Iron – Essential Mineral for Human Health
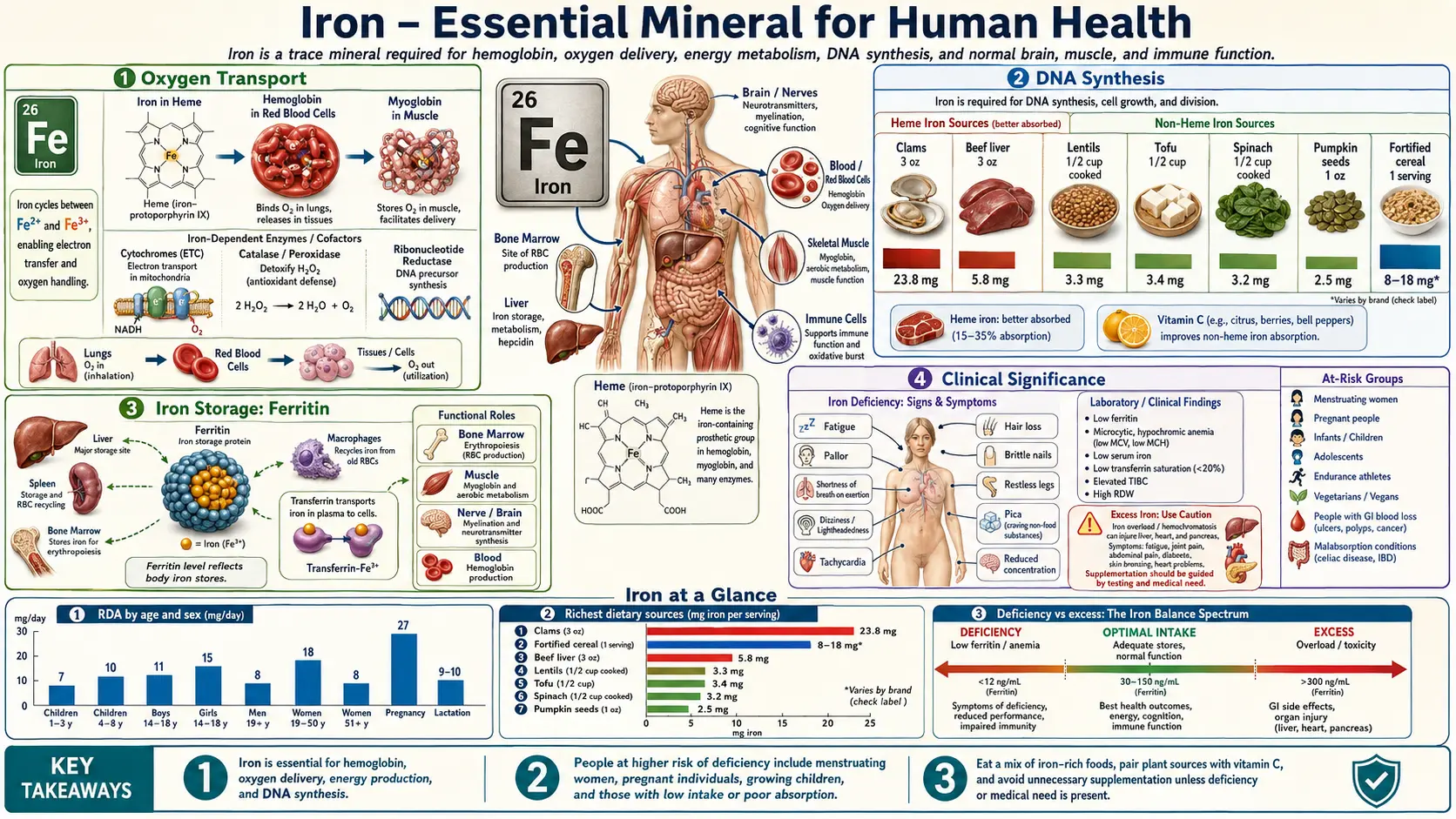
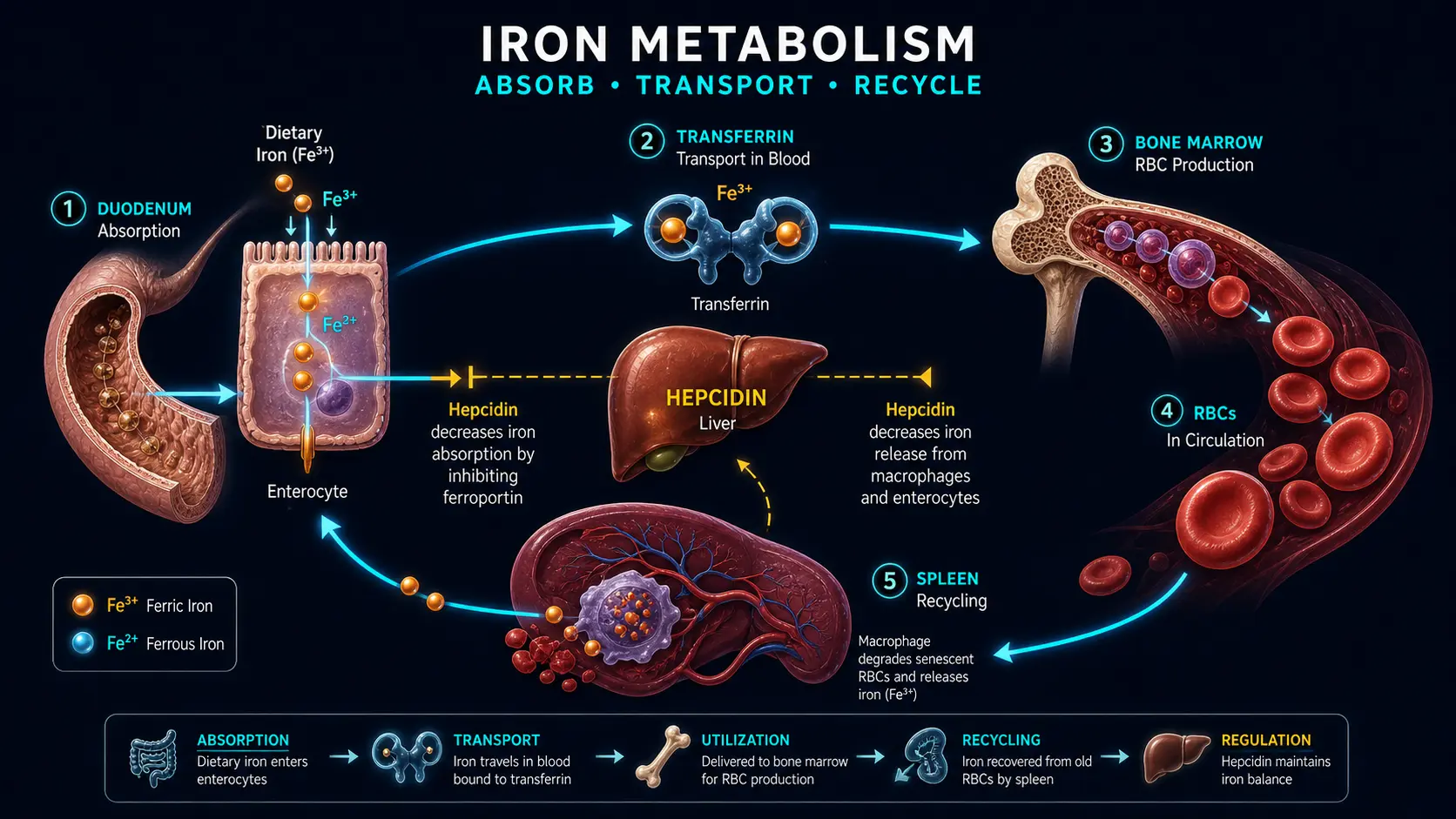
Table of Contents
- Oxygen Transport
- Energy Production
- Enzymatic Functions
- DNA Synthesis
- Immune Function
- Cognitive Development and Neurotransmitter Synthesis
- Iron Storage: Ferritin
- Iron Transport: Transferrin
- Heme Iron vs Non-Heme Iron
- Clinical Significance
- Connections
- Featured Videos
Iron (Fe) is a transition metal and one of the most critical trace minerals in human physiology. The adult human body contains approximately 3 to 5 grams of iron, the majority of which is incorporated into functional proteins. Iron's ability to readily cycle between its ferrous (Fe2+) and ferric (Fe3+) oxidation states makes it indispensable for electron transfer reactions, oxygen binding, and a wide array of enzymatic processes. Because the body lacks a regulated excretory pathway for iron, homeostasis is maintained primarily through the precise control of intestinal absorption.
Oxygen Transport
Hemoglobin
Approximately 65 to 70 percent of total body iron resides in hemoglobin, the oxygen-carrying protein within red blood cells. Each hemoglobin molecule is a tetramer composed of four globin subunits, each containing a heme prosthetic group with a centrally coordinated iron atom in the ferrous (Fe2+) state. This iron atom reversibly binds molecular oxygen in the lungs and releases it in peripheral tissues according to their metabolic demands. The cooperative binding behavior of hemoglobin, described by the oxygen-hemoglobin dissociation curve, allows efficient oxygen loading at high partial pressures in the pulmonary capillaries and unloading at lower partial pressures in metabolically active tissues.
- Normal hemoglobin concentration: 12 to 16 g/dL in adult women; 14 to 18 g/dL in adult men.
- Oxygen-carrying capacity: Each gram of hemoglobin can bind approximately 1.34 mL of oxygen (Huffner's constant).
- Bohr effect: Increased carbon dioxide and decreased pH in metabolically active tissues shift the dissociation curve to the right, promoting oxygen release where it is most needed.
Myoglobin
Myoglobin is a monomeric heme protein found in skeletal and cardiac muscle tissue, accounting for roughly 10 percent of total body iron. Unlike hemoglobin, myoglobin has a hyperbolic oxygen-binding curve, giving it a higher affinity for oxygen at low partial pressures. This property makes myoglobin well suited for accepting oxygen from hemoglobin at the tissue level and storing it within muscle cells for use during periods of intense metabolic activity or transient ischemia.
- Oxygen reservoir: Myoglobin serves as an intracellular oxygen buffer, releasing stored oxygen during muscle contraction when blood flow is temporarily occluded.
- Cardiac significance: Elevated serum myoglobin is an early but non-specific marker of myocardial injury, rising within 1 to 3 hours of acute damage.
Energy Production
Cytochromes and the Electron Transport Chain
Iron is a central component of the mitochondrial electron transport chain (ETC), where it participates in the sequential transfer of electrons from NADH and FADH2 to molecular oxygen, driving the generation of ATP through oxidative phosphorylation. Iron is present in two forms within the ETC:
- Heme-containing cytochromes: Cytochromes b, c1, c, a, and a3 contain iron within their heme prosthetic groups. These proteins shuttle electrons between Complexes III and IV of the respiratory chain. Cytochrome c oxidase (Complex IV) catalyzes the final transfer of electrons to oxygen, reducing it to water.
- Iron-sulfur (Fe-S) clusters: Found in Complexes I, II, and III, iron-sulfur proteins contain iron atoms coordinated with inorganic sulfur. These clusters function as one-electron carriers, facilitating the stepwise transfer of electrons and contributing to proton translocation across the inner mitochondrial membrane.
Iron deficiency impairs mitochondrial function, reducing cellular ATP output and contributing to the fatigue and exercise intolerance characteristic of iron-depleted states, even before overt anemia develops.
Citric Acid Cycle Enzymes
The enzyme aconitase, which catalyzes the interconversion of citrate and isocitrate in the tricarboxylic acid (TCA) cycle, requires an iron-sulfur cluster ([4Fe-4S]) for its catalytic activity. Loss of iron from this cluster inactivates the enzyme and impairs the flow of metabolites through the cycle, further diminishing energy production.
Enzymatic Functions
Beyond oxygen transport and energy metabolism, iron serves as a cofactor or structural component in a diverse array of enzymes:
- Catalase: A heme-containing enzyme that decomposes hydrogen peroxide (H2O2) into water and oxygen, protecting cells from oxidative damage.
- Peroxidases: Including myeloperoxidase in neutrophils, which generates hypochlorous acid as part of the innate antimicrobial defense system.
- Ribonucleotide reductase: A di-iron enzyme essential for the conversion of ribonucleotides to deoxyribonucleotides, a rate-limiting step in DNA synthesis (see below).
- Prolyl and lysyl hydroxylases: Iron-dependent enzymes required for collagen biosynthesis, catalyzing the hydroxylation of proline and lysine residues necessary for proper collagen cross-linking and structural integrity.
- Cytochrome P450 enzymes: A large superfamily of heme-containing monooxygenases involved in drug metabolism, steroid hormone biosynthesis, bile acid formation, and the detoxification of xenobiotics in the liver.
- Tryptophan hydroxylase and phenylalanine hydroxylase: Non-heme iron-dependent enzymes involved in the synthesis of serotonin and tyrosine, respectively.
DNA Synthesis
Iron plays an indispensable role in DNA replication and repair through the enzyme ribonucleotide reductase (RNR). This enzyme catalyzes the reduction of all four ribonucleoside diphosphates to their corresponding deoxyribonucleoside diphosphates, providing the building blocks required for DNA synthesis. The active site of class I RNR contains a di-iron center that generates a stable tyrosyl radical essential for catalysis. Iron depletion reduces RNR activity, slowing DNA replication and contributing to the impaired cell proliferation seen in iron deficiency states, particularly in rapidly dividing cells such as hematopoietic precursors and intestinal epithelium.
- Cell cycle effects: Severe iron depletion causes cell cycle arrest at the G1/S transition, reflecting insufficient deoxyribonucleotide pools for DNA synthesis.
- DNA repair: Several DNA repair enzymes, including certain glycosylases and helicases, contain iron-sulfur clusters that are essential for their function in maintaining genomic integrity.
Immune Function
Iron is required for the proper function of both the innate and adaptive arms of the immune system. However, the relationship between iron and immunity is complex because iron is also an essential nutrient for most pathogenic microorganisms.
- Neutrophil bactericidal activity: Myeloperoxidase, a heme enzyme in neutrophil granules, generates reactive oxygen species that kill ingested bacteria. Iron deficiency impairs this oxidative burst capacity.
- Lymphocyte proliferation: T-cell and B-cell expansion depends on iron-requiring enzymes such as ribonucleotide reductase. Iron deficiency has been associated with reduced T-helper cell numbers and impaired cell-mediated immunity.
- Nutritional immunity: The host sequesters iron from invading pathogens by reducing serum iron levels through hepcidin-mediated mechanisms, upregulating lactoferrin in mucosal secretions, and increasing ferritin synthesis to lock iron in storage. This strategy, known as nutritional immunity, limits microbial access to this essential growth factor.
- Iron overload and infection risk: Conversely, excess free iron can promote bacterial growth. Conditions such as hereditary hemochromatosis or frequent transfusions are associated with increased susceptibility to infections with siderophilic organisms such as Vibrio vulnificus, Yersinia enterocolitica, and Listeria monocytogenes.
Cognitive Development and Neurotransmitter Synthesis
Brain Iron and Neurodevelopment
The developing brain has a particularly high demand for iron. Iron is required for myelination of neuronal axons, synaptogenesis, and the synthesis of monoamine neurotransmitters. The period of greatest vulnerability extends from the third trimester of pregnancy through the first two years of life, during which iron deficiency can produce lasting neurocognitive deficits that may not be fully reversible with later iron repletion.
- Myelination: Oligodendrocytes, the myelin-producing cells of the central nervous system, are among the most iron-rich cells in the brain. Iron is required for cholesterol and lipid biosynthesis, both of which are essential for myelin membrane assembly.
- Hippocampal development: Iron deficiency during critical developmental windows has been shown to alter hippocampal structure and function, resulting in impaired learning and memory.
Neurotransmitter Synthesis
Iron serves as a cofactor for key enzymes in the biosynthesis of monoamine neurotransmitters:
- Tyrosine hydroxylase: Catalyzes the rate-limiting step in the synthesis of dopamine and norepinephrine, converting tyrosine to L-DOPA. This enzyme requires ferrous iron and tetrahydrobiopterin as cofactors.
- Tryptophan hydroxylase: Catalyzes the rate-limiting step in serotonin synthesis, converting tryptophan to 5-hydroxytryptophan. Also iron-dependent.
- Monoamine oxidase: An iron-containing flavoprotein that degrades dopamine, serotonin, and norepinephrine. Iron is bound within the FAD cofactor environment of this enzyme.
Disruption of these pathways may underlie the behavioral symptoms of iron deficiency, including irritability, poor attention, and impaired cognitive performance observed in both children and adults.
Iron Storage: Ferritin
Ferritin is the principal intracellular iron storage protein, found in virtually all cell types but most abundantly in hepatocytes, macrophages of the reticuloendothelial system, and bone marrow. Each ferritin molecule is a spherical shell composed of 24 subunits (a mixture of H and L chains) that can sequester up to 4,500 atoms of iron in a safe, soluble, and bioavailable form as a ferric oxyhydroxide mineral core.
- H-subunit (heavy chain): Possesses ferroxidase activity, catalyzing the oxidation of Fe2+ to Fe3+ for incorporation into the mineral core. Predominates in tissues with high iron utilization such as the heart and brain.
- L-subunit (light chain): Facilitates iron nucleation and long-term storage. Predominates in tissues with primary storage functions such as the liver and spleen.
- Serum ferritin: A small amount of ferritin is released into the bloodstream, and serum ferritin concentration serves as the most sensitive and specific single laboratory marker of total body iron stores. Values below 15 ng/mL are diagnostic of iron deficiency in most clinical contexts, while values above 200 ng/mL in women or 300 ng/mL in men may suggest iron overload.
- Hemosiderin: When iron stores are high, ferritin molecules aggregate and undergo partial lysosomal degradation to form hemosiderin, a water-insoluble iron complex visible on tissue biopsy with Prussian blue staining.
Iron Transport: Transferrin
Transferrin is a glycoprotein synthesized primarily by the liver that serves as the principal plasma iron transport protein. Each transferrin molecule has two high-affinity binding sites for ferric iron (Fe3+), and under normal conditions approximately 20 to 45 percent of these sites are occupied (this ratio is the transferrin saturation).
- Transferrin receptor 1 (TfR1): Expressed on the surface of most cells, with particularly high density on erythroid precursors in the bone marrow. The transferrin-TfR1 complex is internalized by receptor-mediated endocytosis. Acidification of the endosome releases iron from transferrin, and the apoferrin-TfR1 complex is recycled to the cell surface.
- Transferrin receptor 2 (TfR2): Expressed predominantly in hepatocytes, where it acts as an iron sensor that modulates hepcidin expression in response to circulating transferrin saturation levels.
- Soluble transferrin receptor (sTfR): A truncated form of TfR1 released into plasma, the concentration of which reflects total erythropoietic activity and tissue iron demand. Elevated sTfR is a useful marker of iron deficiency, particularly in the setting of concurrent inflammation where ferritin levels may be falsely elevated.
- Hepcidin: The master regulator of systemic iron homeostasis, hepcidin is a peptide hormone produced by the liver that controls iron absorption and recycling by binding to and inducing the degradation of ferroportin, the sole known cellular iron exporter. Hepcidin production is increased by iron loading and inflammation (via IL-6), and decreased by iron deficiency, erythropoietic drive, and hypoxia.
Heme Iron vs Non-Heme Iron
Dietary iron exists in two chemical forms that differ markedly in their absorption mechanisms and bioavailability:
- Heme iron: Derived from hemoglobin and myoglobin in animal-source foods (red meat, poultry, fish). Heme iron is absorbed intact through a dedicated transporter (HCP1/PCFT) on the apical surface of duodenal enterocytes and is largely unaffected by other dietary components. Absorption rates range from 15 to 35 percent.
- Non-heme iron: Found in plant foods, dairy, eggs, and iron-fortified products. Non-heme iron must be reduced from Fe3+ to Fe2+ by duodenal cytochrome b (Dcytb) before transport via the divalent metal transporter 1 (DMT1). Absorption rates range from 2 to 20 percent and are strongly influenced by enhancers (ascorbic acid, meat factor) and inhibitors (phytates, polyphenols, calcium).
For a detailed comparison of these two forms, see Heme vs Non-Heme Iron.
Clinical Significance
Iron Deficiency
Iron deficiency is the most prevalent nutritional deficiency worldwide, affecting an estimated 2 billion people. It progresses through three stages: depletion of iron stores (decreased ferritin), iron-deficient erythropoiesis (decreased transferrin saturation, elevated sTfR), and iron deficiency anemia (decreased hemoglobin with microcytic, hypochromic red blood cells). Clinical consequences extend beyond anemia and include fatigue, impaired work capacity, cognitive dysfunction, restless legs syndrome, pica, and compromised immune function.
For a comprehensive discussion of iron deficiency anemia, see Iron Deficiency Anemia.
Iron Overload
Iron overload occurs when body iron content exceeds storage capacity, leading to the deposition of free iron in parenchymal tissues and generation of reactive oxygen species through Fenton chemistry. The two major categories are:
- Hereditary hemochromatosis: An autosomal recessive disorder, most commonly caused by homozygous C282Y mutations in the HFE gene, leading to inappropriately low hepcidin and excessive intestinal iron absorption. Over decades, iron accumulates in the liver, heart, pancreas, and joints, causing cirrhosis, cardiomyopathy, diabetes mellitus, arthropathy, and skin hyperpigmentation.
- Secondary iron overload (hemosiderosis): Results from chronic transfusion therapy (as in thalassemia major or sickle cell disease), ineffective erythropoiesis, or excessive dietary iron intake. Treatment includes iron chelation therapy with agents such as deferoxamine, deferasirox, or deferiprone.
Recommended Dietary Allowances
- Adult men (19+ years): 8 mg/day
- Adult premenopausal women (19-50 years): 18 mg/day
- Pregnant women: 27 mg/day
- Postmenopausal women (51+ years): 8 mg/day
- Infants (7-12 months): 11 mg/day
- Children (1-3 years): 7 mg/day
- Tolerable Upper Intake Level (adults): 45 mg/day
Connections
- Iron Benefits Deep Dive
- Iron Deficiency Anemia
- Heme vs Non-Heme Iron
- Iron and Cognitive Performance
- Iron and Athletic Performance
- Anemia
- Hemochromatosis
- Copper
- Relationship Between Hemoglobin and Ceruloplasmin — why ceruloplasmin (the copper ferroxidase) is required to load iron into hemoglobin, and why "iron-deficiency anemia" is often functional copper deficiency.
- Zinc
- Vitamin C
- Vitamin B12
- Vitamin B6
- Fatigue
- Thalassemia
- Sickle Cell Disease
- Beef
- Eggs
- Complete Blood Count
- Whole Food Copper Sources
- Celiac Disease
- Selenium