Ankylosing Spondylitis
Deep-Dive Articles
Ankylosing spondylitis is a long game — usually diagnosed young, always chronic, managed for decades. The eight guides below each tackle one piece of the puzzle: the HLA-B27 gene behind it, the biologics that change the disease course, the exercise program that actually slows fusion, how to handle NSAIDs long-term, what to do about that 3 AM morning stiffness, how gut conditions (IBD) fit in, the starch-free-diet controversy, and pregnancy planning. Start wherever matches where you are right now.
HLA-B27 Explained
The gene present in 90–95% of AS patients — what it is, how it’s tested, what a positive result means (and doesn’t), the three competing theories for how it drives disease, and the ethnic prevalence patterns that explain why AS is common in some populations and rare in others.
Biologics Guide
TNF inhibitors (Humira, Enbrel, Remicade, Cimzia, Simponi) vs. IL-17 inhibitors (Cosentyx, Taltz) vs. JAK inhibitors (Rinvoq, Xeljanz). Dose, cost, insurance realities, side effects, and how to pick based on your comorbidities (IBD, uveitis, psoriasis).
AS & IBD Overlap
Up to 10% of AS patients develop Crohn’s or ulcerative colitis, and ~30% have subclinical gut inflammation on colonoscopy. Which biologics treat both, how to screen for IBD as an AS patient, and why this overlap changes drug choices.
Exercise & PT Protocol
Swimming, McKenzie extensions, posture-focused strength work, yoga, and the Back-Pain Liberation programs that slow spinal fusion. The evidence-based routine most rheumatologists never write down, with week-by-week progression.
NSAID Strategy
Daily vs. PRN dosing, which NSAID (meloxicam, naproxen, celecoxib, indomethacin), why continuous NSAIDs may slow spinal fusion, GI protection with PPIs or misoprostol, cardiovascular and kidney risk, and when to step up to biologics.
Morning Stiffness Management
The 2-hour lockup that defines AS mornings — heat, bedtime NSAID timing, sleep position, mattress choice, warm shower-plus-stretch protocols, and how to get out of bed on bad days without pain spikes.
Starch-Free Diet & Ebringer
The Alan Ebringer hypothesis — Klebsiella pneumoniae in the gut molecularly mimics HLA-B27, and cutting starch (the bacteria’s food) reduces flares. Honest coverage of the evidence, the protocol, patient reports, and why mainstream rheumatology remains skeptical.
Pregnancy & AS
AS disease activity through pregnancy (often worsens in the third trimester), which biologics and NSAIDs are safe, postpartum flare management, breastfeeding compatibility, fertility considerations, and the pre-conception planning timeline.
Uveitis & Eye Involvement in AS
HLA-B27 iritis, the anti-TNF class difference, and the 48-hour ophthalmology rule.
1. Overview
Ankylosing spondylitis (AS) is a chronic, progressive inflammatory arthritis that primarily affects the axial skeleton, including the sacroiliac joints and spine. It belongs to a family of related conditions collectively termed spondyloarthropathies (SpA), which also includes psoriatic arthritis, reactive arthritis, enteropathic arthritis, and undifferentiated spondyloarthropathy. AS is characterized by inflammation at sites where ligaments and tendons attach to bone, a process known as enthesitis, which distinguishes it from other forms of inflammatory arthritis.
The hallmark pathological process in AS is progressive osteoproliferation, the paradoxical formation of new bone at sites of inflammation. Over time, this leads to the formation of syndesmophytes (bony bridges between vertebrae) and can ultimately result in complete fusion of the spine, producing the characteristic "bamboo spine" appearance on radiographs. This process can cause significant pain, stiffness, and progressive loss of spinal mobility, potentially leading to severe functional disability.
AS is strongly associated with the HLA-B27 gene, one of the strongest known associations between an HLA antigen and a disease. Approximately 90-95% of patients with AS carry the HLA-B27 allele, compared to roughly 6-8% of the general population in Caucasian populations. However, most individuals who are HLA-B27 positive never develop AS, indicating that additional genetic and environmental factors are necessary for disease expression. The disease typically manifests in young adulthood, with a mean age of onset between 20 and 30 years, and symptom onset before age 45 is a key diagnostic criterion.
2. Epidemiology
The prevalence of ankylosing spondylitis varies considerably across populations, closely mirroring the prevalence of the HLA-B27 allele. In European and North American populations, the prevalence is estimated at 0.1-0.5%, affecting approximately 2.7 million adults in the United States. The condition is 2-3 times more common in men than women, though this ratio has narrowed with improved recognition of the disease in women, who tend to have more peripheral and less radiographic axial disease.
The global incidence rate is approximately 0.4-14 per 100,000 person-years, with significant geographic variation. Prevalence is highest in populations with high HLA-B27 frequency, including Northern European, Native American (Haida, Pima), and certain Siberian populations. The disease is relatively rare in Sub-Saharan African and Japanese populations, where HLA-B27 frequency is low. The mean age at symptom onset is 23 years, with approximately 80% of patients developing symptoms before age 30 and less than 5% presenting after age 45. Diagnostic delay remains a significant problem, with an average delay from symptom onset to diagnosis of 6-8 years globally.
3. Pathophysiology
Enthesitis: The Primary Lesion
The fundamental pathological process in AS is enthesitis, inflammation at the enthesis (the site where tendons, ligaments, and joint capsules insert into bone). The enthesis is now recognized as an "enthesis organ" comprising the insertion itself, adjacent fibrocartilage, bursa, and underlying bone marrow. In AS, immune cells infiltrate the enthesis and adjacent bone marrow, producing pro-inflammatory cytokines that drive both tissue destruction and paradoxical new bone formation.
The Role of HLA-B27
Despite decades of research, the precise mechanism by which HLA-B27 predisposes to AS remains incompletely understood. Three principal hypotheses have been proposed: the arthritogenic peptide hypothesis (HLA-B27 presents self-derived peptides that trigger autoreactive CD8+ T cells), the misfolding hypothesis (HLA-B27 heavy chains misfold in the endoplasmic reticulum, triggering the unfolded protein response and activation of IL-23/IL-17 inflammatory pathways), and the free heavy chain hypothesis (cell-surface expression of HLA-B27 free heavy chain homodimers activates innate immune receptors including KIR3DL2 on natural killer cells and T cells).
The IL-23/IL-17 Axis
The interleukin-23/interleukin-17 (IL-23/IL-17) pathway has emerged as the central cytokine axis in AS pathogenesis. IL-23, produced by dendritic cells and macrophages, promotes the differentiation and expansion of Th17 cells and innate-like T cells (including gamma-delta T cells and mucosal-associated invariant T cells) that produce IL-17A. IL-17A drives inflammation, bone erosion, and paradoxically promotes osteoblast differentiation and new bone formation. Genome-wide association studies have identified polymorphisms in the IL-23 receptor (IL23R) gene as significant risk factors for AS, further implicating this pathway.
New Bone Formation
A distinguishing feature of AS compared to other inflammatory arthritides is the progressive formation of new bone. Following resolution of active inflammation at entheseal sites, a reparative process occurs involving activation of Wnt signaling, bone morphogenetic protein (BMP) pathways, and Hedgehog signaling. These pathways stimulate mesenchymal stem cell differentiation into osteoblasts, leading to formation of syndesmophytes and eventually complete bony ankylosis. This process may be partially uncoupled from active inflammation, which has implications for timing of treatment.
4. Etiology and Risk Factors
Genetic Factors
- HLA-B27 — present in 90-95% of AS patients; confers a 5-16% lifetime risk of developing AS in carriers; more than 200 subtypes identified, with B*27:05 and B*27:02 most strongly associated
- Non-HLA genes — genome-wide association studies have identified more than 100 additional susceptibility loci, including IL23R, ERAP1, ERAP2, IL6R, RUNX3, TBX21, and CARD9
- ERAP1 (endoplasmic reticulum aminopeptidase 1) — trims peptides for HLA class I presentation; variants associated with AS risk only in HLA-B27-positive individuals, supporting the arthritogenic peptide hypothesis
- Heritability — estimated at greater than 90%; concordance rate in monozygotic twins is 63% compared to 12-27% in HLA-B27-positive first-degree relatives
Environmental Factors
- Gut microbiome — dysbiosis has been consistently demonstrated in AS patients; subclinical gut inflammation is present in up to 60% of AS patients; the gut-joint axis is a major area of research
- Mechanical stress — entheses at high-stress sites are preferentially affected, suggesting biomechanical factors contribute to disease localization
- Smoking — associated with worse radiographic progression, higher disease activity, and poorer functional outcomes
- Infections — potential role of Klebsiella pneumoniae and other enteric bacteria has been proposed but remains unproven
Demographic Risk Factors
- Male sex — 2-3 times higher risk than females
- Age of onset — typically between 17-45 years; juvenile onset (before age 16) accounts for 10-20% of cases
- Family history — first-degree relatives of AS patients have a 20-fold increased risk
- Ethnicity — prevalence parallels HLA-B27 frequency across populations
5. Clinical Presentation
Axial Symptoms
The cardinal symptom of AS is chronic inflammatory back pain, which differs from mechanical back pain by the following characteristics: insidious onset, onset before age 45, duration greater than 3 months, morning stiffness lasting more than 30 minutes, improvement with exercise but not rest, nocturnal pain (especially in the second half of the night), and alternating buttock pain (reflecting bilateral sacroiliitis). Progressive spinal involvement leads to loss of lumbar lordosis, increased thoracic kyphosis, and restricted chest expansion. The classic late-stage posture shows fixed forward flexion ("question mark posture" or "stooped posture").
Peripheral Manifestations
- Peripheral arthritis — occurs in approximately 30-40% of patients; typically oligoarticular, asymmetric, and preferentially involves the lower extremities (hips, knees, ankles)
- Hip involvement — occurs in 25-35% of patients; associated with more severe disease and greater functional disability; may require total hip replacement
- Enthesitis — inflammation at tendon and ligament insertions; Achilles tendon and plantar fascia insertion are most commonly affected
- Dactylitis — "sausage digit" swelling of entire fingers or toes; less common in AS than in psoriatic arthritis
Extra-Articular Manifestations
- Acute anterior uveitis (iritis) — the most common extra-articular manifestation, occurring in 25-40% of patients; presents with unilateral eye pain, redness, photophobia, and blurred vision; recurrent episodes are common
- Inflammatory bowel disease — clinical Crohn's disease or ulcerative colitis develops in 5-10% of AS patients; subclinical gut inflammation (detectable on ileocolonoscopy) is present in up to 60%
- Psoriasis — occurs in approximately 10% of AS patients
- Cardiovascular involvement — aortitis, aortic valve insufficiency (1-10%), conduction abnormalities (heart block), and increased risk of atherosclerotic cardiovascular disease
- Pulmonary involvement — restrictive lung disease from chest wall rigidity; rarely, apical pulmonary fibrosis
- Neurological complications — cauda equina syndrome (rare); atlantoaxial subluxation; spinal fractures with minimal trauma
- Osteoporosis — paradoxically common despite new bone formation; increases fracture risk, particularly of the rigid, fused spine
6. Diagnosis
Modified New York Criteria (1984)
The traditional classification criteria for AS require radiographic evidence of sacroiliitis (grade 2 or greater bilaterally, or grade 3-4 unilaterally) plus at least one clinical criterion: (1) low back pain and stiffness for more than 3 months that improves with exercise but is not relieved by rest, (2) limitation of lumbar spine motion in sagittal and frontal planes, or (3) limitation of chest expansion relative to age and sex norms. These criteria remain the gold standard for established AS but miss early disease before radiographic changes develop.
ASAS Classification Criteria for Axial SpA (2009)
The Assessment of SpondyloArthritis International Society (ASAS) developed broader criteria that include non-radiographic axial spondyloarthropathy (nr-axSpA). These require back pain for 3 or more months with onset before age 45, plus either: (1) sacroiliitis on imaging (radiograph or MRI) plus one or more SpA features, or (2) HLA-B27 positive plus two or more SpA features. SpA features include inflammatory back pain, arthritis, enthesitis, uveitis, dactylitis, psoriasis, Crohn's/colitis, good response to NSAIDs, family history of SpA, HLA-B27, and elevated CRP.
Laboratory Studies
- HLA-B27 testing — positive in 90-95% of AS patients; supports but does not confirm diagnosis; negative HLA-B27 does not exclude AS
- C-reactive protein (CRP) — elevated in approximately 40-50% of AS patients; correlates with disease activity and predicts radiographic progression
- Erythrocyte sedimentation rate (ESR) — may be elevated but is less reliable than CRP in AS
- Rheumatoid factor and anti-CCP antibodies — characteristically negative; positivity suggests an alternative diagnosis
Imaging
- Pelvic radiographs — sacroiliitis graded 0-4 (New York criteria); "bamboo spine" with complete syndesmophyte bridging in advanced disease; "shiny corners" (Romanus lesions), squaring of vertebral bodies, and "dagger sign" (ossified supraspinous and interspinous ligaments)
- MRI of sacroiliac joints — critical for early diagnosis; bone marrow edema on STIR or T2-weighted fat-suppressed sequences indicates active sacroiliitis; structural changes (erosions, fat metaplasia, sclerosis, ankylosis) may also be present
- Spinal MRI — detects spondylitis (corner inflammatory lesions), spondylodiscitis, and facet joint inflammation
- Low-dose CT — superior to radiography for detecting structural damage including erosions and ankylosis of sacroiliac joints
Clinical Assessment Tools
- BASDAI (Bath AS Disease Activity Index) — patient-reported measure of disease activity; scores 0-10; score of 4 or greater indicates active disease
- ASDAS (AS Disease Activity Score) — composite measure incorporating CRP and patient-reported outcomes; categories: inactive (less than 1.3), low (1.3-2.1), high (2.1-3.5), very high (greater than 3.5)
- BASFI (Bath AS Functional Index) — assesses functional limitation
- BASMI (Bath AS Metrology Index) — objective measurement of spinal mobility
7. Treatment
Non-Pharmacological Management
- Exercise and physical therapy — cornerstone of AS management; regular stretching, range-of-motion exercises, and aerobic fitness improve pain, stiffness, and function; supervised exercise programs are superior to home exercises alone
- Patient education — understanding the chronic nature of the disease and importance of lifelong exercise
- Smoking cessation — smoking is associated with worse outcomes, poorer treatment response, and accelerated radiographic progression
- Posture training — strategies to maintain upright posture and prevent fixed kyphosis
NSAIDs
NSAIDs are the first-line pharmacological treatment for AS and remain the mainstay of therapy for axial symptoms. Continuous NSAID use may slow radiographic progression compared to on-demand use, though this remains debated. Common choices include indomethacin (75-150 mg/day), naproxen (1000 mg/day), diclofenac (150 mg/day), and etoricoxib (90 mg/day). At least two different NSAIDs should be tried for a minimum of 2-4 weeks each at full anti-inflammatory doses before considering NSAID failure.
Biologic Therapies
- TNF inhibitors — first-line biologic therapy for patients with active disease despite adequate NSAID trial; all five approved agents are effective: infliximab (5 mg/kg IV every 6-8 weeks), adalimumab (40 mg SC every 2 weeks), etanercept (50 mg SC weekly), golimumab (50 mg SC monthly), and certolizumab pegol (200 mg SC every 2 weeks); ASAS40 response rates of approximately 40-50%
- IL-17A inhibitors — secukinumab (150 mg SC monthly after loading) and ixekizumab (80 mg SC every 4 weeks after loading) are effective for axial and peripheral disease; may be preferred when TNFi is contraindicated or as second-line biologic; secukinumab is contraindicated in active IBD
- JAK inhibitors — tofacitinib (5 mg twice daily) and upadacitinib (15 mg daily) have demonstrated efficacy in AS; upadacitinib is FDA-approved for active AS; provides an oral alternative to biologics
Conventional Synthetic DMARDs
- Sulfasalazine — may be useful for peripheral arthritis (2-3 g/day) but has no proven efficacy for axial disease
- Methotrexate — limited evidence of efficacy in AS; may be considered for peripheral arthritis but not recommended for axial symptoms
- Local corticosteroid injections — useful for sacroiliitis (CT-guided), enthesitis, and peripheral arthritis; systemic corticosteroids have no established role in AS
Surgical Management
- Total hip arthroplasty — for severe, refractory hip disease; may be needed in 5-10% of AS patients
- Spinal osteotomy — corrective surgery for severe fixed kyphotic deformity; high-risk procedure performed at specialized centers
- Spinal fracture management — requires high index of suspicion; even minor trauma can cause unstable fractures in the fused spine
8. Complications
- Spinal ankylosis and fixed deformity — progressive fusion of vertebral segments leading to rigid kyphotic posture and severe functional limitation
- Spinal fractures — the rigid, osteoporotic spine is highly susceptible to fracture from minimal trauma; cervical fractures may cause spinal cord injury and quadriplegia
- Cauda equina syndrome — rare but serious complication caused by arachnoid diverticula eroding the lumbar lamina
- Atlantoaxial subluxation — instability of the C1-C2 junction; risk for spinal cord compression
- Recurrent anterior uveitis — can lead to synechiae, glaucoma, cataracts, and visual impairment if undertreated
- Aortic insufficiency and conduction defects — aortitis can cause aortic root dilation, aortic regurgitation, and atrioventricular block requiring pacemaker
- Restrictive lung disease — reduced chest expansion from costovertebral ankylosis impairs ventilatory function
- Apical pulmonary fibrosis — rare; may mimic tuberculosis on imaging
- Secondary amyloidosis (AA amyloidosis) — rare in the era of effective biologic therapy; due to chronic inflammation
- Osteoporosis — increased vertebral fracture risk; standard DXA may underestimate bone loss due to syndesmophyte artifact
- Increased cardiovascular risk — chronic systemic inflammation contributes to accelerated atherosclerosis
9. Prognosis
The prognosis of ankylosing spondylitis is highly variable, ranging from mild disease with minimal functional limitation to severe, progressive ankylosis with significant disability. Key predictors of poor prognosis include hip involvement, elevated CRP at baseline, smoking, early onset of symptoms, male sex, poor response to NSAIDs, and presence of syndesmophytes on baseline radiographs. The mSASSS (modified Stoke AS Spinal Score) is used to quantify radiographic progression.
With modern biologic therapies, the functional outcomes for AS patients have improved dramatically. TNF inhibitors and IL-17A inhibitors can achieve sustained clinical remission or low disease activity in 40-60% of patients and may slow radiographic progression. However, even with optimal treatment, approximately 30-40% of patients experience significant functional limitation over 20-30 years of disease. Life expectancy is slightly reduced in AS, with standardized mortality ratios of approximately 1.3-1.6, primarily driven by cardiovascular disease, spinal fractures, and amyloidosis.
Importantly, the diagnostic delay in AS (average 6-8 years) means that many patients have already developed significant structural damage before treatment begins. Earlier diagnosis and treatment are associated with better outcomes, highlighting the importance of recognizing inflammatory back pain patterns and appropriate use of MRI and HLA-B27 testing in younger patients.
10. Prevention
There is currently no established method for preventing the onset of ankylosing spondylitis. However, several strategies can prevent disease progression, complications, and functional decline:
- Early diagnosis — recognizing the features of inflammatory back pain and pursuing appropriate diagnostic workup reduces the diagnostic delay that allows structural damage to accumulate
- Regular exercise — the single most important self-management strategy; daily stretching and range-of-motion exercises help maintain spinal mobility and prevent fixed deformity
- Smoking cessation — smokers have more severe disease, worse functional outcomes, and poorer treatment responses
- Treatment adherence — consistent use of NSAIDs and biologic therapies as prescribed; premature discontinuation is associated with disease flares and radiographic progression
- Ophthalmologic screening — patients should be educated about symptoms of anterior uveitis and seek immediate treatment to prevent ocular complications
- Cardiovascular risk management — aggressive management of traditional cardiovascular risk factors (hypertension, dyslipidemia, diabetes) given the elevated cardiovascular risk in AS
- Osteoporosis prevention — bone density monitoring, adequate calcium and vitamin D intake, weight-bearing exercise
- Fall prevention — critical in patients with spinal ankylosis due to the catastrophic risk of spinal fractures
- Genetic counseling — HLA-B27-positive individuals with AS should understand the risk to offspring (approximately 10-20% if the child inherits HLA-B27)
Table of Contents
- Deep-Dive Articles
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Research Papers
- Connections
- Featured Videos
Research Papers
Curated PubMed topic searches on Ankylosing Spondylitis. Each link opens a live PubMed query so the result set stays current as new studies are indexed.
- PubMed topic search: Ankylosing spondylitis review
- PubMed topic search: HLA-B27 ankylosing spondylitis
- PubMed topic search: Axial spondyloarthritis classification
- PubMed topic search: TNF inhibitor ankylosing spondylitis
- PubMed topic search: Secukinumab ankylosing spondylitis
- PubMed topic search: Ixekizumab spondylitis trial
- PubMed topic search: JAK inhibitor ankylosing spondylitis
- PubMed topic search: MRI sacroiliitis spondyloarthritis
- PubMed topic search: BASDAI BASFI ankylosing spondylitis
- PubMed topic search: Exercise physical therapy ankylosing spondylitis
- PubMed topic search: Non-radiographic axial spondyloarthritis
- PubMed topic search: Uveitis ankylosing spondylitis
Connections
- Deep Dives: HLA-B27 Explained · Biologics Guide · AS & IBD Overlap · Exercise & PT · NSAID Strategy · Morning Stiffness · Starch-Free Diet · Pregnancy & AS · Uveitis
- Rheumatology
- Arthritis
- Lupus
- Sjogren's Syndrome
- Fibromyalgia
- Turmeric
- Boswellia
- Vitamin D
- Magnesium
Featured Videos
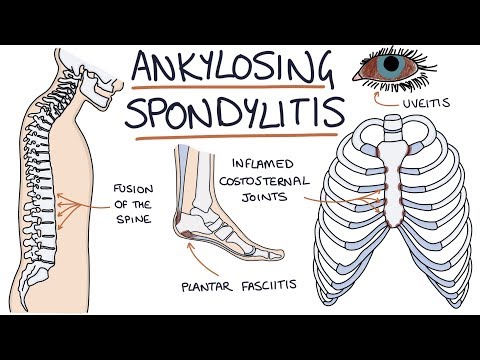
Ankylosing Spondylitis: Visual Explanation for Students
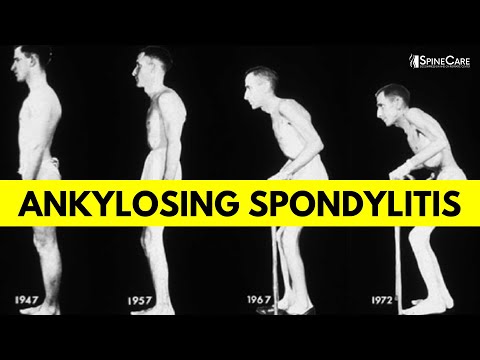
What is Ankylosing Spondylitis? Causes, Symptoms, and Diagnosis EXPLAINED

Q&A With Dr. J | Natural Treatments for Ankylosing Spondylitis

Have chronic back pain? You might have Ankylosing Spondylitis! | Rheumatologist Dr. Micah Yu

Ankylosing Spondylitis (AS) Treatment, Symptoms, Pathophysiology, Diagnosis, Medicine Lecture USMLE

10 Early Signs of Ankylosing Spondylitis You Shouldn’t Ignore!

7 Signs of Ankylosing Spondylitis - A Rheumatologist Review

Apollo Hospitals | What is Ankylosing Spondylitis? | Dr. Sundeep Upadhyaya
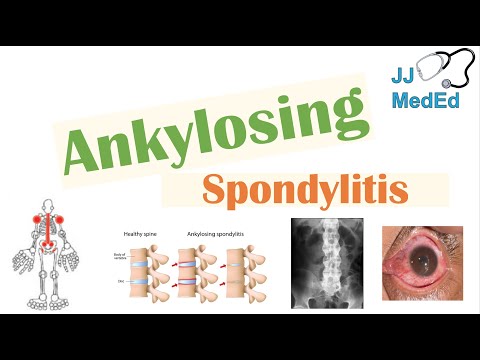
Ankylosing Spondylitis | HLA-B27, Pathophysiology, Signs & Symptoms, Diagnosis, Treatment