Inflammatory Markers
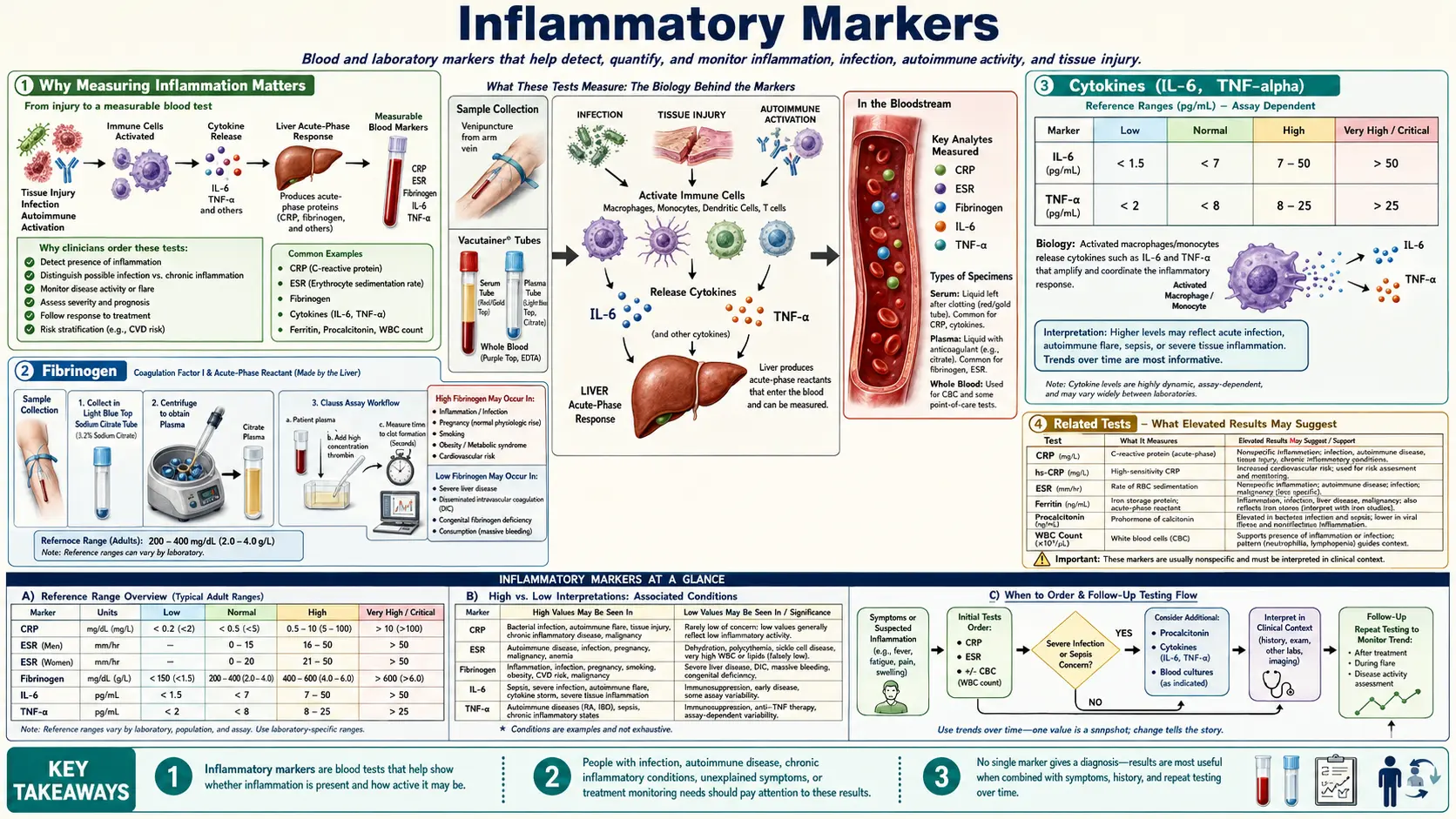
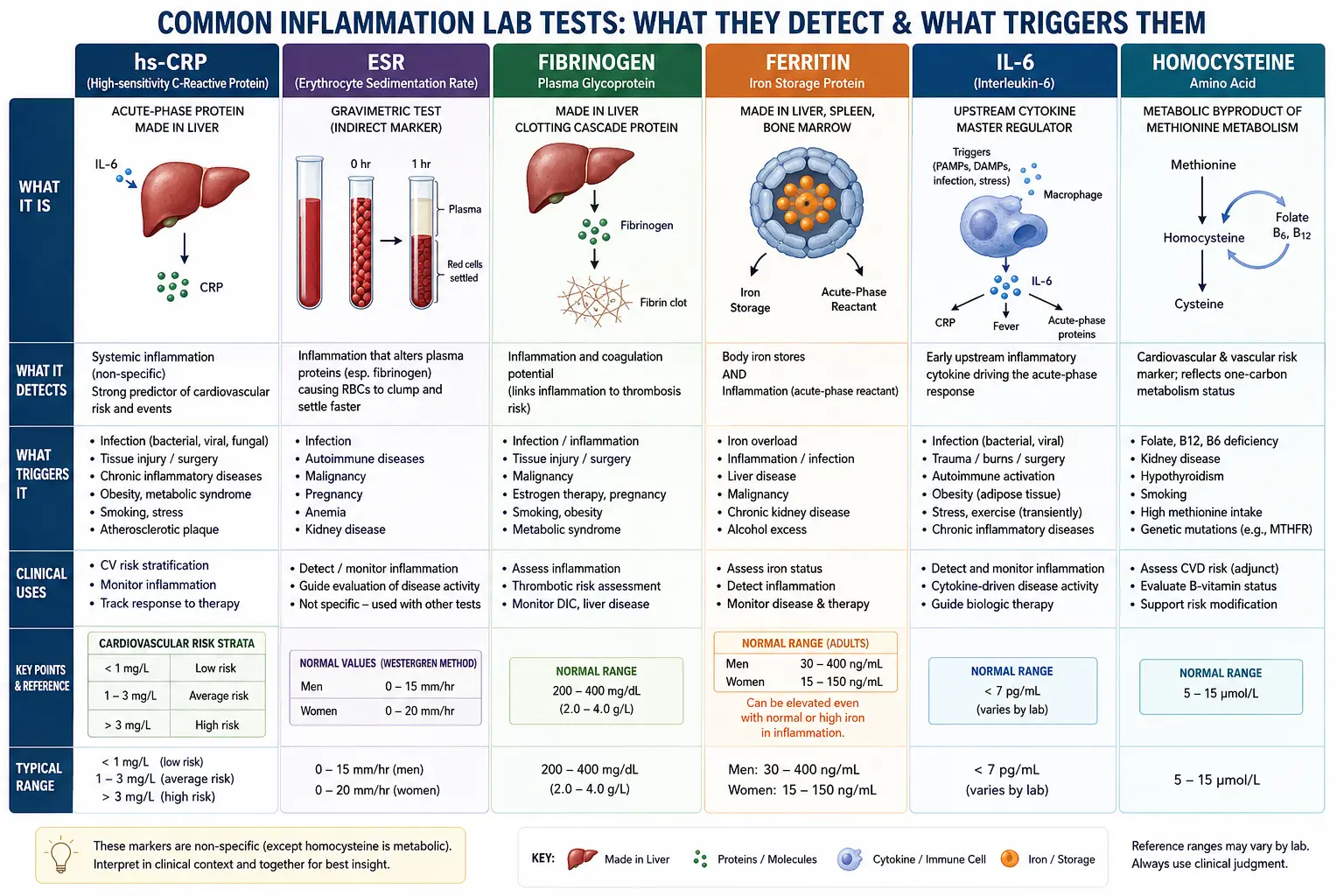
Table of Contents
- Why Measuring Inflammation Matters
- hs-CRP (High-Sensitivity C-Reactive Protein)
- ESR (Erythrocyte Sedimentation Rate)
- Homocysteine
- Fibrinogen
- Ferritin as an Inflammatory Marker
- Uric Acid
- ANA (Antinuclear Antibodies)
- Complement Levels (C3, C4)
- Cytokines (IL-6, TNF-alpha)
- Common Causes of Elevated Inflammation
- Anti-Inflammatory Natural Strategies
- When to Retest
- Related Tests
- Featured Videos
Why Measuring Inflammation Matters
Inflammation is increasingly recognized as the root mechanism underlying the vast majority of chronic diseases. From cardiovascular disease and type 2 diabetes to neurodegenerative conditions, autoimmune disorders, and even cancer, chronic low-grade inflammation acts as a silent driver of tissue damage and organ dysfunction. Unlike the acute inflammation that occurs after an injury or infection — which is a necessary and beneficial healing response — chronic systemic inflammation persists over months and years, gradually eroding health at the cellular level.
As a functional medicine practitioner, I consider inflammatory markers to be among the most valuable tools in clinical assessment. They provide a window into the body's internal environment that goes far beyond what symptoms alone can reveal. A patient may feel relatively well while harboring significant subclinical inflammation that, left unaddressed, will eventually manifest as serious disease.
The key inflammatory markers we evaluate include:
- hs-CRP — the single most important general inflammation marker
- ESR — a complementary measure of systemic inflammation
- Homocysteine — linking inflammation to cardiovascular and neurological risk
- Fibrinogen — bridging coagulation and inflammation
- Ferritin — often elevated in inflammatory states independent of iron status
- Uric acid — a metabolic and inflammatory marker
- ANA — screening for autoimmune-driven inflammation
- Complement (C3, C4) — assessing immune complex activity
- Cytokines (IL-6, TNF-alpha) — advanced markers of immune activation
By tracking these markers over time, we can identify inflammation early, determine its likely source, and measure the effectiveness of interventions — whether dietary, lifestyle-based, or supplemental. The goal is always to address the root cause rather than simply suppress symptoms.
hs-CRP (High-Sensitivity C-Reactive Protein)
High-sensitivity C-reactive protein is the single most important general inflammatory marker available in clinical practice. CRP is an acute-phase protein produced by the liver in response to interleukin-6 (IL-6) and other pro-inflammatory cytokines. The high-sensitivity assay allows detection of very low levels of CRP, making it invaluable for assessing chronic, low-grade inflammation that standard CRP tests would miss entirely.
Conventional vs. Functional Optimal Ranges
Conventional medicine considers hs-CRP values below 3.0 mg/L to be within the normal range. However, in functional medicine, we apply much tighter optimal ranges because research consistently demonstrates that even mildly elevated hs-CRP is associated with increased disease risk:
- Conventional normal range: 0.0 - 3.0 mg/L
- Functional optimal range: below 0.5 mg/L (ideally below 0.3 mg/L)
A patient with an hs-CRP of 2.5 mg/L would be told everything is "normal" by conventional standards, yet from a functional perspective, this level represents significant systemic inflammation that warrants investigation and intervention.
Cardiovascular Risk Stratification
The American Heart Association and Centers for Disease Control have established the following cardiovascular risk stratification based on hs-CRP:
- Low risk: below 1.0 mg/L
- Moderate risk: 1.0 - 3.0 mg/L
- High risk: above 3.0 mg/L
Elevated hs-CRP is an independent predictor of cardiovascular events including heart attack, stroke, and peripheral arterial disease. It adds prognostic value even when LDL cholesterol levels are within normal limits — reinforcing the understanding that atherosclerosis is fundamentally an inflammatory disease, not merely a cholesterol storage problem.
Values above 10.0 mg/L typically indicate an acute infection or significant tissue injury and should be interpreted in the context of the patient's clinical presentation. In such cases, retesting after the acute event resolves provides a more accurate baseline reading.
Clinical Considerations
Factors that can elevate hs-CRP include obesity (adipose tissue is a major source of inflammatory cytokines), smoking, periodontal disease, chronic infections, insulin resistance, poor sleep, high-sugar diets, and sedentary lifestyle. Statin medications lower hs-CRP independent of their cholesterol-lowering effects, which may account for some of their observed cardiovascular benefits. However, natural anti-inflammatory strategies can achieve similar reductions without the side effects associated with statins.
ESR (Erythrocyte Sedimentation Rate)
The erythrocyte sedimentation rate measures how quickly red blood cells settle to the bottom of a standardized tube over one hour. When inflammation is present, the liver produces increased amounts of fibrinogen and immunoglobulins. These proteins cause red blood cells to clump together (forming rouleaux), making them heavier and causing them to settle more rapidly.
What ESR Measures
ESR is a nonspecific marker of inflammation — it does not indicate the source or type of inflammation, only that inflammation is present. It tends to respond more slowly than hs-CRP, rising gradually over days to weeks and declining slowly after the inflammatory stimulus resolves. This makes ESR better suited for monitoring chronic inflammatory conditions rather than detecting acute changes.
Reference Ranges
- Conventional ranges: Men under 50: 0-15 mm/hr; Men over 50: 0-20 mm/hr; Women under 50: 0-20 mm/hr; Women over 50: 0-30 mm/hr
- Functional optimal range: below 10 mm/hr for both sexes
When ESR Is Most Useful
ESR is particularly valuable in the following clinical scenarios:
- Monitoring autoimmune conditions such as rheumatoid arthritis, lupus, and polymyalgia rheumatica
- Screening for temporal arteritis (giant cell arteritis) — markedly elevated ESR above 50 mm/hr is a hallmark finding
- Tracking chronic infections including osteomyelitis and tuberculosis
- Assessing treatment response — declining ESR over time indicates that an anti-inflammatory intervention is working
It is important to note that ESR naturally increases with age, and values tend to be higher in women than men. Anemia and pregnancy can also elevate ESR independent of inflammation, while polycythemia (elevated red blood cell count) can falsely lower it.
Homocysteine
Homocysteine is a sulfur-containing amino acid produced during the metabolism of methionine. While it is a normal intermediate in methylation pathways, elevated levels are directly toxic to blood vessel endothelium and are strongly associated with increased cardiovascular disease risk, neurological dysfunction, and overall inflammatory burden.
Cardiovascular and Neurological Risk
Elevated homocysteine (hyperhomocysteinemia) damages the vascular endothelium, promotes oxidative stress, increases platelet aggregation, and accelerates atherosclerosis. Research has linked high homocysteine to:
- Cardiovascular disease: coronary artery disease, stroke, deep vein thrombosis, and peripheral vascular disease
- Neurological conditions: cognitive decline, dementia, Alzheimer's disease, depression, and migraine
- Pregnancy complications: neural tube defects, preeclampsia, recurrent miscarriage, and placental abruption
- Bone health: increased fracture risk due to impaired collagen cross-linking
The B Vitamin Connection
Homocysteine metabolism depends critically on three B vitamins that serve as cofactors in its conversion pathways:
- Vitamin B12 (methylcobalamin): required by the enzyme methionine synthase to convert homocysteine back to methionine. Deficiency is common in vegans, the elderly, and those with pernicious anemia or impaired gastric acid production.
- Folate (5-MTHF / methylfolate): donates a methyl group in the same methionine synthase reaction. Individuals with MTHFR gene variants (particularly C677T and A1298C) have impaired ability to convert folic acid to its active methylfolate form and are at higher risk for elevated homocysteine. Supplementation with methylfolate (5-MTHF) rather than synthetic folic acid is essential for these individuals.
- Vitamin B6 (pyridoxal-5-phosphate): serves as a cofactor for cystathionine beta-synthase, which converts homocysteine to cystathionine in the transsulfuration pathway. This pathway ultimately produces glutathione, the body's master antioxidant.
Optimal Ranges
- Conventional normal range: 5 - 15 micromol/L
- Functional optimal range: 6 - 8 micromol/L
In clinical practice, I routinely see patients with homocysteine levels of 12-14 micromol/L who have been told their results are "normal." From a functional perspective, these levels represent a modifiable risk factor that responds remarkably well to targeted B vitamin supplementation — often with a combination of methylcobalamin (1000-5000 mcg), methylfolate (800-1000 mcg), and P5P (25-50 mg daily).
Fibrinogen
Fibrinogen is a glycoprotein produced by the liver that plays a dual role as both a clotting factor and an acute-phase reactant. It is the precursor to fibrin, the protein that forms the structural meshwork of blood clots. Because fibrinogen increases in response to inflammatory stimuli, elevated levels serve as both a marker of systemic inflammation and an independent cardiovascular risk factor.
Clotting and Inflammation
The relationship between clotting and inflammation is bidirectional. Inflammatory cytokines (particularly IL-6) stimulate the liver to produce more fibrinogen, which increases blood viscosity, promotes platelet aggregation, and creates a prothrombotic state. This elevated clotting tendency further damages blood vessel walls, triggering additional inflammation — a vicious cycle that drives atherosclerosis and increases the risk of heart attack and stroke.
Reference Ranges
- Conventional normal range: 200 - 400 mg/dL
- Functional optimal range: 200 - 300 mg/dL
Elevated fibrinogen is associated with smoking, obesity, diabetes, chronic kidney disease, and the metabolic syndrome. Nattokinase, a fibrinolytic enzyme derived from the Japanese food natto (fermented soybeans), has shown promise in reducing fibrinogen levels naturally. Omega-3 fatty acids, regular exercise, and smoking cessation also help lower fibrinogen.
Ferritin as an Inflammatory Marker
Ferritin is most commonly known as the body's primary iron storage protein. However, it is also an acute-phase reactant, meaning that serum ferritin levels rise in response to inflammation independent of actual iron stores. This dual nature makes ferritin interpretation more nuanced than many clinicians appreciate.
Beyond Iron Storage
When evaluating ferritin, it is essential to consider the inflammatory context. A patient with elevated ferritin and concurrent elevations in hs-CRP or ESR may not have iron overload at all — their ferritin may be elevated purely as an inflammatory response. Conversely, a patient with "normal" ferritin in the context of significant inflammation may actually be iron deficient, as the inflammatory elevation is masking a true deficiency.
Interpreting Ferritin in Context
- Conventional normal range: 12 - 300 ng/mL (men); 12 - 150 ng/mL (women)
- Functional optimal range: 40 - 100 ng/mL for both sexes
- Iron deficiency likely: below 30 ng/mL (even if within conventional range)
- Possible iron overload or inflammation: above 150 ng/mL in women, above 300 ng/mL in men
Conditions that elevate ferritin through inflammation rather than iron excess include liver disease, chronic infections, autoimmune conditions, malignancies, metabolic syndrome, and non-alcoholic fatty liver disease (NAFLD). To differentiate inflammatory ferritin elevation from true iron overload, additional tests such as serum iron, TIBC, transferrin saturation, and hs-CRP should be ordered concurrently.
Markedly elevated ferritin above 1000 ng/mL warrants urgent evaluation, as this can indicate hemochromatosis, hemophagocytic lymphohistiocytosis (HLH), adult-onset Still's disease, or hepatocellular damage.
Uric Acid
Uric acid is the end product of purine metabolism. While moderate levels have antioxidant properties, elevated uric acid (hyperuricemia) is a significant driver of inflammation, gout, kidney stones, and metabolic dysfunction. It is increasingly recognized as an independent risk factor for cardiovascular disease, hypertension, and chronic kidney disease.
Gout Risk and Metabolic Health
When serum uric acid exceeds the saturation point (approximately 6.8 mg/dL), monosodium urate crystals can deposit in joints and soft tissues, triggering the intensely painful inflammatory attacks characteristic of gout. However, even in the absence of gout, elevated uric acid has far-reaching metabolic consequences:
- Insulin resistance: elevated uric acid impairs nitric oxide production in the endothelium and contributes to insulin resistance, creating a feedback loop with fructose metabolism
- Hypertension: uric acid activates the renin-angiotensin system and directly damages renal vasculature
- Kidney disease: urate crystal deposition in renal tubules causes chronic inflammation and fibrosis
- Cardiovascular risk: elevated uric acid promotes endothelial dysfunction and vascular inflammation
- Non-alcoholic fatty liver disease: uric acid promotes hepatic lipogenesis and oxidative stress
Reference Ranges
- Conventional normal range: 3.5 - 7.2 mg/dL (men); 2.6 - 6.0 mg/dL (women)
- Functional optimal range: 3.5 - 5.5 mg/dL
Dietary factors that raise uric acid include excessive fructose intake (high-fructose corn syrup, fruit juices, agave nectar), alcohol (especially beer), and high-purine foods (organ meats, shellfish, sardines). Dehydration, kidney dysfunction, and certain medications (particularly thiazide diuretics) also contribute. Tart cherry extract, adequate hydration, vitamin C (500-1000 mg daily), and dietary modification are effective natural strategies for lowering uric acid.
ANA (Antinuclear Antibodies)
Antinuclear antibodies are autoantibodies directed against components of the cell nucleus, including DNA, histones, and other nuclear proteins. A positive ANA test indicates that the immune system is producing antibodies against the body's own tissues — a hallmark of autoimmune disease and a driver of chronic inflammation.
Autoimmune Screening
ANA testing is the primary screening tool for systemic autoimmune conditions. Results are reported as a titer (the highest dilution at which the antibody is still detected) and a pattern (which can suggest specific autoimmune conditions):
- Titer of 1:40 — weakly positive, found in up to 25-30% of healthy individuals; often not clinically significant
- Titer of 1:80 — low positive; warrants clinical correlation
- Titer of 1:160 or higher — more likely to be clinically significant
- Titer of 1:320 or higher — strongly suggestive of autoimmune disease
ANA Patterns and Associated Conditions
- Homogeneous pattern: associated with systemic lupus erythematosus (SLE) and drug-induced lupus
- Speckled pattern: associated with mixed connective tissue disease, Sjogren's syndrome, and SLE
- Nucleolar pattern: associated with scleroderma (systemic sclerosis)
- Centromere pattern: associated with limited scleroderma (CREST syndrome)
A positive ANA alone does not diagnose an autoimmune disease — it must be interpreted in the context of symptoms, clinical findings, and additional antibody testing (anti-dsDNA, anti-Smith, anti-SSA/SSB, anti-RNP, anti-Scl-70). From a functional medicine perspective, identifying a positive ANA early — even before a patient meets full diagnostic criteria for a named autoimmune condition — provides an opportunity to intervene with gut healing, dietary modifications, toxin avoidance, and immune modulation before the disease progresses.
Complement Levels (C3, C4)
The complement system is a cascade of proteins that form a critical part of innate immunity. C3 and C4 are the most commonly measured complement components and serve as important markers of immune complex-mediated inflammation and autoimmune activity.
Interpreting C3 and C4
Complement proteins can be either consumed (decreased) during active autoimmune flares or elevated as part of an acute-phase response:
- Low C3 and C4: suggests active complement consumption, commonly seen in lupus (SLE) flares, immune complex-mediated glomerulonephritis, cryoglobulinemia, and severe infections. Low complement with a positive ANA and anti-dsDNA antibodies is highly suggestive of active lupus.
- Low C4 with normal C3: may indicate hereditary C4 deficiency (relatively common) or early complement activation in lupus
- Elevated C3 and C4: as acute-phase reactants, complement levels can rise during infection, inflammation, and tissue injury. Persistent elevation may indicate chronic inflammatory states.
Reference Ranges
- C3 conventional range: 90 - 180 mg/dL
- C4 conventional range: 10 - 40 mg/dL
Serial complement measurements are particularly valuable for monitoring lupus disease activity — declining C3 and C4 levels often precede clinical flares, allowing for proactive treatment adjustments. In functional medicine, we also evaluate complement levels when a patient presents with recurrent infections, vasculitis symptoms, or unexplained inflammatory kidney disease.
Cytokines (IL-6, TNF-alpha)
Cytokines are signaling molecules produced by immune cells that orchestrate the inflammatory response. While dozens of cytokines have been identified, interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-alpha) are the most clinically relevant pro-inflammatory cytokines measured in advanced inflammatory testing.
Interleukin-6 (IL-6)
IL-6 is a pleiotropic cytokine that is the primary driver of CRP production in the liver. It plays a central role in the transition from acute to chronic inflammation and is elevated in virtually all chronic inflammatory conditions. IL-6 levels are associated with:
- Cardiovascular disease severity and prognosis
- Insulin resistance and type 2 diabetes
- Cancer progression and cachexia
- Depression and neuroinflammation
- Age-related inflammatory conditions (sometimes called "inflammaging")
Tumor Necrosis Factor-Alpha (TNF-alpha)
TNF-alpha is a potent pro-inflammatory cytokine produced primarily by macrophages and T cells. It plays a critical role in autoimmune inflammation and is the target of several biologic drugs used in rheumatoid arthritis, Crohn's disease, psoriasis, and ankylosing spondylitis. Elevated TNF-alpha is associated with:
- Autoimmune disease activity
- Chronic fatigue and fibromyalgia
- Insulin resistance (TNF-alpha from adipose tissue impairs insulin signaling)
- Neurodegeneration and blood-brain barrier disruption
- Mast cell activation and chronic inflammatory response syndrome (CIRS)
Advanced Testing Considerations
Cytokine testing is not part of routine bloodwork and is typically ordered through specialty labs. Results must be interpreted carefully, as cytokine levels can fluctuate significantly throughout the day and are affected by recent meals, exercise, stress, and sleep. Despite these limitations, cytokine panels provide invaluable information for patients with complex, multi-system inflammatory conditions that do not respond to standard interventions. Functional optimal levels for both IL-6 and TNF-alpha are as low as possible, ideally in the lower half of the laboratory reference range.
Common Causes of Elevated Inflammation
Identifying and addressing the root causes of inflammation is the cornerstone of functional medicine practice. Elevated inflammatory markers are never the disease themselves — they are signals pointing to underlying triggers that must be investigated systematically.
Dietary Causes
- Refined sugar and high-fructose corn syrup: directly stimulate IL-6 and TNF-alpha production
- Refined seed oils: excessive omega-6 fatty acids (soybean, corn, canola, safflower oil) shift the eicosanoid balance toward pro-inflammatory prostaglandins
- Trans fats: synthetic trans fats are potent activators of NF-kB, a master inflammatory signaling pathway
- Processed foods: contain emulsifiers, preservatives, and artificial additives that damage the gut lining and promote intestinal permeability
- Food sensitivities: gluten, dairy, eggs, soy, and corn are common triggers of immune-mediated inflammation in susceptible individuals
- Alcohol: increases intestinal permeability and promotes hepatic inflammation
Obesity and Metabolic Dysfunction
Visceral adipose tissue is not merely an energy storage depot — it is an active endocrine organ that produces large quantities of inflammatory cytokines (IL-6, TNF-alpha, leptin, resistin). Obesity is one of the most common and potent drivers of chronic systemic inflammation, and weight loss alone frequently normalizes inflammatory markers.
Chronic Infections
- Oral infections: periodontal disease is a major source of chronic inflammation linked to cardiovascular disease
- Gut dysbiosis: small intestinal bacterial overgrowth (SIBO), candida overgrowth, and parasitic infections
- Viral infections: chronic reactivation of Epstein-Barr virus (EBV), cytomegalovirus (CMV), and herpes viruses
- Tick-borne infections: Lyme disease and coinfections (Babesia, Bartonella, Ehrlichia)
- Helicobacter pylori: chronic gastric inflammation
Autoimmune Activation
Autoimmune conditions represent the immune system attacking the body's own tissues, producing chronic inflammation that can affect virtually any organ system. Identifying autoimmune-driven inflammation early through appropriate testing (ANA, specific autoantibodies, complement levels) is critical for implementing interventions before irreversible tissue damage occurs.
Stress and Poor Sleep
Chronic psychological stress activates the hypothalamic-pituitary-adrenal (HPA) axis, leading to cortisol dysregulation. While cortisol is normally anti-inflammatory, chronic stress leads to cortisol resistance at the cellular level, resulting in unchecked inflammation. Sleep deprivation — even a single night of poor sleep — measurably increases IL-6, TNF-alpha, and hs-CRP levels. Chronic sleep disruption compounds this effect dramatically.
Environmental Toxins
- Heavy metals: mercury, lead, arsenic, and cadmium activate NF-kB and promote oxidative stress
- Mold and mycotoxins: biotoxins from water-damaged buildings trigger chronic inflammatory response syndrome (CIRS)
- Pesticides and herbicides: glyphosate and other agrochemicals disrupt the gut microbiome and impair detoxification
- Plasticizers and endocrine disruptors: BPA, phthalates, and PFAS compounds promote immune dysregulation
- Air pollution: particulate matter and volatile organic compounds cause systemic inflammation via pulmonary exposure
Anti-Inflammatory Natural Strategies
Reducing chronic inflammation through natural means is one of the most powerful interventions in functional medicine. Rather than relying solely on anti-inflammatory medications that suppress symptoms without addressing root causes, we focus on strategies that resolve the underlying drivers of inflammation.
Omega-3 Fatty Acids
EPA and DHA from cold-water fish oil are among the most well-researched natural anti-inflammatory compounds. They compete with pro-inflammatory omega-6 fatty acids for incorporation into cell membranes and are converted into specialized pro-resolving mediators (SPMs) — resolvins, protectins, and maresins — that actively resolve inflammation rather than merely suppressing it. Therapeutic doses range from 2000-4000 mg of combined EPA/DHA daily for active inflammation. The omega-3 index (a measure of red blood cell membrane EPA/DHA content) should ideally be above 8%.
Turmeric and Curcumin
Curcumin, the active compound in turmeric, is a potent inhibitor of NF-kB and has been shown to reduce hs-CRP, IL-6, and TNF-alpha in clinical trials. Bioavailability is a critical consideration — standard curcumin powder is poorly absorbed. Formulations with enhanced bioavailability (such as those using piperine, phospholipid complexes, or nano-particle technology) are essential for therapeutic benefit. Effective doses typically range from 500-2000 mg of curcumin daily in a bioavailable form.
Anti-Inflammatory Diet
The foundation of any anti-inflammatory protocol is dietary modification. Key principles include:
- Eliminate refined sugars, processed foods, and refined seed oils
- Increase consumption of deeply colored vegetables and berries (rich in polyphenols and antioxidants)
- Include wild-caught fatty fish at least 3 times per week
- Use extra-virgin olive oil as the primary cooking and dressing fat
- Incorporate anti-inflammatory spices such as turmeric, ginger, rosemary, and cinnamon
- Identify and eliminate individual food sensitivities through an elimination diet or IgG food sensitivity testing
- Increase fiber intake to support a healthy gut microbiome
Exercise
Regular moderate exercise is one of the most effective anti-inflammatory interventions available. Physical activity reduces visceral adiposity, improves insulin sensitivity, and triggers the release of anti-inflammatory myokines from skeletal muscle. The optimal dose is 150-300 minutes per week of moderate aerobic activity combined with 2-3 sessions of resistance training. However, it is important to note that excessive, high-intensity exercise without adequate recovery can paradoxically increase inflammation.
Sleep Optimization
Sleep is a non-negotiable pillar of inflammation management. Adults should aim for 7-9 hours of quality sleep per night in a cool, dark environment. Key strategies include maintaining consistent sleep and wake times, minimizing blue light exposure in the evening, avoiding caffeine after noon, and addressing sleep apnea if present. Melatonin (0.5-3 mg) and magnesium glycinate (200-400 mg) at bedtime may be helpful for those with difficulty falling or staying asleep.
Stress Reduction
Chronic stress management is essential for controlling inflammation. Evidence-based stress reduction techniques include meditation and mindfulness practices, deep breathing exercises (particularly diaphragmatic and box breathing), yoga and tai chi, time in nature (forest bathing), social connection and community, and adaptogenic herbs such as ashwagandha, rhodiola, and holy basil. Even 10-15 minutes of daily meditation practice has been shown to measurably reduce inflammatory markers over time.
Additional Anti-Inflammatory Supplements
- Quercetin: 500-1000 mg daily — a flavonoid that stabilizes mast cells and inhibits inflammatory pathways
- Resveratrol: 200-500 mg daily — activates sirtuins and has broad anti-inflammatory effects
- Vitamin D: maintain serum levels of 50-80 ng/mL — deficiency is strongly associated with increased inflammation
- N-acetylcysteine (NAC): 600-1800 mg daily — supports glutathione production and reduces oxidative stress
- Boswellia (frankincense): 300-500 mg daily — inhibits 5-lipoxygenase and reduces inflammatory leukotrienes
- SPM Active (specialized pro-resolving mediators): supports the resolution phase of inflammation
When to Retest
The timing of retesting inflammatory markers depends on the specific marker, the severity of the initial elevation, and the interventions implemented. General guidelines include:
- hs-CRP: retest in 6-8 weeks after initiating anti-inflammatory interventions. If the initial value was elevated due to an acute infection, wait at least 2-3 weeks after full resolution before retesting to obtain a true baseline.
- ESR: retest in 8-12 weeks, as ESR changes more slowly than hs-CRP. For autoimmune conditions, ESR may be monitored every 3-6 months as part of disease activity assessment.
- Homocysteine: retest in 8-12 weeks after initiating B vitamin supplementation. Most patients will see significant improvement within this timeframe if the correct forms and doses of B12, folate, and B6 are used.
- Fibrinogen: retest in 8-12 weeks. This marker responds to exercise, omega-3 supplementation, and nattokinase over time.
- Ferritin: retest in 3-6 months. If elevated due to inflammation, ferritin should decline as other inflammatory markers normalize. If elevated due to iron overload, therapeutic phlebotomy and retesting every 3 months is standard.
- Uric acid: retest in 6-8 weeks after dietary and supplement interventions.
- ANA and complement levels: retest in 3-6 months for monitoring autoimmune disease activity. ANA titers may not change significantly, but trending complement levels (C3, C4) provides valuable information about disease flares.
- Cytokines: retest in 3-6 months given the higher cost and variability of these tests.
As a general principle, I recommend establishing a comprehensive inflammatory baseline, implementing targeted interventions for 2-3 months, retesting the most relevant markers, and then continuing to monitor quarterly to semi-annually as needed. Tracking trends over time is far more valuable than any single data point.
Related Tests
Inflammatory markers provide the most complete clinical picture when interpreted alongside other functional laboratory assessments. Consider ordering the following related tests to gain a comprehensive understanding of a patient's inflammatory status and its underlying drivers:
- Complete Blood Count (CBC) with differential: elevated white blood cells, neutrophil predominance, or eosinophilia can point to infection, autoimmunity, or allergic inflammation
- Comprehensive Metabolic Panel (CMP): liver enzymes (AST, ALT), kidney function, and glucose provide context for inflammatory findings
- Fasting insulin and glucose: insulin resistance is both a cause and consequence of chronic inflammation
- HbA1c: glycated hemoglobin reflects average blood sugar over 3 months and is associated with inflammatory burden
- Lipid panel (advanced): LDL particle number, oxidized LDL, and Lp(a) provide cardiovascular risk context alongside inflammatory markers
- Vitamin D (25-OH): deficiency is strongly linked to immune dysregulation and increased inflammation
- Omega-3 Index: measures red blood cell EPA/DHA content; low levels correlate with increased inflammation
- Thyroid panel: thyroid dysfunction (particularly Hashimoto's thyroiditis) is a common source of autoimmune inflammation
- Iron studies (serum iron, TIBC, transferrin saturation): essential for differentiating inflammatory ferritin elevation from true iron overload or deficiency
- GI-MAP or comprehensive stool analysis: identifies gut infections, dysbiosis, and intestinal permeability that commonly drive systemic inflammation
- Food sensitivity panel (IgG/IgA): identifies dietary triggers of immune-mediated inflammation
- Heavy metal testing: blood or urine toxic metals (mercury, lead, arsenic, cadmium) to identify environmental inflammatory triggers
- Organic acids test (OAT): evaluates mitochondrial function, neurotransmitter metabolites, and markers of yeast and bacterial overgrowth
- MTHFR and methylation gene panel: identifies genetic variants that impair homocysteine metabolism and detoxification capacity
By combining inflammatory markers with these complementary assessments, practitioners can build a thorough picture of the patient's inflammatory burden, identify the root causes driving inflammation, and design truly individualized treatment protocols that address the underlying dysfunction rather than merely suppressing symptoms.
Research Papers and References
The following are curated PubMed literature searches covering the evidence base for inflammatory markers testing, interpretation, and clinical management. Each link opens a live, filtered PubMed query so the results stay current as new studies are indexed.
- hs-CRP cardiovascular risk — PubMed literature search
- JUPITER trial CRP rosuvastatin — PubMed literature search
- ESR clinical utility — PubMed literature search
- IL-6 chronic inflammation — PubMed literature search
- TNF-alpha disease — PubMed literature search
- Inflammation and aging inflammaging — PubMed literature search
- Fibrinogen cardiovascular — PubMed literature search
- CRP meta-analysis mortality — PubMed literature search
- Omega-3 inflammation CRP — PubMed literature search
- Mediterranean diet inflammation — PubMed literature search
External Authoritative Resources
- MedlinePlus — Laboratory Tests Reference
- Lab Tests Online — Patient-Oriented Lab Test Reference (AACC)
- StatPearls / NCBI Bookshelf — Clinical Laboratory Reference
Connections
- Homocysteine
- Uric Acid
- Atherosclerosis
- Cardiovascular Disease
- Stroke
- Gout
- Lupus
- Arthritis
- Insulin Resistance
- Obesity
- Kidney Disease
- Anti-Inflammatory Diet
- Turmeric
- Omega-3 Fatty Acids
- Iron
- Oxidative Stress
- MCAS
- Chronic Fatigue Syndrome