Post-Viral Triggers: Epstein-Barr Virus and Others in ME/CFS
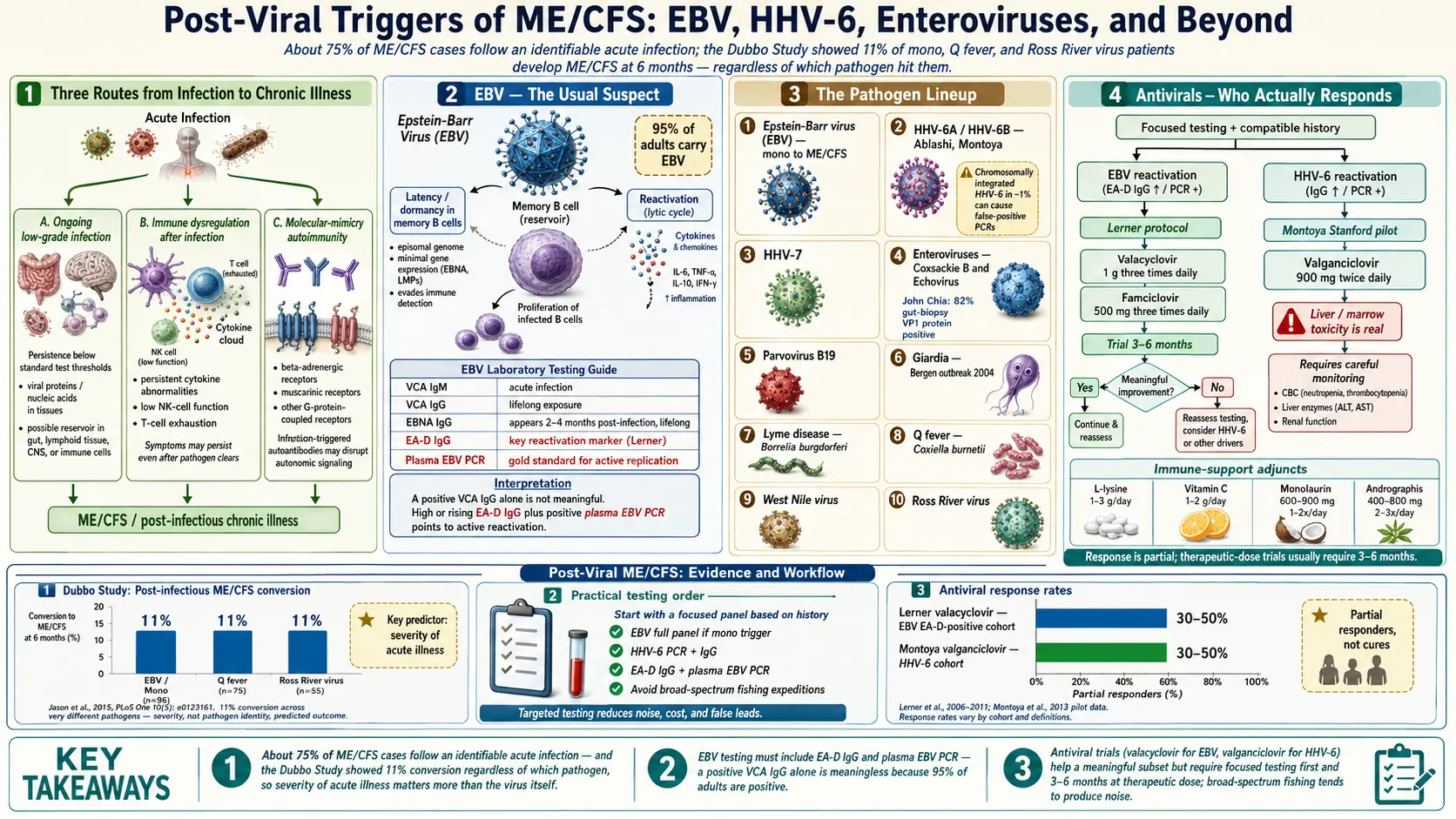
Table of Contents
- What "Post-Viral" Actually Means
- The Dubbo Study — Mono as the Starting Gun
- Epstein-Barr Virus: The Usual Suspect
- Reading Your EBV Panel Without Panic
- HHV-6 and HHV-7 Reactivation
- Enteroviruses: Coxsackie B and Echovirus
- Parvovirus B19
- Giardia and the Bergen Outbreak
- Lyme Disease as an ME/CFS Trigger
- Q Fever (Coxiella burnetii)
- West Nile and Ross River Virus
- Antivirals: Who Actually Responds
- Natural Antivirals and Immune Support
- A Practical Testing and Treatment Order
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
What "Post-Viral" Actually Means
A large fraction of ME/CFS patients can point to a specific illness that started everything. One week they were fine; a few weeks later they had a sore throat, a fever, and swollen glands; six months later they still could not climb the stairs without their heart rate spiking and needing a two-day recovery. That pattern — an acute infection followed by a chronic multi-system collapse — is what clinicians mean by a post-infectious or post-viral onset of ME/CFS.
"Post-viral" does not necessarily mean the virus is still replicating inside you. It can mean any of three things, and all three probably happen in different patients:
- Ongoing low-grade infection — the pathogen is still there, hiding in tissue reservoirs, at levels too low for standard testing.
- Immune dysregulation triggered by the infection — the pathogen is gone, but the immune system never returned to its pre-illness baseline. Cytokines, natural killer cell function, and T-cell exhaustion markers stay abnormal for years.
- Autoimmunity against self-tissue — the infection primed antibodies against molecular mimics of your own receptors (beta-adrenergic, muscarinic, G-protein-coupled), and those antibodies persist.
This article walks through the specific pathogens repeatedly linked to ME/CFS onset, how to test for them sensibly, and what to do if a trigger is confirmed. The goal is not a fishing expedition — random broad-spectrum viral panels tend to produce confusing results — but a focused workup guided by your history.
The Dubbo Study — Mono as the Starting Gun
The single most-cited post-viral ME/CFS study came out of the Australian town of Dubbo, published in the BMJ in 2006 by Ian Hickie and colleagues. The researchers followed 253 people diagnosed with one of three acute infections — Epstein-Barr virus (mononucleosis), Q fever, or Ross River virus — and checked in at six months.
The headline result: 11% of these patients met criteria for ME/CFS six months later, regardless of which of the three pathogens had hit them. The rate was the same across all three infections, which is the crucial finding — it meant the trigger was not EBV-specific. Something about the host's response to a significant infection, not the virus itself, determined who tipped into chronic illness.
The predictors of who became chronic were not the viral load or the strain. They were the severity of the initial acute illness (how high the fever, how swollen the glands, how long the bed rest) and, to a lesser extent, female sex and certain cytokine polymorphisms. A mild case of mono rarely kicked off ME/CFS. A flattening, two-weeks-in-bed case did so in about one in ten.
Dubbo reframed ME/CFS as something that happens to a minority of people after severe infections, not a mysterious condition that appears from nowhere. It also legitimized the experience of patients who knew exactly when their illness started.
Epstein-Barr Virus: The Usual Suspect
EBV is a herpesvirus that roughly 95% of adults carry for life. Most people catch it as children, where it causes nothing memorable. Catch it as a teenager or young adult and you get infectious mononucleosis — the classic sore throat, enormous lymph nodes, exhausted-for-a-month illness. After the acute phase, the virus goes dormant inside memory B cells, where it stays forever, occasionally reactivating and being re-suppressed without you noticing.
In ME/CFS, two things can go wrong with that dormancy:
- Primary infection that never fully clears. The acute mono just drags on and on, evolving into ME/CFS without a clear transition point. This is the classic Dubbo scenario.
- Reactivation in someone with old EBV. You had mono at 17, forgot about it, and then in your 30s a stressful period, surgery, or another infection lets EBV come out of dormancy. Immune surveillance fails to put it back, and a chronic reactivation pattern establishes.
EBV in ME/CFS is not just a passenger. Lab work has shown abnormal antibody profiles, deficient EBV-specific T-cell responses, and in some cohorts detectable viral DNA in blood or saliva when controls have none. The 2018 Rasa review in the Journal of Translational Medicine summarizes the evidence: chronic EBV reactivation is present in a meaningful subset of ME/CFS patients, though not in all of them.
Reading Your EBV Panel Without Panic
The biggest source of confusion in EBV testing is that a positive IgG result alone proves almost nothing. Ninety-five percent of adults are IgG positive. Your doctor saying "your EBV came back positive" is meaningless without the specific antibody subsets.
Here is what each marker means, in plain terms:
- VCA IgM (viral capsid antigen, IgM class) — rises in the first few weeks of acute infection, then fades. A positive VCA IgM in an adult who has been sick for 2–6 weeks suggests recent primary mono.
- VCA IgG — rises during acute infection and stays positive for life. By itself it tells you only that you have been exposed to EBV at some point, which again is true of almost everyone.
- EBNA IgG (Epstein-Barr nuclear antigen, IgG) — appears 2–4 months after the acute infection and stays positive for life. A positive EBNA IgG means the infection is old. A negative EBNA with positive VCA IgG is suspicious for either a recent infection or, in a chronic-illness context, a faulty immune response that has failed to mount a normal memory profile.
- Early Antigen-D (EA-D) IgG — the key reactivation marker. In healthy people with old EBV, EA-D is usually negative or low. A high or rising EA-D titer in someone with chronic fatigue suggests ongoing reactivation. Dr. Martin Lerner built his treatment protocols around this marker.
- EBV PCR — measures actual viral DNA in blood (whole blood or plasma) or saliva. A positive plasma PCR is the strongest evidence of active viral replication, because dormant EBV sits inside B cells and does not spill into plasma. Whole-blood PCR is more sensitive but less specific (it picks up latent genomes inside memory B cells too).
Practical rule: if you suspect EBV reactivation, ask for a full panel including VCA IgM, VCA IgG, EBNA IgG, EA-D IgG, and plasma EBV PCR. A standard "mono spot" or a single IgG is not enough. The whole point is to distinguish old exposure (everyone) from active reactivation (a minority).
HHV-6 and HHV-7 Reactivation
Human herpesvirus-6 and -7 are cousins of EBV. Almost everyone catches HHV-6 as a toddler — it causes roseola, the classic "fever-then-rash" baby illness — and like EBV, it stays dormant for life. HHV-6 comes in two variants: HHV-6A (less common, more often linked to neurological disease) and HHV-6B (the childhood roseola strain).
In ME/CFS cohorts, HHV-6 reactivation has been reported for decades, particularly by Dharam Ablashi (one of HHV-6's original co-discoverers) and by the Stanford group around Jose Montoya. Testing options:
- HHV-6 IgG and IgM — high IgG titers or positive IgM can suggest reactivation, though interpretation is less clean than EBV.
- HHV-6 PCR in plasma — the most specific test; a positive result points to active replication.
- Chromosomally-integrated HHV-6 (ciHHV-6) — in about 1% of people, HHV-6 has integrated into germline DNA. These patients will have positive PCR in every sample they ever give, which does not mean active infection. A specialty lab can distinguish ciHHV-6 from true reactivation.
HHV-7 is less studied in ME/CFS but has been flagged as a potential co-reactivator. Most clinicians test for HHV-6 first and add HHV-7 only if the workup is negative and suspicion remains high.
Enteroviruses: Coxsackie B and Echovirus
Enteroviruses are a large family of RNA viruses that cause summer colds, hand-foot-and-mouth, and occasionally more serious illness. Two subgroups keep surfacing in ME/CFS literature: Coxsackie B (especially CVB3 and CVB4) and echoviruses.
The leading voice on enteroviral ME/CFS is Dr. John Chia in California. His 2008 paper in the Journal of Clinical Pathology studied stomach biopsies from ME/CFS patients and found enteroviral VP1 protein in 82% of patients versus 20% of controls. Chia's argument is that enteroviruses can persist in gut tissue for years, replicating at low levels below the threshold of standard blood tests, and that the gut is the reservoir driving chronic symptoms.
Testing enterovirus is harder than testing EBV:
- Standard antibody panels (neutralizing antibodies against CVB1-6 and the common echovirus types) show only prior exposure.
- A high, persistent titer (1:320 or above) against a single Coxsackie strain in a chronically ill patient is suggestive but not diagnostic.
- The most specific test is stomach biopsy with VP1 immunostaining, but only a handful of labs worldwide run it.
- Stool enterovirus PCR can pick up active shedding but is often negative in chronic cases.
If your ME/CFS started with a gastrointestinal illness — sudden vomiting, diarrhea, body aches in summer — enterovirus belongs on the short list.
Parvovirus B19
Parvovirus B19 causes "fifth disease" in children (the slapped-cheek rash) and can cause a flu-like illness with joint pain in adults. A subset of adults infected with parvovirus B19 develop chronic fatigue and arthralgia that persists for years after the acute phase.
Testing: parvovirus B19 IgM (recent), IgG (old exposure), and PCR for DNA. Chronic-phase patients can be IgG positive and PCR positive simultaneously, suggesting failed clearance. Treatment is mostly supportive; intravenous immunoglobulin (IVIG) has been tried in severely immune-compromised patients but is not standard for ME/CFS.
Giardia and the Bergen Outbreak
In 2004, the drinking-water supply of Bergen, Norway was contaminated with Giardia lamblia. Between 1,300 and 2,500 people developed acute giardiasis — watery diarrhea, cramping, bloating. The water was treated and the parasites cleared. Then the Norwegian health system followed the cohort.
Halvor Naess and colleagues reported the 3-year outcomes in BMC Infectious Diseases in 2012: a substantial fraction of the Giardia cohort developed chronic fatigue syndrome, irritable bowel syndrome, or both, at rates well above unexposed controls. Follow-up at 5 and 10 years confirmed that many of these patients did not recover.
Bergen is important because it is the cleanest real-world experiment ever run on post-infectious ME/CFS outside of viral pathogens. It proved that a protozoan gut infection can trigger the same chronic-fatigue syndrome that EBV does, reinforcing the idea that the trigger matters less than the host response.
If your ME/CFS followed traveler's diarrhea or a known Giardia exposure, a stool Giardia antigen test or PCR is reasonable. Eradication with metronidazole or tinidazole is easy if active infection is found, though clearing the parasite often does not clear the fatigue.
Lyme Disease as an ME/CFS Trigger
The relationship between Lyme disease and ME/CFS is tangled and contested. The clean version: a minority of patients treated for acute Lyme with a standard course of doxycycline remain ill six months later with fatigue, cognitive dysfunction, sleep disturbance, and musculoskeletal pain. The CDC calls this Post-Treatment Lyme Disease Syndrome (PTLDS). The clinical picture overlaps heavily with ME/CFS — in some series, more than half of PTLDS patients meet ME/CFS criteria.
Whether PTLDS represents persistent infection, immune dysregulation, or autoimmunity is an active research question. For the patient, the practical point is:
- If your ME/CFS began after a tick bite, a bulls-eye rash, or a summer flu-like illness in a Lyme-endemic area, pursue two-tier Lyme serology (ELISA followed by Western blot) even if years have passed.
- Seronegative Lyme is controversial but real in a small percentage of cases; clinical judgment matters.
- Co-infections (babesia, bartonella, anaplasma) should be considered, because they can produce their own chronic syndromes.
For a fuller discussion, see Lyme Disease and PTLDS and Chronic Lyme.
Q Fever (Coxiella burnetii)
Coxiella burnetii is a bacterium carried by sheep, goats, and cattle. Humans catch it by inhaling contaminated dust, usually near farms or slaughterhouses. Acute Q fever is a flu-like illness with high fever, headache, and sometimes pneumonia or hepatitis.
A defined minority of Q fever patients develop Q Fever Fatigue Syndrome (QFS), a chronic post-infectious state indistinguishable from ME/CFS in most respects. Q fever was one of the three triggers in the Dubbo study, and the Netherlands had a large Q fever outbreak from 2007–2010 that produced a QFS cohort followed for over a decade.
Testing: Coxiella phase I and phase II IgG. If you grew up near livestock or had an unexplained pneumonia before ME/CFS onset, it is worth checking.
West Nile and Ross River Virus
Both are mosquito-borne RNA viruses. Ross River virus is endemic to Australia and the South Pacific; it causes an acute illness of fever, rash, and prolonged polyarthralgia, and was the third pathogen in the Dubbo study. West Nile virus is endemic across North America; most infections are asymptomatic, but those who develop symptomatic West Nile (flu-like illness, occasionally encephalitis) have a recognized post-viral fatigue syndrome that can persist for years.
Testing is by IgG/IgM serology. Treatment is supportive. These are mostly diagnostic "ah-ha" findings — they explain an onset rather than offering a treatment target — but explaining the start of the illness matters for patients who have been told their symptoms are psychosomatic.
Antivirals: Who Actually Responds
Antiviral medication is not a routine ME/CFS treatment, but in carefully selected subsets it can produce striking improvement. The evidence base is small, uncontrolled in most cases, and drawn mostly from two clinicians — the late Martin Lerner in Michigan and Jose Montoya's group at Stanford.
Valacyclovir (brand name Valtrex) targets EBV, HSV-1, HSV-2, and VZV. Lerner's 2010 paper in the International Journal of Molecular Medicine reported improvement in EBV-subset ME/CFS patients on long-term valacyclovir, using his "EBV subset" criteria: elevated EA-D IgG, high VCA IgG, and abnormal EBV-specific EKG findings. Dose: typically 1–3 g daily for 6–12 months.
Valganciclovir (Valcyte) is a stronger drug, active against HHV-6 and CMV in addition to EBV. It has meaningful side effects — bone-marrow suppression, neutropenia, potential carcinogenicity — and requires close lab monitoring. Montoya's 2013 randomized pilot trial showed improvement in cognitive and physical function in ME/CFS patients with high HHV-6 and EBV titers, though the trial was small. Use is reserved for patients with strong serological evidence of HHV-6 or CMV reactivation and willingness to accept monitoring.
Famciclovir (Famvir) is another option in the valacyclovir family, sometimes chosen for its convenience or tolerability.
Who responds: generally patients with a clear post-viral onset, persistent serological evidence of reactivation (EA-D, plasma PCR, high HHV-6 titers), and a relatively short duration of illness. Patients with 20-year illness and normal serology do not respond, and the drugs are not worth the risks in that scenario.
These medications are prescription-only and require a clinician willing to treat based on the Lerner/Montoya framework. Most general practitioners and even most infectious disease specialists will not prescribe them for ME/CFS. Finding a knowledgeable ME/CFS specialist — through the U.S. ME/CFS Clinician Coalition or Bateman Horne Center — is usually the path.
Natural Antivirals and Immune Support
Several supplements have antiviral activity in vitro or in small trials. None are substitutes for prescription antivirals in confirmed reactivation, but they are cheap, generally low-risk, and some patients report benefit:
- L-Lysine — an amino acid that competes with arginine, which herpesviruses need for replication. Classically used for HSV cold sores at 1–3 g per day. Some ME/CFS patients use it for EBV reactivation. See L-Lysine.
- Monolaurin — a coconut-derived fatty-acid derivative with in vitro activity against enveloped viruses including EBV. Typical dose 600–1,800 mg daily.
- Olive leaf extract — contains oleuropein, which has shown antiviral effects in laboratory studies. Quality varies wildly between brands.
- N-acetylcysteine (NAC) — replenishes glutathione, which is typically depleted in chronic viral illness. 600–1,800 mg daily.
On the immune-modulation side, two prescription interventions have moved from fringe to mainstream discussion:
- Low-Dose Naltrexone (LDN) — 1.5–4.5 mg at bedtime. Modulates microglia and reduces neuroinflammation. A meaningful fraction of ME/CFS patients report reduced pain and brain-fog on LDN. See the sibling article on LDN, Abilify, and experimental therapies for the full protocol.
- Low-Dose IL-2 — experimental, administered by subcutaneous injection, expands regulatory T cells and has been trialed in autoimmunity and a handful of ME/CFS cases. Not yet standard, but on the horizon.
A Practical Testing and Treatment Order
If you believe a post-viral trigger started your ME/CFS, do not order every test listed above at once. Start focused:
- History first. Pin down the month and, if possible, the week the illness started. Write down symptoms of that initial illness: fever, sore throat, swollen glands, rash, diarrhea, tick bite, travel.
- Targeted serology. Based on the history, pick one or two likely pathogens. Classic mono history: EBV full panel (VCA IgM, VCA IgG, EBNA IgG, EA-D IgG, plasma PCR). Summer gastrointestinal onset: enterovirus panel plus stool Giardia. Tick exposure: two-tier Lyme plus co-infections. Farm or livestock exposure: Q fever phase I and II. Mosquito-heavy summer onset: West Nile IgG/IgM.
- Add HHV-6 if EBV is suggestive but you want more detail, particularly with cognitive prominence.
- Interpret with a specialist. An ME/CFS-literate physician can distinguish "old exposure" from "active reactivation" from "chromosomally integrated". General practitioners often cannot.
- If a clear trigger is identified, discuss targeted treatment: metronidazole for Giardia, doxycycline for new-onset Lyme, valacyclovir or valganciclovir for confirmed reactivation. Adjunct with pacing, sleep, and orthostatic-intolerance management, because the non-viral dysfunction in ME/CFS persists even when a pathogen is addressed.
A final word on expectations: identifying a post-viral trigger is meaningful but is not a cure. Many patients see a specific antiviral produce 20–40% improvement, which is real and worth pursuing, but the full ME/CFS syndrome — post-exertional malaise, orthostatic intolerance, cognitive dysfunction — usually requires the full toolkit: pacing, autonomic management, sleep repair, and in some cases experimental therapies. The trigger is the door that opened the illness. Closing it helps. It rarely ends it.
Key Research Papers
- Hickie I, et al. Post-infective and chronic fatigue syndromes precipitated by viral and non-viral pathogens: prospective cohort study (Dubbo). BMJ. 2006.
- Chia JK, Chia AY. Chronic fatigue syndrome is associated with chronic enterovirus infection of the stomach. J Clin Pathol. 2008.
- Naess H, et al. Chronic fatigue syndrome after Giardia enteritis: clinical characteristics of a Bergen cohort. BMC Infect Dis. 2012.
- Lerner AM, et al. Valacyclovir treatment in Epstein-Barr virus subset chronic fatigue syndrome. Int J Mol Med. 2010.
- Rasa S, et al. Chronic viral infections in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). J Transl Med. 2018.
Research Papers
The following PubMed topic searches return current peer-reviewed work on post-viral triggers of ME/CFS and related syndromes:
- EBV and ME/CFS
- HHV-6 and chronic fatigue syndrome
- Enterovirus, Coxsackie B, and ME/CFS
- Dubbo post-infective fatigue cohort
- Giardia and post-infectious fatigue (Bergen cohort)
- Q Fever Fatigue Syndrome
- Post-Treatment Lyme Disease Syndrome
- Valganciclovir trials in ME/CFS
- Ross River virus and chronic fatigue
- Parvovirus B19 and chronic fatigue
Connections
- ME/CFS Overview
- Lyme Disease
- PTLDS and Chronic Lyme
- Post-Exertional Malaise
- ME/CFS Diagnostic Criteria
- 2-Day CPET and Objective Testing
- Orthostatic Intolerance in ME/CFS
- Pacing and the Energy Envelope
- Severe and Very Severe ME
- LDN, Abilify and Experimental Therapies
- Mitochondrial Dysfunction and Energy Metabolism
- Hashimoto's Thyroiditis
- Lysine
- Epstein-Barr Virus
- POTS
- MCAS
- Fibromyalgia
- Fatigue