least annually — contamination sneaks back in over time.
Living With Refractory Disease
Refractory celiac is hard in ways ordinary celiac is not. The diet you trusted to be the cure has not been enough. Immunosuppressive drugs carry their own side effects. The words "T-cell lymphoma" float at the edge of every appointment. None of that is fair, and pretending otherwise would insult your experience.
At the same time, three things are worth holding on to:
- Most people diagnosed with refractory celiac have Type 1, which is a treatable chronic condition rather than a cancer precursor.
- The landscape is moving. Anti-IL-15 antibodies, selective immune-cell depletors, and improved auto-SCT protocols have changed Type 2 outcomes in the last decade and look likely to keep doing so.
- Connecting with a specialty center early — even by telehealth — reliably improves both the diagnostic precision and the treatment options available to you. You do not have to navigate this in a community GI practice alone.
Practical things worth doing: ask for copies of every biopsy report and flow cytometry result so you can carry them to a second opinion; get the open-capsule budesonide formulation specifically (regular budesonide tablets release in the wrong place); keep a food-and-symptom diary for two weeks before each specialist visit so you can show patterns rather than describe them from memory; test with a GIP kit if you are confident you are strict but still sick — the result is genuinely useful either way.
Key Research Papers
- Rubio-Tapia A, Murray JA, et al. Classification and management of refractory celiac disease. Gastroenterology. 2010.
- Malamut G, Cellier C, et al. Refractory celiac disease: From bench to bedside. Gastroenterology. 2009.
- Al-Toma A, Mulder CJ, et al. Autologous hematopoietic stem cell transplantation in refractory celiac disease with aberrant T cells. Blood. 2007.
- Cellier C, Delabesse E, et al. Refractory sprue, coeliac disease, and enteropathy-associated T-cell lymphoma. Lancet. 2000.
Research Papers
For further reading, the following PubMed searches return the current peer-reviewed literature on refractory celiac disease and EATL:
- Refractory celiac disease classification and diagnosis
- Aberrant intraepithelial lymphocytes in RCD Type 2
- Open-capsule budesonide in refractory celiac
- Cladribine treatment in RCD Type 2
- Autologous HSCT in refractory celiac disease
- Enteropathy-associated T-cell lymphoma (EATL)
- Gluten immunogenic peptide testing (GIP, GlutenDetect)
- HLA DQ2.5 homozygosity and refractory disease risk
- Ulcerative jejunitis and celiac disease
- Anti-IL-15 therapies in celiac clinical trials
Connections
- Celiac Disease Overview
- Gluten-Free Diet: A Practical Guide
- Non-Celiac Gluten Sensitivity
- Celiac Serology Testing: tTG, DGP, EMA
- Associated Autoimmune Conditions
- Endoscopy, Biopsy, and Marsh Classification
- HLA-DQ2 and HLA-DQ8 Genetic Testing
- Nutritional Deficiencies and Bone Health
- Lymphoma
- Crohn's Disease
- Inflammatory Bowel Disease
- SIBO Breath Test Guide
- Anemia
- Vitamin D3
- Dermatitis Herpetiformis