IBD Biologics: TNF, IL-23, and Integrin Inhibitors
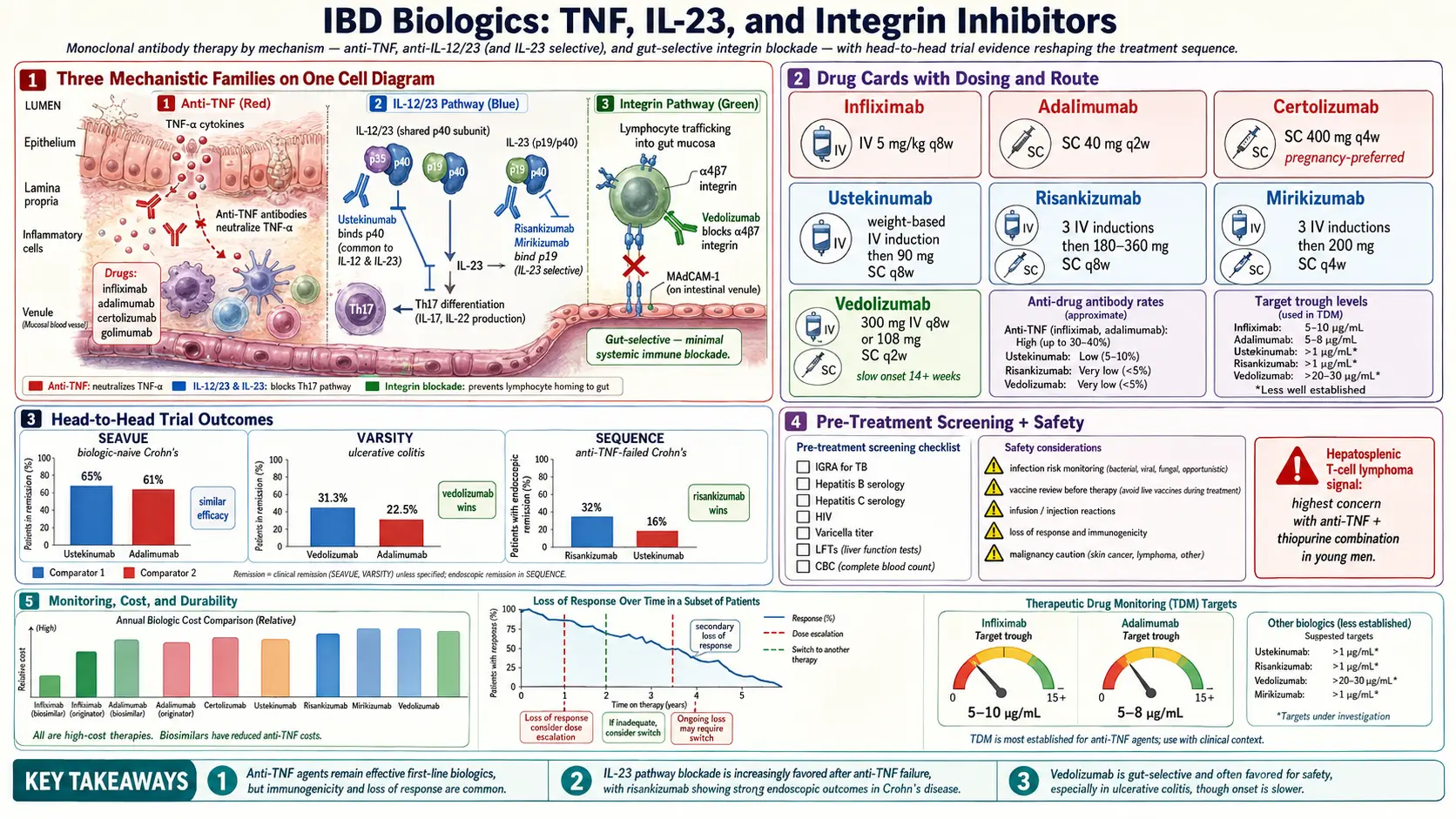
Table of Contents
- What Biologics Are and Why They Changed IBD
- Anti-TNF Agents: Infliximab, Adalimumab, Certolizumab, Golimumab
- Loss of Response, Antibody Formation, and Therapeutic Drug Monitoring
- IL-12/23 Blockade: Ustekinumab
- Selective IL-23 Inhibitors: Risankizumab and Mirikizumab
- Gut-Selective Integrin Blockade: Vedolizumab
- Head-to-Head Trials: SEAVUE, VARSITY, SEQUENCE
- Positioning: First-Line vs Rescue
- Infusion vs Self-Injection: Practical Differences
- Screening Before You Start: TB, Hepatitis B, Vaccines
- Infection Risk and How to Manage It
- The Malignancy Signal
- Insurance, Copay Assistance, and the Cost Reality
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
What Biologics Are and Why They Changed IBD
Biologics are large protein drugs — usually monoclonal antibodies — that lock onto one specific molecule in your immune system and block it. Unlike steroids or methotrexate, which dampen inflammation broadly, biologics hit a single target. That precision is what lets them calm a flaring gut without shutting down your whole defense system.
Before 1998, people with moderate-to-severe Crohn's disease or ulcerative colitis cycled through prednisone, azathioprine, and eventually surgery. Steroids worked but wrecked bones, eyes, skin, and mood. Surgery helped, but Crohn's came back near the new anastomosis, and UC patients faced a lifelong J-pouch. Then infliximab arrived, and for the first time ulcers on colonoscopy could visibly close in a few weeks. That's called mucosal healing, and it's now the benchmark of success.
The modern IBD biologics divide into three mechanistic families:
- Anti-TNF agents — block tumor necrosis factor alpha, the master inflammation cytokine.
- Anti-IL-12/23 and anti-IL-23 agents — block interleukin-23 (and in one case also IL-12), the cytokines that feed the Th17 pathway driving gut inflammation.
- Gut-selective integrin inhibitors — stop inflammatory white blood cells from crossing out of the bloodstream into the bowel wall, without touching immune traffic in the rest of the body.
Each family has different strengths, different risks, and different prices. The right choice depends on your disease behavior, your prior drug history, your other health conditions, and, unavoidably, what your insurance will cover.
Anti-TNF Agents: Infliximab, Adalimumab, Certolizumab, Golimumab
Tumor necrosis factor alpha (TNF-α) is a central inflammatory signal. In IBD, it is over-produced in the bowel wall, where it cranks up immune-cell recruitment, damages the epithelial barrier, and drives ulceration. Anti-TNF drugs are antibodies that mop up TNF before it can bind its receptor.
Infliximab (Remicade, and biosimilars Inflectra, Renflexis, Avsola). The original anti-TNF for IBD, approved for Crohn's in 1998 and UC in 2005. Given by IV infusion over about 2 hours, with a loading schedule at weeks 0, 2, and 6, then a maintenance infusion every 8 weeks. Standard dose is 5 mg/kg, doubled to 10 mg/kg if response is partial. Biosimilars are functionally identical and cost substantially less — most insurers now require a biosimilar first.
Adalimumab (Humira and biosimilars Amjevita, Cyltezo, Hyrimoz, and others). Subcutaneous self-injection with a prefilled pen. Loading dose of 160 mg at week 0, 80 mg at week 2, then 40 mg every other week. Some patients need weekly dosing to stay in remission. Humira lost U.S. exclusivity in 2023 and biosimilar adalimumab is now widely available, though uptake has been slower than expected.
Certolizumab pegol (Cimzia). A pegylated anti-TNF Fab fragment — meaning it lacks the antibody's Fc tail. That makes it the preferred anti-TNF in pregnancy because very little of it crosses the placenta. Given as a 400 mg subcutaneous injection at weeks 0, 2, and 4, then every 4 weeks. Approved in Crohn's, not in UC.
Golimumab (Simponi). Monthly subcutaneous injection, approved for UC but not Crohn's. Less commonly used than infliximab or adalimumab because head-to-head comparisons suggest slightly lower efficacy in moderate-to-severe UC.
Across the class, response rates in moderate-to-severe disease run roughly 60–70% at week 6, with about 40–45% of patients in steroid-free remission at one year. Infliximab is the strongest rescue drug for acute severe ulcerative colitis in hospital, where it competes with cyclosporine as a last-ditch option before colectomy.
Loss of Response, Antibody Formation, and Therapeutic Drug Monitoring
The hardest part of anti-TNF therapy is that it often stops working. Roughly one in three patients loses response within the first year, and about 10–15% per year thereafter. There are two main reasons, and telling them apart changes what you do next.
Mechanistic loss of response. The drug is in your bloodstream at an adequate level, but your gut inflammation is no longer TNF-driven. Adding more drug will not help; you need to switch mechanism (to an IL-23 or integrin inhibitor, or to a JAK inhibitor).
Pharmacokinetic loss of response. The drug level is too low, either because you are metabolizing it fast (heavy inflammation burns through drug) or because you have made anti-drug antibodies — your immune system has decided the biologic is foreign and is clearing it before it can work. About 10–20% of infliximab users and 5–10% of adalimumab users develop neutralizing antibodies, usually within the first year.
The tool to sort this out is therapeutic drug monitoring (TDM): a trough blood level drawn just before the next dose, plus an anti-drug antibody assay. Target trough levels are roughly 5–10 µg/mL for infliximab and 7.5–12 µg/mL for adalimumab in maintenance. If your level is high and you are still flaring, the problem is mechanism — switch drugs. If your level is low and antibodies are negative, you can dose-escalate (shorter interval or higher dose). If your level is low and antibodies are high, the drug is being destroyed — switch within class (to adalimumab if you were on infliximab, or vice versa) or out of class entirely. Adding a low-dose immunomodulator like methotrexate or azathioprine reduces antibody formation by about half but comes with its own side-effect trade-offs.
TDM costs roughly $100–$300 per panel and is covered by most insurers when a drug is failing. Ask for it before you conclude a biologic is not working — plenty of patients are switched off a perfectly viable drug that just needed a dose bump.
IL-12/23 Blockade: Ustekinumab
Ustekinumab (Stelara) targets the p40 subunit shared by interleukin-12 and interleukin-23. Blocking p40 dampens both the Th1 (IL-12 driven) and Th17 (IL-23 driven) arms of the immune response. It is FDA-approved for Crohn's disease (2016) and ulcerative colitis (2019).
The dosing is distinctive. Induction is a one-time weight-based IV infusion (roughly 260–520 mg depending on body weight), then maintenance is a 90 mg subcutaneous injection every 8 weeks at home. That means you touch an infusion chair exactly once — a strong convenience advantage for patients who do not want recurring infusion appointments but dislike the every-other-week cadence of adalimumab.
Clinical response rates sit around 55–65% in moderate-to-severe disease with one-year steroid-free remission near 40%. It is generally better tolerated than anti-TNF, with lower rates of serious infection and no clear lymphoma signal. Ustekinumab is often the choice for patients with a history of recurrent infections, older adults, those with prior TB exposure, and patients who prefer a slower injection schedule. Biosimilar ustekinumab entered the U.S. market in 2025 and is beginning to shift formulary preferences.
Selective IL-23 Inhibitors: Risankizumab and Mirikizumab
Newer research showed that blocking IL-12 probably is not necessary for gut healing — IL-23 alone is the driver. Targeting only the p19 subunit unique to IL-23 turned out to produce stronger remission rates than p40 blockade, with similar safety.
Risankizumab (Skyrizi). Approved for Crohn's (2022) and UC (2024). Induction is three IV infusions at weeks 0, 4, and 8 (600 mg each), then maintenance is a 180 mg or 360 mg subcutaneous injection every 8 weeks from an on-body pump device. In the ADVANCE and MOTIVATE trials, about 40–45% of Crohn's patients reached clinical remission at week 12, higher than historical ustekinumab response in refractory populations.
Mirikizumab (Omvoh). Approved for UC (2023) and Crohn's (2025). Induction is three IV infusions at weeks 0, 4, and 8 (300 mg each), then maintenance is a 200 mg subcutaneous injection every 4 weeks at home. The LUCENT UC trial showed clinical remission at week 52 in about 50% of induction responders. Notably strong on bowel-urgency scores — many UC patients describe it as "getting their bathroom life back" within weeks.
Both drugs have been remarkably clean safety-wise. Rates of serious infection are in the low single digits per 100 patient-years and similar to placebo in trials. No malignancy signal has emerged. Injection-site reactions are the most common nuisance.
Gut-Selective Integrin Blockade: Vedolizumab
Vedolizumab (Entyvio) works by a completely different strategy. It is a monoclonal antibody against the α4β7 integrin, a docking molecule on white blood cells. α4β7 is the specific key that unlocks entry to the gut mucosa through a protein called MAdCAM-1. Block it, and inflammatory T cells physically cannot leave the bloodstream and cross into the bowel wall. The immune system in your brain, lungs, and skin is left alone because those tissues use different integrin partners.
The result is the best overall safety record in the IBD biologic world. Vedolizumab has not shown increased rates of serious infections, tuberculosis reactivation, or malignancy in long-term registries. There was early worry about progressive multifocal leukoencephalopathy (PML) because the related drug natalizumab caused it, but after more than a decade of use with hundreds of thousands of patients, no confirmed PML case has been tied to vedolizumab.
Dosing: 300 mg IV infusion at weeks 0, 2, and 6, then every 8 weeks. A subcutaneous version (108 mg every 2 weeks) was approved in 2023 for maintenance and is a good option for patients who want to escape the infusion center.
The trade-off is slower onset. Vedolizumab can take 14 weeks or longer to show meaningful benefit, particularly in Crohn's. Efficacy is higher in UC than Crohn's, and higher in biologic-naïve patients than in those who have already failed an anti-TNF. For the right patient — older, infection-prone, cancer history, or with latent TB — its safety profile makes it the first-line choice over any alternative.
Head-to-Head Trials: SEAVUE, VARSITY, SEQUENCE
Most IBD trials compare a drug to placebo. Three recent studies did something rarer — they put two biologics against each other, which is what patients and clinicians actually need to decide.
SEAVUE (2022). Ustekinumab vs adalimumab in biologic-naïve Crohn's disease. After one year, clinical remission rates were essentially identical (about 65% vs 61%). Neither drug won. The practical implication: for a patient just starting biologics, either is reasonable, and the choice can come down to route, price, and safety preference.
VARSITY (2019). Vedolizumab vs adalimumab in moderate-to-severe ulcerative colitis. Vedolizumab produced higher clinical remission at week 52 (31.3% vs 22.5%) and higher mucosal healing (39.7% vs 27.7%). This was the study that moved vedolizumab to a genuine first-line position in UC, especially for patients without extraintestinal disease to worry about.
SEQUENCE (2023). Risankizumab vs ustekinumab in Crohn's patients who had failed at least one anti-TNF. Risankizumab was superior for endoscopic remission at week 48 (32% vs 16%) and comparable on clinical remission. That result pushed risankizumab into the second-line slot after anti-TNF failure in Crohn's, where ustekinumab had previously dominated.
These head-to-head data are shifting practice fast. If your gastroenterologist's choice seems to track an older algorithm (TNF first, then ustekinumab, then vedolizumab), ask them about SEQUENCE and VARSITY — a more current sequence in many cases puts vedolizumab or risankizumab earlier.
Positioning: First-Line vs Rescue
There is no single correct order. A reasonable framework, based on 2024–2026 guidelines and real-world practice:
Crohn's disease, moderate-to-severe. First line is typically anti-TNF (infliximab with an immunomodulator, or adalimumab) for patients with perianal or fistulizing disease, or risankizumab for those without fistulas who prefer self-injection. Ustekinumab is a strong first-line alternative in older patients or those with infection concerns. Vedolizumab is often held for later because of slower onset, though it is reasonable first-line in milder disease.
Ulcerative colitis, moderate-to-severe. Vedolizumab has moved to a common first-line position after VARSITY, especially in patients without extraintestinal manifestations. Infliximab remains first-line for acute severe UC and for patients with coexisting axial spondyloarthritis (see ankylosing spondylitis, where TNF works and vedolizumab does not). Mirikizumab is an increasingly popular choice for urgency-predominant UC.
Rescue after anti-TNF failure. Risankizumab (in Crohn's, per SEQUENCE) or ustekinumab. Vedolizumab efficacy drops somewhat after anti-TNF exposure but is still worth trying if the priority is safety. See JAK inhibitors and S1P modulators for the small-molecule alternatives that also come into play at this stage.
Extraintestinal manifestations matter. If you have uveitis, axial arthritis, or erythema nodosum alongside IBD, anti-TNF covers both; vedolizumab usually does not. See extraintestinal manifestations for the full overlap map.
Infusion vs Self-Injection: Practical Differences
This is more than a convenience issue — it affects adherence, cost, and quality of life.
Infusion drugs (infliximab, vedolizumab IV, risankizumab induction). Given at an infusion center or hospital outpatient unit. Each visit is 2–4 hours once the needle is in. Pro: a nurse watches for reactions, your drug is definitely in you, and many patients enjoy the routine and the protected time. Con: travel, parking, time off work, and the facility fee — which can be higher than the drug itself on a hospital bill.
Self-injection drugs (adalimumab, certolizumab, golimumab, ustekinumab maintenance, risankizumab maintenance, mirikizumab maintenance, vedolizumab SC). A prefilled pen or on-body injector you use at home, usually every 2, 4, or 8 weeks. Pro: no travel, no facility fee, schedule flexibility. Con: you have to remember the date, store the pen refrigerated, and some people find the injection stings (most come at refrigerator temperature; letting the pen warm on the counter for 30 minutes before use reduces sting significantly).
If needle phobia is a real issue, infusions can be preferable because the needle is only placed once by someone else. If travel or time off work is the barrier, self-injection wins. Many people end up with a hybrid pattern: IV induction at a center, then self-injection maintenance at home.
Screening Before You Start: TB, Hepatitis B, Vaccines
Every biologic requires a pre-treatment screening workup. Do not skip this — reactivation of latent infection is one of the few genuinely dangerous complications, and it is almost entirely preventable.
- Tuberculosis. Either an interferon-gamma release assay (Quantiferon or T-SPOT) or a tuberculin skin test (PPD). Chest X-ray if there is any positive result or significant exposure history. Latent TB must be treated for at least one month of isoniazid (or another approved regimen) before biologics begin. Anti-TNF drugs carry the highest reactivation risk; vedolizumab the lowest.
- Hepatitis B. HBsAg, anti-HBc, and anti-HBs. If you are a hepatitis B carrier (HBsAg positive or isolated anti-HBc positive), you need prophylactic entecavir or tenofovir alongside the biologic to prevent reactivation hepatitis, which can be fatal.
- Hepatitis C. Screen once. Not a contraindication, but worth knowing.
- HIV. Screen if risk factors or not recently tested.
- Varicella (chickenpox). If you have not had chickenpox or the vaccine, get vaccinated at least 4 weeks before starting a biologic — live vaccines cannot be given once you are immunosuppressed.
- Routine vaccines. Update flu (annually, inactivated), pneumococcal (PCV20 or PCV15 followed by PPSV23), shingles (Shingrix, which is non-live and safe during biologic therapy), HPV if age-appropriate. Ideally update before starting.
- Skin exam. Baseline dermatology evaluation is reasonable given the small melanoma and non-melanoma skin cancer signal on long-term anti-TNF.
Infection Risk and How to Manage It
Biologics raise the risk of serious infection, but less than patients usually fear and less than chronic steroids do. The absolute rate in trials is roughly 2–5 serious infections per 100 patient-years on anti-TNF, 1–3 on ustekinumab, and about 1–2 on vedolizumab — in all cases substantially lower than long-term prednisone.
Practical rules:
- Fever over 101°F or rigors. Call your IBD team the same day. Do not take your next dose until cleared.
- Dental abscess, skin infection, UTI, sinusitis. Treat promptly with antibiotics. Short courses are fine; you do not need to pause your biologic for a simple UTI.
- Surgery. Biologics are generally paused 1–2 half-lives before elective surgery and resumed once the wound is clean. Infliximab is usually held for 4–6 weeks beforehand; adalimumab about 2 weeks. Emergency surgery is not a reason to panic — evidence does not show biologics increase major surgical complications in IBD.
- Travel. Live vaccines (yellow fever, oral typhoid, MMR boost) are off-limits during biologic therapy. Plan travel vaccines before you start, or take an inactivated alternative.
- Pregnancy. Most biologics can be continued through pregnancy — uncontrolled IBD is more dangerous to the fetus than the drug. Certolizumab is preferred in the third trimester because of minimal placental transfer. Discuss with a maternal-fetal medicine specialist.
The Malignancy Signal
A realistic, non-alarming summary: anti-TNF drugs carry a small but real signal for certain rare cancers, particularly hepatosplenic T-cell lymphoma (HSTCL) in young men on combination therapy with a thiopurine. The absolute risk is extremely low — on the order of 1 in 10,000 patient-years — but the cancer is aggressive. This is why many centers now avoid long-term thiopurine co-therapy in young men when possible, using methotrexate or biologic monotherapy instead.
Melanoma and non-melanoma skin cancer rates are modestly increased on anti-TNF. Annual dermatology checks are reasonable. Lymphoma overall is slightly elevated above the background IBD rate, which is itself slightly above the general population, but the absolute numbers remain small.
Ustekinumab, risankizumab, mirikizumab, and vedolizumab have not shown clear malignancy signals in the time they have been monitored. This is one of the genuine advantages of the newer mechanisms and is a significant reason gastroenterologists now reach for them earlier in patients with cancer histories or strong family histories.
Insurance, Copay Assistance, and the Cost Reality
Biologics are expensive. List prices run roughly $40,000 to $90,000 per year per drug at wholesale. Nobody should pay that, but navigating who does pay requires active work.
Commercial insurance. Most plans cover biologics after step therapy — you may be required to fail a less expensive option first, or to use a biosimilar before the brand. Prior authorization is nearly universal. Your GI office should have a dedicated biologic coordinator; if they do not, ask for the manufacturer's patient support line.
Copay assistance cards. Every brand biologic has one. AbbVie's Humira Complete, J&J's With Me (ustekinumab, infliximab), Takeda's EntyvioConnect, and similar programs typically reduce your out-of-pocket cost to $5 per dose if you have commercial insurance. Federally insured patients (Medicare, Medicaid, VA, Tricare) cannot use these cards by law, but manufacturer patient assistance foundations often cover qualifying low-income patients directly.
Copay accumulator / maximizer programs. Watch out. Some insurers have started not counting copay assistance toward your deductible or out-of-pocket maximum. If this applies to your plan, the manufacturer's $5 cost to you can suddenly explode to several thousand dollars mid-year when the assistance cap is reached. Ask your benefits administrator directly whether copay assistance applies to your deductible.
Charitable foundations. The HealthWell Foundation, the PAN Foundation, and the Crohn's & Colitis Foundation all run copay assistance funds, but they open and close quickly as money runs out. Sign up for alerts.
Site-of-care issues. Hospital outpatient infusion charges can be 2–4x higher than freestanding infusion center charges for the exact same infliximab. If your facility fees are eating your deductible, ask about transferring infusions to a community infusion center — many insurers now require this.
Biosimilars are not optional. If your insurer switches you from Remicade to Inflectra or from Humira to Cyltezo, it is almost always fine. Extensive switching studies show no loss of efficacy or change in safety. The savings are real and shared across the system.
Key Research Papers
- Targan SR, et al. A short-term study of chimeric monoclonal antibody cA2 to tumor necrosis factor alpha for Crohn's disease. N Engl J Med. 1997. (Original infliximab trial)
- Feagan BG, et al. Vedolizumab as induction and maintenance therapy for ulcerative colitis. N Engl J Med. 2013. (GEMINI 1)
- Feagan BG, et al. Ustekinumab as induction and maintenance therapy for Crohn's disease. N Engl J Med. 2016. (UNITI)
- Sands BE, et al. Vedolizumab versus adalimumab for moderate-to-severe ulcerative colitis. N Engl J Med. 2019. (VARSITY)
- Sands BE, et al. Ustekinumab versus adalimumab for induction and maintenance therapy in biologic-naïve patients with moderately to severely active Crohn's disease. Lancet. 2022. (SEAVUE)
- D'Haens G, et al. Risankizumab as induction therapy for moderately to severely active Crohn's disease. Lancet. 2022. (ADVANCE / MOTIVATE)
- D'Haens G, et al. Mirikizumab as induction and maintenance therapy for ulcerative colitis. N Engl J Med. 2023. (LUCENT)
- Peyrin-Biroulet L, et al. Risankizumab versus ustekinumab for moderate-to-severe Crohn's disease. N Engl J Med. 2024. (SEQUENCE)
- Baert F, et al. Influence of immunogenicity on the long-term efficacy of infliximab in Crohn's disease. N Engl J Med. 2003. (Antibody formation and loss of response)
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on IBD biologics, positioning, and safety:
- Infliximab in Crohn's and ulcerative colitis maintenance
- Adalimumab and therapeutic drug monitoring in IBD
- Vedolizumab long-term safety
- Ustekinumab real-world outcomes in Crohn's disease
- Risankizumab in Crohn's and the SEQUENCE trial
- Mirikizumab in ulcerative colitis
- Anti-TNF and hepatitis B reactivation
- IBD biologic biosimilar switching studies
- Hepatosplenic T-cell lymphoma risk with thiopurines and anti-TNF
- Vedolizumab subcutaneous maintenance
Connections
- Inflammatory Bowel Disease Overview
- Crohn's vs Ulcerative Colitis: Key Differences
- Diagnostic Workup: Calprotectin, Colonoscopy, MRE
- JAK Inhibitors and S1P Modulators
- Diet for IBD: SCD and Mediterranean
- IBD Surgery Decisions
- Extraintestinal Manifestations: Joints, Skin, Eyes
- IBD Fatigue, Anemia, and Nutrient Deficiencies
- Pregnancy and IBD
- Crohn's Disease
- Ulcerative Colitis
- Arthritis
- Ankylosing Spondylitis
- AS and IBD Overlap
- Cancer
- Tuberculosis