AS and IBD Overlap: Crohn's, UC, and the Shared Biology
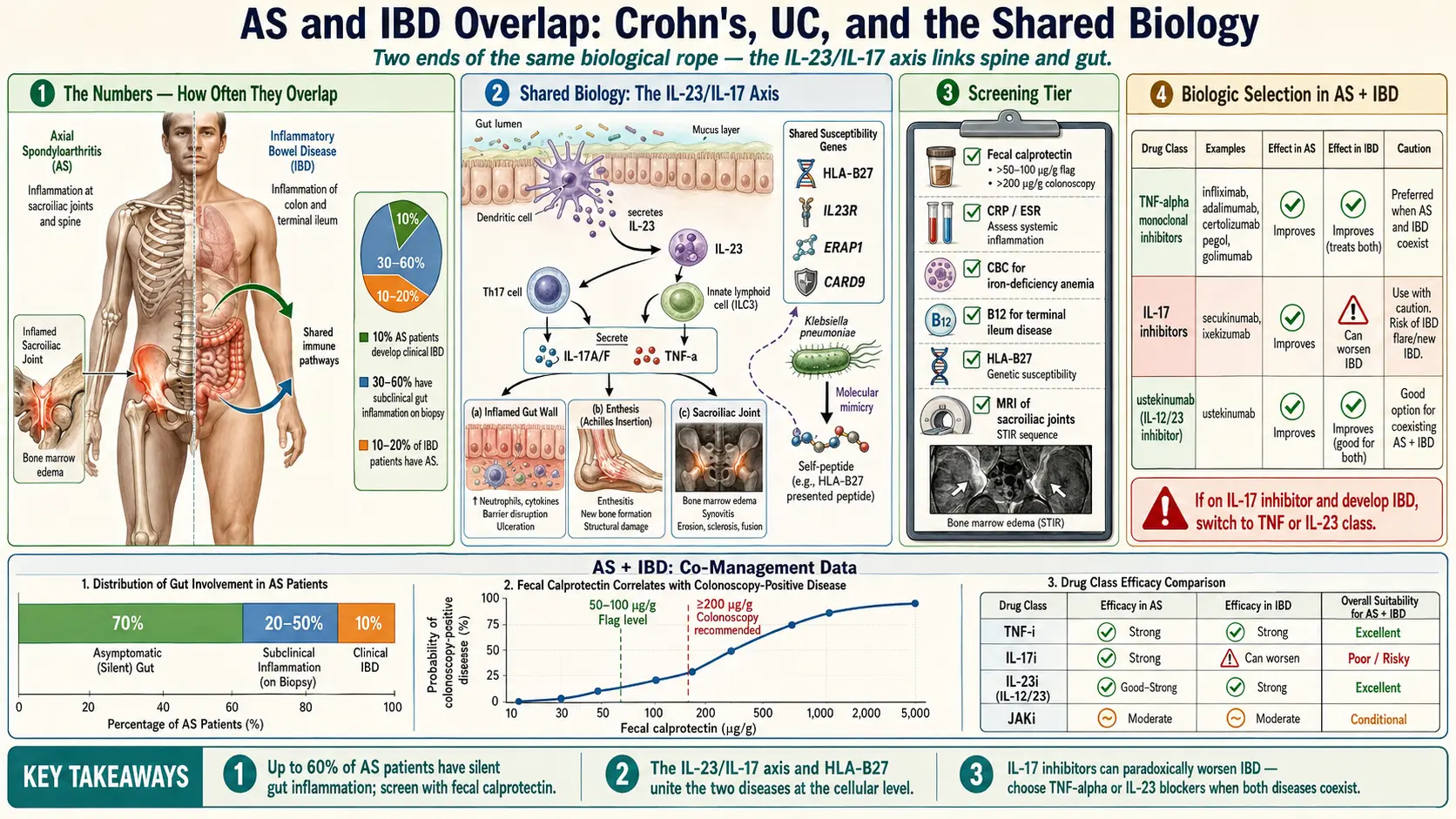
Table of Contents
- The Numbers — How Often They Overlap
- Why They Cluster — The Shared Biology
- Symptoms of IBD to Watch For if You Have AS
- Screening AS Patients for Occult IBD
- Symptoms of AS to Watch For if You Have IBD
- Screening IBD Patients for AS
- How IBD Overlap Changes Biologic Selection
- The “Best Single Drug” Question for AS + IBD
- What to Do If You’re Already on an IL-17 Inhibitor and Develop IBD
- Gut-Directed Approaches That Help Both
- NSAIDs in AS + IBD — The Conflict
- Monitoring Co-Managed Disease
- Other Extraintestinal Manifestations Connecting AS and IBD
- Pregnancy and AS+IBD Overlap
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
1. The Numbers — How Often They Overlap
If you have ankylosing spondylitis (AS) and you’ve noticed you also run to the bathroom more than your friends, you’re not imagining it. AS and inflammatory bowel disease (IBD) — Crohn’s disease and ulcerative colitis — are two ends of the same biological rope.
Roughly 10% of AS patients develop clinical IBD over their lifetime, with Crohn’s being more common than ulcerative colitis. That number climbs dramatically when you look under the microscope: 30–60% of AS patients have subclinical gut inflammation visible on colonoscopy with biopsy, even when they have no GI symptoms at all. The bowel is inflamed; the patient just doesn’t feel it yet.
The traffic runs both ways. 10–20% of IBD patients have AS or another form of spondyloarthritis, and inflammatory back pain is the single most common extraintestinal manifestation of IBD. Both conditions cluster in families that carry HLA-B27, and both live inside the same IL-23/IL-17 cytokine pathway. When you treat one, you are often — whether you know it or not — treating the other.
2. Why They Cluster — The Shared Biology
The IL-23/IL-17 Axis
The dominant unifying theme is the IL-23/IL-17 axis. IL-23 is produced by dendritic cells and macrophages at mucosal surfaces — the gut especially — and it drives a population of T cells and innate lymphoid cells to release IL-17 and TNF-α. Those cytokines inflame the gut wall, the entheses (where tendon meets bone), and the sacroiliac joints. The same inflammatory cocktail, in different tissues.
Gut Dysbiosis
The intestinal microbiome of AS patients looks abnormal in the same way IBD microbiomes look abnormal: reduced diversity, expansion of pro-inflammatory species, loss of butyrate-producing commensals. Whether dysbiosis causes the disease or is caused by it is still argued, but the overlap in microbial signatures is striking.
Intestinal Permeability
Both AS and IBD patients show increased intestinal permeability — the so-called “leaky gut.” Bacterial lipopolysaccharide and peptidoglycan fragments cross the gut wall and enter systemic circulation, where they can trigger inflammation at distant sites, including the spine.
Shared Genetics
Several susceptibility genes are shared: HLA-B27, IL23R, ERAP1, CARD9, STAT3, and others. These genes sit inside the innate immune response and antigen processing pipeline — the exact machinery that decides when to treat a gut bacterium as a threat.
Molecular Mimicry
Some of the most durable hypotheses invoke molecular mimicry. Peptides from gut bacteria — notably Klebsiella pneumoniae and certain E. coli strains — bear structural similarity to self-peptides that HLA-B27 presents, creating a crossfire where an anti-bacterial immune response ends up attacking joint and gut tissue. See Starch-Free Diet and the Ebringer Hypothesis and HLA-B27 Explained for the mechanistic detail.
3. Symptoms of IBD to Watch For if You Have AS
If you have AS, your inflammation doesn’t only live in your spine. Flag any of the following for your rheumatologist and ask for a GI referral:
- Chronic diarrhea lasting more than four weeks, especially if accompanied by blood or mucus.
- Abdominal cramping, particularly if it worsens after meals.
- Unintentional weight loss — not a crash diet, just pounds drifting off.
- Night sweats, fatigue, and low-grade fever, which point to systemic inflammation.
- Perianal disease — fissures, fistulas, or abscesses. These strongly suggest Crohn’s disease and should trigger same-week GI evaluation.
- Iron-deficiency anemia or persistently low ferritin on your AS blood panels.
- Elevated fecal calprotectin above 50–100 μg/g, a direct marker of bowel inflammation.
4. Screening AS Patients for Occult IBD
Because subclinical gut inflammation is so common, a reasonable screening tier looks like this:
- Fecal calprotectin — the single most useful stool test. Inexpensive, non-invasive, and correlates tightly with colonic inflammation.
- CBC — look for low iron, low ferritin, and elevated platelets (a classic inflammation fingerprint).
- CRP and ESR — if these run persistently higher than your AS activity would predict, occult gut inflammation is a leading suspect.
- B12 deficiency — points to terminal ileum disease, a Crohn’s hallmark.
- Colonoscopy with ileoscopy — definitive, indicated if you have GI symptoms or a fecal calprotectin above roughly 200 μg/g. Ask specifically for ileal intubation and random biopsies, since microscopic disease may be invisible to the naked eye.
If any of these light up, loop in a gastroenterologist. See Crohn’s Disease for the full workup pathway.
5. Symptoms of AS to Watch For if You Have IBD
If you already carry an IBD diagnosis and you start noticing back pain, don’t write it off as a desk-chair problem. Inflammatory back pain has a fingerprint:
- Chronic inflammatory back pain — insidious onset, lasting more than three months, typically starting before age 40.
- Morning stiffness lasting more than 30 minutes, and often two hours or more.
- Pain that improves with exercise and NOT with rest — the opposite of a mechanical back problem.
- Night pain waking you in the second half of the night, relieved only by getting up and moving.
- Alternating buttock pain — a classic signature of sacroiliac joint involvement.
- Enthesitis — heel pain (Achilles), plantar fascia pain, chest wall tenderness.
- Dactylitis — “sausage digit” swelling of a whole finger or toe (more characteristic of psoriatic spondyloarthritis).
- Peripheral arthritis, usually in larger joints and asymmetric.
6. Screening IBD Patients for AS
The screening workup for AS in IBD patients is relatively simple and high-yield:
- Clinical exam for spinal range of motion, Schober’s test, SI joint tenderness, and enthesitis at the Achilles, plantar fascia, and costochondral junctions.
- HLA-B27 testing if inflammatory back pain is present or there’s a family history of spondyloarthritis, psoriasis, or uveitis.
- MRI of the SI joints if inflammatory back pain is present. MRI is far more sensitive than plain X-ray for early disease and can show bone marrow edema before structural damage appears.
- Pelvis X-ray for established sacroiliitis if disease has been present for years.
- Referral to rheumatology with any positive finding. Early biologic therapy can halt structural progression.
7. How IBD Overlap Changes Biologic Selection
This is where the overlap matters most practically. A biologic that is a home run for axial AS can be a disaster for IBD — and vice versa. Get this decision wrong and you can trigger a flare in the organ you weren’t even targeting.
Drugs That Treat BOTH Axial AS and IBD
- Adalimumab (Humira) — FDA-approved for both AS and IBD; monoclonal anti-TNF.
- Infliximab (Remicade) — FDA-approved for both; monoclonal anti-TNF given by infusion.
- Certolizumab pegol (Cimzia) — approved for Crohn’s disease and AS; pegylated anti-TNF with no Fc region (relevant in pregnancy).
- Golimumab (Simponi) — approved for ulcerative colitis and AS.
Drugs That Treat AS but Do NOT Treat (and May Worsen) IBD
- Etanercept (Enbrel) — a soluble TNF receptor fusion protein. Works for AS but does NOT treat IBD because of its different TNF-binding mechanism. It usually doesn’t worsen IBD, but it offers no protection either, so it’s a poor choice when overlap exists.
- Secukinumab (Cosentyx) — IL-17A inhibitor. Can trigger or worsen IBD flares and should be avoided if IBD is present or suspected.
- Ixekizumab (Taltz) — same IL-17A class; same caution.
- Bimekizumab (Bimzelx) — IL-17A/F dual inhibitor; similar caution.
Drugs That Treat IBD but Not Axial AS
- Vedolizumab (Entyvio) — gut-selective integrin blocker. Highly effective for IBD but offers no axial AS benefit. Sometimes used for peripheral joint disease.
- Ustekinumab (Stelara) — IL-12/23 inhibitor. Effective for Crohn’s and ulcerative colitis, with modest benefit for peripheral joint disease but little to no axial AS benefit.
Emerging Options
Risankizumab (Skyrizi) and guselkumab (Tremfya) are IL-23 selective inhibitors, effective for IBD and psoriatic arthritis, with evolving but still unclear axial AS data. Stay tuned.
See the Biologics Guide for dosing, safety monitoring, and how to sequence agents.
8. The “Best Single Drug” Question for AS + IBD
For most patients with overlapping AS and IBD, the answer is simple: adalimumab or infliximab. Both have decades of prospective trial and registry data in both conditions, and both are genuinely effective at controlling axial, peripheral, and intestinal disease.
Choosing between them is mostly a lifestyle call. Infliximab is loaded and maintained by infusion every 6–8 weeks in an infusion center, which suits severe or difficult-to-control IBD (the dose can be escalated and the interval shortened quickly). Adalimumab is a self-administered subcutaneous injection every 2 weeks, more convenient for stable disease and for patients who dislike infusion chairs.
Biosimilars are now available for both drugs and are often the insurance-preferred option, typically at a fraction of the branded price with equivalent efficacy.
9. What to Do If You’re Already on an IL-17 Inhibitor and Develop IBD
This is a scenario rheumatologists see more often as IL-17 agents become popular. A patient on secukinumab or ixekizumab for AS starts passing bloody stools and is diagnosed with new-onset IBD. What now?
Stop the IL-17 inhibitor. Switch to a TNF-α inhibitor (most commonly adalimumab or infliximab). IL-17 inhibition-triggered IBD often reverses once the drug is stopped, especially if caught early. Involve a gastroenterologist for induction therapy — sometimes a short course of steroids is needed to quiet the flare while the new biologic takes effect.
10. Gut-Directed Approaches That Help Both
Non-pharmacologic interventions that target the gut have plausible dual benefit:
- Low-FODMAP diet — well-established for IBS-overlap symptoms and sometimes helpful with post-IBD functional pain. See Low-FODMAP Diet for the structured elimination and reintroduction phases.
- Starch-free / low-starch diet (Ebringer protocol) — theoretical benefit for both conditions by starving Klebsiella pneumoniae in the bowel. See Starch-Free Diet and Ebringer.
- Exclusive enteral nutrition (EEN) — strong evidence in pediatric Crohn’s induction. Theoretical AS benefit is unproven but plausible given the gut-joint axis.
- Fecal microbiota transplant (FMT) — experimental for both diseases, with some encouraging small-trial data in UC.
- Probiotics — VSL#3 / Visbiome has modest evidence in ulcerative colitis and pouchitis; evidence in AS is mixed but the side-effect profile is benign.
11. NSAIDs in AS + IBD — The Conflict
NSAIDs are first-line pain control in AS, and for good reason — they work, and they may even slow radiographic progression when taken continuously. But they can worsen IBD flares and cause mucosal ulceration, creating a direct conflict in overlap patients.
Practical rules of thumb:
- Celecoxib (COX-2 selective) is the least gut-toxic option and is preferred when NSAIDs are unavoidable.
- If IBD is active, minimize or avoid NSAIDs entirely. Lean on the biologic for joint inflammation and use acetaminophen for pain bridging.
- When IBD is in deep remission, low-dose intermittent celecoxib with GI-protective co-therapy (e.g., a PPI if indicated for other reasons) is often tolerated.
See NSAID Strategy for the full risk-benefit framework.
12. Monitoring Co-Managed Disease
If you have both conditions, the ideal setup is a rheumatologist and a gastroenterologist who share notes. Typically a single biologic is prescribed and monitored by the rheumatologist while the gastroenterologist tracks disease activity from the IBD side.
- Quarterly visits alternating between specialists for the first year, then annually once stable.
- Labs every 3–6 months: CBC, CRP, fecal calprotectin, LFTs, and a biologic-specific safety panel (e.g., TB screen annually on anti-TNF).
- Imaging: MRI of SI joints or spine as clinically indicated (often annually early, then less frequent once stable); colonoscopy per IBD surveillance guidelines, usually every 1–3 years depending on disease extent and dysplasia history.
- Watch for extraintestinal flares: iritis / anterior uveitis, pyoderma gangrenosum, erythema nodosum, and primary sclerosing cholangitis (PSC).
13. Other Extraintestinal Manifestations Connecting AS and IBD
Both diseases share a portfolio of extraintestinal manifestations that can precede, accompany, or follow the index diagnosis:
- Anterior uveitis (eye inflammation) — affects 30–40% of AS patients over their lifetime and is even more common when IBD overlap is present. Red, painful, photophobic eye — treat as a same-day ophthalmology emergency.
- Psoriasis — a subset of overlap patients develop plaque or inverse psoriasis, reflecting the common IL-23/IL-17 axis.
- Primary sclerosing cholangitis (PSC) — more tightly associated with ulcerative colitis; can present with elevated alkaline phosphatase and pruritus.
- Osteoporosis — both diseases reduce bone density, and any history of systemic steroid use compounds the risk. DXA scanning is appropriate for most overlap patients.
14. Pregnancy and AS+IBD Overlap
Biologic choice becomes even more critical in pregnancy. Certolizumab pegol is the preferred agent for patients trying to conceive or already pregnant because its lack of an Fc region means minimal placental transfer to the fetus. Adalimumab and infliximab are also considered compatible through much of pregnancy but should be timed carefully in the third trimester.
Disease activity tends to improve during the first trimester (pregnancy’s natural immunomodulation) but flares are common postpartum, so plan for close follow-up and early re-dosing of the biologic after delivery. See Pregnancy and AS for the full roadmap.
15. Key Research Papers
- Rudwaleit M, et al. Subclinical gut inflammation in spondyloarthritis. Ann Rheum Dis. 2020.
- Ward MM, et al. 2019 Update of the ACR/SAA/SPARTAN Recommendations for the Treatment of Axial Spondyloarthritis. Arthritis & Rheumatology. 2019.
- Magro F, et al. Third European Evidence-Based Consensus on Extra-Intestinal Manifestations of Inflammatory Bowel Disease. Journal of Crohn’s and Colitis. 2020.
- Hueber W, et al. Secukinumab, a human anti-IL-17A monoclonal antibody, for moderate to severe Crohn’s disease: a randomised, double-blind placebo-controlled trial. Aliment Pharmacol Ther. 2012. (demonstrates IL-17 inhibitor harm in IBD)
16. Research Papers
Curated PubMed topic searches for AS and IBD overlap. Each link opens a live PubMed query so you always see the most current studies.
- PubMed: Ankylosing spondylitis and inflammatory bowel disease
- PubMed: Spondyloarthritis and subclinical gut inflammation
- PubMed: IL-17 inhibitors and Crohn’s disease
- PubMed: Anti-TNF in AS and Crohn’s
- PubMed: Fecal calprotectin in spondyloarthritis
- PubMed: HLA-B27 and inflammatory bowel disease
- PubMed: Gut-joint axis in spondyloarthritis
- PubMed: Vedolizumab and spondyloarthritis
Connections
- Ankylosing Spondylitis
- HLA-B27 Explained
- Biologics Guide
- Exercise and PT Protocol
- NSAID Strategy
- Morning Stiffness Management
- Starch-Free Diet and Ebringer
- Pregnancy and AS
- Uveitis and Eye Involvement
- Crohn's Disease
- Ulcerative Colitis
- Inflammatory Bowel Disease
- IBD Extraintestinal Manifestations
- IBD Biologics Guide
- JAK Inhibitors and S1P Modulators
- SIBO
- Inflammatory Markers
- Crohn's Disease vs Ulcerative Colitis