Crohn's Disease vs Ulcerative Colitis — Key Differences
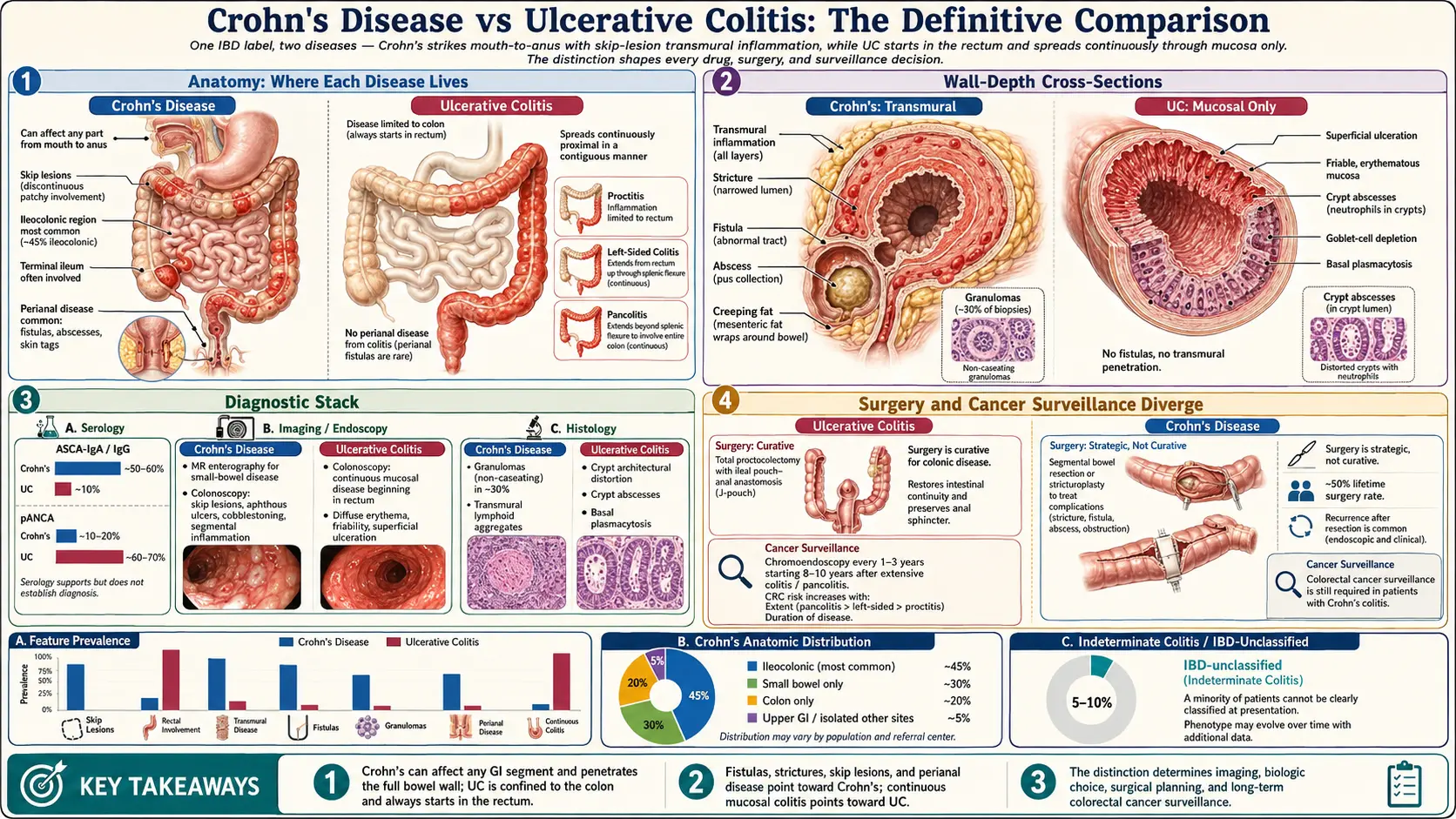
Table of Contents
- Why the Distinction Matters
- Anatomy — Where Each Disease Lives
- Depth of Inflammation — Transmural vs Mucosal
- Crohn's-Specific Complications: Strictures and Fistulas
- UC-Specific Patterns: Backwash Ileitis and Toxic Megacolon
- Symptom Differences Patients Actually Notice
- Serology — ASCA vs pANCA
- Imaging — What MRE, CT, and Colonoscopy Show
- Under the Microscope — Granulomas and Crypt Changes
- Why UC Can Be "Cured" by Surgery and Crohn's Cannot
- Indeterminate Colitis — When the Line Blurs
- How the Diagnosis Changes Your Biologic Choice
- Colorectal Cancer Risk — Different Surveillance Needs
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
Why the Distinction Matters
When someone hears "inflammatory bowel disease," it sounds like a single illness. It is not. Crohn's disease and ulcerative colitis (UC) are two different diagnoses that share a family resemblance — chronic immune-driven inflammation of the gut — and diverge almost everywhere else. They attack different parts of the bowel, go to different depths, cause different complications, respond differently to the same drugs, and have dramatically different surgical options.
Getting the label right is not a bookkeeping exercise. It changes which biologic your insurance will approve, whether surgery is a cure or a damage-control measure, how often you need colonoscopies, what your cancer risk looks like, and what complications your GI team will watch for over the next thirty years. A patient told they have "IBD" without specifying which one should push for clarity. Ask directly: Is this Crohn's, ulcerative colitis, or indeterminate colitis? What's the evidence for that label?
About 1.6 million Americans live with IBD, split roughly in half between the two diseases (with UC very slightly more common). Peak onset is between ages 15 and 35, with a second smaller peak in the 50s and 60s. Both run in families, but neither follows simple Mendelian inheritance — over 240 susceptibility genes have been mapped.
Anatomy — Where Each Disease Lives
The single most important difference is geographic. Think of your gut as a 25-foot tube running from mouth to anus. Where the inflammation sits — and how it's distributed — is the first clue to which disease you have.
Ulcerative colitis lives in the colon. Only the colon. It starts in the rectum (the last few inches of the large intestine) in virtually every case and spreads upward in a continuous unbroken sheet. There is no healthy tissue hiding between diseased patches. The inflammation might stop after a few inches (proctitis), run partway up the left side (left-sided colitis), or involve the entire colon (pancolitis) — but it is always one continuous stretch starting from the bottom. The small intestine is spared. The mouth is spared. The stomach is spared.
Crohn's can strike anywhere from mouth to anus. Mouth ulcers, esophagitis, stomach involvement, duodenal disease — all possible, though uncommon. The most frequent location by far is the terminal ileum, the last segment of the small intestine just before it joins the colon. About 45% of Crohn's patients have disease limited to the ileum and the nearby cecum (ileocolonic Crohn's). Another 25% have only small-bowel disease; 20% have only colonic disease; the rest have patchy involvement scattered across multiple sites.
Crohn's is also famously discontinuous. Diseased segments (sometimes called "skip lesions") alternate with stretches of completely normal bowel. A surgeon opening the abdomen of a Crohn's patient can find an inflamed inch of terminal ileum, three feet of pristine small bowel, and another inflamed patch near the transverse colon. UC never does this.
One more anatomic tell: perianal disease — fistulas, abscesses, skin tags, and fissures around the anus — is a Crohn's signature. About a third of Crohn's patients develop perianal complications at some point. UC does not cause fistulas or abscesses through the bowel wall, period.
Depth of Inflammation — Transmural vs Mucosal
The bowel wall is not a single sheet. It has four layers, arranged like a jelly roll: the mucosa (inner lining that touches food), the submucosa (a layer of connective tissue and blood vessels), the muscularis propria (the muscle that squeezes food along), and the serosa (the outer wrapper).
Ulcerative colitis is a mucosal disease. Inflammation is confined to the innermost lining and the superficial submucosa. The deeper muscle and outer wall are spared. This is why UC bleeds so reliably — the inflamed mucosa is raw and crumbly, loaded with surface blood vessels, and literally weeping into the stool. It's also why UC, however severe on the inside, almost never creates a fistula burrowing through the full wall.
Crohn's is a transmural disease. The inflammation punches through all four layers. That single fact explains almost every Crohn's complication:
- When the full wall gets inflamed and then heals with scar tissue, the healed ring is narrower and stiffer than normal — a stricture.
- When inflammation erodes all the way through, it creates a deep ulcer that can tunnel into neighboring tissue — a fistula.
- When inflammation walls itself off mid-tunnel, it forms an abscess, a pocket of pus inside the abdomen.
- Transmural inflammation recruits fat from outside the bowel to wrap around the inflamed segment — "creeping fat" — a finding surgeons recognize on sight.
UC has none of these. Severe UC can destroy the mucosa so completely that the whole colon becomes a drum-tight, paralyzed tube (toxic megacolon), but it still doesn't burrow sideways into other organs.
Crohn's-Specific Complications: Strictures and Fistulas
Strictures. Roughly a third of Crohn's patients develop at least one stricture during their disease course. Early in the disease strictures are mostly inflammatory — swelling that narrows the lumen but can shrink with drug therapy. Later, strictures become fibrotic: permanent scar tissue that no biologic will fix. Symptoms include cramping abdominal pain 30–90 minutes after meals, audible bowel sounds, bloating, and eventually partial obstruction with vomiting. Management escalates from medical therapy to endoscopic balloon dilation to strictureplasty or resection.
Fistulas. These are abnormal tunnels connecting two surfaces that should never touch. Enterocutaneous fistulas drain to skin. Enteroenteric fistulas link two loops of bowel. Enterovesical fistulas connect bowel to bladder (producing pneumaturia — air bubbles in urine — and recurrent UTIs). Rectovaginal fistulas drain stool into the vagina. About 35% of Crohn's patients develop at least one fistula over a lifetime. Treatment is a combination of antibiotics, anti-TNF biologics (infliximab is the best-studied), setons (surgical drains left in place), and sometimes surgery. See the surgery decisions article.
Abscesses. Walled-off pockets of infection, usually presenting with fever, localized pain, and a tender mass. Drainage (percutaneous or surgical) is mandatory before starting biologics, because pushing immunosuppression into an undrained abscess is dangerous.
UC-Specific Patterns: Backwash Ileitis and Toxic Megacolon
Backwash ileitis. A small proportion of UC patients with pancolitis show mild inflammation in the last few inches of the terminal ileum, just past the ileocecal valve. The appearance can alarm a pathologist — is this secretly Crohn's? — but true backwash ileitis is superficial, limited to a short segment, and presumed to result from reflux of colonic contents across an incompetent ileocecal valve. It does not change the UC diagnosis, doesn't progress, and resolves when colonic inflammation is controlled. The key distinguishers from Crohn's ileitis: continuity with colonic disease, very short length, no strictures, no skip lesions proximally, and a normal valve in between.
Toxic megacolon. A life-threatening UC emergency in which the colon dilates to a diameter of 6 cm or more, loses motility, and becomes systemically toxic (fever, tachycardia, leukocytosis). Risk factors include severe pancolitis, hypokalemia, opioids, anticholinergics, and loperamide. Mortality untreated approaches 30%. Management is urgent: IV steroids, broad-spectrum antibiotics, fluid resuscitation, and if no improvement in 24–72 hours, emergency colectomy. Crohn's colitis can also cause toxic megacolon, but it is classically a UC complication.
Severe bleeding. Because UC is a mucosal bleeding disease, the most severe flares can produce transfusion-dependent hemorrhage. Visible blood in every bowel movement is the norm in active UC — dramatically more common than in Crohn's, where bleeding is often occult or intermittent.
Symptom Differences Patients Actually Notice
Textbooks present these as clean contrasts. In real life there is overlap, but patterns do emerge.
UC symptoms: bloody diarrhea, urgent and frequent bowel movements (sometimes 10–20 per day in severe flares), tenesmus (the painful sensation of needing to go even when the rectum is empty), lower-abdominal cramping that eases after stooling, and weight loss driven mostly by reduced food intake. Pain is usually left-sided or lower-central because of where UC lives.
Crohn's symptoms: abdominal pain that often localizes to the right lower quadrant (terminal ileum territory), chronic diarrhea that is more often non-bloody, weight loss driven by malabsorption (not just reduced intake), fatigue out of proportion to obvious bowel symptoms, perianal pain or drainage, mouth ulcers, and fistula-related symptoms (pneumaturia, vaginal drainage). Crohn's pain often has a colicky post-meal pattern from strictures.
Children and adolescents with Crohn's commonly present with growth failure and delayed puberty — sometimes the only symptom before obvious GI complaints. Pediatric UC more often presents with obvious bloody diarrhea.
Serology — ASCA vs pANCA
Two antibody tests help sort out confusing cases, though neither is good enough to diagnose IBD on its own.
ASCA (anti-Saccharomyces cerevisiae antibodies) are IgA and IgG antibodies against a sugar on baker's yeast. They are positive in roughly 50–60% of Crohn's patients and only about 10% of UC patients. Their presence tilts toward Crohn's.
pANCA (perinuclear anti-neutrophil cytoplasmic antibodies) are positive in roughly 60–70% of UC patients and only about 10–20% of Crohn's patients. Their presence tilts toward UC — particularly a UC-like variant of Crohn's colitis that may behave more like UC after surgery.
The combination is informative: ASCA-positive + pANCA-negative favors Crohn's with about 95% specificity. ASCA-negative + pANCA-positive favors UC with similar specificity. But sensitivity is poor — about a third of IBD patients are negative on both tests. Expanded panels (anti-OmpC, anti-CBir1, anti-I2) add modest information but are not routine. Serology is a tiebreaker, not a substitute for colonoscopy with biopsy plus imaging.
Imaging — What MRE, CT, and Colonoscopy Show
Different diseases need different pictures. See the diagnostic workup article for the full sequence.
Colonoscopy with ileal intubation and biopsies is the cornerstone for both diseases. In UC, the endoscopist sees continuous erythema, loss of the normal vascular pattern, granularity, friability (contact bleeding), and superficial ulcerations starting from the rectum. In Crohn's colitis, the same scope shows patchy disease, aphthous ulcers (small punched-out lesions), deep linear "bear claw" ulcers, cobblestoning (when ulcers separate islands of relatively normal mucosa), and often a diseased terminal ileum with a normal colon between.
MR enterography (MRE) is the preferred test for evaluating small-bowel Crohn's. It shows bowel-wall thickening, enhancement pattern, mesenteric fat stranding, creeping fat, strictures with proximal dilation, fistula tracts, and abscesses — all without radiation. Small-bowel Crohn's is invisible to a colonoscope that can only reach the terminal ileum; MRE sees what the scope cannot.
CT enterography covers similar territory with faster acquisition but uses ionizing radiation — a real concern for young Crohn's patients who will need many scans over decades.
Capsule endoscopy (a swallowable pill camera) images small-bowel mucosa directly. It is useful in Crohn's workup when MRE is equivocal, but it is contraindicated in suspected strictures — the capsule can lodge and require surgery.
Transabdominal ultrasound, heavily used in Europe and increasingly in North America, can detect bowel-wall thickening, hyperemia, and complications with zero radiation and no prep — particularly helpful for children and for monitoring known disease.
Under the Microscope — Granulomas and Crypt Changes
Pathology completes the picture. Features that favor Crohn's: non-caseating granulomas (tight clusters of immune cells, found in about 30% of Crohn's biopsies and essentially never in UC), focal and patchy inflammation, deeper ulceration reaching muscle, transmural lymphoid aggregates, and relatively preserved goblet cells.
Features that favor UC: diffuse continuous inflammation limited to mucosa, crypt architectural distortion (the glands get bent and branched), cryptitis and crypt abscesses, marked goblet cell depletion (the cells that make protective mucus are wiped out), and basal plasmacytosis (plasma cells piled up at the base of the crypts).
A single biopsy can be ambiguous. Multiple biopsies from different regions, read by a pathologist with IBD experience, give the best chance of a confident call.
Why UC Can Be "Cured" by Surgery and Crohn's Cannot
This is the single biggest prognostic difference, and it follows inevitably from anatomy.
UC is confined to the colon. Remove the colon and rectum, and you remove every site where UC can occur. The standard operation is a total proctocolectomy with ileal pouch–anal anastomosis (IPAA, or "J-pouch"): the surgeon removes the entire colon and rectum, then fashions a reservoir from the tail end of the small intestine and connects it to the anus. In the best hands and patients, this restores bowel continuity (no permanent stoma), allows 4–8 bowel movements per day, and eliminates UC disease activity. Roughly 20–30% of UC patients eventually need colectomy, either elective for refractory disease or emergent for complications.
UC after colectomy is effectively "cured" in the disease-elimination sense. Flares, bleeding, and colorectal cancer risk from UC go to zero. New complications can arise — pouchitis (inflammation of the pouch itself, affecting 50% of J-pouch patients at some point), cuffitis, fertility reduction in women, and rare pouch failure — but the underlying UC is gone.
Crohn's can live anywhere in 25 feet of tube. Removing one inflamed segment leaves 24 more feet where the disease can reappear. After an ileocecal resection (the most common Crohn's operation), the endoscopic recurrence rate at the new anastomosis is 70–90% within a year unless aggressive prophylactic therapy is started. Clinical recurrence rates run about 30% at 5 years and 50% at 10 years. Repeated surgery carries a real risk of short-bowel syndrome if too much small intestine is removed.
So surgery in Crohn's is strategic, not curative: it is used to relieve obstructions, drain abscesses, close fistulas, and excise short segments that drugs cannot salvage. It is an essential tool — about 50% of Crohn's patients will have at least one surgery — but no one tells a Crohn's patient the disease is gone after an operation.
Indeterminate Colitis — When the Line Blurs
About 5–15% of IBD patients with isolated colonic disease cannot be firmly classified as Crohn's or UC even after colonoscopy, biopsies, imaging, and serology. The label for this middle ground is indeterminate colitis (IC), or, in newer nomenclature, IBD-unclassified (IBDU).
Indeterminate colitis matters most at surgical decision points. A patient with probable UC offered a J-pouch needs to understand that if the disease is actually Crohn's, the pouch will eventually fail in a sizable minority of cases — somewhere around 30–40% long-term in some series. Many surgeons still offer J-pouches to IC patients, but the counseling is different: the pouch failure rate is higher, and the patient may end up with a permanent ileostomy down the road.
Over years of follow-up, roughly a third of IC patients eventually reclassify as definite UC, another third as definite Crohn's, and the remainder stay indeterminate. Repeat biopsies, careful review of perianal exams, and watching for new small-bowel involvement all help refine the diagnosis over time.
How the Diagnosis Changes Your Biologic Choice
The two diseases have overlapping but distinct drug approvals, and insurance pays attention to the label. See the biologics article and JAK & S1P article for mechanism and positioning.
Anti-TNF agents (infliximab, adalimumab, certolizumab, golimumab): The original IBD biologics, approved for both diseases (certolizumab is Crohn's-only; golimumab is UC-only). Infliximab has the best fistula-closure data in Crohn's — transmural disease demands a drug that penetrates deeply.
Anti-integrin (vedolizumab): Approved for both. Gut-selective, very safe, but slower to work. Often preferred in older patients and those with infection risk.
Anti-IL-12/23 (ustekinumab) and anti-IL-23 (risankizumab, mirikizumab, guselkumab): Ustekinumab is approved for both diseases. The pure IL-23 inhibitors have split approvals: risankizumab for both Crohn's and UC, mirikizumab for UC (and Crohn's in more recent approvals), guselkumab for UC and Crohn's. Dermatology-experienced patients with psoriasis plus IBD often do well on this class because it treats both.
JAK inhibitors (tofacitinib, upadacitinib): Tofacitinib is UC-only. Upadacitinib is approved for both UC and Crohn's. Fast-acting but carry boxed warnings for cardiovascular events, VTE, and malignancy.
S1P modulators (ozanimod, etrasimod): Both UC-only. Oral, once-daily, modest efficacy, useful for patients wanting to avoid injections.
A patient with Crohn's and perianal fistulas is usually started on infliximab — the best fistula data. A patient with moderate-severe UC preferring an oral pill might reasonably start on upadacitinib or an S1P modulator. The same symptoms with a different diagnosis lead to different prescriptions.
Colorectal Cancer Risk — Different Surveillance Needs
Both diseases raise colorectal cancer risk above baseline, but the shape of the risk is different.
UC: Risk begins to rise after about 8–10 years of disease, is highest in patients with pancolitis, and is further elevated by coexisting primary sclerosing cholangitis (PSC). Guidelines recommend surveillance colonoscopy every 1–3 years starting 8 years after diagnosis (or immediately at diagnosis in those with PSC), with chromoendoscopy or high-definition white-light examination and targeted biopsies.
Crohn's: Colonic Crohn's carries a similar colorectal cancer risk to UC when the extent and duration are matched. Small-bowel Crohn's raises small-bowel adenocarcinoma risk (a rare cancer at baseline) by about 20-fold — still an uncommon event but one reason long-segment small-bowel Crohn's warrants attention.
For both diseases, sustained mucosal healing (normal-appearing mucosa on scope plus normal histology) appears to lower the cancer risk, which is one of several reasons aggressive treat-to-target therapy has become the norm.
Key Research Papers
- Baumgart DC, Sandborn WJ. Crohn's disease. Lancet. 2012. (And companion UC review by Ordas et al.)
- Torres J, Mehandru S, Colombel JF, Peyrin-Biroulet L. Crohn's disease. Lancet. 2017.
- Ungaro R, Mehandru S, Allen PB, Peyrin-Biroulet L, Colombel JF. Ulcerative colitis. Lancet. 2017.
- Feuerstein JD, et al. AGA Clinical Practice Guidelines on the Management of Moderate to Severe Ulcerative Colitis. Gastroenterology. 2020.
- Feuerstein JD, et al. AGA Clinical Practice Guidelines on the Medical Management of Moderate to Severe Luminal and Perianal Fistulizing Crohn's Disease. Gastroenterology. 2021.
- Jostins L, et al. Host-microbe interactions have shaped the genetic architecture of inflammatory bowel disease. Nature. 2012.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on the differences between Crohn's disease and ulcerative colitis:
- Crohn's vs ulcerative colitis differential diagnosis
- ASCA and pANCA serology in IBD
- Indeterminate colitis and IBD-unclassified
- Backwash ileitis in ulcerative colitis
- Crohn's perianal fistula management
- Ileal pouch-anal anastomosis outcomes
- MR enterography in Crohn's disease
- IBD colorectal cancer surveillance
Connections
- Inflammatory Bowel Disease Overview
- Diagnostic Workup: Calprotectin, Colonoscopy, MRE
- Biologics: TNF, IL-23, and Integrin Inhibitors
- JAK Inhibitors and S1P Modulators
- Diet for IBD: SCD and Mediterranean
- IBD Surgery Decisions
- Extraintestinal Manifestations: Joints, Skin, Eyes
- IBD Fatigue, Anemia, and Nutrient Deficiencies
- Pregnancy and IBD: Fertility, Medications, Outcomes
- Crohn's Disease
- Ulcerative Colitis
- Irritable Bowel Syndrome
- Celiac Disease
- Ankylosing Spondylitis
- AS and IBD Overlap
- Gut Healing Protocol