Diabetes Complications: Retinopathy, Nephropathy, Neuropathy
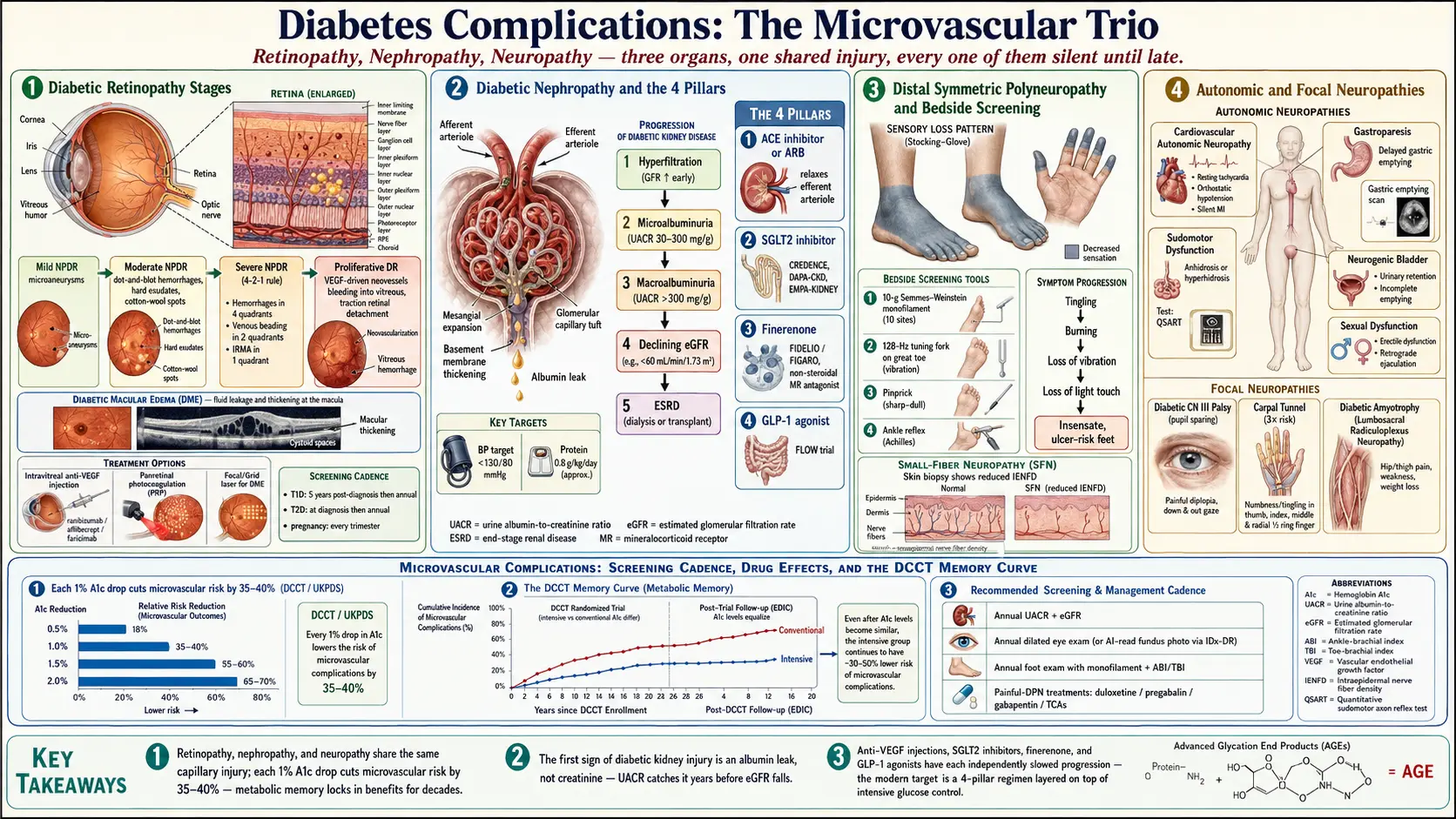
Table of Contents
- The Big Three — Why They Cluster
- Retinopathy — Saving Your Vision
- Retinopathy Screening Cadence
- Retinopathy Treatments
- Nephropathy — Protecting Your Kidneys
- Nephropathy Screening — UACR and eGFR
- Nephropathy Treatments — The Four Pillars
- Neuropathy — Nerves Under Glucose Attack
- Autonomic and Focal Neuropathies
- Neuropathy Screening
- Neuropathy Treatments
- Other Complications — Feet, Bones, Brain, Ears
- Intensive Control Caveats
- A Word on Metformin and B12
- Key Research Papers
- Connections
- Featured Videos
The Big Three — Why They Cluster
Diabetes complications are often called "micro" and "macro" vascular. The microvascular trio — retinopathy, nephropathy, neuropathy — all start in the same place: the smallest blood vessels in your body. High glucose damages the inner lining of capillaries, thickens their basement membranes, and leaks fluid into tissues that depend on pristine microcirculation. The retina, the kidney glomerulus, and the nerve sheath are unusually vulnerable because each of them is fed by delicate capillary beds with almost no collateral blood supply. When those capillaries go, the organ goes.
The good news: all three complications share the same levers. Tight glucose control, tight blood pressure control, and a handful of specific drug classes slow or halt every one of them. The landmark DCCT trial in type 1 diabetes and the UKPDS trial in type 2 diabetes both showed that each 1% drop in A1c cuts microvascular complication risk by roughly 35–40%. Those benefits compound over decades — a concept called metabolic memory. What you do in the first ten years of your diagnosis keeps paying dividends thirty years later.
This article walks through each complication: what it is, how to screen for it, and how modern medicine treats it. Screening is the single most powerful thing you can do, because every one of these diseases is silent in the stage where it is most reversible.
Retinopathy — Saving Your Vision
Diabetic retinopathy is the leading cause of blindness in working-age adults in the developed world. It progresses through predictable stages:
- Mild non-proliferative retinopathy (NPDR). Tiny capillary outpouchings called microaneurysms appear on the retina. You feel nothing; your vision is unchanged. It is visible only on dilated eye exam or fundus photography.
- Moderate NPDR. Dot-and-blot hemorrhages, hard exudates (lipid leaks), and cotton-wool spots (tiny infarcts) accumulate.
- Severe NPDR. The "4-2-1 rule" — hemorrhages in 4 retinal quadrants, venous beading in 2, or intraretinal microvascular abnormalities in 1 — signals a high short-term risk of progression to the proliferative stage.
- Proliferative diabetic retinopathy (PDR). Starved retinal tissue releases vascular endothelial growth factor (VEGF), which drives the growth of fragile new blood vessels. These neovessels bleed into the vitreous, pull on the retina, and can cause traction retinal detachment — the final pathway to blindness.
Overlapping all of these stages is diabetic macular edema (DME) — fluid leaking into the central retina (the macula), blurring reading vision. Macular edema can occur at any NPDR or PDR stage, and it is now the most common cause of vision loss in diabetes, because we catch PDR earlier than we used to.
Retinopathy Screening Cadence
The American Diabetes Association and the American Academy of Ophthalmology agree on the cadence:
- Type 1 diabetes: first dilated eye exam 5 years after diagnosis, then at least annually.
- Type 2 diabetes: first dilated eye exam at diagnosis (because many patients have had undiagnosed hyperglycemia for years), then annually.
- Pregnancy with pre-existing diabetes: exam in the first trimester, then every trimester plus a postpartum check. Pregnancy can rapidly worsen retinopathy.
- Well-controlled diabetes with repeatedly normal exams: screening can safely stretch to every 2 years in some patients — ask your ophthalmologist.
How it is screened. A dilated fundus exam by an ophthalmologist or optometrist remains the gold standard. Fundus photography is increasingly used in primary care — a retinal camera takes high-resolution photos, and either a specialist or an AI reads them. In 2018 the FDA approved IDx-DR, the first autonomous AI medical device ever cleared in any specialty, which reads retinal photos and issues a "refer" or "rescreen in 12 months" result without a human specialist. If your primary care clinic has a retinal camera, the exam takes 10 minutes and does not require pupil dilation. Optical coherence tomography (OCT) is a separate test, performed by eye specialists, that shows cross-sectional layers of the retina; it is the standard for detecting and monitoring macular edema.
Retinopathy Treatments
A generation ago, laser was all we had. Now the toolkit is much deeper.
Anti-VEGF intravitreal injections. The backbone of modern treatment. A tiny needle delivers an anti-VEGF drug directly into the vitreous gel, typically monthly at first and then tapered. The three workhorses are ranibizumab (Lucentis), aflibercept (Eylea), and the newer faricimab (Vabysmo), which blocks both VEGF-A and angiopoietin-2 and can often be dosed every 3–4 months. Bevacizumab (Avastin) is used off-label at much lower cost and works nearly as well for most patients. Anti-VEGF injections both treat macular edema and shrink proliferative neovessels.
Laser photocoagulation. Still the standard for high-risk PDR when injections are not enough. Panretinal photocoagulation (PRP) uses hundreds of laser burns on the peripheral retina to reduce the drive for neovascular growth, trading peripheral vision for preservation of the central vision. Focal/grid laser treats isolated leaking microaneurysms in the macula.
Steroid implants. For macular edema that resists anti-VEGF, sustained-release dexamethasone implants (Ozurdex) or the fluocinolone implant (Iluvien) deliver months of anti-inflammatory effect inside the eye.
Vitrectomy. Surgical removal of the vitreous gel, reserved for vitreous hemorrhage that does not clear or for traction retinal detachment. Modern small-gauge vitrectomy is outpatient and dramatically safer than it was twenty years ago.
Fenofibrate. A surprise from the FIELD and ACCORD-Eye trials: this generic lipid drug, originally prescribed for triglycerides, slowed retinopathy progression by roughly 40% independent of its cholesterol effect. Ask your endocrinologist whether it fits your profile, especially if you also have mild hypertriglyceridemia.
If you are told you need injections, do not panic. The needle is small, the eye is numbed, and most patients describe the experience as strange but not painful. The injections preserve vision that no other treatment can rescue. Refusing them because of needle fear is the single most common preventable path to diabetic blindness.
Nephropathy — Protecting Your Kidneys
Diabetic kidney disease (DKD) is now the number-one cause of end-stage renal disease in the United States, accounting for about 45% of new dialysis starts. It unfolds over years and is almost entirely silent until late.
The classical progression:
- Hyperfiltration. The kidneys actually work harder early on; eGFR can be above normal.
- Microalbuminuria. Small amounts of albumin start leaking into urine — 30–300 mg/g urinary albumin-to-creatinine ratio (UACR). This is the earliest clinically detectable sign, and it usually appears years before creatinine rises.
- Macroalbuminuria. UACR climbs above 300 mg/g. Protein leak becomes detectable on a standard dipstick.
- Declining eGFR. Glomerular filtration rate falls, creatinine climbs, and the kidneys can no longer compensate.
- End-stage renal disease. eGFR below 15 mL/min/1.73m². Dialysis or transplant required.
The take-home: the first sign of diabetic kidney injury is almost always an albumin leak, not a creatinine rise. If you wait for your creatinine to climb, you have already lost half of your nephron reserve. That is why screening is so essential, and why it rests on the UACR, not the basic metabolic panel alone.
Nephropathy Screening — UACR and eGFR
What to order. Two tests, annually:
- Spot urine albumin-to-creatinine ratio (UACR) on a random urine sample. A 24-hour collection is no longer needed.
- Serum creatinine with calculated eGFR (the lab does the math).
When to start. Type 1 diabetes: 5 years after diagnosis, then annually. Type 2 diabetes: at diagnosis, then annually. Pregnancy: early, and then per obstetrician guidance.
Interpreting results. A single elevated UACR does not confirm DKD — exercise, fever, menstruation, or a urinary tract infection can all transiently raise it. Confirm with two out of three positive samples over 3–6 months. Once confirmed, microalbuminuria is the threshold at which drug therapy should begin (see the next section), whether or not your blood pressure is high and whether or not your creatinine has moved.
Nephropathy Treatments — The Four Pillars
Modern DKD treatment rests on four complementary drug classes plus blood pressure and protein targets. Each one independently slows kidney function decline. Used together, they can often stabilize kidney function for years.
1. ACE inhibitors or ARBs. First-line for any patient with albuminuria, regardless of blood pressure. These drugs relax the efferent arteriole of the glomerulus and lower intraglomerular pressure — a pure kidney-protective effect separate from their blood-pressure benefit. Lisinopril, ramipril, enalapril (ACEi), losartan, valsartan, telmisartan (ARB). Do not use them together — combined ACEi + ARB increases harm without benefit.
2. SGLT2 inhibitors. The single biggest advance in DKD in a generation. Empagliflozin, dapagliflozin, and canagliflozin reduce glomerular hyperfiltration, lower intraglomerular pressure, and protect kidneys through mechanisms independent of glucose lowering. Three trials — CREDENCE, DAPA-CKD, and EMPA-KIDNEY — established that these drugs slow kidney decline in DKD and in non-diabetic kidney disease alike. SGLT2 inhibitors are now FDA-approved for chronic kidney disease regardless of diabetes status. See the sibling article on cardio-renal protection for the full trial data.
3. Non-steroidal mineralocorticoid receptor antagonist — finerenone (Kerendia). A newer class. The FIDELIO-DKD and FIGARO-DKD trials showed finerenone reduces both kidney and cardiovascular events in patients with DKD and type 2 diabetes already on maximal ACEi/ARB therapy. It is layered on top of, not instead of, the renin-angiotensin blocker. Watch serum potassium; finerenone can raise it.
4. GLP-1 receptor agonists. Semaglutide and similar drugs were long suspected to help kidneys, and the FLOW trial in 2024 confirmed renal benefit in type 2 DKD. Weight loss, blood pressure reduction, and direct renal anti-inflammatory effects appear to contribute.
Blood pressure target: under 130/80 mm Hg. Lower is not necessarily better — aggressive targets below 120/70 can precipitate falls and acute kidney injury in older patients.
Dietary protein: around 0.8 g/kg body weight per day. This is a moderation target, not a low-protein diet. Going substantially below 0.8 g/kg risks muscle loss and malnutrition without additional kidney benefit. A 70 kg adult eats roughly 56 g of protein a day, which is two modest chicken-breast portions or an equivalent plant-based mix.
Nephrology referral. When eGFR drops below 30 mL/min/1.73m², or when it is falling rapidly (more than 5 mL/min per year), refer to a nephrologist. Early referral cuts the risk of a chaotic dialysis start.
Neuropathy — Nerves Under Glucose Attack
Up to half of all people with long-standing diabetes develop some form of neuropathy. The most common by far is distal symmetric polyneuropathy — the classic "stocking-glove" pattern that begins in the toes and creeps upward, usually symmetrically, over years.
Symptoms progress in a predictable order:
- Tingling, pins-and-needles, and burning in the toes and soles, often worse at night.
- Loss of vibration sense, then loss of light-touch sense.
- Insensate feet — you cannot feel a pebble in your shoe, a blister forming, or hot bathwater. This is where the danger lives, because silent injury leads to foot ulcers.
- Ankle reflexes disappear.
- Hands eventually develop the same pattern, starting in the fingertips.
A painful variant, painful diabetic neuropathy (PDN), affects about a third of patients with polyneuropathy. The pain is often described as burning, stabbing, electric, or "walking on broken glass," and it characteristically worsens at night and with inactivity. Tesfaye and colleagues' 2010 Toronto consensus paper remains the definitive reference on classification and treatment.
Small-fiber neuropathy is a particularly important subtype. Only the tiniest unmyelinated C-fibers are affected; standard nerve conduction studies are normal, and diagnosis requires a skin biopsy showing reduced intraepidermal nerve fiber density. Small-fiber neuropathy overlaps substantially with fibromyalgia, with POTS, and with Long-COVID-like autonomic syndromes — patients can spend years being told their symptoms are psychological before the diagnosis is made. See the small-fiber neuropathy overlap article for more.
Autonomic and Focal Neuropathies
Autonomic neuropathy affects the nerves that run your internal organs. It is under-diagnosed because symptoms are vague and rarely linked to diabetes by patients or primary care teams.
- Cardiovascular autonomic neuropathy (CAN). Resting tachycardia, exercise intolerance, orthostatic hypotension, silent myocardial ischemia (heart attacks without chest pain). CAN is diagnosed on heart rate variability testing or tilt-table testing and overlaps heavily with POTS-like presentations.
- Gastroparesis. Delayed stomach emptying causing early satiety, nausea, vomiting of undigested food, and erratic post-meal glucose. Diagnosed by a gastric emptying scintigraphy scan.
- Sudomotor dysfunction. Abnormal sweating — either gustatory sweating (face sweats during meals) or anhidrosis (feet that cannot sweat, which accelerates skin breakdown). Quantitative sudomotor axon reflex testing (QSART) and skin biopsy confirm it.
- Genitourinary autonomic neuropathy. Erectile dysfunction in men, vaginal dryness and reduced arousal in women, neurogenic bladder with incomplete emptying and recurrent urinary infections.
Focal neuropathies affect a single nerve at a time, often abruptly.
- Third cranial nerve palsy. Sudden painful double vision with a drooping eyelid. The pupil is usually spared — an important distinction from a dangerous aneurysm. Most diabetic CN3 palsies resolve in weeks to months.
- Carpal tunnel syndrome. Diabetes triples the risk. Managed with splinting, steroid injection, or surgical release.
- Diabetic amyotrophy (proximal diabetic neuropathy). Severe thigh pain followed by quadriceps wasting and weakness, usually asymmetric. Frightening but usually partially or fully reversible over 12–24 months.
Neuropathy Screening
Annual foot exam in a primary care or endocrine clinic, using four quick bedside tools:
- 10-gram Semmes-Weinstein monofilament pressed at standardized sites on the plantar surface. If you cannot feel it, protective sensation is gone.
- 128 Hz tuning fork on the bony prominence of the great toe for vibration sense.
- Pinprick for sharp-dull discrimination.
- Ankle reflex testing with a reflex hammer.
When to start. Type 1 diabetes: 5 years after diagnosis, then annually. Type 2 diabetes: at diagnosis, then annually. Any patient with symptoms should be examined immediately regardless of duration.
For suspected autonomic disease, order targeted testing: tilt-table testing for orthostatic intolerance, gastric emptying scintigraphy for gastroparesis, QSART or skin biopsy for small-fiber and sudomotor disease.
Neuropathy Treatments
The uncomfortable truth: no drug currently reverses diabetic nerve damage. What we have are drugs that dampen pain and slow progression. The best-proven disease-modifier is still tight glycemic control.
First-line for painful neuropathy (ADA-preferred):
- Duloxetine (a serotonin-norepinephrine reuptake inhibitor) — starting 30 mg daily, titrating to 60 mg.
- Gabapentin — titrated from 300 mg up to 1800–3600 mg daily in divided doses.
- Pregabalin — 75 mg twice daily, titrating to 300–600 mg daily.
- Tricyclic antidepressants — amitriptyline or nortriptyline at bedtime. Nortriptyline causes fewer anticholinergic side effects and is a better choice in older adults.
Most patients need to try two or three of these before landing on a tolerable combination. All have meaningful side effects — sedation, weight gain, dry mouth, or edema — so start low and go slow.
Newer options. Capsaicin 8% patch (Qutenza) is applied for 30–60 minutes in a clinic and can provide 3 months of pain relief by depleting pain-fiber neurotransmitters. Spinal cord stimulation is now FDA-approved for refractory painful diabetic neuropathy and produces life-changing relief in carefully selected patients whose pain has not responded to medications.
Topical agents. Lidocaine 5% patches, capsaicin cream, and compounded amitriptyline/ketamine creams offer modest benefit for focal pain and spare you some systemic side effects.
Opioids are not first-line and should be avoided long-term. They rarely help neuropathic pain at tolerable doses and carry addiction risk.
Alpha-lipoic acid at 600 mg daily has modest evidence in European trials; it is inexpensive, well-tolerated, and reasonable to try. Benfotiamine (a fat-soluble thiamine analogue) has weaker evidence.
Foot-care infrastructure matters as much as pills: well-fitted shoes, daily foot inspection (use a mirror), immediate attention to any blister or crack, and regular podiatry visits. Diabetic foot ulcers are the leading non-traumatic cause of lower-limb amputation in the developed world, and nearly all begin as a silent insensate injury.
Other Complications — Feet, Bones, Brain, Ears
Beyond the classic triad, diabetes touches nearly every organ system. The ones worth knowing about:
- Peripheral arterial disease (PAD). Macrovascular narrowing of the leg arteries, often coexisting with neuropathy. The combination — poor blood flow plus no sensation — is what drives amputations. Screen with ankle-brachial index testing in long-standing diabetes or any patient with claudication or a non-healing foot wound.
- Foot ulcers. Daily inspection, offloading boots, early podiatry, and wound-care clinic referral for any ulcer that does not shrink within 2 weeks. The goal is to catch every lesion early enough that it never becomes an amputation conversation.
- Osteoporosis and fracture risk. Both type 1 and type 2 diabetes independently raise fracture risk, even when bone density reads as normal. Bone quality is impaired by glycation of collagen. DEXA scanning is still worthwhile, but do not be reassured by a normal T-score.
- Periodontal disease. Diabetes doubles gum disease risk, and severe periodontitis in turn worsens glycemic control — a bidirectional loop. Twice-yearly dental cleanings are not a luxury.
- Hearing loss. Sensorineural hearing loss is about twice as common in diabetics. Get a baseline audiogram if you notice any change.
- Cognitive decline and dementia. Long-standing diabetes roughly doubles dementia risk, both vascular and Alzheimer-type. Tight midlife glucose control appears to be protective; severe recurrent hypoglycemia in older adults may be harmful.
Intensive Control Caveats
The DCCT and UKPDS story — lower A1c, fewer complications — is true but not universal. The ACCORD trial in 2008 enrolled patients with long-standing type 2 diabetes and high cardiovascular risk, randomizing them to an A1c target below 6.0% versus standard 7.0–7.9%. The intensive arm had more deaths, and the trial was stopped early. Subsequent analyses suggest the excess mortality was driven by severe hypoglycemia and by how aggressively the target was pursued, not by low A1c per se.
The lesson: individualize your target. A 35-year-old with new type 1 diabetes, no complications, and no hypoglycemia unawareness should probably aim for an A1c of 6.5% or lower. A 75-year-old with 20 years of type 2 diabetes, coronary disease, and a history of passing out from low sugar should aim for 7.5–8.0% and prioritize avoiding hypoglycemia. Tight control is a gift in the first decade of diabetes and a trap in the fourth. See the sibling article on hypoglycemia awareness and prevention for why severe lows count as a complication in their own right.
A Word on Metformin and B12
Metformin interferes with B12 absorption in the terminal ileum. Patients who have been on metformin longer than about 4 years have a meaningfully higher rate of B12 deficiency, and the symptoms — tingling, numbness, fatigue — are easy to mistake for diabetic neuropathy itself. Check a serum B12 level (or methylmalonic acid if B12 is borderline) at least every 2–3 years on long-term metformin, and sooner if neuropathic symptoms appear. Replacement is cheap, oral, and effective; missing the diagnosis means treating a reversible deficiency as if it were permanent nerve damage.
Key Research Papers
Foundational and recent peer-reviewed publications on diabetic retinopathy, nephropathy, and neuropathy — the microvascular triad. Each citation links to the full text via DOI.
- The Diabetes Control and Complications Trial Research Group. The Effect of Intensive Treatment of Diabetes on the Development and Progression of Long-Term Complications in Insulin-Dependent Diabetes Mellitus (DCCT). New England Journal of Medicine. 1993;329(14):977–986.
- UK Prospective Diabetes Study (UKPDS) Group. Intensive Blood-Glucose Control with Sulphonylureas or Insulin Compared with Conventional Treatment and Risk of Complications in Patients with Type 2 Diabetes (UKPDS 33). Lancet. 1998;352(9131):837–853.
- Wong TY, Cheung CMG, Larsen M, Sharma S, Simó R. Diabetic Retinopathy. Nature Reviews Disease Primers. 2016;2:16012.
- Solomon SD, Chew E, Duh EJ, et al. Diabetic Retinopathy: A Position Statement by the American Diabetes Association. Diabetes Care. 2017;40(3):412–418.
- Alicic RZ, Rooney MT, Tuttle KR. Diabetic Kidney Disease: Challenges, Progress, and Possibilities. Clinical Journal of the American Society of Nephrology. 2017;12(12):2032–2045.
- Perkovic V, Jardine MJ, Neal B, et al. Canagliflozin and Renal Outcomes in Type 2 Diabetes and Nephropathy (CREDENCE). New England Journal of Medicine. 2019;380(24):2295–2306.
- Pop-Busui R, Boulton AJM, Feldman EL, et al. Diabetic Neuropathy: A Position Statement by the American Diabetes Association. Diabetes Care. 2017;40(1):136–154.
- Ziegler D, Tesfaye S, Spallone V, et al. Screening, Diagnosis and Management of Diabetic Sensorimotor Polyneuropathy in Clinical Practice. Diabetes/Metabolism Research and Reviews. 2022;38(3):e3525.
- American Diabetes Association Professional Practice Committee. Standards of Care in Diabetes—2024. Diabetes Care. 2024;47(Suppl 1):S1–S321.
- Tesfaye S, Boulton AJM, Dyck PJ, et al. Diabetic Neuropathies: Update on Definitions, Diagnostic Criteria, Estimation of Severity, and Treatments. Diabetes Care. 2010;33(10):2285–2293.
Live PubMed Searches
Each link opens a live PubMed query so results stay current as new papers are indexed.
- Diabetic retinopathy and anti-VEGF therapy
- Diabetic macular edema and faricimab
- Diabetic retinopathy AI screening
- Diabetic nephropathy and SGLT2 inhibitors
- Finerenone FIDELIO and FIGARO trials
- Semaglutide FLOW trial renal outcomes
- Painful diabetic neuropathy treatment
- Cardiovascular autonomic neuropathy in diabetes
- Small-fiber neuropathy and skin biopsy
- ACCORD intensive glucose control and mortality
- Metformin and vitamin B12 deficiency
Connections
- Alpha Lipoic Acid (Neuropathy Treatment)
- Antioxidants
- Diabetes Overview
- Cardio-Renal Protection: SGLT2 and GLP-1
- Hypoglycemia Awareness and Prevention
- Insulin Types and Dosing Strategies
- Remission and Reversal Strategies
- CGM and Time-in-Range
- A1c, Fructosamine, and Glycation Markers
- Type 1 vs Type 2 vs LADA and MODY
- Diabetic Foot Care and Ulcer Prevention
- Kidney Disease
- eGFR
- Creatinine
- Peripheral Neuropathy
- Edema
- POTS
- Fibromyalgia
- Hemoglobin A1C