Celiac Serology Testing: tTG, DGP, EMA, and the Traps
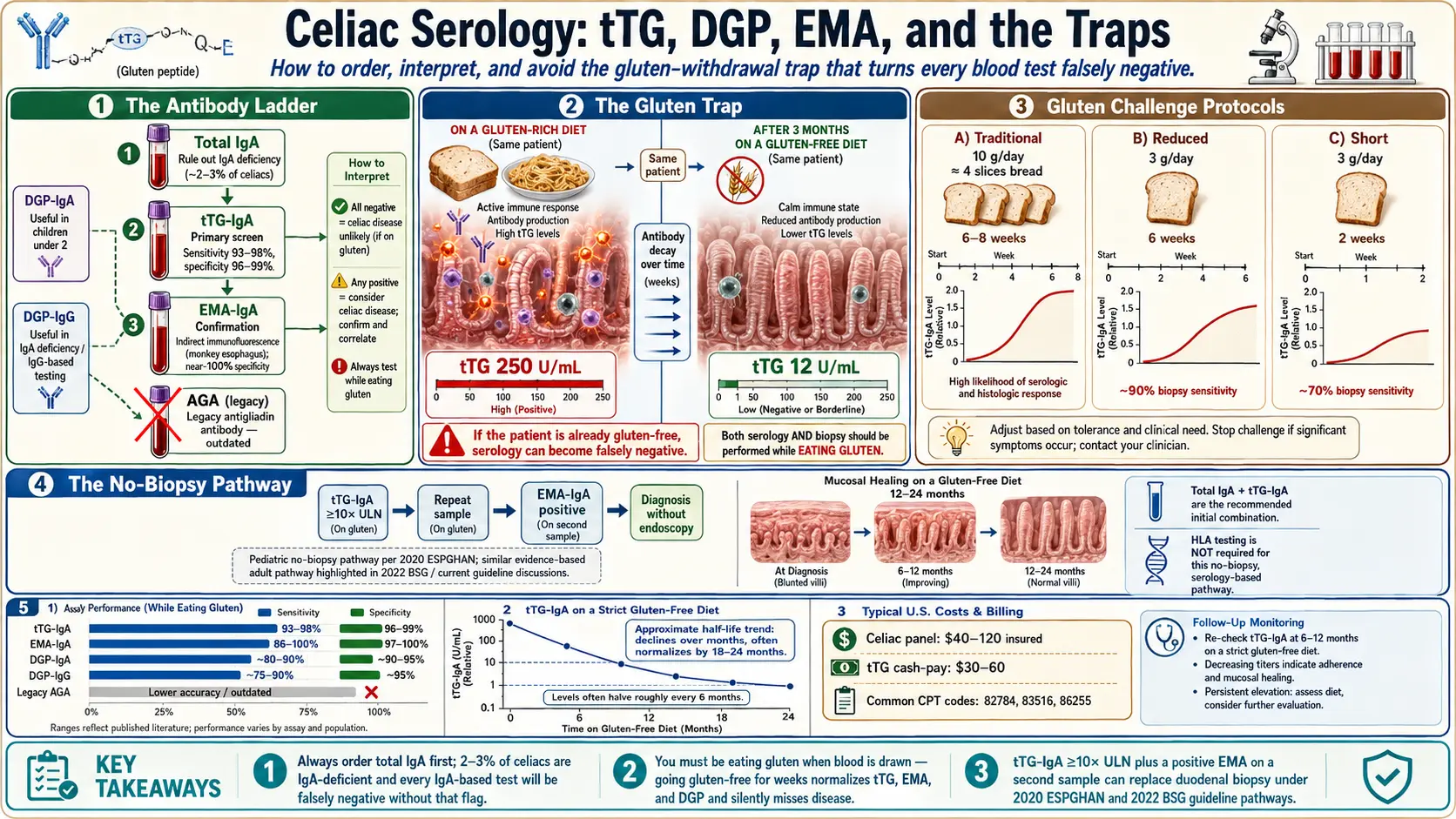
Table of Contents
- Why Getting the Order Right Matters
- Step One: Always Check Total IgA First
- tTG-IgA — The Primary Screen
- EMA-IgA — The Confirmatory Test
- DGP-IgA and DGP-IgG — The Specialists' Test
- Why the Old AGA Test Is Outdated
- Titer Magnitude: When Numbers Replace Biopsy
- The Critical Trap: You Must Be Eating Gluten
- Gluten Challenge Protocols
- Seronegative Celiac — When Blood Tests Miss It
- Post-Diagnosis: Using tTG to Track Healing
- Home and Mail-In Test Kits
- Insurance, Coding, and Cost
- Pediatric vs Adult Panels
- Ordering Pitfalls — What Labs Actually Run
- Why Your GI Won't Just "Test You"
- Key Research Papers
- Connections
- Featured Videos
Why Getting the Order Right Matters
Celiac disease is one of the most under-diagnosed autoimmune conditions in medicine. Roughly 1% of the population has it, but most walk around undiagnosed for years — some for decades. The reason is almost never that the blood tests are bad. The tests are excellent. The reason is that the tests get ordered wrong, run wrong, or interpreted wrong — and that the patient often is not eating gluten when the blood is drawn, which silently destroys every result on the panel.
This article is the map. It walks through every blood test used to screen for and confirm celiac disease, in the order a smart clinician (or a smart patient advocating for themselves) would use them. It flags the traps that cause false negatives, the rules that let you skip the biopsy in certain cases, and the insurance and lab-order details that decide whether your workup costs $40 or $400.
If you read one section, read The Critical Trap: You Must Be Eating Gluten. That single mistake — going gluten-free before testing — is the number-one reason people get told "you don't have celiac" when they actually do.
Step One: Always Check Total IgA First
Before you order any celiac-specific antibody, a competent workup starts with a total serum IgA level. Here is why.
Most celiac antibodies are IgA class antibodies — they belong to the branch of the immune system that patrols mucosal surfaces, including the lining of the gut. That is a design feature: gluten hits the gut first, so the gut's immune system responds first, and the antibodies it makes are mostly IgA. Unfortunately, 2–3% of people with celiac disease are selective IgA deficient, compared to roughly 0.2% of the general population. If you are in that group, your body simply cannot produce meaningful quantities of IgA, and every IgA-based celiac test will come back falsely negative no matter how sick your gut is.
A total IgA measurement costs around $10–$25 and takes a single extra tube of blood. If it comes back low (typically under 7 mg/dL for adults, lower cutoffs for kids), your clinician switches to IgG-based celiac tests: tTG-IgG and DGP-IgG. If it is normal, you proceed with the standard IgA panel. Skipping this step is the second most common mistake in celiac testing, after drawing blood off-gluten.
tTG-IgA — The Primary Screen
Tissue transglutaminase IgA (tTG-IgA) is the single most important celiac test. It is the first blood test every major guideline body — the American College of Gastroenterology, the European Society for Paediatric Gastroenterology Hepatology and Nutrition (ESPGHAN), the British Society of Gastroenterology — recommends for anyone with suspected celiac disease. If you are going to order one test, this is it.
What it measures: tissue transglutaminase is the enzyme inside your intestinal lining that modifies gluten peptides after you eat them. In celiac disease, the immune system mistakenly starts producing antibodies against this enzyme. Those antibodies circulate in blood and are easy to detect with an ELISA assay.
The performance is remarkable. In adults eating a normal gluten-containing diet, tTG-IgA has a sensitivity of 93–98% (it catches most cases) and a specificity of 96–99% (it rarely lights up when celiac is absent). No other single gastrointestinal antibody test comes close.
A few caveats you should know:
- Young children under 2 sometimes mount a weaker tTG response. Pediatric panels therefore add DGP (see below).
- Mild false positives (usually 1–3× upper limit) can occur in chronic liver disease, heart failure, type 1 diabetes, and other autoimmune conditions — a reason EMA or biopsy is used to confirm.
- The test is IgA-based, so selective IgA deficiency will falsely lower it. Always pair with a total IgA.
EMA-IgA — The Confirmatory Test
Endomysial antibody IgA (EMA-IgA) is celiac serology's gold standard for specificity. It is reported as a titer on a dilution series (1:5, 1:10, 1:20, etc.) and uses an old-school immunofluorescence assay, where a technician looks through a microscope at monkey esophagus or human umbilical cord tissue and scores the pattern of antibody binding. It is labor-intensive and operator-dependent, which is why it costs more (roughly $100–$200) and is not the first-line screen.
What it buys you is near-perfect specificity — essentially 100% in experienced labs. A positive EMA-IgA in a symptomatic patient, with a positive tTG-IgA, is so specific for celiac that under the 2020 ESPGHAN pediatric guidelines and the 2022 BSG adult guidelines it can replace the duodenal biopsy in carefully selected cases (see Titer Magnitude).
EMA is not used in isolation. The standard workflow is: screen with tTG-IgA, and if positive (especially if borderline), confirm with EMA-IgA on the same sample or a repeat draw.
DGP-IgA and DGP-IgG — The Specialists' Test
Deamidated gliadin peptide (DGP) antibodies target a chemically modified form of gliadin — the fragment of gluten that the enzyme tissue transglutaminase has "deamidated" inside the gut wall. Think of it as the crime scene residue: if your immune system has been reacting to gluten as processed by celiac gut chemistry, you will usually have anti-DGP antibodies in your blood.
DGP has two main uses:
- Children under 2 years old. Their tTG response can be sluggish; DGP-IgG catches cases that tTG-IgA misses.
- IgA-deficient patients. DGP-IgG is one of the two go-to tests (along with tTG-IgG) when IgA is unavailable.
- Suspected seronegative celiac. When a patient's symptoms scream celiac but tTG-IgA is negative, adding DGP-IgA/IgG occasionally catches a case that would otherwise go to biopsy without a positive antibody.
DGP is not routinely better than tTG for the average adult screen. If your lab offers a "celiac panel" that runs tTG-IgA, EMA-IgA, total IgA, and DGP-IgG all at once, that is fine — comprehensive. If your insurer only pays for one test, tTG-IgA is still the right pick.
Why the Old AGA Test Is Outdated
Older charts sometimes still list anti-gliadin antibody (AGA) — the original celiac blood test from the 1980s. Do not order it. AGA targets native (unprocessed) gliadin and lights up in a wide range of conditions, including non-celiac gluten sensitivity, wheat allergy, Crohn's disease, and healthy wheat eaters with no gut disease at all. Its specificity is poor enough that modern guidelines explicitly recommend against it.
If a provider orders "AGA" or "anti-gliadin IgG/IgA" as part of your celiac workup in 2026, politely ask whether they meant DGP (the modern replacement). Many electronic health records still carry both order sets and the defaults drift.
Titer Magnitude: When Numbers Replace Biopsy
For decades, a duodenal biopsy was mandatory to confirm celiac disease. Then in 2012, ESPGHAN changed pediatric practice: children with a tTG-IgA at or above 10 times the upper limit of normal (≥10× ULN), confirmed by a positive EMA on a second blood draw, plus HLA-DQ2 or DQ8 positivity, could be diagnosed without endoscopy. The 2020 ESPGHAN update dropped the HLA requirement, further simplifying the path.
In 2022, the British Society of Gastroenterology extended a similar option to adults: in carefully selected symptomatic adults with tTG-IgA ≥10× ULN and a positive EMA on a second sample, biopsy is no longer mandatory — though the U.S. American College of Gastroenterology still recommends biopsy for adults in most cases.
Practical takeaway: if your tTG-IgA comes back at, say, 250 U/mL on an assay whose normal is under 20 U/mL, that is a 12× ULN result. Ask your GI whether the biopsy-optional pathway applies in your region. Many countries, including the UK, the Netherlands, and parts of Scandinavia, now routinely skip scopes in high-titer adults. In the U.S., biopsy remains the default but a strong case can be made.
The Critical Trap: You Must Be Eating Gluten
This is the most important paragraph in this article. Every celiac blood test depends on an active immune reaction to gluten. If you stop eating gluten, your tTG-IgA, EMA, and DGP levels begin to fall within weeks, and they will often normalize within three to six months. At that point, the tests cannot distinguish you from a healthy person. A GI who runs celiac serology on a patient who has been gluten-free for months is running a test that is guaranteed to be negative regardless of whether the underlying disease is there.
This happens all the time. A patient feels better after going gluten-free on their own, later asks their doctor "could I have celiac?" and gets blood drawn that same week. The result comes back negative and the patient is told they just have "gluten sensitivity." Months or years later, symptoms return, damage accumulates in the small intestine, and the real diagnosis is missed — sometimes until a vitamin deficiency or osteoporosis forces the question open again.
Rule: do not go gluten-free until celiac has been ruled in or out on an adequate diet. If you are already gluten-free when you decide to get tested, you have two choices: accept that your blood work will be uninformative, or do a gluten challenge to reactivate the antibody response.
Gluten Challenge Protocols
A gluten challenge means eating gluten deliberately, at a meaningful dose, for a defined period so that celiac serology can detect the disease if it is present. The traditional protocol, validated in research settings and still used by most U.S. academic centers:
- Dose: roughly 10 grams of gluten per day — the equivalent of about 4 slices of normal wheat bread, or 2 slices plus a bowl of pasta, daily.
- Duration: 6 to 8 weeks before blood is drawn. Some protocols go to 12 weeks if serology is still negative but suspicion is high.
- End with blood work, and a biopsy if serology is positive.
That is a brutal protocol for someone whose symptoms go away on a gluten-free diet. More recent studies have looked at whether shorter, lower-dose challenges can still trigger detectable antibody and histologic changes. Two reduced protocols now appear in the literature:
- 3 g/day (about one slice of bread) for 2 weeks — catches roughly 70% of cases on biopsy; lower sensitivity on serology.
- 3 g/day for 6 weeks — catches closer to 90% of cases on biopsy; still not a perfect substitute for the full 10g/8-week protocol for serology.
If you cannot tolerate a full challenge (some patients get so sick they abandon it), a short challenge followed by both serology and biopsy is still better than nothing — and HLA genetic testing can help: if you are negative for HLA-DQ2 and DQ8, celiac disease is essentially ruled out without any gluten at all (see the HLA genetic testing article).
Seronegative Celiac — When Blood Tests Miss It
Roughly 5–10% of adults with biopsy-proven celiac disease have negative or borderline serology despite eating gluten. The Marsh lesions are there. The villi are blunted. The tTG is stubbornly under the cutoff.
Why this happens is not fully understood. Some patients have very localized, low-grade disease. Some have mild IgA deficiency that falls inside the "normal" range but low enough to suppress tTG-IgA. Some have partially treated celiac from inadvertently reduced-gluten diets before testing.
The practical implication: a negative blood test in a high-suspicion patient is not the end of the workup. If you have iron-deficiency anemia that will not respond to oral iron, unexplained elevated liver enzymes, a first-degree relative with celiac, type 1 diabetes, Hashimoto's thyroiditis, osteoporosis before age 50, or dermatitis herpetiformis, a referral for endoscopic biopsy is appropriate regardless of blood results. See the biopsy and Marsh classification article for what the procedure finds.
Post-Diagnosis: Using tTG to Track Healing
Once you are diagnosed and start a strict gluten-free diet, tTG-IgA becomes one of the cheapest and most useful follow-up tools in medicine. Antibody levels should roughly halve every six months and normalize within 12 to 24 months in most patients on a truly gluten-free diet.
What that means in practice:
- If your tTG was 250 at diagnosis and is 15 (normal) eighteen months later, your diet is working and your gut is healing.
- If your tTG was 250 at diagnosis and is still 180 a year later, you are getting gluten from somewhere — cross-contamination at home, hidden gluten in medications or supplements, eating out mistakes. This is a diet-detective problem, not a treatment-failure problem.
- If tTG normalizes and then rises again, you have slipped. Common culprits: new medication with wheat starch, switching to a restaurant that has stopped being careful, a new partner or roommate cross-contaminating the kitchen.
Most guidelines recommend tTG-IgA retesting at 6 months, 12 months, and annually thereafter. It does not replace a follow-up endoscopy in complicated cases — villous atrophy can persist even when antibodies normalize — but it catches the majority of dietary slips cheaply.
Home and Mail-In Test Kits
Several direct-to-consumer companies now sell celiac serology by mail: imaware, Everlywell, and a handful of others. You order online, receive a finger-prick or mail-in venous kit, and get results back in one to two weeks. Typical price: $70–$150.
Caveats you should understand before using one:
- They use real tTG assays, usually the same ELISA kits hospitals use. The science is legitimate.
- Most do not include a total IgA. If you are IgA-deficient, the test will be falsely negative and the company will tell you "you're fine."
- None can interpret your diet. If you were on a reduced-gluten diet when you tested, the result is uninformative regardless of assay quality.
- A positive mail-in result still requires a real GI referral for EMA confirmation and biopsy planning. Insurance will usually re-run the panel.
- A negative mail-in result in a symptomatic patient is not reassurance. See seronegative celiac.
Home kits are most useful as a screening nudge — "am I in the neighborhood?" — for people whose insurance hassles them about ordering the lab traditionally. They are not a substitute for a proper workup.
Insurance, Coding, and Cost
In the United States, a standard celiac panel (total IgA + tTG-IgA, sometimes with EMA reflex) runs about $40–$120 at LabCorp or Quest on a commercial insurance plan, and commonly costs nothing out-of-pocket when ordered with a documented clinical indication. Relevant CPT codes include 82784 (total IgA), 83516 or 86364 (tTG-IgA), 86255/86256 (EMA titer), and 83516 (DGP variants).
If your insurer pushes back, the magic words in the order are "chronic diarrhea," "iron deficiency anemia," "unexplained weight loss," "family history of celiac disease," or "autoimmune thyroid disease" — all recognized indications that meet medical-necessity criteria.
Without insurance, a cash-pay tTG at a direct-lab service like Ulta Lab Tests or Walk-In Lab runs about $30–$60 — often cheaper than a home kit and including a real draw. This is a reasonable path for uninsured patients.
Pediatric vs Adult Panels
Pediatric and adult panels overlap but are not identical.
- Under 2 years old: tTG-IgA alone is unreliable. The standard workup is tTG-IgA + DGP-IgG + total IgA. If still equivocal, proceed to biopsy.
- Ages 2–18: tTG-IgA + total IgA is the primary screen. Positive results trigger EMA-IgA confirmation. Under 2020 ESPGHAN rules, tTG-IgA ≥10× ULN with positive EMA on a second sample can skip the biopsy entirely.
- Adults: tTG-IgA + total IgA is the screen. In the U.S., a positive result virtually always triggers endoscopy. In the UK and parts of Europe, high-titer cases may skip biopsy under 2022 BSG guidance.
Pediatric GI practices tend to be more aggressive about testing first-degree relatives, particularly siblings, because early detection changes growth trajectory and bone density outcomes. Parents of a diagnosed child should expect their other children to be offered testing over the next several years.
Ordering Pitfalls — What Labs Actually Run
A "celiac panel" means different things at different labs. Common variations you may see:
- Minimal panel: tTG-IgA only. Cheap, but misses IgA-deficient patients.
- Standard panel: tTG-IgA + total IgA. This is the bare-minimum competent order.
- Comprehensive panel: tTG-IgA + tTG-IgG + DGP-IgA + DGP-IgG + total IgA + EMA-IgA reflex. This catches essentially everything. Often the right order for a patient with strong suspicion and a history of intermittent gluten avoidance.
- Legacy panels that still include AGA — skip the AGA interpretation.
When you see your GI or primary care physician, ask explicitly: "Are you ordering tTG-IgA, total IgA, and DGP-IgG? And EMA if tTG is positive?" That five-second clarification prevents 90% of mis-ordered panels.
Be aware that some electronic health record systems have a default "Celiac Screen" order that includes only tTG-IgA, no total IgA, and no DGP. That is not wrong, but it is not ideal — especially for a first workup.
Why Your GI Won't Just "Test You"
Patients frequently walk into a GI office saying "I've been gluten-free for a year and I want to know if I have celiac. Can you just test me?" The answer, almost always, is no — not usefully. Here is what the GI hears and why they push back:
- Your tTG will be normal whether you have celiac or not. The test has nothing to react to because you are not eating the trigger.
- A duodenal biopsy on a long-term gluten-free patient often shows a normal or near-normal villous architecture, even in real celiac disease. The gut heals. Biopsy without a gluten challenge is not a reliable diagnostic either.
- HLA-DQ2/DQ8 genetic testing can be run off-gluten. A negative HLA essentially rules celiac out. A positive HLA is consistent with celiac but also with 30–40% of the general population — it does not diagnose anyone.
The realistic options, if you are already gluten-free and want a definitive answer:
- HLA-DQ2/DQ8 testing first. If negative, celiac is off the table and you can stay gluten-free for symptomatic reasons without medical surveillance. See the HLA article.
- Gluten challenge + serology + biopsy. The gold-standard path if HLA is positive and you want certainty. Plan for 6–8 weeks of feeling terrible.
- Accept uncertainty. If symptoms are controlled and you have no red-flag features (anemia, osteoporosis, family history, failure to thrive, associated autoimmune disease), some patients reasonably choose to remain gluten-free without a formal diagnosis. The trade-off is that you will not qualify for celiac-specific services, some insurance accommodations, or post-diagnosis monitoring.
A good GI will lay out all three paths and let you choose. If yours refuses to test you but will not explain the reasoning, bring this article to the next appointment.
Key Research Papers
Peer-reviewed work on celiac antibody assay performance, the no-biopsy pathway, IgA-deficiency workarounds, HLA-DQ2/DQ8 typing, and the histopathology that serology is meant to predict.
- Rubio-Tapia A, Hill ID, Kelly CP, Calderwood AH, Murray JA. ACG Clinical Guidelines: Diagnosis and Management of Celiac Disease. American Journal of Gastroenterology. 2013;108(5):656-676.
- Husby S, Koletzko S, Korponay-Szabó IR, et al. ESPGHAN Guidelines for the Diagnosis of Coeliac Disease (2020 update). Journal of Pediatric Gastroenterology and Nutrition. 2020;70(1):141-156.
- Lebwohl B, Sanders DS, Green PHR. Coeliac Disease. Lancet. 2018;391(10115):70-81.
- Singh P, Arora A, Strand TA, et al. Global Prevalence of Celiac Disease: Systematic Review and Meta-Analysis. Clinical Gastroenterology and Hepatology. 2018;16(6):823-836.e2.
- Werkstetter KJ, Korponay-Szabó IR, Popp A, et al. Accuracy in Diagnosis of Celiac Disease without Biopsies in Clinical Practice. Gastroenterology. 2017;153(4):924-935.
- Tonutti E, Bizzaro N. Diagnosis and Classification of Celiac Disease and Gluten Sensitivity. Autoimmunity Reviews. 2014;13(4-5):472-476.
- Lewis NR, Scott BB. Meta-Analysis: Deamidated Gliadin Peptide Antibody and Tissue Transglutaminase Antibody Compared as Screening Tests for Coeliac Disease. Alimentary Pharmacology & Therapeutics. 2010;31(1):73-81.
- Sblattero D, Berti I, Trevisiol C, et al. Human Recombinant Tissue Transglutaminase ELISA: An Innovative Diagnostic Assay for Celiac Disease. American Journal of Gastroenterology. 2000;95(5):1253-1257.
- Volta U, Granito A, Parisi C, et al. Deamidated Gliadin Peptide Antibodies as a Routine Test for Celiac Disease: A Prospective Analysis. Journal of Clinical Gastroenterology. 2010;44(3):186-190.
- Megiorni F, Pizzuti A. HLA-DQA1 and HLA-DQB1 in Celiac Disease Predisposition: Practical Implications of the HLA Molecular Typing. Journal of Biomedical Science. 2012;19(1):88.
- Marsh MN. Gluten, Major Histocompatibility Complex, and the Small Intestine: A Molecular and Immunobiologic Approach to the Spectrum of Gluten Sensitivity. Gastroenterology. 1992;102(1):330-354.
- Oberhuber G, Granditsch G, Vogelsang H. The Histopathology of Coeliac Disease: Time for a Standardized Report Scheme for Pathologists. European Journal of Gastroenterology & Hepatology. 1999;11(10):1185-1194.
- Schyum AC, Rumessen JJ. Serological Testing for Celiac Disease in Adults. United European Gastroenterology Journal. 2013;1(5):319-325.
- Caio G, Volta U, Sapone A, et al. Celiac Disease: A Comprehensive Current Review. BMC Medicine. 2019;17(1):142.
Live PubMed Searches
Live PubMed queries that update as new papers are indexed.
- tTG-IgA sensitivity and specificity for celiac disease
- Endomysial antibody testing in celiac disease
- Deamidated gliadin peptide (DGP) antibodies
- IgA deficiency and celiac disease screening
- Gluten challenge protocols and short-course challenges
- Seronegative celiac disease
- tTG follow-up and gluten-free diet adherence
- ESPGHAN no-biopsy pathway and high-titer tTG
Connections
- Celiac Disease Overview
- Gluten-Free Diet: A Practical Guide
- Endoscopy, Biopsy, and Marsh Classification
- HLA-DQ2 and HLA-DQ8 Genetic Testing
- Nutritional Deficiencies and Bone Health
- Non-Celiac Gluten Sensitivity
- Associated Autoimmune Conditions
- Refractory Celiac Disease and EATL
- Hashimoto's Thyroiditis
- TPO and TgAb Antibodies Explained
- Irritable Bowel Syndrome
- SIBO
- Homocysteine Lab Test
- Anemia
- Dermatitis Herpetiformis