Tick-Borne Co-Infections
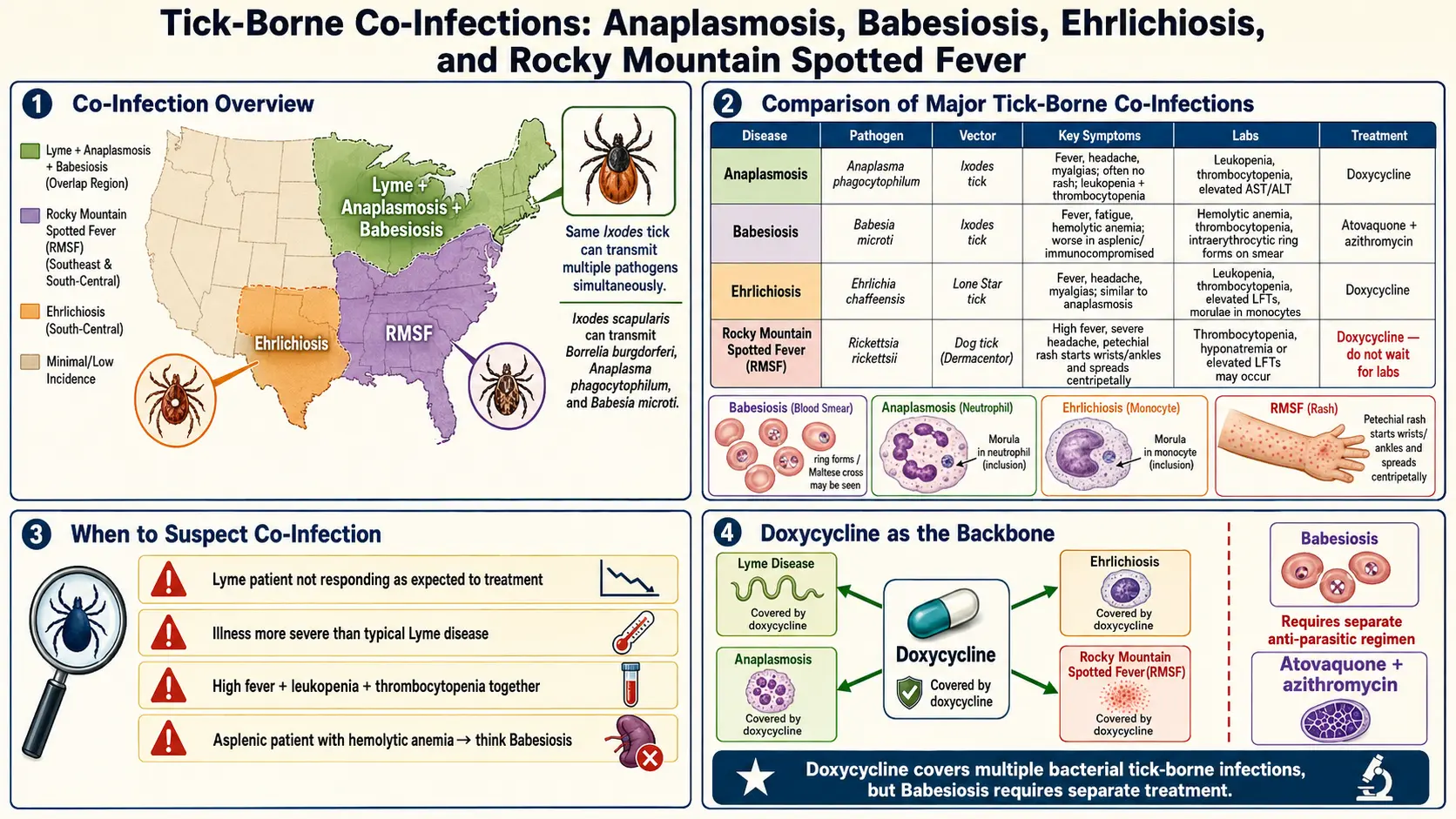
Table of Contents
- Why Co-Infections Matter
- Babesiosis
- Human Granulocytic Anaplasmosis (HGA)
- Human Monocytic Ehrlichiosis (HME)
- Bartonellosis
- Powassan Virus
- Hard-Tick Relapsing Fever (Borrelia miyamotoi)
- Rocky Mountain Spotted Fever
- Alpha-Gal Syndrome
- Tularemia, Colorado Tick Fever, Tick Paralysis
- When to Test for Co-Infections
- Testing Panels and Reputable Labs
- Empirical Treatment Decisions
- Geographic Risk
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
Why Co-Infections Matter
When a tick bites you, it does not necessarily deliver only one pathogen. A single Ixodes scapularis nymph in the Northeast may simultaneously carry Borrelia burgdorferi (Lyme), Babesia microti, Anaplasma phagocytophilum, Borrelia miyamotoi, and occasionally Powassan virus. Regional surveys have reported co-infection rates of 2–12% in adult Ixodes ticks across Lyme-endemic states, and human co-infection rates of 4–10% among confirmed Lyme cases in the Northeast.
For patients, the practical consequences are three-fold:
- Prolonged or atypical symptoms. A classic early Lyme case usually looks like flu plus an expanding rash. When drenching night sweats, air hunger, severe headache, hemolytic anemia, or low blood counts appear, something else is in the picture.
- Treatment failure. Doxycycline — the standard early-Lyme drug — covers Borrelia burgdorferi, Anaplasma, Ehrlichia, and Rickettsia, but has no activity against Babesia. A patient who improves only partially on doxycycline may be harboring untreated babesiosis.
- Severity out of proportion. Lyme disease is rarely lethal in otherwise healthy adults. Severe babesiosis, fulminant anaplasmosis, Powassan encephalitis, and Rocky Mountain spotted fever all can kill quickly. Recognizing a co-infection is the difference between an outpatient visit and an ICU admission.
This page is organized pathogen-by-pathogen so you can use it as a reference. Each section tells you what the bug is, how it makes you feel, how doctors confirm it, and how it is treated. The last third of the article covers when to ask for testing, which labs to trust, and how empirical antibiotic choices stack up when multiple pathogens are on the table.
Babesiosis
The bug: Babesia microti (dominant in the Northeast and Upper Midwest) and Babesia duncani (Pacific Coast). These are intraerythrocytic protozoa — parasites that live inside red blood cells — closely related to the agents of malaria. The same Ixodes scapularis tick that transmits Lyme also transmits B. microti.
How it feels: Babesiosis produces a malaria-like illness. The hallmarks are:
- Drenching night sweats — patients wake up needing to change sheets.
- Air hunger — a distinctive shortness of breath with ordinary activity, driven by hemolytic anemia.
- Dark (cola-colored) urine from hemoglobinuria.
- Profound fatigue, high fever, chills, headache, and myalgia.
- Lab findings: hemolytic anemia, thrombocytopenia, elevated LDH, low haptoglobin, indirect hyperbilirubinemia.
Who gets severe disease: People without a functioning spleen (surgical splenectomy, sickle cell disease), the immunocompromised (HIV, chemotherapy, rituximab), and adults over 50 are at highest risk for fulminant disease with parasitemia above 10%, ARDS, DIC, and multi-organ failure. Healthy young adults often have a milder, flu-like course and can recover on their own, which is exactly why the diagnosis gets missed.
Diagnosis: Thin-film Giemsa-stained blood smear is the bedside gold standard — the parasites look like small rings or the classic tetrad ("Maltese cross") inside red cells. Because parasitemia can be low, follow up a suspicious smear with PCR, which is more sensitive. Serology (IgM/IgG) supports the diagnosis but lags behind the acute illness.
Treatment: Standard outpatient regimen for mild-to-moderate disease is atovaquone (750 mg twice daily) plus azithromycin (500 mg day 1, then 250–500 mg daily) for 7–10 days. For severe disease, hospital-based clindamycin plus oral quinine (or IV azithromycin) is used, sometimes with exchange transfusion when parasitemia exceeds 10% or end-organ injury appears. Immunocompromised patients often need 6 weeks or more of therapy.
Human Granulocytic Anaplasmosis (HGA)
The bug: Anaplasma phagocytophilum, an obligate intracellular bacterium that infects neutrophils. Shares the same Ixodes scapularis / pacificus vector as Lyme.
How it feels: Abrupt fever, rigors, severe headache, myalgia, malaise — a "summer flu" that appears 1–2 weeks after an outdoor exposure. Rash is uncommon (unlike RMSF or Lyme EM). Labs classically show leukopenia, thrombocytopenia, and elevated transaminases — a useful triad to recognize.
Severity: Most cases are self-limited, but older adults and immunocompromised patients can develop respiratory failure, rhabdomyolysis, or opportunistic superinfection. Mortality is roughly 0.5–1%.
Diagnosis: PCR on whole blood in the acute phase is the most reliable test. Peripheral smear may show morulae (clusters of bacteria) inside neutrophils in 20–80% of cases. Paired acute and convalescent serology (IgG four-fold rise) confirms retrospectively.
Treatment: Doxycycline 100 mg twice daily for 10 days. Fever should break within 48 hours — a failure to defervesce on doxycycline is a reason to reconsider the diagnosis. Fortunately, standard Lyme-dose doxycycline covers anaplasmosis simultaneously, which is why empirical dual coverage is so forgiving.
Human Monocytic Ehrlichiosis (HME)
The bug: Ehrlichia chaffeensis (dominant) and Ehrlichia ewingii, intracellular bacteria that infect monocytes. Transmitted primarily by the lone star tick (Amblyomma americanum) rather than Ixodes, which shifts the geographic range toward the Southeast, South Central, and lower Midwest states.
How it feels: Fever, severe headache, myalgia, nausea, and fatigue, with the same leukopenia / thrombocytopenia / elevated LFT triad seen in anaplasmosis. A maculopapular or petechial rash appears in about a third of adults and two-thirds of children — more often than in HGA. Severe cases can progress to meningoencephalitis, ARDS, or a sepsis-like picture with DIC.
Diagnosis: PCR on whole blood is the first-line acute test. Morulae may be seen in monocytes on smear but are less commonly found than in HGA. Serology confirms retrospectively.
Treatment: Doxycycline 100 mg twice daily for 10–14 days, regardless of age. The CDC position is explicit: doxycycline is the drug of choice even in young children when ehrlichiosis or RMSF is suspected, because delayed treatment kills and the short course does not stain teeth meaningfully.
Bartonellosis
The bugs: Bartonella henselae (cat-scratch disease), Bartonella quintana (trench fever, louse-borne), and a handful of other species. These are small, fastidious, intraerythrocytic/endothelial bacteria.
The controversy: Bartonella transmission by cat scratches and flea bites is well established. Transmission by Ixodes ticks is biologically plausible but not proven. PCR surveys have detected Bartonella DNA in questing ticks, but experimental transmission to mammals through tick bite has not been cleanly demonstrated. Mainstream U.S. infectious disease societies (IDSA) do not consider Bartonella a confirmed tick-borne pathogen. ILADS and many Lyme-literate clinicians treat it as a probable co-infection in patients with the right symptom cluster. We note the disagreement honestly and leave the clinical judgment to you and your physician.
How it feels (the cluster most often attributed to tick-associated Bartonella): prolonged low-grade fever, tender lymphadenopathy, sore soles in the morning, ocular involvement (neuroretinitis, uveitis), striae-like skin stretch marks in unusual distributions, and a neuropsychiatric component that can include anxiety, rage attacks, insomnia, and brain fog.
Diagnosis: Serology (IgM/IgG) and PCR are both insensitive. Specialty labs offer enriched culture and droplet-digital PCR with better yield but imperfect validation. Diagnosis is often clinical when it is made at all.
Treatment: Typical regimens use azithromycin or doxycycline, often combined with rifampin, for 4–6 weeks. Severe neurological or ocular disease may require longer IV therapy. There is no single guideline-endorsed regimen for suspected tick-borne bartonellosis, and response is often incomplete.
Powassan Virus
The bug: A flavivirus (same family as West Nile and tick-borne encephalitis virus), transmitted by Ixodes scapularis and Ixodes cookei. Unusual among tick-borne infections in that transmission can occur within as little as 15 minutes of tick attachment — there is no safe "prompt removal" window as there is for Lyme.
How it feels: Early symptoms are nonspecific — fever, headache, vomiting, weakness — arising 1 to 4 weeks after bite. Roughly half of recognized cases progress to encephalitis or meningoencephalitis with seizures, focal neurologic deficits, and altered consciousness. Mortality in severe cases is 10–15%, and roughly half of survivors are left with permanent neurologic sequelae.
Diagnosis: CSF and serum IgM by reference laboratories (state health departments, CDC). MRI may show characteristic abnormalities in the basal ganglia, thalami, and cerebellum.
Treatment: There is no specific antiviral. Supportive care in a neurocritical-care setting. IVIG and high-dose steroids have been used anecdotally without proven benefit. Prevention — permethrin-treated clothing, DEET or picaridin on skin, and careful tick checks — is the only real control.
Hard-Tick Relapsing Fever (Borrelia miyamotoi)
The bug: Borrelia miyamotoi, a relapsing-fever-group spirochete distinct from the Borrelia burgdorferi that causes Lyme. Shares the Ixodes vector.
How it feels: High fever with chills, severe headache, myalgia, fatigue, and sometimes relapsing fever spikes every few days. Rash and classic EM are uncommon. Immunocompromised patients can develop meningoencephalitis. Because the early syndrome looks flu-like without an EM rash, B. miyamotoi is under-diagnosed — a surprising fraction of "summer fever of unclear cause" in the Northeast is traceable to this organism.
Diagnosis: PCR on whole blood during a febrile episode is the most sensitive test. Standard Lyme two-tier serology does not detect B. miyamotoi reliably — a negative Lyme Western blot does not exclude it. Specialty B. miyamotoi-specific serology is available at reference labs.
Treatment: Doxycycline 100 mg twice daily for 14 days for outpatient disease; ceftriaxone IV for meningoencephalitis. Same drugs as Lyme, which again means empirical doxycycline for a tick-bite flu covers this organism too.
Rocky Mountain Spotted Fever
The bug: Rickettsia rickettsii, transmitted by Dermacentor variabilis (American dog tick) in the East, Dermacentor andersoni (Rocky Mountain wood tick) in the West, and Rhipicephalus sanguineus (brown dog tick) in Arizona and Mexico. Not an Ixodes co-infection — it travels with a different tick in different habitats.
How it feels: Abrupt fever, severe headache, myalgia, nausea. The hallmark rash begins 2–5 days into illness: small pink macules on the wrists and ankles, then spreading centripetally to the trunk, palms, and soles, and often becoming petechial. The rash is absent in 10–15% of cases ("Rocky Mountain spotless fever"), which is one reason this infection is missed.
Severity: A medical emergency. Untreated mortality is 20–30%; even with treatment, mortality remains 5–10%, and delays beyond day 5 of illness dramatically worsen outcomes. The pathology is a systemic small-vessel vasculitis — shock, renal failure, pulmonary edema, and encephalitis all follow.
Diagnosis: Clinical. Serology only becomes positive in the second week, long after the window to prevent death has closed. Do not wait for laboratory confirmation. If fever plus headache plus tick exposure plus any compatible rash is present in an endemic area, start treatment immediately and send paired serology for retrospective confirmation.
Treatment: Doxycycline 100 mg twice daily for at least 3 days after defervescence (typically 5–7 days total) — in all ages, including children under 8. The CDC reversed the old pediatric doxycycline concern specifically because RMSF kills fast and the drug works.
Alpha-Gal Syndrome
What it is: Not an infection at all — an IgE-mediated allergy to galactose-alpha-1,3-galactose, a sugar found in most non-primate mammalian meat. A bite from the lone star tick (Amblyomma americanum) introduces the sugar in the tick's saliva and primes the immune system to react.
How it feels: Delayed-onset urticaria, angioedema, GI symptoms, or anaphylaxis 3–6 hours after eating beef, pork, lamb, venison, or other mammalian meat (poultry and fish are safe). The delay between meal and reaction is what makes the diagnosis tricky — patients wake at 2 a.m. with hives after dinner at 8 p.m., and never suspect the burger.
See the full Alpha-Gal Syndrome article for management, diagnostic testing (specific IgE to galactose-alpha-1,3-galactose), and hidden sources of mammalian products in food and medications (gelatin capsules, magnesium stearate, some vaccines, heparin).
Tularemia, Colorado Tick Fever, Tick Paralysis
Tularemia (Francisella tularensis). A small Gram-negative bacterium transmitted by Dermacentor and Amblyomma ticks, as well as by handling infected rabbits. The ulceroglandular form produces a skin ulcer at the bite with regional lymphadenopathy and fever. Severe pneumonic disease can follow inhalation. Treatment is streptomycin or gentamicin; doxycycline or ciprofloxacin are alternatives. Consider in hunters, landscapers, and anyone handling wild lagomorphs.
Colorado tick fever (Colorado tick fever virus). A reovirus transmitted by Dermacentor andersoni in the western mountain states above 4,000 feet elevation. Biphasic fever, headache, myalgia over 5–10 days, sometimes with leukopenia. Supportive care only — no antiviral.
Tick paralysis. A toxin-mediated paralysis caused by a saliva neurotoxin from engorged female ticks (most often Dermacentor species). Ascending flaccid paralysis mimics Guillain-Barré syndrome over hours to days. The cure is simply finding and removing the tick — patients typically recover within 24 hours. Always check the scalp and hairline when an unexplained acute paralysis appears in a child.
When to Test for Co-Infections
Not every Lyme patient needs an expensive co-infection panel. Test when any of the following is present:
- Severity out of proportion to expected Lyme — high fever beyond a few days, shortness of breath, confusion, jaundice, or a rapidly falling hemoglobin.
- Symptoms persisting or worsening after 48–72 hours of appropriate doxycycline for Lyme. Babesiosis does not respond to doxycycline and is the most common reason for this pattern.
- Fevers continuing after completion of standard EM-stage Lyme therapy.
- Cytopenias — thrombocytopenia, leukopenia, or hemolytic anemia on the CBC.
- Elevated transaminases or LDH without another explanation.
- Drenching night sweats, air hunger, or dark urine — a babesiosis-specific cluster.
- Asplenia, immunosuppression, or age over 50 combined with any tick-borne illness.
- Petechial rash, wrists and ankles first — RMSF until proven otherwise.
- Neurologic decline (confusion, seizures, focal deficits) after tick exposure — Powassan, neuroborreliosis, or severe anaplasmosis/ehrlichiosis.
Testing Panels and Reputable Labs
Use established diagnostic laboratories. Reasonable choices:
- CDC and state public health laboratories. Gold standard for Powassan, tularemia, and confirmatory Rickettsial serology. Your physician orders through the state health department.
- LabCorp and Quest Diagnostics. Commercial labs with validated PCR and serology for Babesia microti, Anaplasma, Ehrlichia, Rickettsia, and Lyme.
- ARUP Laboratories, Mayo Clinic Laboratories. Reference laboratories with high-quality tick-borne panels including Borrelia miyamotoi-specific serology.
- IGeneX, Galaxy Diagnostics, T-Lab. Specialty labs used by ILADS-oriented clinicians. Validation and reproducibility are more variable — the expanded panels sometimes catch true infections missed elsewhere and sometimes generate false-positives on marginal bands. Interpret results with a clinician familiar with their assays.
Avoid unvalidated "tick panels" that bundle a dozen assays with poorly characterized specificity. A single well-chosen PCR or serology on the pathogen your symptoms actually suggest will serve you better than a shotgun panel that generates ambiguous bands requiring more testing to sort out.
Empirical Treatment Decisions
The practical value of understanding co-infections is that it sharpens empirical antibiotic choices when a patient is sick and the lab is hours or days behind the bedside.
- Doxycycline 100 mg twice daily covers Borrelia burgdorferi, Anaplasma phagocytophilum, Ehrlichia chaffeensis, Borrelia miyamotoi, and Rickettsia rickettsii. For most tick-bite febrile syndromes in the Northeast and Southeast, it is a single-drug, near-universal empiric regimen. Give it early.
- Add atovaquone plus azithromycin when babesiosis is suspected — drenching sweats, air hunger, hemolysis, thrombocytopenia, or an asplenic/immunocompromised patient. Doxycycline alone is insufficient.
- Switch to IV ceftriaxone when neuroborreliosis, severe B. miyamotoi CNS involvement, or disseminated Lyme with carditis is suspected. See the Antibiotic Treatment Protocols page for detailed regimens.
- Consider rifampin plus azithromycin or doxycycline for suspected bartonellosis, acknowledging the diagnostic uncertainty.
- Supportive care only for Powassan virus, Colorado tick fever, and tick paralysis (remove the tick).
Treat empirically when delay is dangerous. This is especially true for RMSF and severe babesiosis — waiting for serology can cost lives.
Geographic Risk
Tick-borne pathogens do not distribute uniformly. Knowing the local ecology changes the pretest probability of each infection:
- Northeast (Maine through Virginia) and Upper Midwest (Wisconsin, Minnesota). Ixodes scapularis territory. Expect Lyme, Babesia microti, Anaplasma phagocytophilum, Borrelia miyamotoi, and sporadic Powassan.
- Southeast and South Central (Virginia through Texas and Oklahoma). Amblyomma americanum (lone star) territory. Ehrlichiosis, alpha-gal syndrome, tularemia, STARI (southern tick-associated rash illness, a Lyme mimic).
- Mid-Atlantic and Southeast. Dermacentor variabilis (American dog tick) territory. Rocky Mountain spotted fever is concentrated here despite the name — North Carolina, Oklahoma, Arkansas, Missouri, and Tennessee report the majority of U.S. RMSF cases.
- Rocky Mountain states and Pacific Northwest. Dermacentor andersoni territory — RMSF, Colorado tick fever, tick paralysis, tularemia.
- Pacific Coast (California, Oregon, Washington). Ixodes pacificus territory. Western Lyme, anaplasmosis, and the Babesia duncani clade (rather than B. microti).
- Arizona. Rhipicephalus sanguineus (brown dog tick) is the RMSF vector in peri-urban settings with stray dog populations, especially on tribal lands.
When a traveler presents with a febrile illness, ask where they have been in the last month. Co-infection probabilities ride on geography as much as on the bug's biology.
Key Research Papers
- Lantos PM, et al. Clinical Practice Guidelines by the Infectious Diseases Society of America, American Academy of Neurology, and American College of Rheumatology: 2020 Guidelines for the Prevention, Diagnosis and Treatment of Lyme Disease. Clin Infect Dis. 2021.
- Vannier E, Krause PJ. Human babesiosis. N Engl J Med. 2012;366:2397–2407.
- Biggs HM, et al. Diagnosis and Management of Tickborne Rickettsial Diseases: Rocky Mountain Spotted Fever and Other Spotted Fever Group Rickettsioses, Ehrlichioses, and Anaplasmosis — United States. MMWR Recomm Rep. 2016;65(2):1–44.
- Molloy PJ, et al. Borrelia miyamotoi Disease in the Northeastern United States: A Case Series. Ann Intern Med. 2015;163(2):91–98.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on tick-borne co-infections, diagnosis, and management:
- Babesia microti co-infection with Lyme disease
- Anaplasma phagocytophilum diagnosis and treatment
- Ehrlichia chaffeensis and human monocytic ehrlichiosis
- Powassan virus and tick-borne encephalitis
- Borrelia miyamotoi hard-tick relapsing fever
- Rocky Mountain spotted fever and empirical doxycycline
- Bartonella and tick-borne transmission controversy
- Alpha-gal syndrome and the lone star tick
Connections
- Alpha-Gal Syndrome
- Antibiotic Treatment Protocols
- Lyme Disease Overview
- Malaria
- Lyme Neuroborreliosis
- Prevention and Tick Removal
- PTLDS and Chronic Lyme
- Tick Bite First Aid
- Lyme Testing Explained
- Erythema Migrans Rash
- Alpha-Gal Natural History
- Headache
- Thrombocytopenia
- Anemia
- Fatigue
- Lyme Carditis
- Parasites
- Tick Bite Prevention