Biologics Guide for Ankylosing Spondylitis
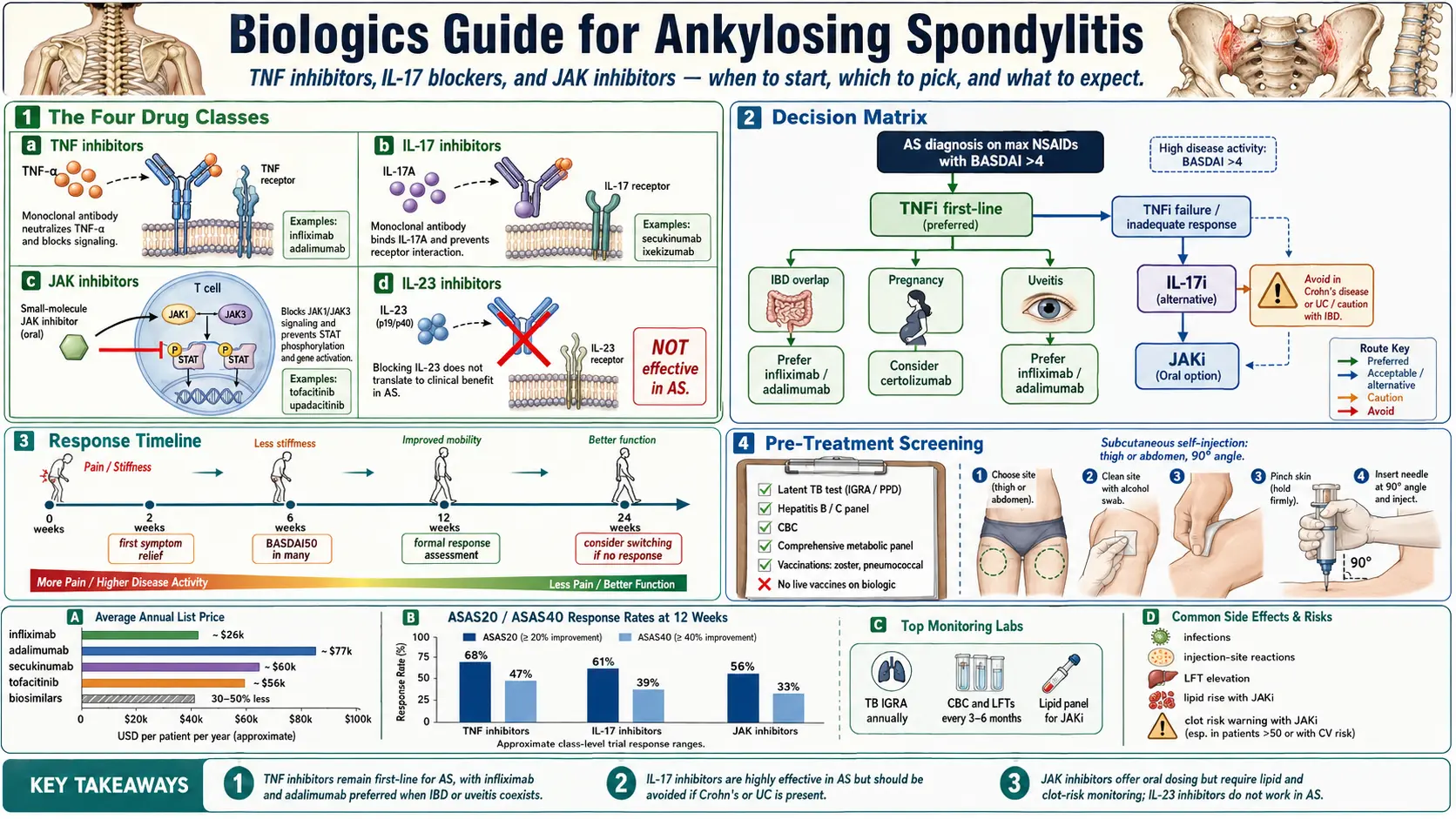
Table of Contents
- Why Biologics Are Different from NSAIDs
- When to Start Biologics
- The Four Drug Classes in AS
- TNF Inhibitors — First-Line Biologics
- IL-17 Inhibitors
- JAK Inhibitors — Oral Option
- IL-23 Inhibitors — Not Effective for AS
- Picking Your Biologic — Decision Matrix
- Cost and Insurance Reality
- How to Give Yourself an Injection
- What to Expect — Timeline of Response
- Side Effects — What to Watch
- Pre-Treatment Screening Checklist
- Monitoring During Treatment
- Stopping or Switching
- Combining Biologics with Other Drugs
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
1. Why Biologics Are Different from NSAIDs
When your rheumatologist says “we’re going to start a biologic,” they are talking about a fundamentally different class of drug than the NSAIDs you have probably been taking. NSAIDs (ibuprofen, naproxen, celecoxib, indomethacin) manage pain and inflammation by blocking cyclooxygenase enzymes. They help you function. They do not, in any meaningful way, change the long-term course of ankylosing spondylitis.
Biologics block specific cytokines — the molecular messengers driving the disease. In AS these are primarily TNF-alpha, IL-17, and, in the case of JAK inhibitors, the intracellular signaling pathway (JAK-STAT) that many cytokines use to deliver their messages. Turning off these pathways can slow or halt spinal fusion when started early, before syndesmophytes (bony bridges between vertebrae) form.
The trade-off is real. Biologics mean injections or infusions instead of pills. They suppress parts of the immune system. They cost thousands of dollars a month. They involve insurance battles, prior authorizations, specialty pharmacies, and baseline infectious-disease screening. For the right patient — especially a young person with active inflammation — the trade-off is worth it. For someone with mild disease well-controlled on NSAIDs, it usually is not.
2. When to Start Biologics
The 2019 ACR/SAA/SPARTAN guidelines (American College of Rheumatology / Spondylitis Association of America) recommend moving to a biologic after failing 2 or more NSAIDs at adequate doses for 2–4 weeks each. “Failure” means active disease despite full anti-inflammatory dosing. For the details of what counts as an adequate NSAID trial, see NSAID Strategy.
But there are several situations where rheumatologists push to start a biologic earlier:
- Peripheral arthritis or enthesitis (joint swelling outside the spine, inflammation where tendons attach to bone) — NSAIDs often don’t touch these.
- Uveitis flares — recurrent eye inflammation strongly predicts structural progression and responds well to TNF inhibitors.
- IBD overlap (Crohn’s or ulcerative colitis) — NSAIDs can actively worsen IBD; a single drug can treat both diseases. See AS and IBD Overlap.
- BASDAI score above 4 (the Bath Ankylosing Spondylitis Disease Activity Index, a validated questionnaire).
- Elevated CRP or ESR despite NSAIDs — systemic inflammation that NSAIDs are failing to suppress.
- Structural damage visible on imaging — MRI showing active sacroiliitis, or plain films showing early syndesmophyte formation.
If you have any of the above, don’t let a provider keep you on NSAIDs for six months “just to be sure.” Time on inflamed vertebrae is time spent building irreversible fusion.
3. The Four Drug Classes in AS
Four cytokine/pathway-targeted drug classes are relevant to AS, and only three of them actually work for the axial (spine) disease:
- TNF inhibitors (TNFi) — first-line biologic. Block tumor necrosis factor-alpha.
- IL-17 inhibitors (IL-17i) — second-line or first-line with specific comorbidities.
- JAK inhibitors (JAKi) — oral pills; block intracellular signaling. Reserved for TNFi/IL-17i failures.
- IL-23 inhibitors — effective in psoriatic arthritis and psoriasis, but not in axial AS. Skip for AS.
4. TNF Inhibitors (TNFi) — First-Line Biologics
TNF inhibitors are the oldest, best-studied, and usually the first biologic class offered for AS. They work in about two-thirds of patients.
Adalimumab (Humira)
40 mg subcutaneous every 2 weeks. The most prescribed biologic in the world. Self-injected at home as either a pre-filled pen (Humira Pen) or syringe. Refrigerated shipping, shelf-stable at room temperature for up to 14 days.
Etanercept (Enbrel)
50 mg subcutaneous weekly. Self-injected. Etanercept is a soluble TNF receptor fusion protein (slightly different mechanism from the monoclonal antibodies). Important caveat: etanercept is not effective for IBD, so avoid it in AS patients with concurrent Crohn’s or UC.
Infliximab (Remicade)
5 mg/kg IV infusion at weeks 0, 2, and 6, then every 6–8 weeks. Requires an infusion center — roughly a 2-hour appointment each time. No self-injection. Doses can be escalated in poor responders (up to 7.5–10 mg/kg or shortened intervals). Excellent evidence in AS + IBD and AS + uveitis.
Certolizumab pegol (Cimzia)
400 mg subcutaneous at weeks 0, 2, and 4, then 200 mg every 2 weeks or 400 mg every 4 weeks. Certolizumab is pegylated and lacks the Fc portion of a normal antibody — which means it does not actively cross the placenta. This makes it the safest TNFi in pregnancy. See Pregnancy and AS.
Golimumab (Simponi, Simponi Aria)
50 mg subcutaneous monthly (Simponi), or IV 2 mg/kg at weeks 0 and 4, then every 8 weeks (Simponi Aria). Monthly dosing is a selling point for patients who hate weekly or bi-weekly injections.
Biosimilars for TNFi
Biosimilars are FDA-approved, near-identical copies of the originator biologic — comparable to generic drugs for small molecules, although the regulatory pathway is different.
- Adalimumab biosimilars: Amjevita, Cyltezo, Hadlima, Hyrimoz, Idacio, Hulio, Abrilada, Yuflyma.
- Infliximab biosimilars: Inflectra, Renflexis, Avsola, Ixifi.
- Etanercept biosimilars: Erelzi, Eticovo.
Biosimilars are often cheaper and have equivalent efficacy in head-to-head studies. Insurance companies may force a switch to whichever biosimilar they contract with this year — this is annoying but usually clinically fine.
TNFi Response and Structural Effects
60–70% of AS patients achieve a BASDAI 50 response (50% improvement in disease activity score) by week 12. Long-term studies show slower spinal fusion with sustained TNFi use, especially when started before significant syndesmophyte formation.
5. IL-17 Inhibitors (IL-17i) — When TNFi Fails or for Specific Comorbidities
IL-17 is a cytokine central to the spondyloarthritis disease process. Blocking it works well in AS and psoriasis, and somewhat less well in IBD (where it can actually cause trouble).
Secukinumab (Cosentyx)
150 mg or 300 mg subcutaneous weekly for 5 loading doses, then monthly. The 300 mg dose is typically used when psoriasis coexists.
Ixekizumab (Taltz)
80 mg subcutaneous at weeks 0, 2, 4, 6, 8, 10, and 12, then every 4 weeks. Aggressive loading front-loads the response.
Bimekizumab (Bimzelx)
Newer. Targets both IL-17A and IL-17F (where secukinumab and ixekizumab only target IL-17A). 160 mg subcutaneous with loading and maintenance schedules per label. Early data suggest higher response rates than single-target IL-17i.
When to Prefer IL-17i Over TNFi
- Concurrent plaque psoriasis — IL-17i is more effective for skin clearance than TNFi.
- Prior TNFi failure (primary non-response or secondary loss of response).
- Specific TNFi contraindications — demyelinating disease history, advanced heart failure.
IL-17i CAUTION in IBD
This is crucial. Secukinumab and ixekizumab can trigger or worsen Crohn’s disease and ulcerative colitis flares. If you have AS plus any form of IBD, choose a TNFi (adalimumab or infliximab) instead. See AS and IBD Overlap.
IL-17i Side Effects
Most characteristic: increased rate of candidiasis (oral thrush, vaginal yeast infections). Also injection site reactions and upper respiratory infections. Typically manageable, but tell your rheumatologist at the first sign of recurrent yeast.
6. JAK Inhibitors (JAKi) — Oral Option
JAK inhibitors are small molecules, not biologics in the strict sense, but they occupy the same therapeutic niche and are often grouped with biologics in clinical conversation.
Tofacitinib (Xeljanz)
5 mg twice daily. FDA approved for AS in 2021.
Upadacitinib (Rinvoq)
15 mg once daily. FDA approved for AS. Many rheumatologists prefer upadacitinib for its once-daily dosing and slightly more selective JAK1 inhibition.
Advantages of JAKi
- Oral — no injections, no infusion center.
- Faster onset — some patients feel improvement within 1–2 weeks.
- No need for refrigeration or sharps disposal.
Disadvantages of JAKi
The FDA has placed a boxed warning on all oral JAKi for MACE (major adverse cardiovascular events), malignancy, thrombosis, and serious infections. The warning is based on the ORAL-SURVEILLANCE trial, which was conducted in rheumatoid arthritis patients over age 50 with at least one cardiovascular risk factor. Whether the same risk applies to younger AS patients is debated, but the warning stands.
A baseline cardiovascular risk assessment is essential before starting a JAKi. They are usually reserved for patients who have failed at least one TNFi and one IL-17i, or who genuinely cannot do injections.
Cost and Monitoring
JAKi require quarterly lab monitoring: CBC, LFTs, and lipid panel. JAK inhibitors reliably raise LDL cholesterol and occasionally suppress neutrophil or lymphocyte counts.
7. IL-23 Inhibitors — Currently NOT Effective for AS
Ustekinumab (Stelara) and risankizumab (Skyrizi) block IL-23, a cytokine upstream of IL-17 in the inflammatory cascade. They work beautifully in psoriasis and, to a lesser degree, peripheral psoriatic arthritis. Randomized trials in ankylosing spondylitis showed no efficacy for axial disease. This was a genuine surprise to the field given the mechanistic logic.
If a provider suggests ustekinumab or risankizumab for your AS, push back. These drugs are appropriate for psoriatic arthritis with peripheral joint disease, not axial spondyloarthritis.
8. Picking Your Biologic — Decision Matrix
Use your comorbidities to pick the drug:
- AS + IBD (Crohn’s or UC): Adalimumab or infliximab — both treat both diseases. Avoid IL-17i and avoid etanercept.
- AS + severe psoriasis: IL-17i (secukinumab, ixekizumab, bimekizumab) — superior skin clearance.
- AS + recurrent uveitis: Adalimumab or infliximab have the best evidence; certolizumab is also effective. Avoid etanercept (less effective for uveitis).
- AS + pregnancy planning: Certolizumab — doesn’t cross the placenta.
- AS + reluctance to inject: JAK inhibitor (tofacitinib or upadacitinib) — oral.
- AS + prior TNFi failure: IL-17i or JAKi.
- First-line, no comorbidities: Any TNFi — choose based on dosing frequency, patient preference, and insurance formulary.
9. Cost and Insurance Reality
Brace yourself. These drugs are expensive.
- TNFi retail list prices: $4,000–$7,000 per month.
- IL-17i: $5,000–$10,000 per month.
- JAKi: $4,000–$6,000 per month.
- Biosimilars: 20–50% cheaper than the originator.
Insurance coverage is usually available with prior authorization. The PA packet typically requires: documented NSAID failure (2+ NSAIDs for 2–4 weeks each), a BASDAI score, CRP or ESR results, and in some cases imaging. If your rheumatologist’s office does not help with this, ask specifically for their PA coordinator.
Manufacturer copay assistance programs dramatically reduce out-of-pocket costs for commercially-insured patients:
- Humira Complete, Cosentyx Connect, Taltz Together, Rinvoq Complete, Enbrel Support, Cimzia MySupport, Simponi One.
- Most bring the copay down to $5–$10 per month for eligible patients.
- Medicare and Medicaid patients are usually not eligible (federal anti-kickback rules).
Patient assistance programs provide free drug to uninsured patients meeting income criteria — typically up to 400–500% of the federal poverty level. Apply directly through the manufacturer.
Specialty pharmacy is mandatory for most biologics — Accredo, CVS Specialty, and Walgreens Specialty are the big three. Retail pharmacies generally cannot fill these drugs. Expect monthly phone calls to confirm each shipment.
Switching biologics: insurance often enforces step therapy — you must fail a cheaper TNFi before they will pay for an IL-17i or JAKi. Document failures carefully so appeals go smoothly.
10. How to Give Yourself an Injection
If you have never given yourself a shot, the first one is nerve-wracking. After three or four, it’s boring. Tips that matter:
- Warm the pen to room temperature for 30 minutes before injecting. Cold drug stings badly; room-temperature drug is nearly painless.
- Rotate sites — front of thigh, abdomen (avoid a 2-inch radius around the navel), or upper outer arm. Rotating prevents lipohypertrophy and bruising.
- Pinch up a fold of skin, insert at 90 degrees, and hold the pen or syringe for the full 10 seconds (or whatever the device manual specifies) to deliver the complete dose.
- Ice the site afterward for 1–2 minutes to reduce bruising.
- Do the first injection in clinic and have a nurse walk you through it. Every manufacturer has a nurse-educator program; use it.
- Dispose in a sharps container, which your pharmacy provides free. Do not throw pens in the regular trash.
11. What to Expect — Timeline of Response
- Weeks 1–4: Many patients notice morning stiffness improving first. Fatigue may lift.
- Weeks 4–12: BASDAI score drops, CRP normalizes, functional capacity improves.
- Full response: 12–16 weeks is the standard window to judge response. Don’t give up at week 6.
- Non-responders at 16 weeks: time to switch drug class (or to a different drug within the class, if there are logistical or side-effect reasons).
- Secondary loss of response: occurs in 20–30% of TNFi users over 2–3 years, often due to anti-drug antibody formation. Switching within the class (e.g., Humira to Cimzia) or to a different class (TNFi to IL-17i) typically recaptures response.
12. Side Effects — What to Watch
Common and usually manageable:
- Injection site reactions (redness, itching, bruising).
- Upper respiratory tract infections.
- Mild fatigue the day of injection.
Serious but rare:
- Reactivation of latent tuberculosis — screen before starting with QuantiFERON-Gold or T-SPOT.TB; add chest X-ray if positive or risk factors.
- Hepatitis B reactivation — screen for HBsAg, anti-HBc, anti-HBs.
- Drug-induced lupus — rare, seen with TNFi; typically resolves on stopping the drug.
- Heart failure worsening — avoid TNFi if ejection fraction below 40%.
- Demyelinating disease — avoid TNFi in patients with multiple sclerosis or optic neuritis history.
Cancer risk: TNFi use is associated with a slightly increased risk of lymphoma — roughly a doubling of baseline risk, but because baseline risk is very low the absolute numbers remain small. Non-melanoma skin cancers are more common; annual skin checks recommended.
Vaccines:
- Live vaccines are contraindicated during biologic therapy — avoid MMR, varicella (chickenpox), yellow fever, and the intranasal live flu vaccine (FluMist).
- Killed and inactivated vaccines are fine and recommended — annual injected flu shot, pneumococcal (PCV20 or PCV15 + PPSV23), Shingrix, Tdap, and any of the non-live respiratory vaccines.
13. Pre-Treatment Screening Checklist
Before the first dose:
- TB screening — QuantiFERON-Gold or T-SPOT.TB; chest X-ray if the test is positive or risk factors suggest it.
- Hepatitis B serology (HBsAg, anti-HBc, anti-HBs) and hepatitis C antibody.
- HIV screening if any risk factors.
- Baseline labs — CBC, CMP, ESR, CRP.
- Pregnancy test for women of childbearing age.
- Update vaccines before starting — pneumococcal, Shingrix, annual flu, Tdap if due. Live vaccines must be completed 4+ weeks before the first biologic dose.
- Dental exam — fix any active infections first. Dental abscesses during biologic therapy are dangerous.
- Malignancy screen appropriate for age (mammogram, colonoscopy, skin exam, Pap smear) — particularly if over 50 or with family history.
14. Monitoring During Treatment
- Labs: CBC, CMP, ESR/CRP every 3–6 months. LFTs and lipid panel quarterly on JAKi.
- BASDAI scoring every rheumatology visit to track response trend.
- MRI of SI joints or spine: not routinely repeated, but useful if response is in question or to document structural stability over years.
- Annual skin check for non-melanoma skin cancer.
- Annual flu shot (injected, not FluMist); updated COVID-19 vaccines per current CDC guidance.
15. Stopping or Switching
Never stop a biologic cold-turkey. Work with your rheumatologist to taper or bridge. Biologics can be paused for surgery — typically one dosing interval before and held until wounds are healed, often 1–2 weeks post-op — with low risk of losing response. Longer pauses (months) can cause loss of response that may require re-titration or a switch.
Switching within a class is common and often works: Humira to Cimzia, Cosentyx to Taltz. Switching across classes (TNFi to IL-17i, IL-17i to JAKi) is the standard move after primary or secondary failure.
16. Combining Biologics with Other Drugs
- NSAIDs — compatible with biologics and frequently continued. They handle breakthrough stiffness the biologic hasn’t fully quelled.
- DMARDs — methotrexate is rarely added to AS biologic therapy because it does not help axial disease, although it can be useful for concurrent peripheral arthritis. Sulfasalazine can help peripheral arthritis and in some cases IBD overlap. Neither does much for the spine.
- Corticosteroids — short courses (oral prednisone 10–20 mg tapers, or intra-articular injections) are fine for acute flares. Avoid long-term systemic steroids in AS — they don’t meaningfully help axial disease and they accelerate osteoporosis, a major concern in this population.
For non-pharmacologic adjuncts, see Exercise and PT Protocol — exercise remains foundational no matter which biologic you’re on.
17. Key Research Papers
- van der Heijde D, et al. 2016 update of the ASAS-EULAR management recommendations for axial spondyloarthritis. Annals of the Rheumatic Diseases. 2017.
- Ward MM, et al. 2019 Update of the ACR/SAA/SPARTAN recommendations for the treatment of axial spondyloarthritis. Arthritis & Rheumatology. 2019.
- Baeten D, et al. Secukinumab, an interleukin-17A inhibitor, in ankylosing spondylitis (MEASURE 1 and MEASURE 2). New England Journal of Medicine. 2015.
- van der Heijde D, et al. Ixekizumab, an interleukin-17A antagonist in radiographic axial spondyloarthritis (COAST-V). Lancet. 2018.
18. Research Papers
Curated PubMed topic searches — each link opens a live query so you always see the most recent literature.
- PubMed: Ankylosing spondylitis biologic therapy
- PubMed: TNF inhibitor ankylosing spondylitis
- PubMed: Secukinumab ankylosing spondylitis
- PubMed: Ixekizumab axial spondyloarthritis
- PubMed: Tofacitinib ankylosing spondylitis
- PubMed: Upadacitinib ankylosing spondylitis
- PubMed: Biosimilar spondyloarthritis
Connections
- Ankylosing Spondylitis
- HLA-B27 Explained
- AS and IBD Overlap
- Exercise and PT Protocol
- NSAID Strategy
- Morning Stiffness Management
- Starch-Free Diet and Ebringer
- Pregnancy and AS
- Uveitis and Eye Involvement
- Arthritis
- Psoriasis
- Crohn's Disease
- Ulcerative Colitis
- Inflammatory Markers