Type 2 Diabetes Remission and Reversal Strategies
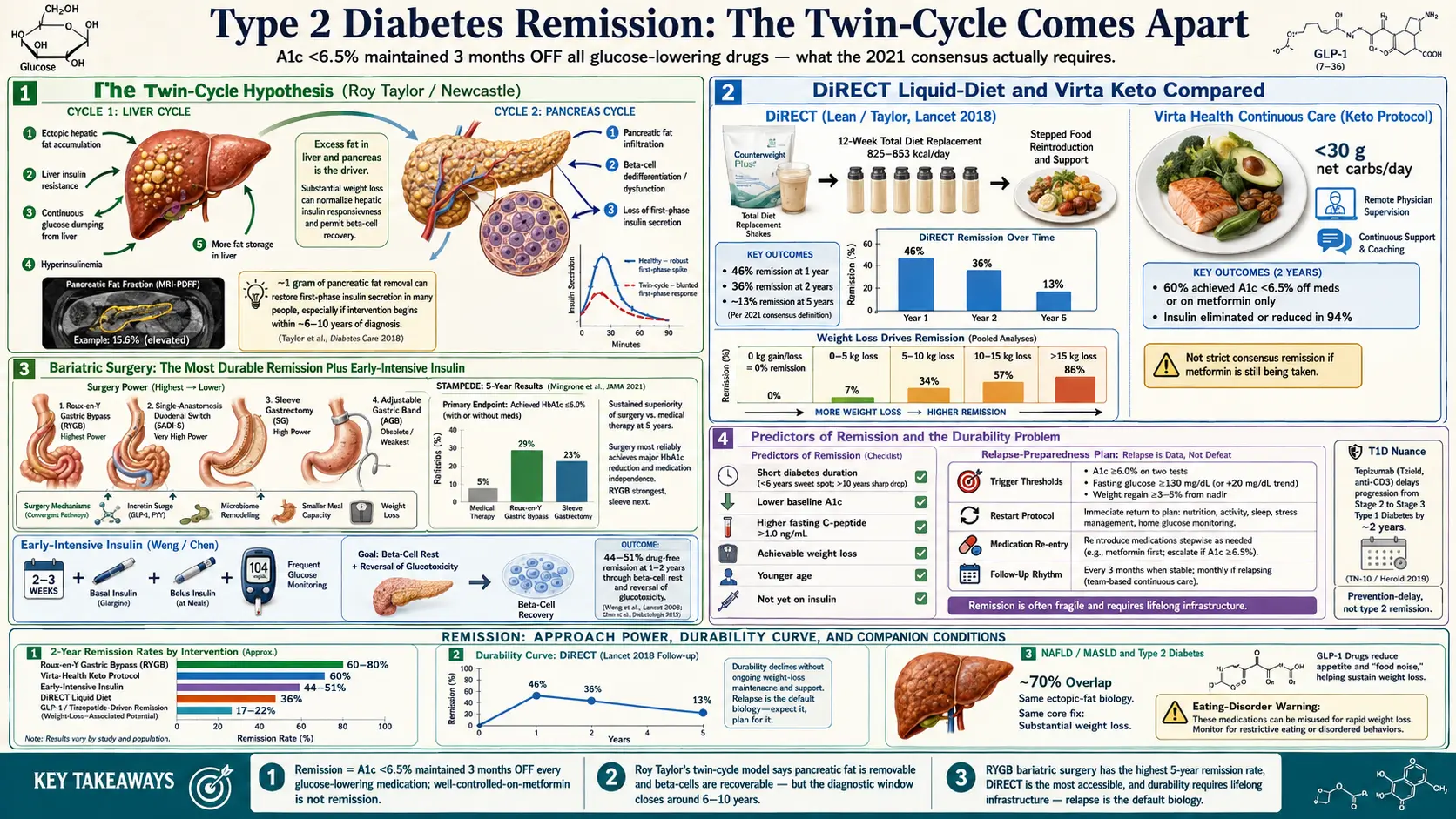
Table of Contents
- What Remission Officially Means — The 2021 Consensus
- The Twin-Cycle Hypothesis — Why Fat Matters More Than Weight
- The DiRECT Trial — Liquid-Diet Remission in Primary Care
- Virta Health — The Ketogenic Route
- Bariatric Surgery — The Most Durable Remission
- Early-Intensive Insulin — The Two-Week Reset
- Time-Restricted Eating and Prolonged Fasting
- GLP-1s, Tirzepatide, and the "Remission-Like" Question
- Who Actually Achieves Remission — Predictors That Matter
- The Durability Problem — Five-Year Relapse Rates
- Relapse Preparedness — Not Failure, Just Data
- Type 1 Diabetes — Teplizumab and the Honeymoon
- Pediatric Type 2 — The Hardest Case
- NAFLD as a Companion Goal
- Mental Health, Food Noise, and Relapse
- Measurement Rigor and Insurance Implications
- Key Research Papers
- Connections
- Featured Videos
What Remission Officially Means — The 2021 Consensus
Until 2021 there was no agreed definition of type 2 diabetes remission. Doctors used words like "reversal," "cure," "resolved," and "well-controlled" interchangeably, and nobody — including patients — knew exactly what the goal line was. That changed when a joint panel of the American Diabetes Association, the European Association for the Study of Diabetes, Diabetes UK, the Endocrine Society, and Diabetes Canada published a consensus statement led by Matthew Riddle in 2021.
The new definition is strict and simple: remission of type 2 diabetes is an A1c below 6.5% maintained for at least three months after stopping all glucose-lowering medications. That is it. No metformin, no SGLT2, no GLP-1, no insulin. If the A1c drifts back above 6.5% — or if any glucose-lowering drug is resumed — remission ends and is classified as relapse.
A few nuances matter. The consensus deliberately avoided the words "cure" and "reversal" because both imply permanence, and diabetes almost never vanishes permanently. "Remission" is the same word oncology uses: the disease is quiet, the numbers are normal, but the underlying vulnerability is still there. You still need A1c testing at least annually, and ideally every three to six months, because silent relapse is common.
An older, looser definition — "partial remission" at A1c under 6.5% while still taking non-insulin medications — was explicitly retired by the 2021 consensus. If you are still on metformin and your A1c is 6.0%, you have well-controlled diabetes, not remission. That distinction is not semantic hair-splitting. It changes how you and your doctor set targets, how the condition is coded on your chart, and how you think about your own progress.
The Twin-Cycle Hypothesis — Why Fat Matters More Than Weight
The reason remission is even possible traces back to a model proposed by Roy Taylor at Newcastle University: the twin-cycle hypothesis. In this model, type 2 diabetes is not a disease of the pancreas alone, or the muscles alone, or the liver alone — it is a feedback loop between two organs caked in fat.
Cycle one lives in the liver. When the liver accumulates ectopic fat (fat stored where it does not belong), it becomes resistant to insulin's signal to stop producing glucose. The liver keeps dumping sugar into the bloodstream even after meals, and chronically elevated insulin drives more fat storage, which worsens hepatic insulin resistance. Cycle two lives in the pancreas. Fat accumulating inside the pancreas itself suffocates the beta cells and strips them of their ability to secrete insulin in sharp, on-demand bursts. Fasting glucose climbs, post-meal spikes get worse, and the whole system tips into diabetes.
The radical claim from Taylor's group is this: pancreatic fat is removable, and beta-cell function is recoverable, but only if you lose fat specifically from the pancreas itself, not just general body weight. In MRI studies, losing about 1 gram of pancreatic fat was enough to restore first-phase insulin secretion in patients who had been diagnosed within the last six to ten years. Beyond that diagnostic window, beta cells appear to die off permanently, and remission becomes much harder.
This explains something clinicians have observed for decades without a good mechanism: people diagnosed recently have much higher remission rates than people who have lived with type 2 diabetes for fifteen years, even at identical weight loss. The clock on your pancreas starts ticking at diagnosis, and the sooner you intervene, the more beta-cell capacity is still recoverable.
The DiRECT Trial — Liquid-Diet Remission in Primary Care
The Diabetes Remission Clinical Trial (DiRECT), led by Mike Lean in Scotland and Roy Taylor in Newcastle, was the study that turned the twin-cycle hypothesis into a mainstream remission protocol. Published in The Lancet in 2018, it was a cluster-randomized trial run entirely in ordinary UK general-practice clinics — not specialist centers, not teaching hospitals.
The protocol was austere. Participants replaced all food with a total-diet replacement formula of 825 to 853 kcal per day — the commercial Counterweight Plus shakes — for 12 weeks. Glucose-lowering and blood-pressure drugs were stopped on day one. After 12 weeks there was a stepped, six-week food reintroduction, then an ongoing maintenance program of monthly weigh-ins, coaching, and rescue plans if weight started creeping back.
The results were startling: 46% of the intervention group achieved remission at 12 months, compared with 4% in the usual-care group. Remission was tightly linked to weight loss — 86% of people who lost more than 15 kg hit remission, versus only 7% of those who lost less than 5 kg. At two years, 36% were still in remission, and Taylor's five-year follow-up published in 2024 reported about 13% sustained remission — a sharp drop that frames the durability problem clearly.
What DiRECT proved is that remission is not reserved for bariatric surgery or elite nutrition clinics. Ordinary GPs, ordinary patients, and a meal-replacement shake program were enough to achieve drug-free remission in almost half of participants at one year. The UK National Health Service has since rolled out the DiRECT protocol as a standard primary-care program.
Virta Health — The Ketogenic Route
A very different protocol with remarkably similar outcomes came out of Indiana University and Virta Health, led by Sarah Hallberg. Instead of a 12-week liquid diet, Virta used a continuous ketogenic diet — under 30 grams of net carbs per day, moderate protein, fat to satiety — with remote physician supervision, daily biometric tracking, and aggressive medication deprescription as glucose dropped.
The 2018 one-year results (Hallberg et al.) and the 2019 two-year follow-up are the reference data. At two years: 60% of completers had A1c below 6.5% without medication or on metformin alone, insulin was eliminated or reduced in 94% of patients who started on it, and median weight loss was around 10% of body weight. Critically, these were patients with an average diabetes duration of 8 years — well past the "early window" most remission trials select for — which is one reason the data is important.
The Virta model differs from DiRECT in two practical ways. First, it is indefinite rather than time-limited — patients stay on the eating pattern long-term, and relapse is framed as drifting off the pattern rather than "finishing" a program. Second, it leans on continuous biometric reporting (glucose, ketones, weight, food) through an app with clinician oversight. This infrastructure is not cheap, but it is what allowed Virta to publish two-year retention and medication numbers that are still rare in dietary-intervention trials. See the reversal protocol deep dive for the practical day-to-day structure.
Bariatric Surgery — The Most Durable Remission
If you want the best odds of long-term remission, metabolic (bariatric) surgery is still the strongest single intervention. The four main procedures, roughly in order of remission power:
- Roux-en-Y gastric bypass (RYGB) — a small stomach pouch is connected directly to the mid-small intestine, bypassing the duodenum. Highest remission rates, strongest incretin and bile-acid changes, most technically demanding.
- Single-anastomosis duodenal switch (SADI-S) / biliopancreatic diversion — the most aggressive rearrangement, with the highest remission and weight-loss numbers but also the highest nutritional-deficiency risk.
- Sleeve gastrectomy — about 80% of the stomach is removed, leaving a banana-shaped tube. Less anatomically disruptive than bypass, now the most common U.S. bariatric operation. Remission rates are solid but slightly below RYGB.
- Adjustable gastric band — a restrictive silicone ring around the upper stomach. Essentially obsolete now; lowest remission rate and high late-complication burden.
The STAMPEDE trial (Schauer et al., NEJM 2017, five-year results) compared intensive medical therapy with RYGB or sleeve gastrectomy in 150 patients with uncontrolled type 2 diabetes. At five years, the surgical arms showed sustained A1c below 6.0% without medications in a meaningful fraction of patients, with RYGB outperforming sleeve. Long-term observational cohorts show 60–80% remission at five years after RYGB in carefully selected patients.
Surgery works through more than just weight loss. Within days of RYGB — before any meaningful weight has come off — glucose normalizes. Mechanisms include altered incretin signaling (a surge of GLP-1 and PYY from the distal gut reached by undigested food), shifts in bile-acid composition and circulation, microbiome remodeling, and a physical reduction in meal size. The Diabetes Surgery Summit guidelines now recommend considering surgery for type 2 diabetes at a BMI as low as 30 with inadequate glycemic control, far earlier than the old BMI-40 threshold.
Early-Intensive Insulin — The Two-Week Reset
One of the most counterintuitive remission strategies is a short, intensive course of insulin at the moment of diagnosis. The most-cited data comes from Weng et al. (Lancet 2008) and a follow-up by Chen et al. (2012), both in Chinese cohorts. Patients newly diagnosed with type 2 diabetes were given two to three weeks of basal-bolus insulin (continuous subcutaneous infusion or multiple daily injections) with aggressive titration to normal glucose, then all medications were stopped.
The result: 44–51% were still in drug-free remission at one to two years. The proposed mechanism is "beta-cell rest" — letting exhausted pancreatic cells recover function without the demand of chronic hyperglycemia. The strategy is standard practice in parts of China and used selectively in Europe, but it has not been widely adopted in U.S. practice, partly because the protocol requires careful inpatient or close-outpatient supervision to avoid hypoglycemia. If you are newly diagnosed with a high A1c, this is a conversation worth having with an endocrinologist.
Time-Restricted Eating and Prolonged Fasting
Less dramatic than DiRECT or surgery, but easier to start on a Monday. Time-restricted eating (TRE) compresses all calories into a window of 6 to 10 hours per day, with the rest of the 24 hours in water-only fasting. The best-quality trial is Sutton et al. (2018) on early time-restricted feeding (eTRF) — a six-hour eating window ending before 3 p.m. — in prediabetic men. Even without weight loss, insulin sensitivity, beta-cell function, blood pressure, and oxidative stress all improved.
Longer supervised fasts (three to seven days, repeated monthly) have shown dramatic A1c and weight effects in small cohorts, but they require medical supervision if you are on any glucose-lowering medication. SGLT2 inhibitors are particularly dangerous during prolonged fasting because of ketoacidosis risk, and sulfonylureas and insulin must be stopped or dramatically reduced. Fasting is a remission lever, not a lifestyle hack — do it with a doctor, not alone.
GLP-1s, Tirzepatide, and the "Remission-Like" Question
The GLP-1 class (semaglutide) and the dual GIP/GLP-1 agonist tirzepatide have opened a new possibility: weight loss of 17 to 22% of body weight in the SURPASS and SURMOUNT trial programs, comparable to mid-tier bariatric surgery. At those weight-loss magnitudes, A1c routinely drops into the non-diabetic range.
Technically, this is not remission under the 2021 consensus definition, because the patient is still on a glucose-lowering drug. If you stop the drug, weight comes back and A1c usually follows within 6 to 12 months — that is the signal from the STEP and SURMOUNT extension data. What these drugs may genuinely do, though, is act as a bridge: the weight loss they produce can be large enough to cross into the pancreatic-fat threshold Taylor described. A carefully designed protocol of GLP-1/tirzepatide to induce weight loss, followed by structured taper and dietary maintenance, is an active area of research. See the metformin, berberine, and GLP-1s deep dive for the drug-specific details.
Who Actually Achieves Remission — Predictors That Matter
Not everyone benefits equally from the same protocol. Across DiRECT, Virta, STAMPEDE, and the Chinese early-insulin cohorts, the same short list of predictors keeps appearing:
- Short diabetes duration. Under six years is the sweet spot. Over ten years, remission rates fall sharply regardless of intervention.
- Lower baseline A1c. Easier to cross the 6.5% line if you started at 7.5% than at 11%.
- Higher baseline C-peptide. C-peptide is a proxy for preserved beta-cell function. A fasting C-peptide above about 1.0 ng/mL predicts better response.
- Achievable weight loss. Across DiRECT, remission probability was 7% at under 5 kg lost, 34% at 5–10 kg, 57% at 10–15 kg, and 86% above 15 kg.
- Younger age. Beta-cell reserve and metabolic flexibility decline with age.
- Not on insulin yet. Insulin dependence usually signals advanced beta-cell loss.
None of these are absolute disqualifiers. Patients with 15-year diabetes on insulin have achieved remission after bariatric surgery. But if you are in the "easy" category — newly diagnosed, moderate A1c, overweight but not on insulin — the odds are very good, and the case for aggressive early action is strong.
The Durability Problem — Five-Year Relapse Rates
Here is the honest truth nobody markets well: most people who achieve remission relapse within five years. DiRECT dropped from 46% at year one to 36% at year two and roughly 13% at year five. Virta's retention declined. Post-surgical cohorts show a slow creep upward in A1c starting around year three.
Relapse is not a character flaw or proof that the strategy "didn't work." It is the default biology of a chronic metabolic disorder. The twin-cycle fat-accumulation process that caused diabetes the first time is still latent, and in a food environment engineered to promote weight regain, the cycle restarts easily.
What protects durability: ongoing lifestyle infrastructure. Monthly weigh-ins. Continuous glucose monitoring (CGM) even in remission, used episodically. Continuing program membership (DiRECT maintenance, Virta ongoing coaching, post-bariatric clinic visits). Scheduled six-month check-ins with primary care. The strategies that produce remission in year one are rarely the same strategies that maintain it in year five — maintenance is its own discipline.
Relapse Preparedness — Not Failure, Just Data
If you achieve remission, plan for relapse the way an addiction-recovery program plans for lapse. Decide in advance:
- Trigger threshold. What A1c, fasting glucose, or weight gain triggers a restart of the full remission protocol? Writing it down before it happens prevents months of drift.
- Restart protocol. For DiRECT alumni, this is usually another 4- to 12-week total-diet replacement. For Virta, it is a re-induction back to strict ketosis. For post-surgery, it often means resuming GLP-1 or re-engaging with the bariatric clinic.
- Medication re-entry. Sometimes the right answer is restarting metformin early rather than trying to white-knuckle a diet-only fix. Metformin is cheap, safe, and does not disqualify you from another remission attempt later.
- Mindset. Relapse is data, not defeat. Your body re-accumulated pancreatic fat faster than your maintenance plan could prevent. Adjust the plan.
Type 1 Diabetes — Teplizumab and the Honeymoon
Type 1 diabetes is an autoimmune disease, and the picture is very different. True, durable remission of established type 1 diabetes is extremely rare and usually brief. Two partial exceptions are worth knowing:
The honeymoon period. In the first months after diagnosis, residual beta cells sometimes recover enough function that insulin requirements drop dramatically, occasionally to zero. This is temporary — usually weeks to a few months, rarely beyond a year. It is not remission in the type 2 sense. Autoantibody attack continues in the background.
Teplizumab (Tzield). FDA-approved in 2022, teplizumab is an anti-CD3 monoclonal antibody that, given as a 14-day infusion course to people with Stage 2 type 1 diabetes (two or more autoantibodies, dysglycemia, no symptoms yet), delays clinical onset by a median of about two years — based on the Herold et al. TN-10 trial published in 2019. Teplizumab does not reverse established type 1 diabetes. It interrupts the autoimmune attack enough to buy time, and that time can matter enormously, particularly in children.
Pediatric Type 2 — The Hardest Case
Childhood and adolescent type 2 diabetes is a distinct clinical problem, not just small-adult type 2. The landmark TODAY trial (Treatment Options for Type 2 Diabetes in Adolescents and Youth) showed that over half of adolescents failed glycemic control within a few years regardless of whether they were on metformin alone, metformin plus rosiglitazone, or metformin plus intensive lifestyle intervention.
Pediatric type 2 progresses faster, responds less well to lifestyle change, and slides to insulin dependence earlier than adult type 2. Bariatric surgery in carefully selected adolescents (Teen-LABS cohort) produces strong remission rates, but the decision is heavier because it is permanent and affects decades of life. If your child has been diagnosed with type 2 diabetes, push for a multidisciplinary pediatric endocrinology program rather than managing it in general pediatrics.
NAFLD as a Companion Goal
Non-alcoholic fatty liver disease — now often called MASLD — is present in roughly 70% of people with type 2 diabetes, and the same twin-cycle fat that sits in the pancreas sits in the liver too. Remission protocols that work for diabetes essentially always also resolve NAFLD. DiRECT participants had meaningful reductions in liver fat on MRI. Virta ketogenic protocols show liver fat improvements at three to six months. Bariatric surgery resolves steatosis in the large majority of patients and can partially reverse early fibrosis.
If you have both conditions (most people with type 2 diabetes do), treat them as one metabolic problem rather than two separate ones. See the NAFLD-MASLD connection deep dive for the shared-mechanism details and imaging follow-up strategy.
Mental Health, Food Noise, and Relapse
Remission protocols are hard emotionally, not just physically. Three mental-health dimensions are worth naming:
Food noise. Many patients on GLP-1s and tirzepatide describe a sudden quieting of constant intrusive thoughts about food. For people who have lived with food noise their whole lives, the silence itself is therapeutic. It is also one reason people feel psychologically worse when they come off the drug — the noise returns before weight does.
Eating disorders. Very-low-calorie and ketogenic protocols are not appropriate for patients with active or recent eating disorders. Restrictive dieting can trigger relapse of anorexia nervosa, binge-eating disorder, or orthorexia. Screen yourself honestly, or ask a primary care physician to screen, before starting.
Body image and maintenance. Dramatic weight loss changes how you look, how people treat you, and how you see yourself — not always comfortably. Loose skin, identity shifts, altered relationships, and grief about years spent at a higher weight are all real. A therapist familiar with bariatric or weight-loss psychology is a reasonable add-on to any serious remission program.
Measurement Rigor and Insurance Implications
Two technical points every patient should understand before declaring remission.
You must be off all glucose-lowering medications for at least three months, confirmed by a lab-drawn A1c under 6.5%. A fingerstick in your kitchen does not count. A1c while still taking metformin does not count. CGM time-in-range is not a substitute. If your doctor will not stop your meds, you are not yet in remission — you are well-controlled on therapy, which is a good place to be but not the consensus definition.
Insurance and billing complexity. Once diagnosed with type 2 diabetes, the ICD-10 code tends to stick to your chart even after remission. Some insurers and life-insurance underwriters treat "history of diabetes" as equivalent to "active diabetes" for premium purposes. Others will update if you provide documented remission data (two A1c values under 6.5% at least three months apart, off all diabetes medications, plus a physician letter). This is worth pushing on — the premium differences can be substantial — but it is a paperwork battle, not an automatic update. Disability paperwork, commercial driver medical certification, and occupational medical clearances also vary widely in how they treat remission. Ask specifically.
Key Research Papers
Foundational peer-reviewed work on type 2 diabetes remission — the consensus definition, the DiRECT and Virta trials, bariatric surgery durability, and the metabolic mechanism behind reversal. Each citation links to the full text via DOI.
- Riddle MC, Cefalu WT, Evans PH, et al. Consensus Report: Definition and Interpretation of Remission in Type 2 Diabetes. Diabetes Care. 2021;44(10):2438–2444.
- Lean ME, Leslie WS, Barnes AC, et al. Primary Care-Led Weight Management for Remission of Type 2 Diabetes (DiRECT): An Open-Label, Cluster-Randomised Trial. Lancet. 2018;391(10120):541–551.
- Lean MEJ, Leslie WS, Barnes AC, et al. Durability of a Primary Care-Led Weight-Management Intervention for Remission of Type 2 Diabetes: 2-Year Results of the DiRECT Open-Label, Cluster-Randomised Trial. Lancet Diabetes & Endocrinology. 2019;7(5):344–355.
- Hallberg SJ, McKenzie AL, Williams PT, et al. Effectiveness and Safety of a Novel Care Model for the Management of Type 2 Diabetes at 1 Year: An Open-Label, Non-Randomized, Controlled Study. Diabetes Therapy. 2018;9(2):583–612.
- Mingrone G, Panunzi S, De Gaetano A, et al. Bariatric-Metabolic Surgery versus Conventional Medical Treatment in Obese Patients with Type 2 Diabetes: 5 Year Follow-Up of an Open-Label, Single-Centre, Randomised Controlled Trial. Lancet. 2015;386(9997):964–973.
- Schauer PR, Bhatt DL, Kirwan JP, et al. Bariatric Surgery versus Intensive Medical Therapy for Diabetes — 5-Year Outcomes (STAMPEDE). New England Journal of Medicine. 2017;376(7):641–651.
- UK Prospective Diabetes Study (UKPDS) Group. Intensive Blood-Glucose Control with Sulphonylureas or Insulin Compared with Conventional Treatment and Risk of Complications in Patients with Type 2 Diabetes (UKPDS 33). Lancet. 1998;352(9131):837–853.
- American Diabetes Association Professional Practice Committee. Standards of Care in Diabetes—2024. Diabetes Care. 2024;47(Suppl 1):S1–S321.
- DeFronzo RA, Ferrannini E, Groop L, et al. Type 2 Diabetes Mellitus. Nature Reviews Disease Primers. 2015;1:15019.
Live PubMed Searches
Each link opens a live PubMed query so results stay current as new papers are indexed.
- Type 2 diabetes remission definition and consensus
- DiRECT trial and primary-care remission programs
- Taylor twin-cycle hypothesis and pancreatic fat
- Ketogenic diet and type 2 diabetes remission
- Bariatric surgery and long-term diabetes remission
- Early-intensive insulin in new-onset type 2 diabetes
- Tirzepatide, SURPASS/SURMOUNT, and metabolic outcomes
- Teplizumab and delay of type 1 diabetes onset
- TODAY trial and pediatric type 2 diabetes outcomes
- Diabetes remission durability and relapse
Connections
- Diabetes Overview
- Type 1 vs Type 2 vs LADA and MODY
- Cardio-Renal Protection: SGLT2 and GLP-1
- Hypoglycemia Awareness and Prevention
- CGM and Time-in-Range
- A1c, Fructosamine, and Glycation Markers
- Complications: Retinopathy, Nephropathy, Neuropathy
- Insulin Types and Dosing Strategies
- Insulin Resistance
- Obesity
- Metabolic Syndrome
- NAFLD and MASLD
- Fasting
- GLP-1 Receptor Agonists
- Berberine
- Coffee
- Diabetic Foot Care and Ulcer Prevention