JAK Inhibitors and S1P Modulators for IBD
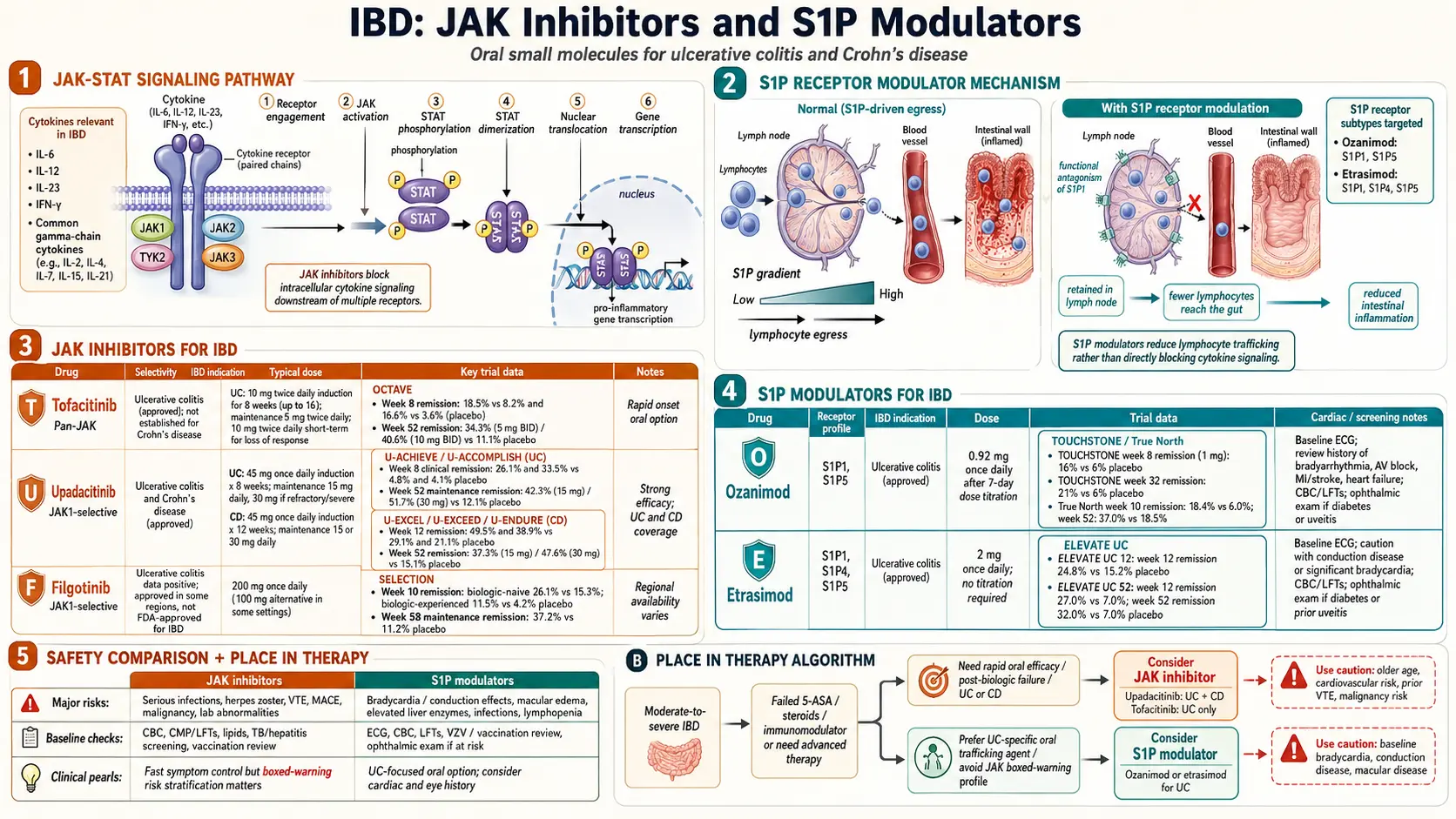
Table of Contents
- Why Pills Now Matter — Small Molecules vs Biologics
- JAK Inhibitors: How They Work
- Tofacitinib (Xeljanz) and the OCTAVE Trials
- Upadacitinib (Rinvoq) and the U-ACHIEVE/U-ACCOMPLISH Program
- Filgotinib — Approved Abroad, Not in the U.S.
- JAK Black-Box Warnings — MACE, VTE, Malignancy
- Cardiovascular Screening Before Starting
- S1P Receptor Modulators: How They Work
- Ozanimod (Zeposia) and True North
- Etrasimod (Velsipity) and ELEVATE UC
- S1P Cardiac Monitoring and Lymphocyte Drop
- Where Oral Agents Fit in Your Treatment Plan
- Pregnancy and Breastfeeding
- Cost Realities and Insurance
- Practical Questions to Ask Your GI
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
Why Pills Now Matter — Small Molecules vs Biologics
For twenty years, advanced IBD therapy meant injections or infusions. TNF inhibitors (infliximab, adalimumab), IL-23 inhibitors (ustekinumab, risankizumab), and integrin inhibitors (vedolizumab) are all biologics — large, engineered antibody proteins that must be delivered under the skin or into a vein because your stomach would digest them. If you want the science on those agents, see the biologics article.
JAK inhibitors and S1P receptor modulators are different. They are small-molecule drugs you swallow once or twice a day. No injections, no infusion chairs, no needles. A small molecule is exactly what it sounds like — a compact chemical (hundreds of daltons, not the 150,000 of a monoclonal antibody) that survives the gut, crosses cell membranes, and blocks a specific intracellular target.
The trade-offs are real. Biologics hit one precise target outside the cell; small molecules hit pathways inside the cell and tend to have broader downstream effects. That is why the FDA has placed boxed warnings on the JAK class and why cardiac monitoring is required for some S1P drugs. For many patients, though, the convenience and speed of response make these agents life-changing — especially when biologics have failed, when needles are a dealbreaker, or when disease has to be controlled fast.
JAK Inhibitors: How They Work
JAK stands for Janus kinase. Inside your immune cells, JAKs sit under the receptors that inflammatory cytokines dock onto. When a cytokine like IL-6, IL-2, IL-15, or interferon-gamma binds its receptor, the JAK enzyme gets switched on and phosphorylates a family of messenger proteins called STATs. The STATs march into the nucleus and turn on inflammatory genes. Block JAK, and you mute dozens of cytokine signals at once.
There are four JAKs: JAK1, JAK2, JAK3, and TYK2. Each pairs with specific receptors. The IBD-relevant drugs try to hit JAK1 preferentially — because hitting JAK2 too hard interferes with red-blood-cell production (causing anemia) and hitting JAK3 too hard suppresses immune development. Newer JAK inhibitors are more selective for JAK1, which is why upadacitinib and filgotinib tend to have cleaner side-effect profiles than older, less selective tofacitinib.
Because JAK inhibitors are pills, they kick in fast. Clinical improvement is often visible within 2–3 days — unusual in a field where biologics take weeks. That makes JAKs attractive when a UC flare is closing in on surgery and time matters.
Tofacitinib (Xeljanz) and the OCTAVE Trials
Tofacitinib was the first JAK inhibitor approved for ulcerative colitis (FDA, May 2018). It blocks JAK1 and JAK3 roughly equally. The approval rested on the OCTAVE trial program — three large randomized studies published by Sandborn and colleagues in the New England Journal of Medicine in 2017.
- OCTAVE Induction 1 and 2. Moderate-to-severe UC patients received 10 mg twice daily for 8 weeks. Remission at week 8: 18.5% of tofacitinib patients vs 8.2% on placebo in one trial, 16.6% vs 3.6% in the other.
- OCTAVE Sustain. Responders were re-randomized to 5 mg twice daily, 10 mg twice daily, or placebo for 52 weeks. Remission at one year was 34.3% on 5 mg, 40.6% on 10 mg, and only 11.1% on placebo.
The dosing matters. Standard induction is 10 mg twice daily for 8 weeks, then step down to 5 mg twice daily for maintenance. Patients who do not fully respond can stay on 10 mg twice daily, but the FDA now recommends using the lowest effective dose because of the cardiovascular and thrombosis signal discussed below.
Tofacitinib is approved for UC only, not Crohn's disease. Early Crohn's trials did not hit endpoints, and the drug never received that indication.
Upadacitinib (Rinvoq) and the U-ACHIEVE/U-ACCOMPLISH Program
Upadacitinib is a selective JAK1 inhibitor, FDA-approved for ulcerative colitis (March 2022) and Crohn's disease (May 2023). The UC data come from the twin U-ACHIEVE and U-ACCOMPLISH induction trials plus U-ACHIEVE Maintenance; the Crohn's data come from U-EXCEL, U-EXCEED, and U-ENDURE.
- U-ACHIEVE induction. 8 weeks of 45 mg once daily produced clinical remission in 26% vs 5% on placebo.
- U-ACCOMPLISH. Similar design, similar numbers — remission rates around 33% vs 4%.
- U-ACHIEVE Maintenance. Responders re-randomized to 15 mg or 30 mg daily. Remission at 52 weeks: 42% on 15 mg, 52% on 30 mg, vs 12% on placebo.
- U-EXCEL and U-ENDURE (Crohn's). 45 mg induction produced clinical remission in about 50% of patients by week 12, and 15–30 mg maintenance sustained it in roughly 40% at one year.
Upadacitinib's JAK1 selectivity means fewer hematologic side effects than tofacitinib, but the same class warnings still apply. It is the only JAK inhibitor currently approved for both UC and Crohn's, which is a real advantage for patients whose diagnosis shifts or whose disease straddles the line. For a refresher on those differences, see the Crohn's vs UC article.
Filgotinib — Approved Abroad, Not in the U.S.
Filgotinib is another selective JAK1 inhibitor, developed by Galapagos and Gilead. The SELECTION trial showed meaningful benefit in UC — induction remission of 26% with 200 mg daily vs 15% on placebo, and maintenance remission around 37% at one year.
The European Medicines Agency approved filgotinib for UC in 2021. In the United States, however, the FDA declined approval because of concerns about testicular toxicity seen in animal studies at high doses. Clinically, the MANTA safety study did not confirm those concerns in humans, but the regulatory path stalled and Gilead returned U.S. rights to Galapagos. As of 2026, U.S. patients cannot access filgotinib for IBD; European and UK patients can.
JAK Black-Box Warnings — MACE, VTE, Malignancy
Every JAK inhibitor used in IBD carries an FDA boxed warning — the most serious regulatory warning available. The warning was added in 2021 after the ORAL Surveillance trial of tofacitinib in rheumatoid arthritis showed excess events compared with TNF inhibitors. That trial was not in IBD, but the FDA applied the warning across the whole JAK class:
- Major adverse cardiovascular events (MACE). Heart attack, stroke, or cardiovascular death. The absolute excess risk in ORAL Surveillance was roughly 0.33 events per 100 patient-years on tofacitinib vs 0.18 on TNF blockers — small in absolute terms but real.
- Venous thromboembolism (VTE). Blood clots in the legs (DVT) or lungs (pulmonary embolism). The signal was strongest on high-dose tofacitinib (10 mg twice daily) in patients over 50 with cardiovascular risk factors.
- Malignancy. Higher rates of lymphoma and non-melanoma skin cancer compared with TNF inhibitors. Lung cancer in people who smoke or have smoked was specifically flagged.
- Serious infections. Herpes zoster (shingles) is the most common and is a class effect — roughly 3–5% of JAK-treated patients develop zoster over a year. Tuberculosis reactivation is possible but rare.
- Death from any cause. ORAL Surveillance showed a small all-cause mortality increase in older RA patients.
Context matters. The ORAL Surveillance population was older RA patients with baseline cardiovascular disease — not the typical UC patient. IBD-specific cohorts have so far shown milder signals, and many gastroenterologists argue the JAK boxed warning overestimates risk for the average IBD patient under 50 with no smoking history. Still, the warning is there, and your doctor is required to discuss it with you.
Cardiovascular Screening Before Starting
Most gastroenterologists now run a short checklist before starting any JAK inhibitor:
- Age and smoking. Over 50 plus current or former smoker = extra caution; many clinicians will recommend a different class first.
- Cardiovascular risk scoring. An ASCVD 10-year risk calculator result above roughly 7.5% prompts cardiology input before starting.
- Lipid panel. JAK inhibitors raise total, LDL, and HDL cholesterol. Baseline and 8-week follow-up lipids are standard.
- History of blood clots. Prior DVT, PE, or a known clotting disorder is a relative contraindication.
- Shingles vaccine. The recombinant zoster vaccine (Shingrix) should be given before starting if you have not already had it. It is a non-live vaccine and safe on immunosuppression, but it works better given before the drug is on board.
- Tuberculosis screening. Quantiferon or T-SPOT blood test once before initiation.
- Hepatitis B panel. Surface antigen, surface antibody, and core antibody to rule out chronic or resolved infection that could reactivate.
- Baseline labs. CBC, liver enzymes, creatinine.
S1P Receptor Modulators: How They Work
S1P stands for sphingosine-1-phosphate, a lipid molecule your body uses to control where immune cells go. Specifically, S1P acts on receptors on the surface of lymphocytes that tell them when to leave lymph nodes and enter the bloodstream. If you block one of those receptors — particularly S1P1 — the lymphocytes get trapped in their home lymph nodes and cannot reach the inflamed colon.
S1P modulators are essentially functional antagonists. They bind, trigger internalization of the S1P1 receptor, and leave the lymphocyte blind to its own exit signal. The practical effect is a targeted, reversible drop in circulating lymphocytes (usually 50–70% from baseline), with fewer disease-relevant T and B cells reaching gut tissue.
The concept is not new. The multiple sclerosis drug fingolimod pioneered the class. IBD S1P modulators are more selective, focusing on S1P1 and S1P5 while sparing S1P2, S1P3, and S1P4 — which reduces the cardiac conduction and retinal side effects that limited fingolimod.
Ozanimod (Zeposia) and True North
Ozanimod is an S1P1 and S1P5 modulator, FDA-approved for moderate-to-severe UC (May 2021). The pivotal data come from the True North trial published by Sandborn and colleagues in NEJM in 2021.
- True North induction. 10 weeks of 0.92 mg once daily produced clinical remission in 18% vs 6% on placebo, with endoscopic improvement in 27% vs 12%.
- True North maintenance. Responders re-randomized at week 10 to continue ozanimod or switch to placebo. At week 52, remission was 37% on ozanimod vs 19% on placebo.
Ozanimod uses a 7-day dose-titration pack — 0.23 mg days 1–4, 0.46 mg days 5–7, then 0.92 mg from day 8 onward. This slow ramp is designed to blunt the first-dose drop in heart rate. Do not skip it; restarting after a missed dose longer than 14 days requires starting the titration pack over.
Ozanimod is metabolized partly by monoamine oxidase. That triggers a real drug interaction warning — MAO inhibitors are contraindicated, and patients on certain antidepressants (especially older MAOIs for depression or Parkinson's drugs like selegiline) cannot take ozanimod without a careful washout.
Etrasimod (Velsipity) and ELEVATE UC
Etrasimod is a newer, once-daily S1P1, S1P4, and S1P5 modulator, FDA-approved for moderate-to-severe UC (October 2023). The approval was based on the ELEVATE UC 52 and ELEVATE UC 12 trials published by Sandborn and colleagues in The Lancet in 2023.
- ELEVATE UC 12. 12-week induction. Clinical remission: 25% on etrasimod 2 mg vs 15% on placebo.
- ELEVATE UC 52. 52-week treat-through design. Week 12 remission was 27% vs 7%; week 52 remission was 32% vs 7%.
Etrasimod's key practical advantage is that it does not require a titration pack. You start at the full 2 mg daily dose. The half-life and pharmacology still produce a transient dip in heart rate after the first dose, so a baseline ECG is required, but the whole initiation process is simpler than ozanimod.
Etrasimod is not metabolized through MAO, so the antidepressant interaction profile is cleaner.
S1P Cardiac Monitoring and Lymphocyte Drop
Both S1P drugs require baseline workup because the S1P1 receptor lives on cardiac tissue as well as lymphocytes. Before starting:
- Baseline ECG. Required. The main concern is a pre-existing heart block, sinus bradycardia, or long QT interval. Patients with recent myocardial infarction, unstable angina, stroke, class III/IV heart failure, or Mobitz II/third-degree AV block (without a pacemaker) cannot take S1P modulators.
- First-dose heart-rate drop. Expect heart rate to fall by 5–10 beats per minute in the first few hours after the first dose. Most patients do not notice it. Patients with borderline bradycardia may need in-office first-dose observation.
- Baseline eye exam. S1P modulators can (rarely) cause macular edema, especially in diabetics or those with uveitis history. Ophthalmology baseline is standard if you have either risk factor.
- Baseline liver enzymes. ALT and AST can rise, usually mild and reversible.
- Lymphocyte count at baseline and 3 months. Expect a drop to 500–800/µL — below the "normal" range but part of how the drug works. A drop below 200/µL is an indication to stop or reduce the dose.
- Varicella immunity. Check antibodies if you do not know your chickenpox status; vaccinate with the non-live Shingrix series before starting.
- Pregnancy test and reliable contraception. These drugs are pregnancy category concerns (see below).
Skin checks are also recommended, with a baseline dermatology exam and yearly follow-up. Basal cell and squamous cell skin cancer rates are slightly elevated over years on S1P modulators.
Where Oral Agents Fit in Your Treatment Plan
Ten years ago, UC and Crohn's treatment went 5-ASA, steroids, immunomodulator, biologic, surgery. JAK inhibitors and S1P modulators have scrambled that sequence. The current American Gastroenterological Association (AGA) and ACG guidance is pragmatic: in moderate-to-severe UC, you can reasonably start with a TNF inhibitor, an IL-23 inhibitor, vedolizumab, ozanimod, etrasimod, or upadacitinib. The best first choice depends on disease severity, speed needed, age, smoking history, comorbidities, and insurance.
Typical scenarios where oral small molecules make sense:
- Biologic failure or loss of response. The most common use. If you have cycled through a TNF inhibitor plus vedolizumab or ustekinumab without durable remission, a JAK or S1P modulator offers a completely different mechanism. Upadacitinib in particular has strong data after TNF failure.
- Needle-averse patients. A real reason. Some people will not stay on an injectable long-term, and partial adherence to a biologic leads to antibody formation and loss of response. A pill taken every day can outperform a shot taken sometimes.
- Acute severe UC on the surgical edge. JAK inhibitors act fast. Emerging data suggest tofacitinib or upadacitinib can rescue patients who are not responding to IV steroids and are being wheeled toward colectomy. This is off-label sequencing in most places but is becoming more common at tertiary IBD centers.
- Extraintestinal symptoms. JAK inhibitors hit broad cytokine pathways, so peripheral arthritis, iritis, and some skin manifestations often improve alongside the gut. See the extraintestinal article. Rheumatologists already use the same drugs for rheumatoid arthritis.
- S1P for mild-to-moderate UC where the safety profile matters more. Ozanimod and etrasimod have cleaner cardiovascular signals than JAKs and are often preferred in younger or lower-risk patients who still need advanced therapy.
What these oral agents are not usually first-line for: fistulizing perianal Crohn's (TNF inhibitors still own this), severe extensive pan-ileal Crohn's with stricture, or any patient with a fresh history of blood clots, recent heart attack, active cancer, or uncontrolled infection.
Pregnancy and Breastfeeding
This is where JAK inhibitors and S1P modulators differ sharply from TNF biologics. Anti-TNF agents like infliximab and adalimumab have two decades of pregnancy data and are generally considered safe to continue through pregnancy. Vedolizumab and ustekinumab have solid data too.
The oral small molecules are different. Both classes cross the placenta and both have signals in animal reproductive studies:
- JAK inhibitors. Animal studies showed embryofetal toxicity. Human data are limited but concerning enough that all IBD JAK inhibitors are currently recommended to be stopped before conception. Washout periods vary: tofacitinib about 4 weeks, upadacitinib about 4 weeks. Reliable contraception is required throughout treatment and for at least 4 weeks after stopping.
- S1P modulators. Fingolimod (the MS prototype) caused birth defects in animal studies and a small excess of malformations in human exposure registries. Ozanimod and etrasimod carry similar warnings. Washout is at least 3 months before conception for ozanimod and about 7 days for etrasimod, reflecting half-life differences.
- Breastfeeding. Discouraged on all four drugs due to lack of safety data.
If you are planning pregnancy or there is any chance you could become pregnant, talk to your GI before starting. Many patients switch to a biologic pre-conception, deliver, breastfeed, and return to the small molecule later. Others stay on biologics long-term specifically to preserve pregnancy options.
Cost Realities and Insurance
Sticker prices for all four drugs are high — roughly $5,000 to $7,000 per month without insurance, or $60,000 to $85,000 per year. Almost no one actually pays this. The real question is what your insurance will cover, what the copay looks like, and whether the manufacturer's assistance program can bridge the rest.
- Commercial insurance. Almost always requires prior authorization. Most plans require documented failure of at least one biologic (or contraindication to biologics) before approving a JAK or S1P drug. A detailed letter from your GI citing trial numbers and your specific history is essential.
- Manufacturer copay cards. Pfizer (tofacitinib), AbbVie (upadacitinib), Bristol Myers Squibb (ozanimod), and Pfizer (etrasimod) all offer copay assistance that drops the monthly cost to around $5–$10 for commercially insured patients. These cards cannot be used with Medicare or Medicaid due to federal anti-kickback rules.
- Medicare. Part D covers oral IBD drugs but typically puts them on specialty tiers with 25–33% coinsurance. Annual out-of-pocket caps under the Inflation Reduction Act have improved this situation as of 2025 — the annual Part D out-of-pocket max is now $2,000.
- Patient Assistance Programs (PAPs). If you are uninsured or underinsured, each manufacturer has a PAP that can provide the drug free or at deeply reduced cost based on income. Income thresholds are usually 400–500% of the federal poverty level.
- Step therapy. Many plans require you to fail a cheaper biologic first. If you have a medical reason why a TNF inhibitor is unsafe (prior lymphoma, severe heart failure, demyelinating disease), document it clearly — it can help you skip the step.
Practical Questions to Ask Your GI
- Do I qualify for a small-molecule agent, or am I better served starting with a biologic given my other conditions?
- What is my ASCVD 10-year risk score? Does it push me away from a JAK inhibitor?
- Have I had shingles? Am I up to date on Shingrix?
- Have I ever had a DVT, PE, or clotting disorder?
- Are there medications I take (especially antidepressants or MAO inhibitors) that interact with ozanimod?
- What is the plan if I improve in 8 weeks? What if I don't?
- Am I planning pregnancy in the next 1–2 years? If so, what is the switch plan?
- What are the copay numbers with my specific insurance plan, and has the office submitted for prior authorization?
- Who watches my lipids and lymphocyte count once I start?
Key Research Papers
- Sandborn WJ, et al. Tofacitinib as induction and maintenance therapy for ulcerative colitis (OCTAVE). N Engl J Med. 2017.
- Ytterberg SR, et al. Cardiovascular and cancer risk with tofacitinib in rheumatoid arthritis (ORAL Surveillance). N Engl J Med. 2022.
- Danese S, et al. Upadacitinib as induction and maintenance therapy for moderately to severely active ulcerative colitis (U-ACHIEVE, U-ACCOMPLISH). The Lancet. 2022.
- Loftus EV, et al. Upadacitinib induction and maintenance therapy for Crohn's disease (U-EXCEL, U-EXCEED, U-ENDURE). N Engl J Med. 2023.
- Feagan BG, et al. Filgotinib as induction and maintenance therapy for ulcerative colitis (SELECTION). The Lancet. 2021.
- Sandborn WJ, et al. Ozanimod as induction and maintenance therapy for ulcerative colitis (True North). N Engl J Med. 2021.
- Sandborn WJ, et al. Etrasimod as induction and maintenance therapy for ulcerative colitis (ELEVATE UC 52 and ELEVATE UC 12). The Lancet. 2023.
- Singh S, et al. AGA Clinical Practice Guidelines on the management of moderate to severe ulcerative colitis. Gastroenterology. 2020.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on JAK inhibitors and S1P receptor modulators in IBD:
- Tofacitinib in ulcerative colitis
- Upadacitinib in UC and Crohn's disease
- Filgotinib in inflammatory bowel disease
- JAK inhibitors and cardiovascular risk (MACE)
- JAK inhibitors and venous thromboembolism
- Ozanimod and the True North trial
- Etrasimod and the ELEVATE UC program
- S1P receptor modulators in IBD
- JAK inhibitors in pregnancy and IBD
- Positioning advanced therapies in ulcerative colitis
Connections
- Inflammatory Bowel Disease Overview
- Crohn's vs Ulcerative Colitis: Key Differences
- Diagnostic Workup: Calprotectin, Colonoscopy, MRE
- Biologics: TNF, IL-23, and Integrin Inhibitors
- Diet for IBD: SCD and Mediterranean
- IBD Surgery Decisions
- Extraintestinal Manifestations: Joints, Skin, Eyes
- IBD Fatigue, Anemia, and Nutrient Deficiencies
- Pregnancy and IBD
- Crohn's Disease
- Ulcerative Colitis
- Arthritis
- Ankylosing Spondylitis
- AS and IBD Overlap
- Cancer
- Shingles (Herpes Zoster)
- Lymphoma