Lyme Neuroborreliosis — When Lyme Affects the Nervous System
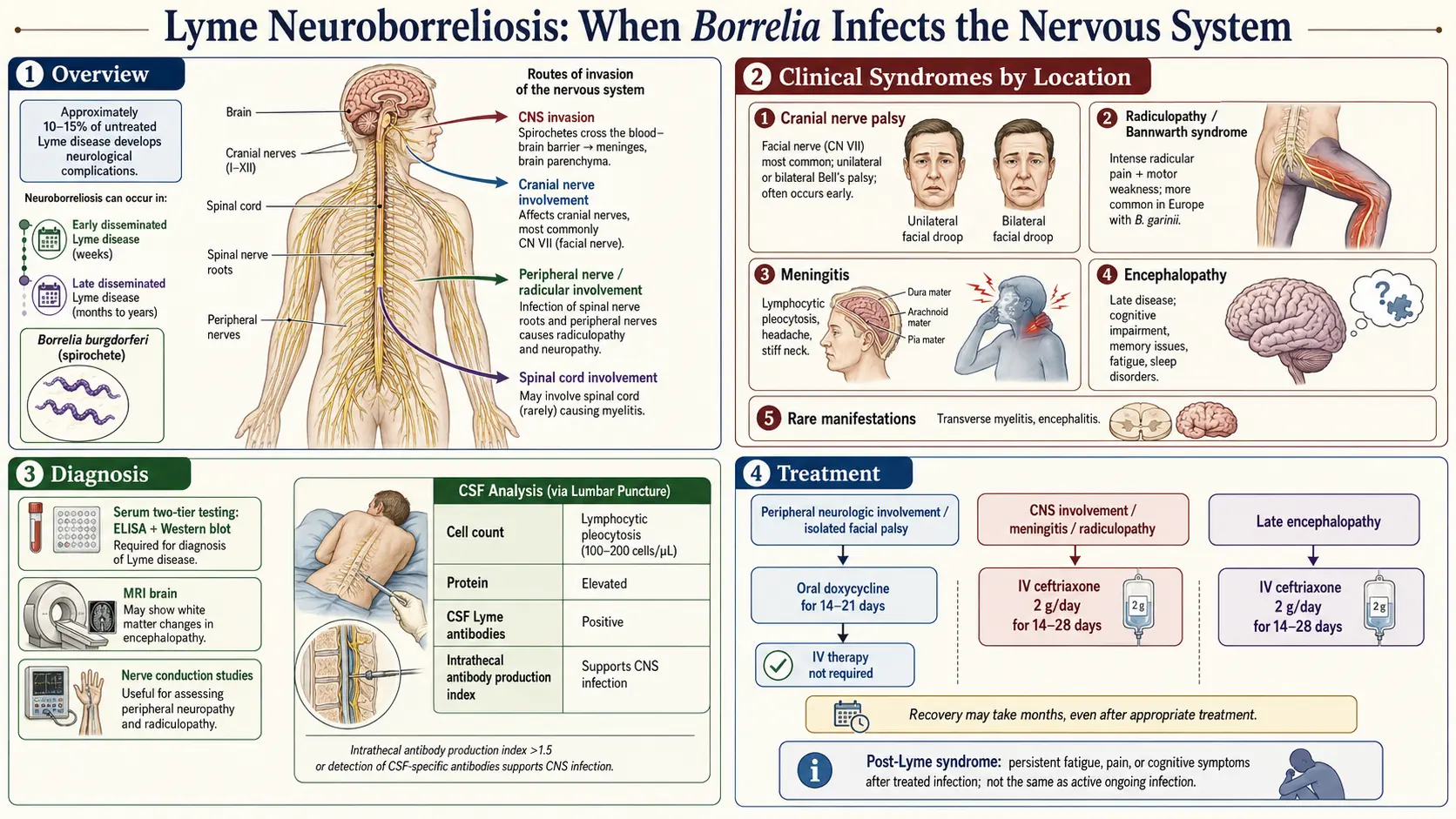
Table of Contents
- What Neuroborreliosis Is
- Cranial Neuropathy — The Most Common U.S. Presentation
- Lymphocytic Meningitis
- Painful Radiculoneuritis (Bannwarth Syndrome)
- Late-Stage Encephalomyelitis
- Lyme Encephalopathy
- Peripheral Neuropathy
- Diagnostic Workup
- When to Do a Lumbar Puncture — and When Not To
- Treatment — IV vs Oral
- Bell's Palsy Specifically
- Recovery Timeline
- Residual Symptoms After Treatment
- Red Flags Requiring the ER
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
What Neuroborreliosis Is
Neuroborreliosis is what doctors call it when the Lyme disease bacterium, Borrelia burgdorferi (and its European cousins B. garinii and B. afzelii), crosses into the nervous system. The bug can slip past the blood-brain barrier within weeks of the tick bite, seed the cranial nerves, the meninges (the lining around the brain and spinal cord), and the nerve roots where they exit the spine. From there it sets off an inflammatory response that produces the specific patterns described below.
Roughly 10–15% of untreated U.S. Lyme patients develop some neurologic involvement, usually in the early-disseminated phase two to twelve weeks after the bite. In Europe the number is higher, because B. garinii is noticeably more neurotropic — it has a stronger affinity for nerve tissue — and roughly a third of European neuroborreliosis cases present with painful nerve-root inflammation, something rarely seen in North America.
Neurologic symptoms are frightening. A drooping face, a splitting headache that will not quit, shooting pains that wake you at 3 a.m. — these feel like a stroke, a tumor, or something permanent. The reassuring truth is that the vast majority of neuroborreliosis cases respond completely to a two-to-three-week course of antibiotics. This article walks through what each pattern looks like, how doctors sort them out, and what recovery realistically looks like.
Cranial Neuropathy — The Most Common U.S. Presentation
The single most common neurologic manifestation of Lyme in North America is facial nerve palsy — a Bell's palsy appearance caused by inflammation of cranial nerve VII. One side of the face droops: the eyelid won't close fully, the corner of the mouth sags, food collects in the cheek, and patients drool when they drink. Taste on the front two-thirds of the tongue may be blunted, and sounds can seem uncomfortably loud on the affected side.
There is one clinical rule worth memorizing if you live in an endemic area: bilateral facial palsy — both sides drooping at once or in quick succession — is Lyme disease until proven otherwise. Idiopathic Bell's palsy is almost always unilateral. Bilateral simultaneous palsy is so uncommon in non-Lyme settings that it forces a serology test and, often, empirical treatment before results return.
Other cranial nerves can be affected, though much less often:
- Cranial nerve III (oculomotor) — droopy eyelid, eye turned outward, double vision.
- Cranial nerve V (trigeminal) — facial numbness or sharp lancinating pain.
- Cranial nerve VI (abducens) — inability to turn the eye outward, double vision worse on lateral gaze.
- Cranial nerve VIII (vestibulocochlear) — hearing loss, tinnitus, vertigo.
More than one cranial nerve can be involved at the same time. Any cranial neuropathy appearing in summer or early fall in an endemic region should prompt Lyme testing, even without a remembered tick bite or rash.
Lymphocytic Meningitis
Lyme meningitis looks and feels like other viral meningitides: headache, neck stiffness, photophobia (light hurts the eyes), nausea, and mild fever. The key distinguishing feature from bacterial meningitis is time course. Bacterial meningitis hits in hours and makes patients desperately ill; Lyme meningitis smolders for days to weeks. Many patients describe a relentless headache that started quietly and just refused to quit, not the thunderclap onset of a true medical emergency.
On lumbar puncture the CSF shows a lymphocytic pleocytosis — typically 100 to 200 white cells per microliter, predominantly lymphocytes rather than neutrophils — along with an elevated protein and a normal glucose. This triad is shared with viral meningitis, but in summer months in an endemic area with a co-existing cranial nerve palsy or radicular pain, Lyme rockets up the differential.
Kids are more likely than adults to present with meningitis as their leading symptom, often combined with a facial palsy on the same admission.
Painful Radiculoneuritis (Bannwarth Syndrome)
Bannwarth syndrome is the European signature of Lyme. In the U.S. it is rare; in central and northern Europe it is one of the two most common neurologic presentations (alongside meningitis). The classic description:
- Severe radicular pain — deep, burning, shooting pain that follows the distribution of a spinal nerve root. It often involves the trunk, back, or one limb, and it frequently sits on the same side of the body as a recent tick bite or erythema migrans rash.
- Worse at night. Patients describe pain so severe they cannot sleep, cannot find a comfortable position, and sometimes pace at 3 a.m. rather than lie down. This nocturnal pattern is one of the most useful clinical clues.
- Motor and sensory deficits — weakness, numbness, or loss of reflexes develop over the following days to weeks, again in a nerve-root distribution.
- CSF lymphocytic pleocytosis on lumbar puncture, usually with intrathecal antibody production.
Bannwarth is frequently misdiagnosed as disc disease — a pinched nerve from a herniated lumbar or cervical disc. Patients are sent for MRIs, given muscle relaxants, even offered epidural injections, while the real culprit is a treatable infection. In endemic regions, severe night-predominant radicular pain with no clear mechanical trigger should earn a Lyme serology.
Late-Stage Encephalomyelitis
This is the rarest and most serious form — parenchymal invasion, meaning Borrelia inflames the brain and spinal cord tissue itself, not just their coverings. It is almost exclusively a European phenomenon, tied to B. garinii, and it typically appears months to years after an untreated infection.
Features include white-matter lesions on MRI that can mimic multiple sclerosis, spastic paraparesis (stiff, weak legs with brisk reflexes), bladder dysfunction, and cognitive changes that go beyond "brain fog" into measurable deficits on formal testing. It is one of the few Lyme presentations that reliably requires intravenous antibiotics.
Lyme Encephalopathy
Lyme encephalopathy is a more controversial category than the ones above. Patients describe subtle but real cognitive complaints: memory lapses, word-finding difficulty, slowed processing, concentration problems, sometimes mood changes. These symptoms are real — but they do not always come with abnormal CSF, abnormal imaging, or clear evidence of active Borrelia in the nervous system.
Much of what is called Lyme encephalopathy in the literature ends up overlapping with post-treatment Lyme disease syndrome (PTLDS) — the cluster of persistent fatigue, pain, and cognitive symptoms that can follow an otherwise well-treated infection (see the PTLDS article). Whether the mechanism is lingering low-grade infection, immune dysregulation, or a post-infectious autonomic/neurologic syndrome remains genuinely debated. What is not debated by mainstream infectious-disease physicians is that long courses of repeat antibiotics do not help and carry real harm.
Peripheral Neuropathy
A minority of Lyme patients develop a mild distal sensory neuropathy: tingling, numbness, or burning in the feet and hands, sometimes with patchy areas of altered sensation. Nerve-conduction studies may show a mild axonal pattern. A less common variant is mononeuritis multiplex — individual peripheral nerves picked off one at a time, producing asymmetric weakness or sensory loss in a patchwork distribution.
Peripheral neuropathy from Lyme usually improves with standard antibiotic treatment, though recovery is slower than for cranial neuropathies because peripheral nerves regenerate at only about a millimeter per day.
Diagnostic Workup
Neuroborreliosis is diagnosed through a combination of clinical picture, blood serology, and — when indicated — CSF analysis.
- Serum two-tier serology. A screening ELISA followed by a confirmatory Western blot (or a second-generation ELISA under the modified two-tier protocol). By the time neurologic symptoms appear, most patients have seroconverted and testing is reliable. See the Lyme testing article for the details.
- Lumbar puncture with CSF antibody index. The gold standard for confirming CNS invasion is demonstrating that anti-Borrelia antibodies are being produced inside the central nervous system rather than simply leaking in from the blood. The lab calculates a CSF:serum antibody ratio, corrected for total IgG. A ratio above 1.0 (antibody concentration in CSF higher than predicted from blood) confirms intrathecal production.
- CSF pleocytosis and protein. Elevated white cells (lymphocyte-predominant) and elevated protein support the diagnosis, though neither is specific.
- CSF PCR. Can detect Borrelia DNA but has low sensitivity (10–30%). A negative PCR does not rule out neuroborreliosis.
- MRI brain and spine. Usually normal in cranial neuropathy and meningitis. Reserved for patients with focal deficits, suspected encephalomyelitis, or to exclude alternative diagnoses (stroke, tumor, MS).
When to Do a Lumbar Puncture — and When Not To
An LP is clearly indicated when:
- Meningitis signs are present (headache, neck stiffness, photophobia).
- Parenchymal CNS disease is suspected (focal deficits, encephalopathy with abnormal MRI, myelopathy).
- Radicular pain with motor or sensory deficits suggests Bannwarth.
- Cranial neuropathy comes with significant headache or multiple cranial nerves involved (the single-nerve, no-headache case is a judgment call and is debated).
An LP is generally not necessary for an isolated unilateral Bell's palsy with mild symptoms in an endemic area. The practical approach in that scenario is a serum serology, empirical oral doxycycline, and follow-up. Sticking a needle into a spine to confirm what you are already treating rarely changes the plan.
Treatment — IV vs Oral
For decades the reflex answer to "Lyme in the nervous system" was IV ceftriaxone 2 g once daily for 14 to 21 days, usually through a PICC line with home-infusion nursing. That approach still works, but it is no longer the only — or even the preferred — option for most patients.
A series of European randomized controlled trials, most notably Ljøstad et al. (2008) in Lancet Neurology, demonstrated that oral doxycycline 200 mg per day for 14 days was just as effective as IV ceftriaxone for European neuroborreliosis, including Bannwarth syndrome and meningitis. The IDSA/AAN/ACR 2020 guidelines (Lantos et al.) explicitly endorse oral doxycycline as equivalent for most neuroborreliosis presentations.
Current practical guidance:
- Oral doxycycline 100 mg twice daily for 14–21 days is the first choice for cranial neuropathy (including Bell's palsy), meningitis, and radiculoneuritis.
- IV ceftriaxone 2 g daily for 14–28 days is reserved for parenchymal CNS disease (encephalomyelitis, myelitis), severely ill or hospitalized patients, documented treatment failure on oral therapy, pregnancy, or children too young for doxycycline (though under current guidelines doxycycline is now considered safe for short courses even in young children).
- IV cefotaxime and IV penicillin G are acceptable alternatives to ceftriaxone if it cannot be used.
See the antibiotic treatment protocols article for full dosing, pediatric adjustments, and allergy workarounds.
Bell's Palsy Specifically
Lyme-associated Bell's palsy deserves a focused note because it is so common and because patients naturally ask: "Do I also need prednisone?"
In idiopathic (non-Lyme) Bell's palsy, a short prednisone taper started within 72 hours clearly improves recovery. Whether the same is true for Lyme-associated facial palsy is genuinely unresolved. Some studies have suggested steroids may delay recovery when Lyme is the cause; others have found no harm. Most clinicians in endemic areas take a pragmatic middle path: start doxycycline, and also give a short prednisone taper (commonly 60 mg for 5 days, then a rapid taper), on the reasoning that if the diagnosis turns out to be idiopathic they have covered that too, and if it is Lyme the antibiotic handles the infection.
The prognosis is good. Roughly 80–90% of Lyme-associated Bell's palsy recovers fully over weeks to months. A minority are left with mild residual weakness, asymmetry when smiling, or subtle synkinesis (the eye twitches when the mouth moves). Eye care during the acute phase is essential: artificial tears by day, lubricating ointment and a patch or taped lid at night, to prevent corneal damage while the lid will not close.
Recovery Timeline
Realistic expectations help enormously. Typical patterns:
- Meningitis symptoms — headache and stiff neck usually resolve within days to two weeks of starting antibiotics.
- Cranial nerve palsies (including Bell's) — begin improving in 2–4 weeks, usually complete by 2–3 months. A few patients continue to improve out to 6 months.
- Radiculoneuropathy (Bannwarth) — pain often eases within days of starting antibiotics; motor and sensory deficits resolve over weeks to 6 months, sometimes longer.
- Peripheral neuropathy — slow, often months to a year, limited by the rate of nerve regeneration.
- Encephalopathy — variable. Some patients recover fully, others report lingering cognitive and fatigue symptoms that blur into PTLDS territory.
Residual Symptoms After Treatment
Even after a complete, guideline-concordant course of antibiotics, a meaningful minority of patients continue to report fatigue, cognitive complaints, and intermittent pains for weeks to months. This is common enough that it has its own name: post-treatment Lyme disease syndrome. Read the PTLDS article for a full discussion.
Two points matter here. First, residual symptoms do not automatically mean the antibiotics failed or that Borrelia is still active. Multiple high-quality randomized trials have shown that prolonged or repeat antibiotic courses do not produce sustained improvement and do cause real harm (Clostridium difficile colitis, PICC-line infections, gallstones from ceftriaxone). Second, the absence of a quick antibiotic fix does not mean you are dismissed. Graded exercise, sleep restoration, targeted treatment of pain and mood symptoms, and patience are the real tools. Most patients continue to improve over 6–12 months.
Red Flags Requiring the ER
Most neuroborreliosis can be worked up in an outpatient clinic. The following scenarios warrant same-day emergency evaluation:
- Severe headache with neck stiffness plus fever — could be bacterial meningitis, which kills quickly if untreated.
- Altered mental status — confusion, drowsiness, personality change, disorientation.
- Acute focal neurologic deficit — sudden weakness on one side, sudden speech trouble, sudden vision loss. Stroke must be excluded first.
- New seizure.
- Rapidly progressing weakness of the legs or arms, or new loss of bladder or bowel control.
Lyme is a treatable infection. Catching neurologic involvement early, treating with the right antibiotic at the right dose, and giving your nervous system time to heal will resolve symptoms completely for the great majority of patients. A droopy face is not a death sentence, a splitting headache is not a stroke, and a night of radicular pain is not a disc you need surgery for — it is, more often than people realize, a bacterial infection that a two-week prescription will put right.
Key Research Papers
- Halperin JJ, et al. Practice Parameter: Treatment of nervous system Lyme disease (AAN guideline). Neurology. 2007.
- Dattwyler RJ, Luft BJ, Kunkel MJ, et al. Ceftriaxone Compared with Doxycycline for the Treatment of Acute Disseminated Lyme Disease. N Engl J Med. 1997;337(5):289–294. (Oral doxycycline vs IV ceftriaxone in neuroborreliosis.)
- Ljøstad U, et al. Oral doxycycline versus intravenous ceftriaxone for European Lyme neuroborreliosis: a multicentre, non-inferiority, double-blind, randomised trial. Lancet Neurol. 2008.
- Lantos PM, et al. Clinical Practice Guidelines by the IDSA, AAN, and ACR: 2020 Guidelines for the Prevention, Diagnosis, and Treatment of Lyme Disease. Clin Infect Dis. 2021.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on neuroborreliosis diagnosis, treatment, and outcomes:
- Lyme neuroborreliosis treatment
- Bannwarth syndrome
- Facial palsy and Lyme disease
- Ljøstad doxycycline neuroborreliosis trial
- Lyme meningitis and CSF antibody index
- Borrelia garinii neurotropism
- Lyme encephalopathy and cognitive symptoms
- Bilateral facial palsy and Lyme disease
Connections
- Antibiotic Treatment Protocols
- Lyme Testing Explained
- PTLDS and Chronic Lyme
- Lyme Disease Overview
- Meningitis
- Erythema Migrans Rash
- Prevention and Tick Removal
- Tick Bite First Aid
- Tick-Borne Co-Infections
- Silver Nanoparticles and Meningitis
- Headache
- Peripheral Neuropathy
- Stroke
- Fatigue
- Lyme Carditis
- Brain Fog