Lyme Disease
Deep-Dive Articles
Lyme disease rewards readers who go deep. The eight guides below each tackle one piece — how to read a tick bite, what that weird rash actually means, why the two-tier blood test misses early cases, which antibiotic for which stage, the controversy over “chronic Lyme”, the co-infections that travel with Borrelia, neuroborreliosis, and prevention that actually works. Start wherever you are right now.
Erythema Migrans Rash Guide
The bullseye isn’t always a bullseye — 20–30% of EM rashes are solid, and look-alikes (spider bites, cellulitis, ringworm, nummular eczema) trip people up. How to time the rash, what to photograph, and why rash alone is enough to start antibiotics.
Tick Bite First Aid & Prophylaxis
Step-by-step tick removal (fine-tipped tweezers, no petroleum jelly), saving the tick for ID, and the single-dose 200 mg doxycycline prophylaxis criteria — engorgement time, endemic area, 72-hour window.
Lyme Testing Explained
CDC two-tier serology, the modified two-tier (ELISA-then-ELISA) update, the early-window false negative, Western blot band interpretation, PCR and culture, and which “chronic Lyme” lab panels to avoid.
Antibiotic Treatment Protocols
Doxycycline, amoxicillin, cefuroxime, ceftriaxone — dose, duration, and stage-matched choices. Pregnancy and pediatric pivots. Why 10–14 days is usually enough, and when IV is truly indicated.
PTLDS & “Chronic Lyme”
Post-Treatment Lyme Disease Syndrome vs. the “chronic Lyme” label — what the RCTs on long-term antibiotics actually showed, why IDSA and ILADS disagree, and evidence-based symptom management that helps.
Tick-Borne Co-Infections
Babesia, Anaplasma, Ehrlichia, Bartonella, Powassan virus, hard-tick relapsing fever — when to suspect, what to test, and how co-infection changes the antibiotic plan.
Lyme Neuroborreliosis
Cranial neuropathy (Bell’s palsy), lymphocytic meningitis, painful radiculoneuritis (Bannwarth syndrome), late encephalopathy — how it presents, when to do LP, and IV vs. oral antibiotics.
Prevention & Tick Removal
DEET vs. picaridin vs. IR3535, permethrin-treated clothing, the daily tick check, landscape and yard tactics, and the clothing-and-shower drill that works in endemic areas.
Lyme Carditis & Cardiac Complications
AV block, syncope, and the reversible cardiac manifestation of early Lyme.
Table of Contents
- Deep-Dive Articles
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- References
- Featured Videos
1. Overview
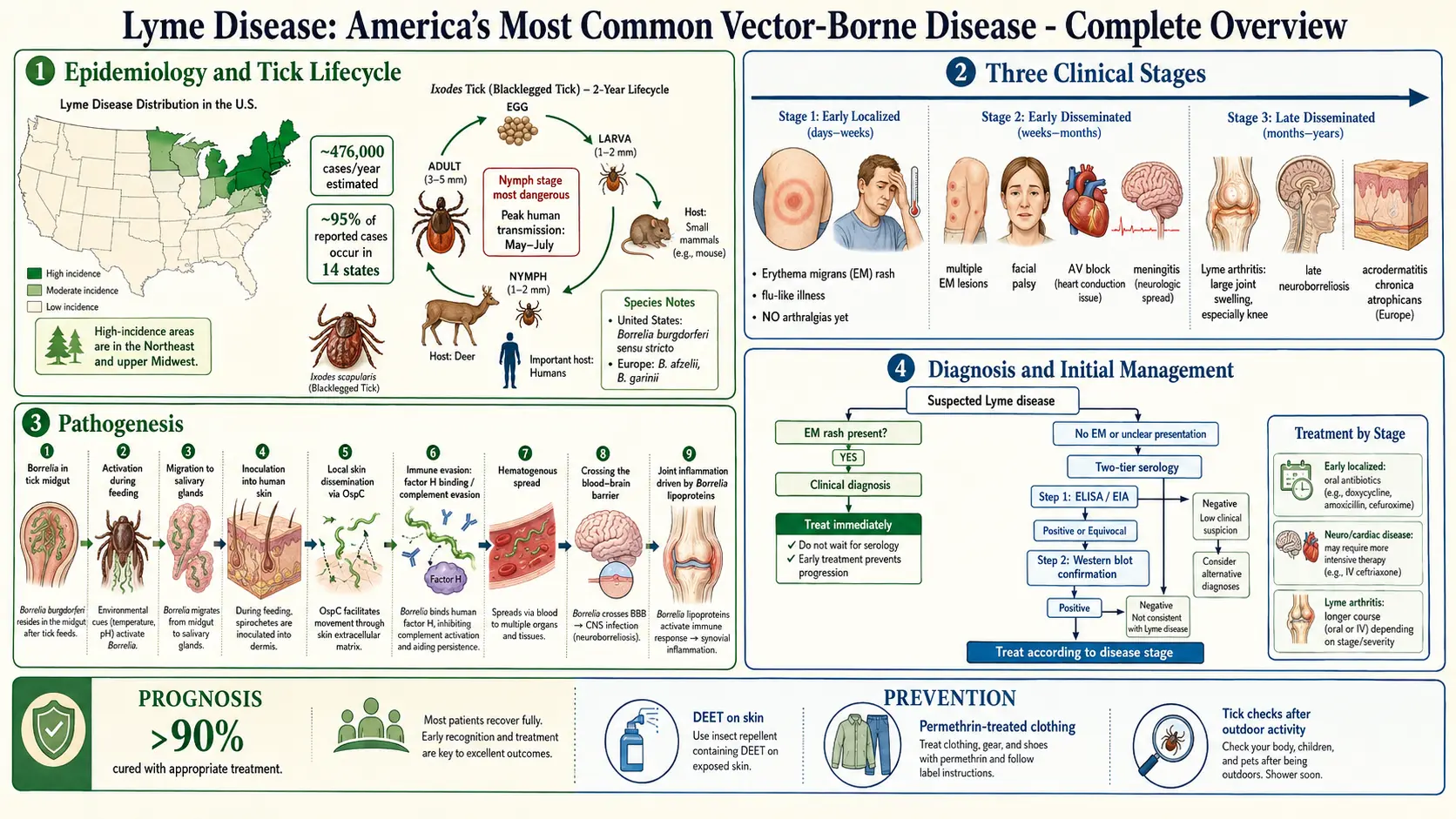
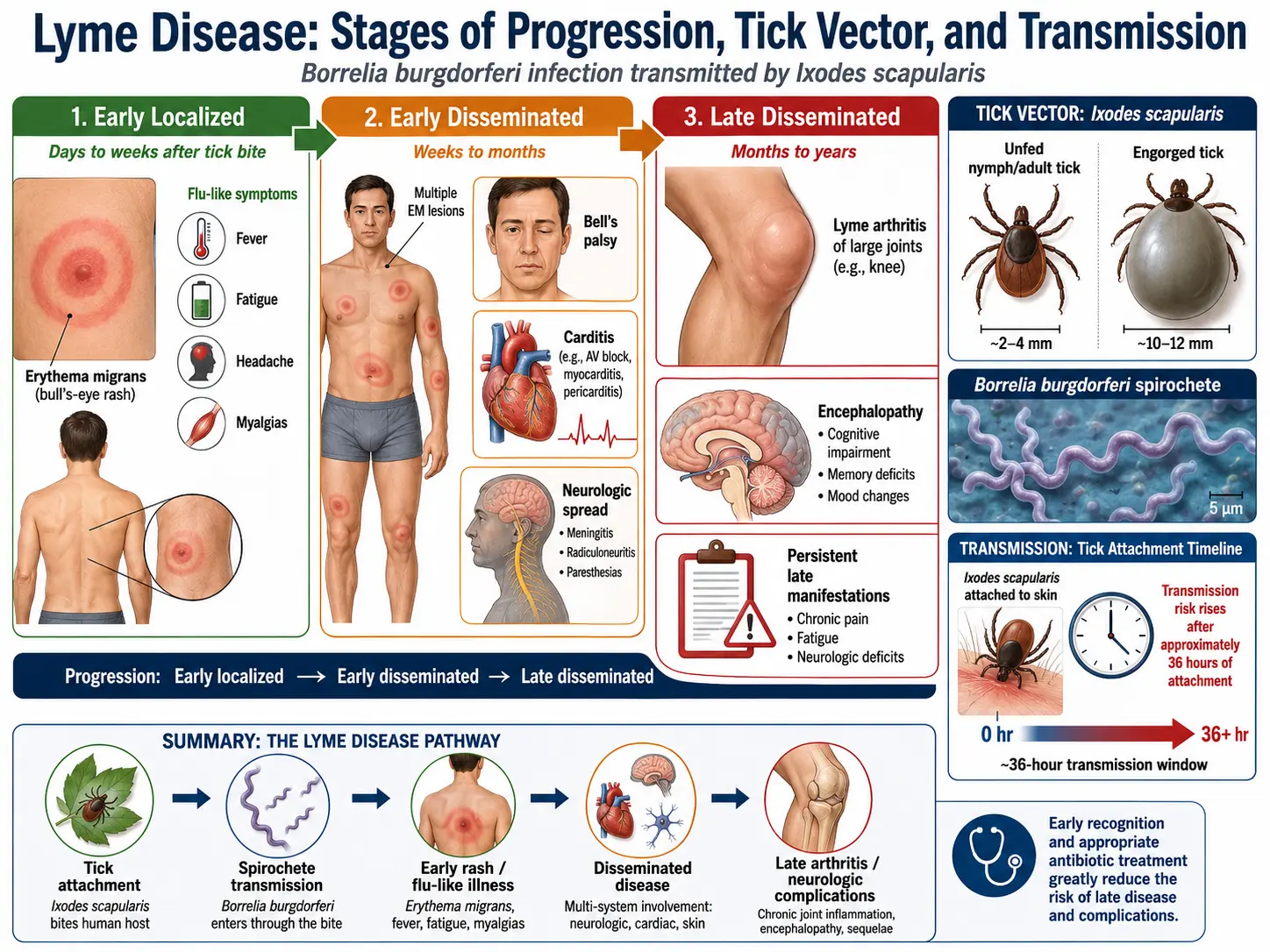
Lyme disease is the most common vector-borne infectious disease in the Northern Hemisphere, caused by spirochetes of the Borrelia burgdorferi sensu lato complex and transmitted by the bite of infected Ixodes species hard-bodied ticks. The disease was first formally described in 1977 following an investigation of an unusual cluster of juvenile arthritis cases in Lyme, Connecticut, by Allen Steere and colleagues; the causative organism was identified by Willy Burgdorfer in 1982.
Lyme disease is a multisystem illness that progresses through three clinical stages if untreated: early localized (erythema migrans), early disseminated (neurologic, cardiac, and musculoskeletal involvement), and late disseminated (Lyme arthritis, chronic neuroborreliosis). The vast majority of cases are curable with standard antibiotic therapy when diagnosed and treated promptly.
A significant minority of patients — approximately 10–20% — experience persistent symptoms (fatigue, cognitive difficulties, musculoskeletal pain) lasting months after completion of antibiotic therapy, a condition termed Post-Treatment Lyme Disease Syndrome (PTLDS) or, controversially, "chronic Lyme disease." The etiology of PTLDS remains debated; evidence does not support ongoing infection as the primary mechanism.
2. Epidemiology
Lyme disease is endemic across distinct geographic regions defined by the distribution of competent tick vectors and reservoir hosts:
- United States: Approximately 476,000 Americans are diagnosed and treated for Lyme disease each year (CDC estimate combining confirmed and probable cases). Concentrated in two geographic foci: the Northeast and Upper Midwest. Fourteen states account for >95% of confirmed cases, with highest incidence in Pennsylvania, New York, New Jersey, Connecticut, Massachusetts, Wisconsin, and Minnesota.
- Europe: Estimated 300,000 cases annually across Europe; B. afzelii and B. garinii predominate and cause different clinical syndromes (acrodermatitis chronica atrophicans and Lyme neuroborreliosis, respectively).
- Asia: Cases reported across northeastern China, Japan, Korea, and Russia; predominantly B. garinii and B. afzelii.
- Seasonality: Peak incidence May–August corresponding to nymphal Ixodes activity, when ticks are small (poppy-seed sized) and frequently undetected.
- Demographics: Bimodal age distribution with peaks in children aged 5–14 and adults aged 45–64; slight male predominance; highest risk in persons with outdoor occupational or recreational exposure in endemic areas.
- Emerging range: I. scapularis and I. pacificus geographic ranges are expanding northward into Canada, driven by climate warming — correspondingly expanding the geographic risk zone.
3. Pathophysiology
Borrelia burgdorferi is a highly motile, microaerophilic spirochete with a distinctive flat-wave morphology generated by periplasmic flagella. Its pathogenesis involves several distinct mechanisms:
Transmission and Early Dissemination
Transmission requires tick attachment for a minimum of 36–48 hours (nymphal tick) to allow spirochetes to migrate from the tick midgut to salivary glands and be inoculated into the host dermis. Tick saliva contains immunomodulatory molecules (prostaglandins, anti-complement proteins, anti-platelet factors) that facilitate spirochete survival at the bite site.
In the dermis, B. burgdorferi induces local inflammation via TLR1/2 and TLR5 recognition of borrelial lipoproteins and flagellin, activating NF-κB–mediated cytokine production (IL-6, TNF-α, IL-8). Spirochetes invade through the dermis, producing the pathognomonic erythema migrans (EM) rash.
Hematogenous Dissemination
Spirochetes enter the bloodstream and disseminate to multiple tissues including joints, CNS, heart, and skin. Dissemination involves binding to host fibronectin, decorin, and plasminogen via borrelial surface lipoproteins (OspA, OspC, DbpA/B, BBK32). BBB traversal occurs via transcellular and paracellular mechanisms, facilitated by spirochete-induced MMP activation and CXCL8 production.
Immune Evasion
B. burgdorferi employs multiple immune evasion strategies: VlsE (variable major protein-like sequence, expressed) undergoes antigenic variation in the mammalian host, altering surface lipoprotein expression to evade adaptive immunity. OspC/BBA68 binding to complement factor H confers serum resistance. Intracellular survival within macrophages and endothelial cells creates a partially protected niche. Persistence in the extracellular matrix (particularly joint collagen) reduces antibiotic penetrance.
Lyme Arthritis Immunopathology
In late Lyme arthritis, spirochete-induced TLR2 activation and molecular mimicry (between OspA and leukocyte function antigen-1, LFA-1) drive a Th1/Th17-dominant synovial inflammation characterized by IFN-γ, IL-17, and macrophage activation. In antibiotic-refractory Lyme arthritis, ongoing immunopathology driven by innate immune activation persists despite bacterial clearance — a mechanism distinct from active infection.
4. Etiology and Risk Factors
Causative Organisms
- Borrelia burgdorferi sensu stricto: Predominant cause in North America; also present in Europe.
- Borrelia afzelii: Predominant in Europe and Asia; typically causes milder disease with skin-predominant manifestations (ACA).
- Borrelia garinii: Europe and Asia; higher neurotropism, associated with Lyme neuroborreliosis.
- Borrelia spielmanii, bavariensis, mayonii: Additional genospecies causing clinical disease in Europe or North America.
Tick Vectors
- Ixodes scapularis (black-legged or deer tick): Primary vector in northeastern and upper midwestern US.
- Ixodes pacificus (western black-legged tick): Vector in western US; lower infection prevalence.
- Ixodes ricinus (castor bean tick): Primary vector in Europe.
- Ixodes persulcatus: Vector in Asia and eastern Russia.
Risk Factors
- Residence in or travel to endemic areas (Northeast/Upper Midwest US, Europe)
- Occupational or recreational exposure to tick habitats (leaf litter, tall grass, woodland edges, gardens)
- Duration of tick attachment (risk substantially lower with prompt tick removal)
- Tick density and infection prevalence in local area
- Outdoor activities without protective clothing or repellent use
- Pet ownership (dogs may carry ticks into the home)
- Not performing tick checks after outdoor exposure
5. Clinical Presentation
Stage 1: Early Localized Disease (Days to Weeks)
Erythema migrans (EM): Pathognomonic rash occurring in approximately 70–80% of infected individuals; appears 3–30 days (median 7 days) after tick bite at or near the bite site. Begins as a small red macule or papule, expands centrifugally to a diameter ≥5 cm (average 15 cm). Classic target or "bull's-eye" appearance (central clearing with concentric rings) occurs in a minority; most EM lesions are uniformly erythematous. Rash may be warm to touch but is usually painless and non-pruritic. Systemic symptoms may accompany EM: fatigue, myalgias, arthralgias, headache, fever (low-grade), regional lymphadenopathy — mimicking viral illness.
Stage 2: Early Disseminated Disease (Weeks to Months)
Multiple EM lesions: Secondary EM lesions at distant skin sites (hematogenous dissemination), smaller and often lacking central clearing.
Lyme neuroborreliosis (early): Occurs in approximately 10–15% of untreated cases. Classic triad: lymphocytic meningitis, cranial neuropathies (facial nerve palsy — bilateral in 25–30% of Lyme CN VII palsy), and painful radiculopathy (Bannwarth syndrome, particularly prominent in European disease). Facial nerve palsy in a child in an endemic area during summer-fall should be presumed Lyme until proven otherwise. Less common: encephalitis, transverse myelitis, mononeuritis multiplex, cerebellar ataxia.
Lyme carditis: Occurs in approximately 1–4% of untreated patients. Classically presents as varying degrees of atrioventricular (AV) block — first-degree (PR prolongation), Wenckebach second-degree, or complete (third-degree) AV block. The AV block is typically transient and fluctuating; high-degree block may require temporary pacing but rarely permanent pacing. Also myopericarditis and, rarely, pancarditis. Cardiac Lyme disease can cause sudden cardiac death.
Musculoskeletal: Migratory arthralgias, myalgias, and periarticular pain without frank arthritis; brief episodes of joint swelling may occur before established Lyme arthritis.
Ocular: Rare — conjunctivitis, uveitis, keratitis, optic neuritis, periorbital edema.
Stage 3: Late Disseminated Disease (Months to Years)
Lyme arthritis: Most common late manifestation in North America (60% of untreated patients). Mono- or oligoarticular arthritis predominantly affecting large joints — the knee is involved in >90% of cases. Episodes of swelling and pain lasting weeks to months, often intermittent early (with symptom-free intervals) but eventually becoming persistent. Synovial fluid shows inflammatory changes (WBC 10,000–100,000/mm³, predominantly neutrophils).
Late neuroborreliosis: Lyme encephalopathy (subtle cognitive deficits, memory impairment, concentration difficulties), chronic axonal polyneuropathy, and rarely progressive encephalomyelitis (more common with European genospecies).
Acrodermatitis chronica atrophicans (ACA): Characteristic of European B. afzelii infection; bluish-red discoloration and skin atrophy predominantly on distal extremities, progressing over months to years to parchment-like, atrophic skin with fibrosis. Rare in North America.
6. Diagnosis
Lyme disease diagnosis requires integration of epidemiologic exposure (endemic area, outdoor activity), clinical findings, and serologic testing. Diagnosis of early localized Lyme disease with classic EM is clinical — serology is negative in up to 50% of early EM cases.
CDC-Recommended Two-Tier Serologic Testing
- Tier 1: Sensitive enzyme immunoassay (EIA) or immunofluorescence assay (IFA): Screen for IgM and IgG antibodies to B. burgdorferi. If negative, testing stops (disease excluded in correct clinical context). If equivocal or positive, proceed to Tier 2.
- Tier 2: Western blot (IgM and IgG):
- IgM Western blot (first 4 weeks of illness): Positive if ≥2 of 3 bands present (23, 39, 41 kDa).
- IgG Western blot (after 4 weeks): Positive if ≥5 of 10 bands present (18, 23, 28, 30, 39, 41, 45, 58, 66, 93 kDa).
Modified two-tier testing (MTTT): FDA-cleared alternative using two EIAs (e.g., C6 ELISA + VlsE-based EIA) in place of Western blot; non-inferior sensitivity and specificity, reduced subjectivity, now endorsed by IDSA and CDC.
Important serologic caveats: Serology remains positive for years after successful treatment and cannot distinguish active infection from past exposure. IgM Western blot is less specific and should only be interpreted if symptom duration is <30 days. False positives occur with other spirochetal infections, EBV, autoimmune conditions. Seronegativity does not exclude early EM (diagnose clinically and treat).
Cerebrospinal Fluid (CSF) Analysis — Lyme Neuroborreliosis
LP indicated for suspected CNS involvement beyond isolated facial palsy. Findings: lymphocytic pleocytosis (50–500 cells/mm³), elevated protein, normal glucose. CSF Lyme antibody index (intrathecal antibody production ratio >1.3–2.0) is preferred over CSF serology alone for European neuroborreliosis. B. burgdorferi PCR on CSF has low sensitivity (<40%) but high specificity.
Synovial Fluid and Tissue PCR
PCR for B. burgdorferi on synovial fluid or tissue is highly sensitive (85%) and specific for Lyme arthritis; useful particularly for seronegative cases or when diagnosis is uncertain.
Cardiac Evaluation
12-lead ECG and continuous telemetry for all patients with suspected Lyme carditis (PR >300 ms warrants hospitalization and monitoring).
7. Treatment
All stages of Lyme disease are treatable with antibiotics. Early treatment is associated with better outcomes and lower risk of complications.
Early Localized Disease (Erythema Migrans)
- Doxycycline 100 mg PO twice daily × 10–14 days — first-line (also provides coverage for anaplasmosis, often co-transmitted).
- Amoxicillin 500 mg PO three times daily × 14 days — alternative (pregnancy, children <8 years).
- Cefuroxime axetil 500 mg PO twice daily × 14 days — alternative.
- Azithromycin is not recommended — inferior efficacy in clinical trials.
Early Disseminated Disease
Lyme neuroborreliosis (meningitis, radiculopathy, encephalitis):
- Ceftriaxone 2 g IV once daily × 14–28 days — preferred for CNS disease requiring IV access.
- Doxycycline 100–200 mg PO twice daily × 14–28 days — alternative for facial palsy without clinical meningitis (equivalent CNS penetration; non-inferior in European trials).
Isolated facial nerve palsy (Lyme): Oral doxycycline 100 mg twice daily × 14–21 days; corticosteroids not recommended.
Lyme carditis: First-degree AV block (PR <300 ms): oral doxycycline 100 mg twice daily × 14–21 days. High-degree AV block or PR >300 ms: ceftriaxone 2 g IV once daily until stabilized, then complete 21–28 days with oral agent. Temporary transvenous pacing for hemodynamically significant complete heart block.
Late Lyme Disease
Lyme arthritis: Doxycycline 100 mg PO twice daily × 28 days — first-line. Amoxicillin 500 mg TID × 28 days — alternative. If synovitis persists after initial oral course: second 28-day oral course OR ceftriaxone 2 g IV once daily × 2–4 weeks. NSAIDs and intra-articular corticosteroids may provide symptomatic relief for inflammatory arthritis.
Antibiotic-refractory Lyme arthritis (persistent after ≥2 courses, PCR-negative synovial fluid): Treat with DMARDs (hydroxychloroquine, methotrexate) or synovectomy — antibiotic-refractory arthritis is immunologically mediated, not caused by persistent infection.
Late neurologic Lyme disease: Ceftriaxone 2 g IV once daily × 14–28 days.
Post-Exposure Prophylaxis
Single dose doxycycline 200 mg PO within 72 hours of removal of an identified I. scapularis tick that was attached for ≥36 hours in an endemic area — approximately 87% efficacy in preventing EM (Nadelman et al., N Engl J Med 2001).
8. Complications
- Antibiotic-refractory Lyme arthritis: Approximately 10% of Lyme arthritis patients; requires immunomodulatory therapy.
- Lyme carditis complications: Complete heart block with syncope or sudden cardiac death (rare); reported cases of fatal Lyme carditis in young adults with undiagnosed disease.
- Neurocognitive sequelae: Cognitive impairment, fatigue, and mood disturbance may persist as PTLDS after adequate antibiotic treatment; not attributable to ongoing borrelial infection.
- Jarisch-Herxheimer reaction: Febrile exacerbation within 24 hours of starting antibiotics due to cytokine release from spirochetal killing; more common with IV therapy; managed supportively.
- Co-infections: Ixodes ticks may simultaneously transmit Anaplasma phagocytophilum (HGA), Babesia microti, Borrelia miyamotoi, and Powassan encephalitis virus — all capable of causing severe or fatal illness, particularly in immunocompromised patients.
- Post-Treatment Lyme Disease Syndrome (PTLDS): Persistent symptoms (fatigue, cognitive difficulties, musculoskeletal pain) >6 months after antibiotic treatment; four NIH-funded randomized controlled trials show no benefit of prolonged antibiotic therapy.
9. Prognosis
The overall prognosis of Lyme disease treated with standard antibiotic courses is excellent:
- Early localized EM: >95% cure rate with 10–14 days of doxycycline; virtually no long-term sequelae.
- Early disseminated (neurologic, cardiac): Complete recovery in most patients; AV block resolves in >90% within 6 weeks with treatment; facial palsy resolves completely in majority.
- Late Lyme arthritis: Resolution in approximately 85–90% after one or two antibiotic courses; antibiotic-refractory arthritis requires months of immunotherapy but generally responds.
- PTLDS: Symptoms typically improve spontaneously over months; severe or disabling PTLDS persists in a minority. Prolonged antibiotic therapy shows no benefit and exposes patients to significant harm (superinfection, line complications, C. difficile, antibiotic resistance).
- Untreated: Progression through disseminated stages is common; late neurologic disease may result in permanent deficits.
10. Prevention
- Personal protective measures: Wear long sleeves and pants (light-colored for tick visibility), tuck pants into socks in tick-infested areas; use EPA-registered repellents (DEET 20–30%, picaridin 20%, IR3535, oil of lemon eucalyptus, permethrin-treated clothing).
- Tick checks: Perform thorough full-body tick check after outdoor exposure, paying special attention to scalp, hairline, behind ears, axillae, groin, and popliteal fossa; check pets and gear. Shower within 2 hours of outdoor exposure.
- Tick removal: Use fine-tipped tweezers, grasp close to skin, pull steadily upward. Do not twist, crush, or apply heat/petroleum. Cleanse with alcohol or soap and water. Attached tick should be submitted for species identification and infection testing if available.
- Landscaping modifications: Keep grass mowed, clear leaf litter and brush, create wood-chip or gravel barriers between lawn and wooded areas, apply acaricides (tick-killing pesticides).
- Post-exposure prophylaxis: Single-dose doxycycline 200 mg within 72 hours of high-risk tick bite (as above).
11. Recent Research and Advances
- Modified two-tier testing (MTTT): Widely adopted, replacing Western blot with a second EIA — FDA-cleared MTTT algorithms show improved sensitivity for early disease and equivalent specificity.
- CXCL13 as CSF biomarker: Elevated CSF CXCL13 (B-cell attracting chemokine) is a sensitive and specific marker of active Lyme neuroborreliosis, potentially useful when serology is indeterminate.
- Borrelia persistence and PTLDS mechanisms: Studies of spirochete persistence in immune-privileged sites (brain, collagen-rich tissues) using primate models; immune activation and autoimmune mechanisms (molecular mimicry) rather than persistent spirochetal infection appear to drive PTLDS.
- Emerging tick-borne co-infections: Borrelia miyamotoi (relapsing fever group), Ehrlichia muris eauclairensis, and Heartland phlebovirus expanding in range; clinical significance increasing.
- Tick population genomics: Whole-genome sequencing of I. scapularis and B. burgdorferi populations is informing our understanding of range expansion and genomic diversity.
- Lyme disease biobank: SLICE (Surveillance, Lyme disease and Infectious Conditions Epidemiology) cohort enrolling prospective samples to identify PTLDS biomarkers and predictors of disease severity.
12. References
- Steere AC, Malawista SE, Hardin JA, et al. Erythema chronicum migrans and Lyme arthritis. Ann Intern Med. 1977;86:685–698.
- Burgdorfer W, Barbour AG, Hayes SF, et al. Lyme disease — a tick-borne spirochetosis? Science. 1982;216:1317–1319.
- Lantos PM, Rumbaugh J, Bockenstedt LK, et al. Clinical Practice Guidelines by the Infectious Diseases Society of America (IDSA), American Academy of Neurology (AAN), and American College of Rheumatology (ACR): 2020 Guidelines for the Prevention, Diagnosis and Treatment of Lyme Disease. Clin Infect Dis. 2021;72:e1–e48.
- Nadelman RB, Nowakowski J, Fish D, et al. Prophylaxis with single-dose doxycycline for the prevention of Lyme disease after an Ixodes scapularis tick bite. N Engl J Med. 2001;345:79–84.
- Wormser GP, Dattwyler RJ, Shapiro ED, et al. The clinical assessment, treatment, and prevention of Lyme disease, human granulocytic anaplasmosis, and babesiosis: clinical practice guidelines by the IDSA. Clin Infect Dis. 2006;43:1089–1134.
- Steere AC, Strle F, Wormser GP, et al. Lyme borreliosis. Nat Rev Dis Primers. 2016;2:16090.
- Klempner MS, Hu LT, Evans J, et al. Two controlled trials of antibiotic treatment in patients with persistent symptoms and a history of Lyme disease. N Engl J Med. 2001;345:85–92.
- Rebman AW, Aucott JN. Post-treatment Lyme disease as a model for persistent symptoms in Lyme disease. Front Med. 2020;7:57.
- Barbour AG, Fish D. The biological and social phenomenon of Lyme disease. Science. 1993;260:1610–1616.
- Strle F, Stanek G. Clinical manifestations and diagnosis of Lyme borreliosis. Curr Probl Dermatol. 2009;37:51–110.
- Shapiro ED. Lyme disease. N Engl J Med. 2014;370:1724–1731.
- Bockenstedt LK, Gonzalez DG, Haberman AM, et al. Spirochete antigens persist near cartilage after murine Lyme borreliosis therapy. J Clin Invest. 2012;122:2652–2660.
- CDC. Lyme Disease Surveillance Data. https://www.cdc.gov/lyme/datasurveillance/index.html
- Krause PJ, Narasimhan S, Wormser GP, et al. Human Borrelia miyamotoi infection in the United States. N Engl J Med. 2013;368:291–293.
- Bamm VV, Ko JT, Mainprize IL, et al. Lyme disease frontiers: reconciling Borrelia biology and clinical conundrums. Pathogens. 2019;8:299.
- Mead P, Petersen J, Hinckley A. Updated CDC recommendation for serologic diagnosis of Lyme disease. MMWR Morb Mortal Wkly Rep. 2019;68:703.
Research Papers
The following PubMed topic searches retrieve current peer-reviewed literature on Lyme Disease. Each link opens a live PubMed query so you always see the most recent publications.
- Lyme disease review
- Borrelia burgdorferi pathogenesis
- Lyme disease doxycycline treatment
- Post-treatment Lyme disease syndrome
- Lyme disease erythema migrans
- Lyme neuroborreliosis diagnosis
- Lyme carditis atrioventricular block
- Lyme arthritis chronic
- IDSA Lyme disease guideline
- Ixodes tick Lyme epidemiology
- Lyme disease vaccine OspA
- Cat's claw uncaria Lyme
Connections
- Lyme Neuroborreliosis
- Erythema Migrans Rash
- Tick-Borne Co-Infections
- Antibiotic Treatment Protocols
- Lyme Testing Explained
- Lyme Carditis
- Prevention and Tick Removal
- PTLDS and Chronic Lyme
- Tick Bite First Aid
- Tick-Origin Science
- Cat's Claw
- Alpha-Gal Syndrome
- Tick Bite Prevention
- Garlic
- Immune Boosting
- Japanese Knotweed
- Andrographis
- Vitamin D3