Extraintestinal Manifestations of IBD: Joints, Skin, Eyes, Liver
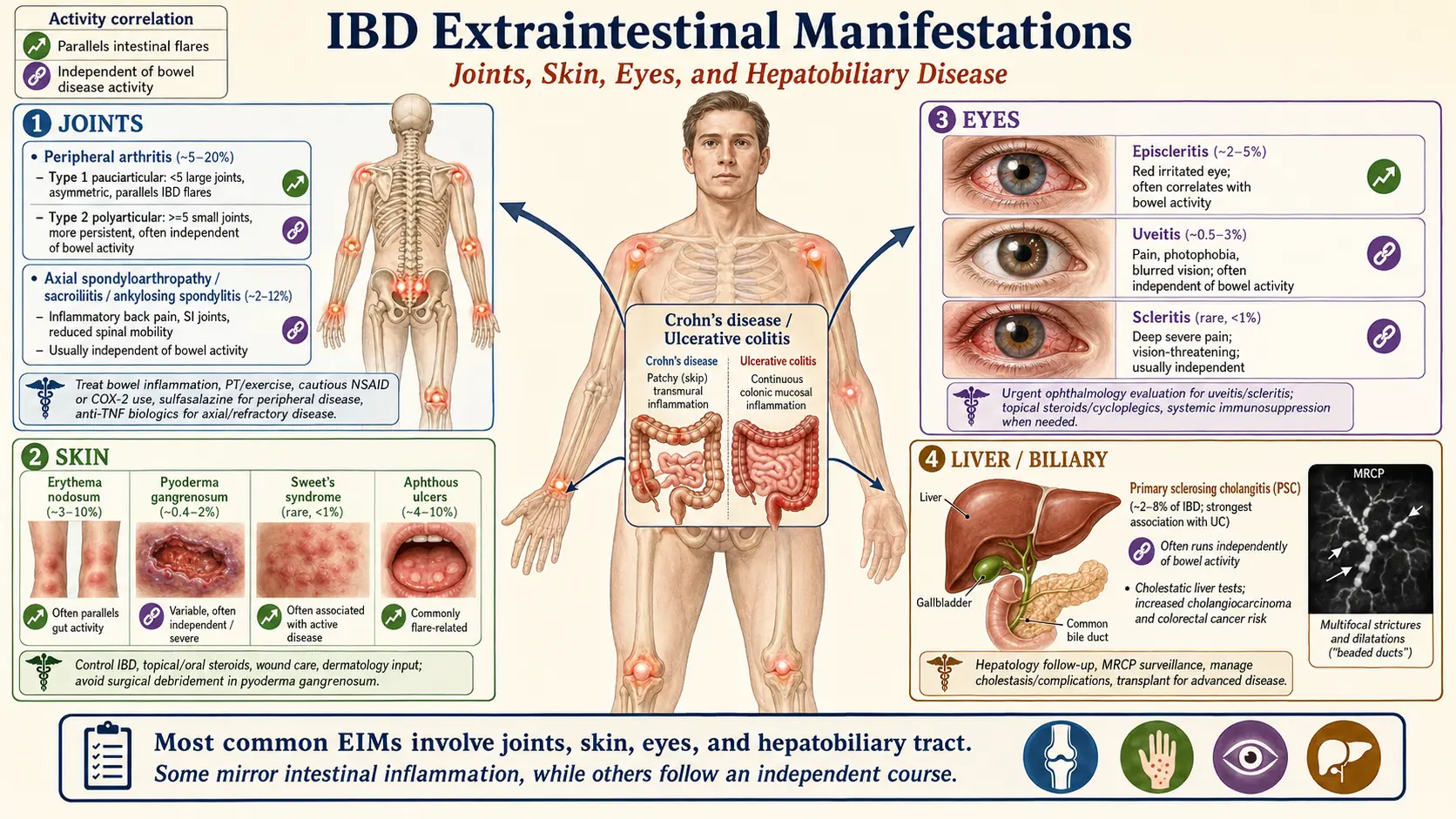
Table of Contents
- The Gut Is Not the Whole Disease
- Peripheral Arthritis — Type 1 and Type 2
- Axial Spondyloarthritis and Ankylosing Spondylitis Overlap
- Erythema Nodosum
- Pyoderma Gangrenosum
- Aphthous Stomatitis and Other Oral Lesions
- Uveitis, Episcleritis, and Scleritis
- Primary Sclerosing Cholangitis (PSC)
- Metabolic Bone Disease — Osteopenia and Osteoporosis
- Thromboembolism Risk During Flares
- Treatment Overlap — One Drug, Many Problems
- Red Flags — When to Call Someone Today
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
The Gut Is Not the Whole Disease
Inflammatory bowel disease is named for the bowel, but for roughly 25–40% of patients the disease also shows up somewhere else: joints that swell, skin that ulcerates, eyes that go red and painful, or a liver whose bile ducts slowly scar. These are called extraintestinal manifestations (EIMs), and they matter for three reasons.
First, some EIMs appear before bowel symptoms and can be the clue that gets you diagnosed at all. A young adult with recurrent painful red eyes or a stubborn shin ulcer plus intermittent diarrhea may spend years bouncing between specialists before anyone orders a calprotectin test. Second, some EIMs — primary sclerosing cholangitis is the headline example — carry their own cancer and transplant risks that have almost nothing to do with how bad your colitis feels today. Third, the choice of IBD drug often hinges on which EIMs you have. Anti-TNF biologics treat gut and joints and uveitis and pyoderma in one shot. Vedolizumab (gut-selective) is excellent for the bowel but weaker on joints and skin. JAK inhibitors help some EIMs and worsen others. See the biologics article for the drug-by-drug breakdown.
This page walks through each of the big EIMs: what it looks like, how fast you need to move, and how treatment decisions interlock with your IBD plan.
Peripheral Arthritis — Type 1 and Type 2
Joint inflammation is the most common EIM in IBD, affecting 15–30% of patients at some point. Rheumatologists split IBD peripheral arthritis into two distinct patterns, and the distinction actually matters for treatment.
Type 1 (Pauciarticular) — The Flare Companion
- Few joints — fewer than five, usually large (knee, ankle, wrist, elbow).
- Asymmetric — one knee but not the other.
- Acute attacks that last days to a few weeks.
- Parallels bowel activity — joints flare when the colon flares, settle when the colon settles.
- Non-erosive — does not damage the joint permanently.
Type 1 is essentially a symptom of uncontrolled gut inflammation. Treating the bowel usually treats the joints. If you and your gastroenterologist step up IBD therapy (corticosteroid taper, biologic, or immunomodulator) and the colitis goes into remission, the knees and ankles calm down with it. NSAIDs can help symptomatically but carry a real risk of triggering bowel flares — discuss short courses only. Intra-articular steroid injections are a safer bridge for a single hot joint.
Type 2 (Polyarticular) — The Independent One
- Many joints — five or more, often small (MCPs, PIPs, wrists).
- Symmetric — both hands look the same.
- Chronic — months to years, often lifelong.
- Independent of bowel activity — can flare while the colon is in total remission.
- Usually non-erosive but can occasionally cause damage.
Type 2 behaves more like its own rheumatologic disease. Controlling the gut alone is not enough. Treatment leans on anti-TNF biologics (infliximab, adalimumab), which target both inflammations at once, or sulfasalazine, which has mild activity in both bowel and joints. Methotrexate is reasonable if the IBD is Crohn's (it has bowel evidence) and weaker if the IBD is ulcerative colitis. Rheumatology co-management is usually needed.
Axial Spondyloarthritis and Ankylosing Spondylitis Overlap
Separately from peripheral arthritis, 5–10% of IBD patients develop inflammation of the spine and sacroiliac joints — what rheumatologists now call axial spondyloarthritis, with ankylosing spondylitis as the radiographic-stage endpoint. The hallmark symptom is inflammatory back pain: worse at rest, wakes you in the second half of the night, stiffness over an hour in the morning, and — critically — better with movement rather than worse. If your back hurts most after walking, it is probably mechanical. If it hurts most when you wake up and loosens with activity, it is inflammatory.
The genetic marker HLA-B27 is positive in roughly 50–70% of IBD-associated axial disease — lower than in classical AS (90–95%) but still far above general-population rates. See the HLA-B27 article and the AS and IBD overlap article for the full explanation of how these diseases share genetics and gut-driven immunity.
Axial disease is independent of bowel activity — like Type 2 peripheral arthritis. Your back can fuse slowly while your colon looks pristine on colonoscopy. That is why treatment selection matters:
- Anti-TNF biologics (infliximab, adalimumab, golimumab, certolizumab) treat both gut and spine. First choice when both are active.
- IL-17 inhibitors (secukinumab, ixekizumab) work beautifully for AS but can worsen or trigger IBD. Avoid in patients with known IBD.
- IL-23 inhibitors (risankizumab, ustekinumab) treat Crohn's disease; ustekinumab also has modest evidence in psoriatic and axial spondyloarthritis.
- Vedolizumab (gut-selective integrin blocker) does not reach the spine and generally does not treat axial disease — some patients even develop new joint symptoms on it.
- NSAIDs are AS first-line but can aggravate IBD. Use the lowest effective dose for the shortest duration, and prefer COX-2 selective agents (celecoxib) if tolerated.
The ankylosing spondylitis overview covers imaging (MRI sacroiliac joints before X-ray changes appear), physical-therapy protocols, and the full rheumatologic workup.
Erythema Nodosum
Erythema nodosum (EN) is the more common of the two classic IBD skin lesions, affecting roughly 3–15% of patients. It looks like tender, warm, red-to-purple nodules typically 1–5 cm across, most often on the shins but sometimes on thighs, forearms, or trunk. They feel bruised and hot; pressing them hurts.
EN tracks gut activity closely. Lesions appear during flares and fade as the flare resolves, usually over 3–6 weeks, often leaving a transient bruise-like discoloration but no scar. New crops during active colitis are common.
Treatment is simple: treat the bowel. Rest, leg elevation, and cool compresses help with pain. NSAIDs relieve tenderness but again carry bowel-flare risk — short courses only. Stubborn, painful EN that does not respond to gut control may warrant systemic corticosteroids or escalation to a biologic, decisions that usually match the gastroenterologist's plan for the colon anyway.
EN is not specific to IBD. Strep throat, sarcoidosis, tuberculosis, pregnancy, oral contraceptives, and certain drugs can trigger it. First-time EN deserves a chest X-ray (sarcoid, TB) and a strep swab even in a patient with known IBD.
Pyoderma Gangrenosum
Pyoderma gangrenosum (PG) is rarer than EN (1–5% of IBD patients) and far more serious. It begins as a small painful pustule or bump — often mistaken for an insect bite or minor wound — that rapidly breaks down into a deep, necrotic ulcer with a characteristic violaceous (purple) undermined border. The pain is disproportionate to what the lesion looks like; patients describe it as burning or searing. Lesions favor the legs but can appear anywhere, including around stoma sites (peristomal pyoderma).
Three pitfalls patients and doctors must know:
- Pathergy. PG can be triggered or worsened by surgical debridement. The instinct to cut away the dead tissue is wrong. A surgeon who does not recognize PG can convert a 3 cm ulcer into a 20 cm one. If a wound is expanding rapidly with a purple border and hurts far more than it should, ask the team to consider PG before operating.
- Activity independence. Unlike erythema nodosum, PG does not reliably parallel bowel activity. It can flare when your colitis is quiet.
- Diagnosis is clinical and one of exclusion. There is no confirmatory biopsy finding — biopsy is done mainly to rule out infection, vasculitis, and malignancy.
Treatment escalates with severity:
- Topical or intralesional corticosteroids / tacrolimus for small, early lesions.
- Systemic corticosteroids (prednisone 0.5–1 mg/kg/day) for larger or spreading disease, usually with rapid response.
- Cyclosporine as a steroid-sparing alternative.
- Biologics — especially infliximab — are the modern game-changer. A landmark randomized trial by Brooklyn et al. (2006) established infliximab as an effective PG therapy. Adalimumab, ustekinumab, and JAK inhibitors have also shown benefit in case series.
Peristomal PG around an ostomy site is particularly frustrating because pouching systems chronically traumatize the skin and perpetuate pathergy. Stoma nurses experienced with PG, convex wafers, and anti-inflammatory therapy are essential. See the surgery decisions article for more on ostomy care.
Aphthous Stomatitis and Other Oral Lesions
Painful mouth ulcers are one of the most under-recognized IBD manifestations. Up to 20–50% of patients with Crohn's disease and 10% of those with ulcerative colitis develop recurrent aphthae — small (2–10 mm), round, yellow-white ulcers with red halos on the inside of the lips, cheeks, or tongue. They sting with acidic foods and usually heal in 7–14 days without scarring. They often flare with the bowel.
More Crohn's-specific oral findings include cobblestone mucosa (lumpy, fissured buccal surface), deep linear ulcers, pyostomatitis vegetans (a snail-track pustular lesion seen mainly in UC), and angular cheilitis from iron, B12, or folate deficiency — see the nutrient deficiencies article.
Management is mostly symptomatic: topical anesthetics (lidocaine viscous), topical corticosteroids (triamcinolone in orabase, dexamethasone rinse), and treatment of the underlying IBD. Severe or persistent Crohn's oral disease responds to systemic biologics, just like the gut.
Uveitis, Episcleritis, and Scleritis
Eye involvement affects 2–10% of IBD patients and ranges from nuisance to sight-threatening. The three main patterns differ in how fast you need to move.
Episcleritis — Uncomfortable, Not Dangerous
Inflammation of the thin vascular layer just under the conjunctiva. The eye looks pink or red in a wedge-shaped patch, feels gritty or mildly achy but not deeply painful, and vision is normal. Episcleritis mirrors bowel activity and usually settles with topical lubricants or a short topical NSAID drop. Treating the flare resolves it. Not sight-threatening.
Scleritis — Deep, Painful, Serious
Inflammation of the deeper scleral wall. The eye is deeply red with a bluish or purplish tint, the pain is severe and boring (wakes you at night, radiates to the forehead or jaw), and vision may be blurred. Scleritis can thin the sclera and, in rare cases, perforate the eye. This is a same-day ophthalmology referral, not a "next available appointment." Treatment usually requires systemic NSAIDs, systemic corticosteroids, and often a biologic.
Uveitis — The Sight-Threatening One
Inflammation of the uveal tract (iris, ciliary body, choroid). IBD-associated uveitis is typically anterior (iritis), often bilateral, insidious, and chronic — different from the classical HLA-B27 AS-pattern uveitis, which is unilateral, acute, and alternates eyes.
Red-flag symptoms:
- Eye pain (often worse with bright light)
- Photophobia so severe you squint in normal room light
- Blurred vision or floaters
- Circumlimbal redness (ciliary flush) — the redness ringed tightly around the cornea, not diffuse
- Pupil that looks small, irregular, or sluggish
Any of these warrants same-day ophthalmology evaluation. Untreated uveitis causes synechiae (iris stuck to the lens), glaucoma, cataract, macular edema, and permanent vision loss. Treatment includes topical corticosteroid drops, cycloplegic drops (to prevent synechiae), and in refractory cases systemic corticosteroids or anti-TNF biologics. Infliximab and adalimumab have strong evidence in non-infectious uveitis.
If you have IBD and develop red eye with pain or light sensitivity, go to an emergency department or an ophthalmologist the same day. Do not wait.
Primary Sclerosing Cholangitis (PSC)
PSC is the most serious extraintestinal manifestation of IBD. It is a chronic, progressive inflammation and scarring of the intra- and extrahepatic bile ducts. Roughly 70–80% of PSC patients have IBD, overwhelmingly ulcerative colitis (pancolitis pattern, often with backwash ileitis and rectal sparing). Conversely, only 2–8% of IBD patients develop PSC — but those who do face a very different long-term prognosis.
How It Presents
Many patients are discovered incidentally through abnormal liver blood tests — a persistently elevated alkaline phosphatase (ALP), often 2–3 times the upper limit of normal, with less elevated AST/ALT. GGT is usually up, bilirubin is often normal early on. Others present with fatigue, pruritus (itching), right-upper-quadrant pain, or jaundice. Some present with bacterial cholangitis: fever, chills, abdominal pain — a medical emergency.
Screening in IBD
Current AASLD and ACG guidance supports annual liver function tests in everyone with IBD. If ALP is persistently elevated with no other explanation, the next step is MRCP (magnetic resonance cholangiopancreatography) — a non-invasive MRI of the bile ducts that shows the characteristic beaded or pruned-tree pattern of strictures and dilations. ERCP is reserved for diagnosis when MRCP is equivocal or when a dominant stricture needs intervention.
Cholangiocarcinoma and Colon Cancer Risk
PSC dramatically raises two cancer risks, and both change your surveillance schedule:
- Cholangiocarcinoma. Lifetime risk is roughly 10–15% in PSC patients, compared to essentially zero in the general population. Annual surveillance with MRI/MRCP plus CA 19-9 is typical, though the optimal strategy remains debated.
- Gallbladder cancer. Elevated risk; any gallbladder polyp in PSC is generally a reason for cholecystectomy regardless of size.
- Colorectal cancer. PSC plus ulcerative colitis is the highest-risk combination in IBD. Surveillance colonoscopy every year from the time of PSC diagnosis, not the standard 1–3-year intervals used for UC without PSC. Chromoendoscopy or high-definition white-light with targeted biopsies is preferred.
Treatment
There is no proven disease-modifying medical therapy for PSC. Ursodeoxycholic acid at moderate doses (13–15 mg/kg/day) is widely used and improves liver numbers, but high doses paradoxically worsen outcomes and are avoided. IBD biologics control the colon but do not halt bile-duct disease. Dominant strictures can sometimes be dilated endoscopically. The only cure for advanced PSC is liver transplantation, with generally excellent outcomes, though PSC can recur in the graft.
A PSC diagnosis does not mean imminent disaster — many patients live decades with slowly progressive disease — but it does change the surveillance intensity and the overall risk picture. Insist on a hepatologist, not just a gastroenterologist, if PSC is confirmed.
Metabolic Bone Disease — Osteopenia and Osteoporosis
Bone loss is common and under-diagnosed in IBD. Roughly 30–50% of IBD patients have osteopenia and 15% have frank osteoporosis. The drivers stack up:
- Chronic systemic inflammation — cytokines (TNF, IL-6) directly activate bone-resorbing osteoclasts.
- Corticosteroid exposure — any cumulative prednisone burden raises fracture risk; this is the single most modifiable factor.
- Vitamin D and calcium malabsorption, especially in Crohn's disease of the small bowel or after resection.
- Low body weight, undernutrition, and hypogonadism from chronic illness.
- Reduced weight-bearing activity during flares.
Baseline DEXA is reasonable for any IBD patient with a significant steroid course (≥3 months of prednisone equivalent), postmenopausal status, low BMI, or chronic active disease. Repeat every 1–2 years as indicated. Manage with:
- Adequate calcium (1000–1200 mg/day) and vitamin D (target 25-OH-D above 30 ng/mL).
- Weight-bearing and resistance exercise.
- Smoking cessation, moderate alcohol.
- Minimizing cumulative steroid exposure — a major argument for starting biologics sooner.
- Bisphosphonates (alendronate, zoledronic acid) for established osteoporosis or T-score ≤ -1.5 on chronic steroids.
Thromboembolism Risk During Flares
One of the most dangerous and least-recognized EIMs is not a visible lesion at all — it is a blood clot. Active IBD is a hypercoagulable state. During flares, IBD patients have roughly a threefold elevated risk of venous thromboembolism (VTE) — deep-vein thrombosis and pulmonary embolism — compared with the general population, and hospitalized IBD patients have even higher rates. The risk persists for months after the flare resolves.
Implications:
- Any hospitalized IBD patient should receive pharmacologic DVT prophylaxis (heparin or LMWH) unless actively hemorrhaging. Do not accept "but they have bloody stools" as an excuse — the evidence says prophylaxis is still net-beneficial in most flare hospitalizations.
- Learn the symptoms of DVT (unilateral calf swelling, warmth, tenderness) and PE (sudden shortness of breath, pleuritic chest pain, unexplained tachycardia, syncope). Any of these during or shortly after a flare is an emergency.
- Be cautious with estrogen-containing contraceptives during severe flares; progestin-only options or IUDs may be safer. Discuss with your gastroenterologist and gynecologist.
- Long-distance travel during a flare warrants extra hydration, calf exercises, and compression stockings. Some clinicians recommend prophylactic LMWH for flights over 6–8 hours in actively flaring patients.
- The JAK inhibitor class (tofacitinib, upadacitinib) carries a boxed warning for VTE. Balance that against the thromboembolic risk of uncontrolled flares — see the JAK and S1P article.
Treatment Overlap — One Drug, Many Problems
Because multiple EIMs share inflammatory pathways with the gut, well-chosen IBD therapy can collapse several problems into one prescription. A rough map:
- Anti-TNF (infliximab, adalimumab). Excellent for gut + peripheral arthritis (both types) + axial disease + uveitis + pyoderma gangrenosum + erythema nodosum. The broadest EIM coverage of any class.
- Ustekinumab (IL-12/23). Good for gut + psoriasis + peripheral arthritis; modest for axial disease.
- Risankizumab / Mirikizumab (IL-23). Strong gut efficacy; emerging data for skin and joint EIMs.
- Vedolizumab (gut-selective). Excellent gut safety, poor EIM coverage. Consider combining with targeted EIM therapy (topical eye drops, local injections) or switching classes if EIMs dominate.
- Tofacitinib / Upadacitinib (JAK). Helpful for gut + peripheral arthritis + axial disease + pyoderma. Watch for VTE, zoster, lipid changes.
- Sulfasalazine. Mild gut + peripheral joint coverage; cheap, old, still useful for overlap cases.
- Avoid in IBD: IL-17 inhibitors (secukinumab, ixekizumab) can trigger or worsen colitis. NSAIDs can trigger flares — use briefly and cautiously.
Red Flags — When to Call Someone Today
Most EIMs are chronic annoyances. A few are same-day emergencies:
- Red, painful eye with light sensitivity or blurred vision — same-day ophthalmology.
- Rapidly expanding, painful skin ulcer with a purple border — possible pyoderma gangrenosum; do not let anyone debride it until PG is ruled out.
- Unilateral leg swelling, warmth, and tenderness — possible DVT; emergency evaluation.
- Sudden shortness of breath, pleuritic chest pain, unexplained tachycardia, or fainting — possible pulmonary embolism; call 911.
- Fever + jaundice + right-upper-quadrant pain — possible bacterial cholangitis in PSC; emergency department.
- New-onset severe, progressive back pain with bowel or bladder changes — rule out cauda equina (rare) or spinal fracture in osteoporotic IBD patients.
When in doubt, contact your gastroenterology team or go to urgent care. IBD patients earn the right to a low threshold.
Key Research Papers
- Rogler G, et al. Extraintestinal manifestations of inflammatory bowel disease: current concepts, treatment, and implications for disease management. Gastroenterology. 2021.
- Orchard TR, Wordsworth BP, Jewell DP. Peripheral arthropathies in inflammatory bowel disease: their articular distribution and natural history. Gut. 1998. (Type 1 vs Type 2 classification)
- Brooklyn TN, et al. Infliximab for the treatment of pyoderma gangrenosum: a randomised, double-blind, placebo-controlled trial. Gut. 2006.
- Bowlus CL, et al. AASLD practice guidance on primary sclerosing cholangitis and cholangiocarcinoma. Hepatology. 2023.
- Grainge MJ, West J, Card TR. Venous thromboembolism during active disease and remission in inflammatory bowel disease: a cohort study. Lancet. 2010.
- Harbord M, et al. The First European Evidence-Based Consensus on Extra-Intestinal Manifestations in Inflammatory Bowel Disease. J Crohns Colitis. 2016.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on IBD extraintestinal manifestations:
- IBD extraintestinal manifestations overview
- IBD peripheral arthritis Type 1 and Type 2
- IBD, axial spondyloarthritis, and HLA-B27
- Pyoderma gangrenosum and infliximab in IBD
- Erythema nodosum in inflammatory bowel disease
- Uveitis and inflammatory bowel disease
- PSC, UC, and colonoscopy surveillance
- PSC cholangiocarcinoma risk and MRCP screening
- IBD osteoporosis and bone mineral density
- IBD and venous thromboembolism
Connections
- Inflammatory Bowel Disease Overview
- Crohn's vs Ulcerative Colitis: Key Differences
- Biologics: TNF, IL-23, and Integrin Inhibitors
- JAK Inhibitors and S1P Modulators
- IBD Surgery Decisions
- IBD Fatigue, Anemia, and Nutrient Deficiencies
- Pregnancy and IBD
- Diagnostic Workup: Calprotectin, Colonoscopy, MRE
- Diet for IBD: SCD and Mediterranean
- Ankylosing Spondylitis
- AS and IBD Overlap
- HLA-B27 Explained
- Crohn's Disease
- Ulcerative Colitis
- Arthritis
- Osteoporosis
- Psoriasis
- Jaundice