HLA-B27 Explained
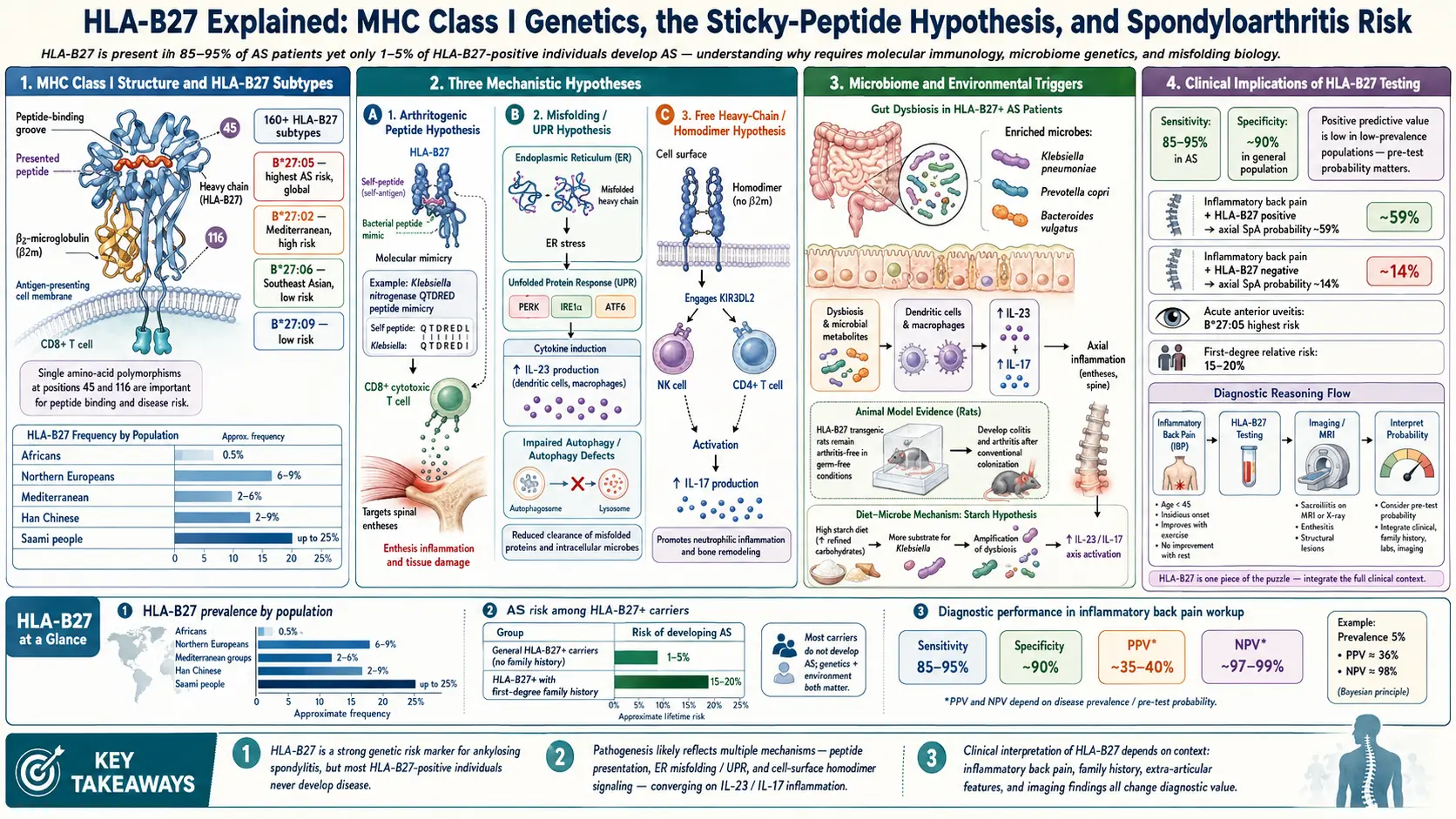
Table of Contents
- What HLA-B27 Is
- The AS Connection by the Numbers
- Testing — How It Works
- Interpreting Your Result
- The 200+ Subtypes — Not All Equal
- Why HLA-B27 Causes AS: The Three Leading Theories
- The Klebsiella Molecular Mimicry Angle
- HLA-B27 and Other Related Diseases
- Extra-Articular Associations
- What a Positive HLA-B27 Changes About Your Life
- Can You Change HLA-B27?
- Should Healthy Family Members Get Tested?
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
What HLA-B27 Is
HLA-B27 is a human leukocyte antigen — a protein sitting on the surface of almost every nucleated cell in your body. It is coded by a gene on the short arm of chromosome 6, inside the dense cluster of immune genes called the Major Histocompatibility Complex (MHC). Specifically, HLA-B27 is a class I MHC molecule, in the same family as HLA-A and HLA-C.
Its normal job is surveillance. Every cell continuously chops up a sample of the proteins it is producing and threads short peptide fragments (8–10 amino acids long) through its HLA-B molecule, presenting them on the cell surface like a tiny identification card. Passing CD8+ T cells inspect these cards. If the peptide looks foreign — viral, bacterial, or cancer-mutated — the T cell destroys the cell. If the peptide looks like normal self, the T cell moves on.
HLA-B27 is just one of hundreds of HLA-B variants. Everyone inherits two HLA-B alleles (one from each parent), and the combination defines part of your immune fingerprint. HLA-B27 is unusual only because of what it does — or fails to do — in the spines of the people who carry it.
The AS Connection by the Numbers
HLA-B27 is the strongest known HLA-disease association in medicine. The raw statistics:
- 90–95% of AS patients carry HLA-B27.
- In the general population, prevalence varies enormously by ancestry: 6–8% of Caucasians, 4% of African Americans, 1% of Japanese, and up to 50% of Haida Native Americans on the Pacific Northwest coast.
- A carrier's lifetime risk of developing AS is only 5–16%.
That last number is the one most patients miss. Having HLA-B27 is necessary for AS in most people, but it is very far from sufficient. Roughly 85–95% of HLA-B27 carriers live a full life without ever developing spondyloarthritis. Something else — likely a combination of additional genes, gut microbes, and environmental exposures — pushes a minority across the threshold into disease.
Testing — How It Works
HLA-B27 testing is a simple blood draw. The laboratory runs the sample on one of two platforms: flow cytometry, which uses a fluorescent antibody that binds HLA-B27 on white-blood-cell surfaces, or PCR, which amplifies the HLA-B gene directly from DNA and checks for the B27 sequence. PCR is more specific; flow cytometry can occasionally cross-react with the closely related HLA-B7 allele and require confirmation.
The cost ranges from $50 to $150 at most U.S. commercial labs (LabCorp, Quest), and insurance typically covers it when ordered for AS workup. The relevant billing codes are CPT 86828 (HLA antibody screening) or CPT 86812 (HLA typing, class I, single antigen).
You only need the test once in your life. HLA-B27 status is genetic and cannot change. To order it, ask your primary care physician or rheumatologist and state the purpose: "evaluation for spondyloarthritis." Any board-certified rheumatologist will order it without resistance if you have chronic inflammatory back pain.
Interpreting Your Result
A raw positive or negative is almost useless without the clinical picture. Four common scenarios:
Positive + AS symptoms. If you already have chronic inflammatory back pain (worse at rest, better with movement, lasting more than three months) plus MRI evidence of sacroiliitis, a positive HLA-B27 strongly supports the AS diagnosis. Combined with imaging, it usually closes the diagnostic question.
Positive + no symptoms. This is the most common scenario for family members who get tested. The correct response is reassurance. Your lifetime risk of developing AS is 5–16%, and the remaining 84–95% of you will never need a rheumatologist. No prophylactic treatment is indicated. You simply know to take chronic back pain seriously if it ever appears.
Negative + AS symptoms. A negative HLA-B27 does not rule out AS. Between 5% and 10% of AS patients are HLA-B27 negative, with higher proportions in populations where B27 is rare (e.g., Middle Eastern or African cohorts). The diagnostic gold standard is MRI of the sacroiliac joints, not genetics. If clinical suspicion is high, push for imaging.
Family screening. First-degree relatives (parents, siblings, children) of an AS patient have roughly a 20% chance of being HLA-B27 positive and an 8–10% chance of eventually developing AS — higher than the general population but still a minority outcome.
The 200+ Subtypes — Not All Equal
"HLA-B27" is not one allele but a family. More than 200 sequence variants have been catalogued, named with a colon-separated suffix: B*27:01, B*27:02, B*27:03, and so on. A handful are clinically important:
- B*27:05 — the most common variant in people of European descent; strongly associated with AS.
- B*27:02 — seen in Mediterranean populations; also AS-associated.
- B*27:04 — the dominant East Asian variant; AS-associated.
- B*27:06 — common in Southeast Asia (Thailand, Indonesia); appears to be protective, meaning carriers rarely develop AS even though they are "HLA-B27 positive" by standard testing.
- B*27:09 — almost exclusive to Sardinia; also protective.
Standard clinical HLA-B27 testing reports only positive or negative. It does not distinguish which subtype you carry. Subtype resolution requires sequence-based HLA typing, usually only available through research labs or transplant centers. For most patients, subtyping does not change management, but it explains why some populations (particularly Southeast Asian) have high HLA-B27 prevalence yet low AS rates.
Why HLA-B27 Causes AS: The Three Leading Theories
After fifty years of research, nobody has definitively proven the mechanism. Three hypotheses dominate the literature, and they are not mutually exclusive — all three may be partially right.
Arthritogenic Peptide Hypothesis
The oldest and most intuitive theory. HLA-B27 has an unusually shaped peptide-binding groove that happens to present a specific self-peptide — possibly derived from joint cartilage, enthesis tissue, or gut epithelium. CD8+ T cells that recognize this peptide become autoreactive and attack the tissues where it appears. The starting trigger may be molecular mimicry: a bacterial peptide that closely resembles the self-peptide primes T cells during an infection, and those primed T cells then cross-react with self. Klebsiella pneumoniae is the most-studied candidate (see the starch-free diet article).
Misfolding Hypothesis
HLA-B27 heavy chains fold inefficiently inside the endoplasmic reticulum of the cell that makes them. A fraction of every batch fails to pair properly with beta-2-microglobulin and instead misfolds or aggregates. The cell senses the pileup of misfolded protein and triggers the unfolded protein response (UPR), a stress pathway that activates inflammatory cytokines — particularly the IL-23/IL-17 axis. This is the same cytokine cascade targeted by modern AS biologics (secukinumab, ixekizumab, risankizumab). See the biologics guide for how these drugs interrupt the pathway downstream.
Free Heavy Chain Hypothesis
On the surface of HLA-B27-positive cells, a minority of molecules appear not as the normal heterodimer (heavy chain plus beta-2-microglobulin) but as abnormal free heavy chain homodimers. These dysfunctional dimers bind receptors such as KIR3DL2 on natural killer cells and a subset of T-helper-17 cells. Binding activates these innate-like cells, feeding them survival signals and tilting them toward producing IL-17. This theory links HLA-B27 to innate immunity rather than classical CD8+ T-cell surveillance.
The Klebsiella Molecular Mimicry Angle
In the 1980s, Alan Ebringer and colleagues at King's College London noticed that short amino-acid sequences in two Klebsiella pneumoniae proteins (nitrogenase and pullulanase) closely resemble sequences in HLA-B27 itself and in human type I, III, and IV collagen. In laboratory studies, antibodies raised against Klebsiella cross-reacted with HLA-B27-positive cells and with connective tissue proteins.
Ebringer's group further argued that Klebsiella thrives in a gut environment rich in dietary starch, and proposed a low-starch diet to reduce the bacterial load and, with it, the immune cross-reaction. Small uncontrolled cohorts reported symptom improvement.
Mainstream rheumatology considers the Klebsiella hypothesis suggestive but not proven. Randomized controlled trials are lacking. The mechanism remains plausible, the dietary intervention is cheap and low-risk, and some patients swear by it — but it is not standard of care. See the starch-free diet page for the full protocol and evidence review.
HLA-B27 and Other Related Diseases
Ankylosing spondylitis is the flagship HLA-B27 disease, but the entire spondyloarthropathy family clusters around the gene:
- Ankylosing spondylitis — 90–95% HLA-B27 positive.
- Reactive arthritis (formerly Reiter syndrome) — 60–80% positive. Triggered by gut or genitourinary infections.
- Psoriatic arthritis with axial involvement — 50–70% positive.
- IBD-associated spondyloarthropathy — 50–70% positive. See the AS & IBD overlap article.
- Acute anterior uveitis — about 50% positive. One of the most common non-AS presentations.
- Undifferentiated spondyloarthropathy — roughly 70% positive. A catch-all for patients with inflammatory features that do not yet meet criteria for a specific diagnosis.
Extra-Articular Associations
HLA-B27 predisposes to a small set of conditions beyond the joints. Patients and physicians should know about them because they often appear years before or after spinal disease.
- Acute anterior uveitis — sudden painful red eye, light sensitivity, blurred vision. Can occur decades before AS back pain or long after diagnosis. Requires urgent ophthalmology evaluation; untreated episodes can damage vision.
- Aortitis and aortic valve disease — inflammation of the ascending aorta leading, in rare cases, to aortic regurgitation.
- IgA nephropathy — a kidney disorder in which IgA antibodies deposit in the glomeruli; increased prevalence in HLA-B27 cohorts.
- Cardiac conduction abnormalities — most commonly atrioventricular (AV) block. Routine ECG is reasonable in long-standing AS.
- Pulmonary apical fibrosis — a late, rare complication seen in advanced AS; mimics tuberculosis on chest imaging.
What a Positive HLA-B27 Changes About Your Life
For most people, surprisingly little. But a small set of practical habits is worth adopting:
- Long-term vigilance. Take chronic inflammatory back pain seriously — meaning back pain that lasts more than three months, starts before age 45, is worse at rest, and improves with movement. Get it evaluated promptly.
- Know your family risk. First-degree relatives share a roughly 20% chance of being HLA-B27 positive themselves.
- Lower threshold for imaging. If inflammatory back pain does appear, push for an MRI of the sacroiliac joints rather than waiting years for radiographs to change.
- Routine eye exams. Uveitis is vision-threatening if untreated. Know the symptoms (sudden eye pain, redness, light sensitivity) and seek same-day ophthalmology care if they appear.
- Not a reason for panic. 85–95% of HLA-B27 carriers never develop AS. The gene is a probability, not a sentence.
Can You Change HLA-B27?
No. HLA-B27 is a fixed genetic variant you inherited at conception. Nothing — no diet, no supplement, no drug, no epigenetic intervention — can remove it or silence it.
What you can influence is the environment in which that gene operates:
- Gut health and infections. Avoiding prolonged antibiotic courses when not necessary, managing enteric infections promptly, and supporting a diverse microbiome may reduce the inflammatory triggers that push HLA-B27 toward disease.
- Exercise and posture. Regular movement, extension-based physiotherapy, and daily spinal mobility work slow the progression of spinal fusion once AS is established. See the exercise and PT protocol.
- Diet. The Klebsiella-starch hypothesis remains controversial but is cheap and low-risk to trial for a few months.
- Biologic therapy timing. Evidence is growing that earlier initiation of TNF or IL-17 inhibitors may alter long-term disease trajectory. See the biologics guide.
Should Healthy Family Members Get Tested?
Generally not routinely. A positive result in a healthy person changes very little about their care — they still watch for back pain and uveitis, which anyone should do anyway. There are a few legitimate reasons to test:
- Planning a family and wanting a risk estimate for children.
- Already developing symptoms suggestive of AS (inflammatory back pain over three months, onset before age 45).
- A personal desire for certainty that the anxiety of not knowing outweighs.
Insurance considerations. A positive HLA-B27 is part of your medical record and may affect some life insurance, disability insurance, or long-term care applications. The U.S. Genetic Information Nondiscrimination Act (GINA) protects against employment and health-insurance discrimination but does not cover life, disability, or long-term care insurance. State laws vary. If insurance underwriting is a concern, consider sorting out coverage before testing. This is a real trade-off, not paranoia — discuss it with your physician before ordering the test.
Key Research Papers
- International Genetics of Ankylosing Spondylitis Consortium. Identification of multiple risk variants for ankylosing spondylitis through high-density genotyping. Nat Genet. 2013.
- Colbert RA, et al. HLA-B27 misfolding and ankylosing spondylitis. Arthritis Res Ther. 2010.
- Bowness P. HLA-B27. Annu Rev Immunol. 2015.
- Ebringer A, Rashid T, Wilson C. Rheumatoid arthritis: proposal for the use of anti-microbial therapy in early cases. Scand J Rheumatol. 2003;32(1):2–11. (Klebsiella-AS link reference.)
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on HLA-B27 biology, disease association, and management:
- HLA-B27 and ankylosing spondylitis
- HLA-B27 subtypes and protective variants
- HLA-B27 misfolding and the unfolded protein response
- HLA-B27, Klebsiella, and molecular mimicry
- HLA-B27 and acute anterior uveitis
- HLA-B27 family screening and first-degree relative risk
- HLA-B27 population prevalence and ancestry
- HLA-B27 free heavy chains and KIR3DL2
Connections
- Ankylosing Spondylitis
- Biologics Guide
- AS and IBD Overlap
- Exercise and PT Protocol
- NSAID Strategy
- Morning Stiffness Management
- Starch-Free Diet and Ebringer
- Pregnancy and AS
- Uveitis and Eye Involvement
- Arthritis
- Crohn's Disease
- Ulcerative Colitis
- Psoriasis
- Klebsiella Pneumoniae
- Inflammatory Markers
- Extraintestinal Manifestations of IBD
- Chronic Low Back Pain Evidence Based Care
- Genetic Testing HLA DQ2 and DQ8