Cardio-Renal Protection: SGLT2 Inhibitors and GLP-1 Agonists
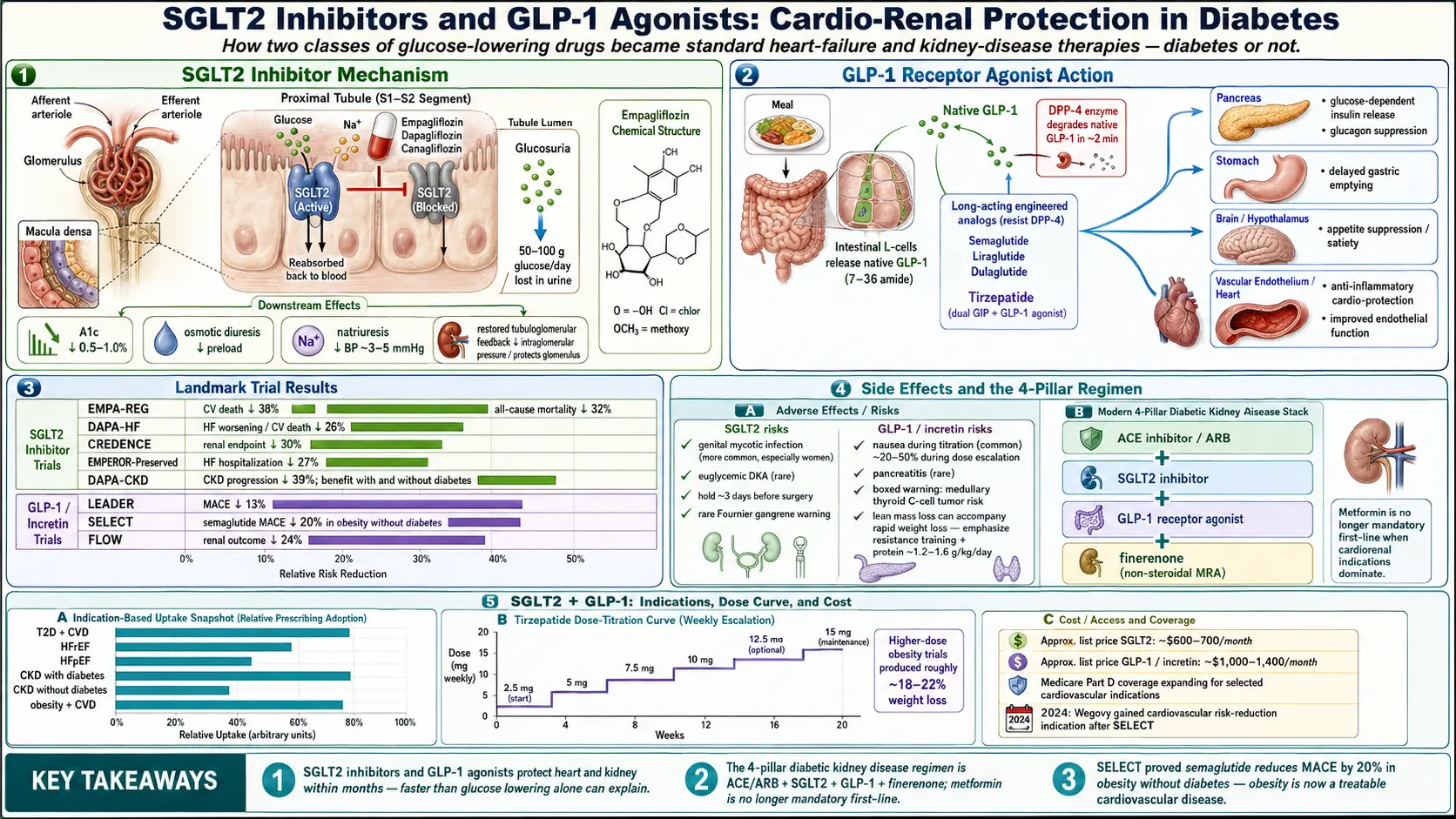
Table of Contents
- The Paradigm Shift — From Safety Trials to Lifesaving Drugs
- SGLT2 Inhibitors — How They Work
- The SGLT2 Drug List
- Landmark Cardiovascular Trials (SGLT2)
- Heart Failure Trials — DAPA-HF, EMPEROR, DELIVER
- Renal Trials — CREDENCE, DAPA-CKD, EMPA-KIDNEY
- SGLT2 Side Effects & Safety
- GLP-1 Receptor Agonists — How They Work
- The GLP-1 Drug List
- GLP-1 Cardiovascular & Renal Trials
- Weight Loss, SELECT, and Obesity as a Disease
- GLP-1 Side Effects & Safety
- Combining SGLT2 + GLP-1 — The Emerging Standard
- Finerenone — The Third Pillar for Diabetic Kidney Disease
- Residual Risk — Icosapent Ethyl and REDUCE-IT
- Cost, Insurance, and Access
- Non-Diabetic Uses
- Who Should Be on One — The 2024 Guideline View
- Key Research Papers
- Connections
- Featured Videos
The Paradigm Shift — From Safety Trials to Lifesaving Drugs
In 2008, after the rosiglitazone controversy suggested a diabetes drug could lower blood sugar while raising heart attack risk, the FDA issued a new mandate: every new glucose-lowering drug for type 2 diabetes had to prove cardiovascular safety in a large outcome trial before approval. The goal was modest — show the drug does not kill people. The result was one of the most important accidents in modern medicine.
Starting with EMPA-REG OUTCOME in 2015, these mandatory safety trials began showing the opposite of what regulators expected. The new drugs did not just fail to harm the heart — they prevented heart attacks, hospitalizations for heart failure, kidney failure, and death. Benefits appeared within months, too fast to be explained by glucose lowering alone. The trials forced cardiologists and nephrologists to rewrite guidelines that had stood for decades, and forced pharmaceutical companies to re-run the same trials in patients without diabetes — where the drugs worked just as well.
Today, two drug classes dominate this revolution: SGLT2 inhibitors (the “-flozins”) and GLP-1 receptor agonists (the “-tides” plus tirzepatide). If you have type 2 diabetes with any cardiovascular or kidney risk, the question is no longer whether you should be on one. It is which one, and when to add the second.
SGLT2 Inhibitors — How They Work
Every day your kidneys filter about 180 grams of glucose out of the blood — and then, under normal circumstances, reabsorb essentially all of it before it reaches the urine. The reabsorption is done mostly by a protein called sodium-glucose cotransporter 2 (SGLT2), sitting in the early part of the proximal tubule. SGLT2 pulls glucose back into the bloodstream and drags a sodium ion along with it.
SGLT2 inhibitors block that protein. The result is that roughly 50–100 grams of glucose spill into the urine every day, along with the sodium that would have been reabsorbed with it. This triggers four simultaneous effects, all therapeutic:
- Glucose lowering — A1c drops by about 0.5 to 1.0 percentage points, without causing hypoglycemia (the drug stops working as blood sugar approaches normal).
- Osmotic diuresis — the spilled glucose drags water with it, gently reducing fluid overload. This is why the heart-failure benefit kicks in within weeks.
- Natriuresis — sodium loss lowers blood pressure by roughly 3–5 mmHg systolic, adding to cardiovascular benefit.
- Tubuloglomerular feedback restoration — the sodium reaching the distal tubule signals the glomerulus to relax its overworked filtration pressure, protecting the kidney long term. This is the mechanism behind the renal outcomes.
Beyond the kidney, SGLT2 inhibitors modestly shift fuel metabolism toward ketones, reduce cardiac preload, lower uric acid, and improve erythropoiesis (hematocrit rises 2–3% on therapy). The whole package adds up to something that behaves less like a glucose drug and more like a gentle diuretic, antihypertensive, and cardiac protectant rolled into one.
The SGLT2 Drug List
- Empagliflozin (Jardiance) — Boehringer Ingelheim / Lilly. The most thoroughly studied. Approved for type 2 diabetes, HFrEF, HFpEF, and chronic kidney disease. Dose: 10 mg once daily, optional increase to 25 mg.
- Dapagliflozin (Farxiga / Forxiga) — AstraZeneca. Approved for type 2 diabetes, HFrEF, HFpEF, and CKD. Dose: 10 mg once daily.
- Canagliflozin (Invokana) — Janssen. The first to show renal benefit (CREDENCE). Dose: 100–300 mg once daily. Carries a slightly higher amputation signal in CANVAS, which has not been reproduced consistently.
- Ertugliflozin (Steglatro) — Merck / Pfizer. Approved for type 2 diabetes only; its outcome trial (VERTIS CV) was neutral on MACE, so it sits below the other three for cardio-renal indications.
- Bexagliflozin (Brenzavvy) — the newest entrant, approved 2023. Works the same way as the others but has no completed outcomes trial yet, so it is usually chosen for cost rather than evidence.
Landmark Cardiovascular Trials (SGLT2)
EMPA-REG OUTCOME (Zinman and colleagues, NEJM 2015). 7,020 patients with type 2 diabetes and established cardiovascular disease were randomized to empagliflozin or placebo on top of standard care. After a median 3.1 years, empagliflozin reduced cardiovascular death by 38%, all-cause mortality by 32%, and heart failure hospitalizations by 35%. The mortality curves separated within three months — far too fast for glucose lowering to explain. Cardiology was forced to pay attention.
CANVAS (canagliflozin, 2017) confirmed the composite MACE benefit in a broader population including primary-prevention diabetics. DECLARE-TIMI 58 (dapagliflozin, 2019) extended the evidence to lower-risk patients and showed a particularly strong heart-failure hospitalization benefit even when MACE reduction was modest. Together, these three trials established SGLT2 inhibition as a disease-modifying therapy in type 2 diabetes with cardiovascular risk.
Heart Failure Trials — DAPA-HF, EMPEROR, DELIVER
The next wave of trials asked a question nobody had taken seriously: do these drugs work in people without diabetes?
DAPA-HF (McMurray and colleagues, NEJM 2019) randomized 4,744 patients with heart failure and reduced ejection fraction (HFrEF) to dapagliflozin or placebo, regardless of diabetes status. Dapagliflozin cut the composite of heart-failure death or hospitalization by 26%, with nearly identical benefit in diabetics and non-diabetics. EMPEROR-Reduced (2020) confirmed the effect with empagliflozin. HFrEF guidelines rewrote themselves in under a year.
EMPEROR-Preserved (2021) tackled the much harder condition of heart failure with preserved ejection fraction (HFpEF) — a disease with no effective drugs until that point. Empagliflozin cut heart-failure hospitalizations by 27% and was the first drug ever to show any mortality signal in HFpEF. DELIVER (2022) replicated the finding with dapagliflozin. For millions of patients with “diastolic dysfunction” who had been told there was nothing to do, the answer became: start an SGLT2 inhibitor.
Renal Trials — CREDENCE, DAPA-CKD, EMPA-KIDNEY
CREDENCE (Perkovic and colleagues, NEJM 2019) randomized 4,401 patients with type 2 diabetes and overt diabetic nephropathy to canagliflozin or placebo. The trial was stopped early for benefit: canagliflozin reduced the composite renal endpoint (end-stage kidney disease, doubling of creatinine, or renal death) by 30%. Dialysis rates fell. It was the first dedicated renal-outcome trial in diabetes to show a kidney-protective drug effect in a generation.
DAPA-CKD (2020) extended the benefit to patients without diabetes. EMPA-KIDNEY (2023) broadened the evidence to an even wider CKD population, including many with proteinuria below the diabetic-nephropathy threshold. Today SGLT2 inhibitors are FDA-approved for CKD whether the patient has diabetes or not.
SGLT2 Side Effects & Safety
- Genital mycotic infections — the most common side effect. Sugar in the urine feeds yeast. Incidence is roughly 5–10% in women, 2–5% in men (uncircumcised men higher). Most episodes respond to a single dose of oral fluconazole or topical antifungal. Good local hygiene prevents recurrence.
- Euglycemic DKA — a rare but dangerous ketoacidosis that can occur at near-normal blood glucose, so the diagnosis is often missed. Triggered by fasting, surgery, severe illness, low-carb diets, or sudden insulin reduction. Hold SGLT2 inhibitors for at least 3 days before any planned surgery and during any acute illness that stops you eating.
- Volume depletion — the diuretic effect can cause dizziness, especially in older adults or those on loop diuretics. Loop-diuretic doses often need to be reduced when an SGLT2 inhibitor is started.
- Fournier’s gangrene — a necrotizing infection of the perineum. Extraordinarily rare but a black-box warning. Seek immediate care for severe pain, swelling, or redness in the genital or perianal area.
- Small rise in LDL cholesterol (roughly 3–5 mg/dL) — clinically minor and outweighed by the cardiovascular benefit.
- Transient eGFR dip in the first 2–4 weeks (3–5 mL/min), followed by long-term preservation of kidney function. This is the drug working, not harming.
GLP-1 Receptor Agonists — How They Work
Glucagon-like peptide-1 is a hormone released by the gut within minutes of a meal. It tells the pancreas to release insulin (but only when blood sugar is elevated — so it does not cause hypoglycemia on its own), tells the pancreas to stop releasing glucagon, tells the stomach to empty more slowly (so a meal lasts longer as a drip-feed instead of a flood), and acts on brain centers in the hypothalamus and brainstem to reduce appetite.
Native GLP-1 is destroyed within two minutes by the enzyme DPP-4. GLP-1 receptor agonists are engineered versions of the hormone — or entirely synthetic molecules that bind the same receptor — modified to resist breakdown. Half-lives range from hours (exenatide twice daily) to a week or more (semaglutide, dulaglutide). In addition to the metabolic effects, GLP-1 receptors sit on vascular endothelium, cardiomyocytes, and inflammatory cells, which is where the cardiovascular benefit is thought to originate.
The GLP-1 Drug List
- Semaglutide — Novo Nordisk. Three brands, same molecule: Ozempic (weekly injection for T2D), Rybelsus (oral daily tablet for T2D — the first oral GLP-1), Wegovy (higher-dose weekly injection for obesity).
- Tirzepatide (Mounjaro / Zepbound) — Eli Lilly. A dual GIP + GLP-1 agonist, not a pure GLP-1. Adds a second incretin receptor and produces the strongest weight loss of any drug ever approved (15–22% in trials). Mounjaro for T2D, Zepbound for obesity.
- Liraglutide (Victoza / Saxenda) — Novo Nordisk. Once-daily injection. Victoza for T2D, Saxenda for obesity. The first GLP-1 to show a CV mortality benefit.
- Dulaglutide (Trulicity) — Lilly. Weekly injection; simple auto-injector. Best CV data in primary prevention (REWIND).
- Exenatide (Bydureon, Byetta) — AstraZeneca. The original GLP-1. Weekly extended-release (Bydureon) or twice-daily (Byetta). Rarely first-line now.
GLP-1 Cardiovascular & Renal Trials
LEADER (Marso and colleagues, NEJM 2016) randomized 9,340 patients with type 2 diabetes at high cardiovascular risk to liraglutide or placebo. Liraglutide reduced the composite MACE endpoint (cardiovascular death, non-fatal MI, non-fatal stroke) by 13%, with a particularly clear cardiovascular mortality benefit. It was the first GLP-1 outcomes trial to turn positive.
SUSTAIN-6 (semaglutide injectable, 2016) and PIONEER-6 (oral semaglutide, 2019) extended the benefit. REWIND (dulaglutide, 2019) was important because most of its enrolled patients had no prior cardiovascular event — it demonstrated benefit in primary prevention, broadening the treatable population substantially.
On the renal side, the FLOW trial (semaglutide, 2024) was stopped early for overwhelming benefit: semaglutide slowed the progression of chronic kidney disease in type 2 diabetes by 24%, meaning GLP-1 agonists now join SGLT2 inhibitors as nephroprotective drugs rather than just metabolic ones.
Weight Loss, SELECT, and Obesity as a Disease
Weight loss on these drugs is not a side effect — it is now a primary reason for prescription. The STEP trials (semaglutide 2.4 mg weekly) produced average weight losses of 12–15% in people with obesity without diabetes. The SURMOUNT trials (tirzepatide 10–15 mg weekly) produced 18–22%, approaching bariatric-surgery territory.
The SELECT trial (semaglutide 2.4 mg, published 2023) was the first cardiovascular outcomes trial in people with obesity who did not have diabetes. It enrolled 17,604 adults with a BMI of 27 or higher and pre-existing cardiovascular disease. Semaglutide reduced the three-point MACE composite by 20%, independent of how much weight each person lost. That result reframed obesity itself as a treatable cardiovascular disease, not merely a risk factor, and accelerated Medicare and insurance coverage for GLP-1 use in obesity with established heart disease.
GLP-1 Side Effects & Safety
- Nausea, vomiting, constipation, diarrhea — the headline side effects, affecting 20–50% of users at dose escalations. Usually manageable with slow dose titration (months, not weeks), small frequent low-fat meals, and temporary dose reduction. Most people adapt within 1–3 months.
- Pancreatitis — a rare association (incidence under 1%). Stop the drug for persistent severe abdominal pain radiating to the back.
- Gallbladder disease — increased rates of gallstones and cholecystitis, especially with rapid weight loss. Any rapid weight loss does this; GLP-1s do not cause it uniquely, but they magnify it.
- Medullary thyroid cancer — a black-box warning based on rodent studies. Contraindicated in anyone with a personal or family history of medullary thyroid carcinoma or MEN-2 syndrome. No human signal has been confirmed in two decades of use, but the warning stands.
- Diabetic retinopathy worsening — a paradoxical signal in SUSTAIN-6 attributed to rapid A1c improvement. People with pre-existing proliferative retinopathy should have an eye exam before starting and avoid dropping A1c too fast.
- Muscle loss — roughly 25–40% of weight lost on GLP-1s is lean mass. This is not unique to these drugs (all weight loss does this), but it matters for long-term function. Resistance training 2–3 times weekly and protein intake of 1.2–1.6 g/kg ideal body weight are essential — not optional — for anyone on a GLP-1 who wants to preserve strength.
- Injection-site reactions and, rarely, hair shedding during rapid weight loss (telogen effluvium, reversible).
Combining SGLT2 + GLP-1 — The Emerging Standard
The two drug classes work through completely separate mechanisms. SGLT2 inhibitors pull glucose out through the kidney and protect the heart and kidneys mechanically and metabolically. GLP-1 agonists reduce appetite, improve insulin secretion, and protect the heart through vascular and inflammatory pathways. The cardio-renal benefits in trials are additive, not overlapping.
The 2023–2024 ADA Standards of Care and the AACE algorithm now recommend starting both drug classes in any patient with type 2 diabetes who has established cardiovascular disease, heart failure, or chronic kidney disease — regardless of baseline A1c. Metformin is still useful but no longer mandatory first-line; for a high-risk patient, starting an SGLT2 inhibitor or GLP-1 agonist (or both) before metformin is now acceptable. See Metformin, Berberine, and GLP-1s for the full comparison of glucose-lowering agents.
Finerenone — The Third Pillar for Diabetic Kidney Disease
Finerenone (Kerendia) is a non-steroidal mineralocorticoid receptor antagonist — a cousin of spironolactone, but designed to block the aldosterone receptor selectively without the sex-hormone side effects that make spironolactone intolerable for many men (gynecomastia) and women (menstrual irregularity).
The FIDELIO-DKD and FIGARO-DKD trials showed that finerenone added to standard care (including an ACE inhibitor or ARB) reduces kidney-failure progression and cardiovascular events in type 2 diabetic kidney disease. The benefit stacks on top of an SGLT2 inhibitor — the two drugs target different pieces of the same problem. For a patient with type 2 diabetes, albuminuric CKD, and an eGFR above 25, the current “four-pillar” package is: an ACE inhibitor or ARB, an SGLT2 inhibitor, a GLP-1 agonist, and finerenone. Potassium must be monitored — finerenone raises it modestly.
Residual Risk — Icosapent Ethyl and REDUCE-IT
Even on maximum statin therapy, an SGLT2 inhibitor, and a GLP-1 agonist, some patients keep having cardiovascular events. In the REDUCE-IT trial, patients with elevated triglycerides (135–499 mg/dL) and either established cardiovascular disease or diabetes-plus-risk-factors were randomized to icosapent ethyl (Vascepa, 4 grams daily of purified EPA) or mineral oil placebo. Icosapent ethyl reduced MACE by 25%. The mechanism is not fully understood — it is not just triglyceride lowering — but for patients with persistent hypertriglyceridemia on statins, it is a reasonable fourth layer on top of the cardio-renal regimen.
Cost, Insurance, and Access
These drugs are expensive. U.S. list prices for brand-name SGLT2 inhibitors run around $600–$700 per month; GLP-1 agonists are $1,000–$1,400 per month; tirzepatide is similar. Commercial insurance and Medicare Part D cover both classes for type 2 diabetes, usually with prior authorization. For obesity without diabetes, Medicare has traditionally excluded anti-obesity drugs, but in 2024 the CMS allowed Part D coverage of semaglutide (Wegovy) specifically for patients with established cardiovascular disease — the first crack in a decades-old exclusion, driven entirely by the SELECT results.
Manufacturer copay cards often bring commercial out-of-pocket cost to $25 per month but are typically barred for Medicare and Medicaid enrollees. When GLP-1 agonists went into FDA-declared shortage in 2022–2024, compounding pharmacies legally produced semaglutide and tirzepatide at a fraction of the cost. The FDA removed both drugs from the shortage list in 2024–2025, which in turn restricted most compounded versions. Patients should buy only from licensed pharmacies — counterfeit injectables sold online have caused overdoses.
Non-Diabetic Uses
- Heart failure (HFrEF and HFpEF) — SGLT2 inhibitors are standard, diabetes or not. See Heart Failure.
- Chronic kidney disease — SGLT2 inhibitors reduce progression with or without diabetes. See Kidney Disease.
- Obesity with cardiovascular disease — semaglutide 2.4 mg is FDA-approved for MACE reduction. See Obesity.
- MASLD (metabolic-associated steatotic liver disease, formerly NAFLD) — GLP-1 agonists and SGLT2 inhibitors both reduce liver fat and inflammation; tirzepatide and semaglutide have the strongest data in MASH.
- Polycystic ovary syndrome — GLP-1 agonists improve insulin resistance, weight, and menstrual regularity.
Who Should Be on One — The 2024 Guideline View
If you have type 2 diabetes, ask your clinician the four questions below. If you answer yes to any of them, guidelines now say an SGLT2 inhibitor or GLP-1 agonist (often both) is indicated regardless of what your A1c is:
- Do I have established cardiovascular disease (prior heart attack, stroke, stent, bypass, or symptomatic peripheral artery disease)?
- Do I have heart failure of any type — reduced or preserved ejection fraction?
- Do I have chronic kidney disease (eGFR under 60 or albumin-to-creatinine ratio over 30 mg/g)?
- Am I at high 10-year cardiovascular risk (multiple risk factors, family history, obesity with metabolic syndrome)?
If the answer is no to all four and your A1c is well-controlled on metformin alone, that remains a perfectly reasonable place to stay. But for the majority of adults with long-standing type 2 diabetes, at least one of those answers is yes — and the evidence for SGLT2 and GLP-1 is now strong enough that the burden has shifted to justifying why someone is not on one.
Key Research Papers
The cardiovascular and renal outcome trials that turned SGLT2 inhibitors and GLP-1 receptor agonists from glucose-lowering drugs into disease-modifying therapies for the heart and kidney.
- The Diabetes Control and Complications Trial Research Group. The Effect of Intensive Treatment of Diabetes on the Development and Progression of Long-Term Complications in Insulin-Dependent Diabetes Mellitus (DCCT). New England Journal of Medicine. 1993;329(14):977-986.
- UK Prospective Diabetes Study (UKPDS) Group. Intensive Blood-Glucose Control with Sulphonylureas or Insulin Compared with Conventional Treatment and Risk of Complications in Patients with Type 2 Diabetes (UKPDS 33). Lancet. 1998;352(9131):837-853.
- American Diabetes Association Professional Practice Committee. Standards of Care in Diabetes—2024. Diabetes Care. 2024;47(Suppl 1):S1-S321.
- Zinman B, Wanner C, Lachin JM, et al. Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes (EMPA-REG OUTCOME). New England Journal of Medicine. 2015;373(22):2117-2128.
- Neal B, Perkovic V, Mahaffey KW, et al. Canagliflozin and Cardiovascular and Renal Events in Type 2 Diabetes (CANVAS). New England Journal of Medicine. 2017;377(7):644-657.
- Wiviott SD, Raz I, Bonaca MP, et al. Dapagliflozin and Cardiovascular Outcomes in Type 2 Diabetes (DECLARE-TIMI 58). New England Journal of Medicine. 2019;380(4):347-357.
- Perkovic V, Jardine MJ, Neal B, et al. Canagliflozin and Renal Outcomes in Type 2 Diabetes and Nephropathy (CREDENCE). New England Journal of Medicine. 2019;380(24):2295-2306.
- Heerspink HJL, Stefansson BV, Correa-Rotter R, et al. Dapagliflozin in Patients with Chronic Kidney Disease (DAPA-CKD). New England Journal of Medicine. 2020;383(15):1436-1446.
- Bakris GL, Agarwal R, Anker SD, et al. Effect of Finerenone on Chronic Kidney Disease Outcomes in Type 2 Diabetes (FIDELIO-DKD). New England Journal of Medicine. 2020;383(23):2219-2229.
- Marso SP, Daniels GH, Brown-Frandsen K, et al. Liraglutide and Cardiovascular Outcomes in Type 2 Diabetes (LEADER). New England Journal of Medicine. 2016;375(4):311-322.
- Marso SP, Bain SC, Consoli A, et al. Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes (SUSTAIN-6). New England Journal of Medicine. 2016;375(19):1834-1844.
- Gerstein HC, Colhoun HM, Dagenais GR, et al. Dulaglutide and Cardiovascular Outcomes in Type 2 Diabetes (REWIND). Lancet. 2019;394(10193):121-130.
- Frias JP, Davies MJ, Rosenstock J, et al. Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes (SURPASS-2). New England Journal of Medicine. 2021;385(6):503-515.
- McMurray JJV, Solomon SD, Inzucchi SE, et al. Dapagliflozin in Patients with Heart Failure and Reduced Ejection Fraction (DAPA-HF). New England Journal of Medicine. 2019;381(21):1995-2008.
- Anker SD, Butler J, Filippatos G, et al. Empagliflozin in Heart Failure with a Preserved Ejection Fraction (EMPEROR-Preserved). New England Journal of Medicine. 2021;385(16):1451-1461.
Live PubMed Searches
Live PubMed queries that update as new papers are indexed.
- SGLT2 inhibitors and cardiovascular outcomes
- Empagliflozin and HFpEF
- Dapagliflozin and chronic kidney disease
- GLP-1 receptor agonists and cardiovascular outcomes
- Semaglutide, SELECT trial, and obesity cardiovascular risk
- Tirzepatide and the SURMOUNT weight-loss trials
- Finerenone, FIDELIO, and FIGARO in diabetic kidney disease
- Icosapent ethyl and REDUCE-IT residual triglyceride risk
- Euglycemic DKA and SGLT2 inhibitors
- GLP-1 weight loss and muscle preservation
Connections
- Diabetes Overview
- Type 1 vs Type 2 vs LADA and MODY
- Insulin Types and Dosing Strategies
- Hypoglycemia Awareness and Prevention
- Remission and Reversal Strategies
- CGM and Time-in-Range
- A1c, Fructosamine, and Glycation Markers
- Complications: Retinopathy, Nephropathy, Neuropathy
- Diabetic Foot Care and Ulcer Prevention
- Insulin Resistance
- Obesity
- Metabolic Syndrome
- Kidney Disease
- Hypertension
- Heart Failure
- eGFR
- GLP-1 Receptor Agonists
- Cardiovascular Disease