CGM and Time-in-Range for Diabetes
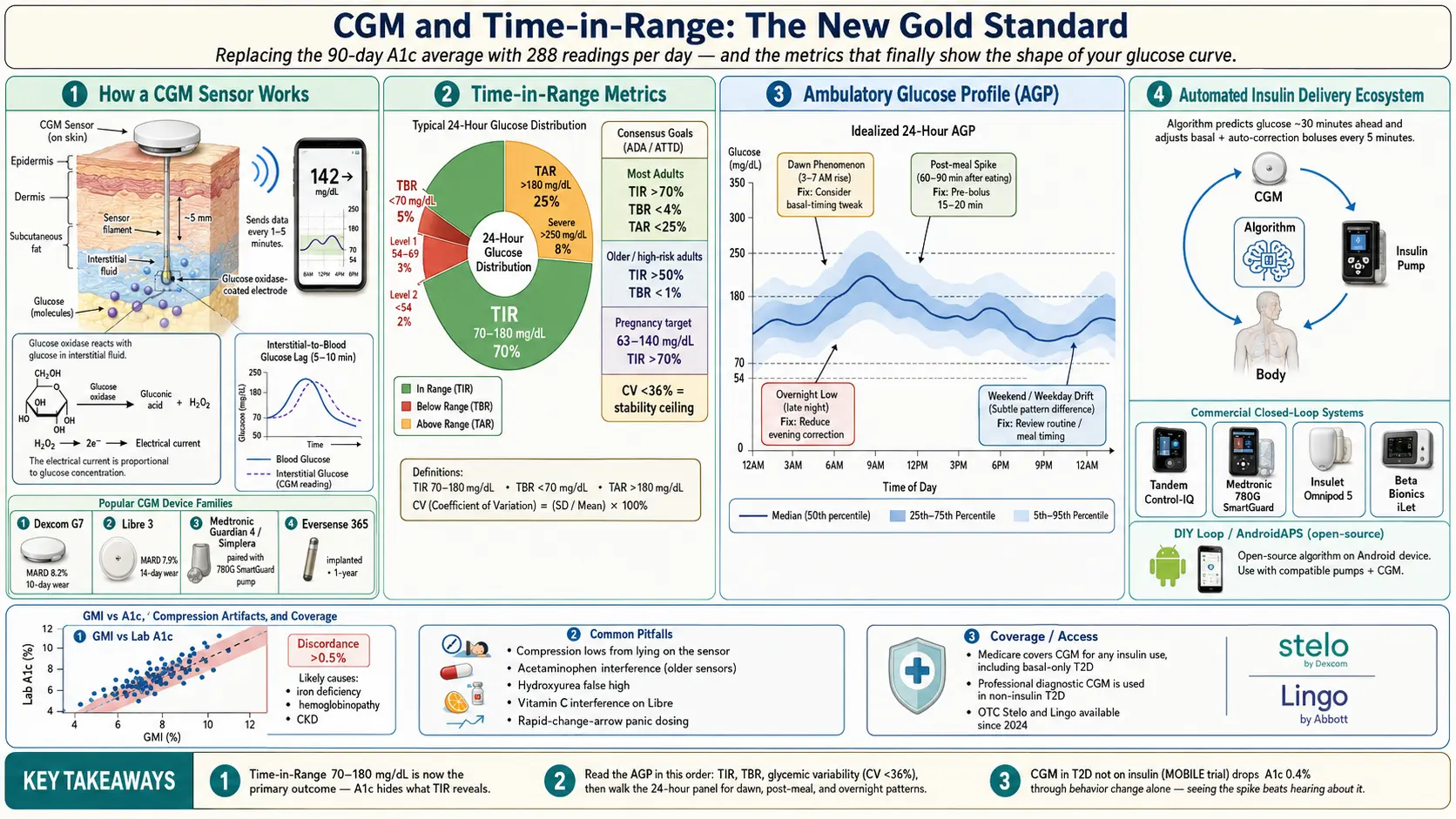
Table of Contents
- Why CGM Changed Everything
- Who Qualifies — Coverage in 2026
- The Four Device Families Compared
- Time-in-Range — The New Gold Standard
- Your TIR Targets by Situation
- GMI — Your CGM-Derived A1c
- Reading the Ambulatory Glucose Profile
- Automated Insulin Delivery Systems
- CGM in T2D Not on Insulin
- Common Pitfalls and Artifacts
- Skin, Adhesive, and Site Rotation
- Data Sharing and Remote Monitoring
- Insurance Tactics
- Your Weekly Interpretation Workflow
- Key Research Papers
- Connections
- Featured Videos
Why CGM Changed Everything
For most of the history of diabetes, glucose control was judged by one number collected four times a year: hemoglobin A1c. A1c is a three-month average — useful, but blind to everything that actually happens to you hour by hour. Two people can have identical A1c values of 7.0% and live in completely different metabolic worlds. One is flat, steady, rarely hypo, rarely spiking. The other swings wildly from 40 mg/dL at 3 a.m. to 280 after lunch, averaging out on paper but wrecking their vessels and nerves in practice.
Continuous glucose monitors (CGMs) replaced the guesswork. A small sensor filament sits in interstitial fluid just under the skin and reports a glucose reading every one to five minutes, day and night, for the life of the sensor. Instead of four fingersticks a day you get 288 readings per day, every day, on your phone. That changed what was possible to ask of diabetes care — and what is now expected of it.
CGM became the standard of care for type 1 diabetes (T1D) in the 2017 American Diabetes Association Standards, which explicitly recommended CGM use for any adult or child with T1D meeting glycemic targets or not. Since then coverage has expanded rapidly to type 2 diabetes (T2D) on insulin, and the frontier is now T2D not on insulin. Wearing a CGM is no longer an enthusiast's choice; for most people on insulin it is the minimum standard.
Who Qualifies — Coverage in 2026
The single biggest change in the last five years is who pays. Medicare expanded CGM coverage in 2022 to include anyone on any insulin regimen — not just multiple daily injections, not just T1D. If you take one dose of basal insulin at bedtime for T2D, you now qualify. In 2023 CMS began piloting coverage for certain non-insulin-treated T2D with documented hypoglycemia, and through 2024–2025 most commercial insurers quietly followed.
The practical rules today for most U.S. patients:
- T1D, any age — universally covered, no prior-authorization fight.
- T2D on any insulin — covered by Medicare and almost all commercial plans. One documented insulin prescription in the chart is enough.
- T2D on sulfonylureas or glinides with hypoglycemia history — increasingly covered with a prior-auth letter citing hypoglycemic events.
- T2D, diet-controlled or on metformin/GLP-1/SGLT2 only — spotty coverage. Options: cash pay (see the device section), a professional "diagnostic CGM" worn for 10–14 days through an endocrinologist (often covered as a procedure), or an over-the-counter sensor. Dexcom Stelo and Abbott Lingo launched OTC in 2024 explicitly for this population. For a deeper discussion of non-diabetic use, see the sibling article CGM for Non-Diabetics.
- Pregnancy with T1D or insulin-treated GDM — covered and strongly recommended. See also Gestational Diabetes.
CGM is billed either as Durable Medical Equipment (DME) through a supplier or as a pharmacy benefit at a retail pharmacy. The pharmacy channel is almost always faster and cheaper in 2026 — ask your prescriber to send it to your regular pharmacy first, and fall back to DME only if the pharmacy benefit is denied.
The Four Device Families Compared
Four CGM platforms cover essentially the entire U.S. market. Each has trade-offs in accuracy, wear time, calibration, pump integration, and cost.
Dexcom G7
The G7 sensor is a compact all-in-one disc worn on the upper arm or abdomen. 10-day wear, 30-minute warmup (the shortest on the market), factory calibrated (no fingersticks required), and an overall mean absolute relative difference (MARD) of 8.2% in adult trials — meaning on average the sensor reading is within 8.2% of a venous lab value. G7 sends data to an iPhone or Android phone, an Apple Watch via the new Direct-to-Watch feature (no phone needed in range), and integrates with Tandem t:slim X2 and Beta Bionics iLet pumps for closed-loop insulin delivery. Customizable high/low alerts, urgent-low-soon predictive alerts, and sharing with up to ten followers.
Freestyle Libre 3 and Libre 3 Plus
The smallest sensor on the market (roughly the size of two stacked pennies), worn on the upper arm. 14-day wear (Libre 3 Plus extends to 15 days), one-hour warmup, no calibration, and the best published accuracy of any over-the-counter-class sensor at MARD 7.9%. Real-time readings stream to phone every minute. Integrates with the Insulet Omnipod 5 closed-loop system. Generally the cheapest at retail cash price, and the simplest to start for a new user. Alerts are less customizable than Dexcom, and the follower ecosystem (LibreLinkUp) is more limited.
Medtronic Guardian 4 and Simplera
Medtronic's current sensor pairs tightly with their own pump ecosystem. Guardian 4 pairs with the MiniMed 780G pump running SmartGuard automated insulin delivery — the only system on the market with fully automatic correction boluses for post-meal highs. Guardian 4 eliminated the old fingerstick calibrations of earlier Medtronic sensors. Simplera Sync is Medtronic's next-generation disposable, all-in-one sensor with no calibration, shorter warmup, and simpler insertion. Choose Medtronic primarily if you want the 780G's aggressive auto-correction behavior; for pure sensor performance, G7 and Libre 3 lead.
Eversense 365
The outlier — a fully implantable sensor placed under the skin of the upper arm by a clinician in a brief office procedure. One sensor lasts 365 days. A removable transmitter is worn on top of the skin over the sensor site and is charged for about 15 minutes daily. The transmitter can be removed (for showers, swimming, sports) without losing the sensor underneath. Eversense is excellent for people with adhesive allergies or frequent sensor-adhesion failures and for those who simply do not want to change a sensor every one to two weeks. Trade-offs: an in-office insertion and removal each year, limited pump integration, and higher upfront logistics.
Time-in-Range — The New Gold Standard
A1c tells you an average but not the shape of your glucose day. CGM made it possible to measure that shape directly, and in 2019 an international consensus group led by Tadej Battelino formalized a new metric set that has now replaced A1c as the primary outcome in most CGM-enabled diabetes care.
The core metric is Time-in-Range (TIR): the percentage of CGM readings in the target glucose band of 70–180 mg/dL (3.9–10.0 mmol/L). Around TIR sit two companion metrics:
- Time Below Range (TBR) — percent of readings under 70 mg/dL, subdivided into Level 1 (54–69, alert) and Level 2 (<54, clinically significant hypoglycemia).
- Time Above Range (TAR) — percent over 180 mg/dL, subdivided at 250 mg/dL for severe hyperglycemia.
- Glycemic variability — expressed as the coefficient of variation (CV). Below 36% is considered stable; above 36% is labile.
Why TIR matters more than a single A1c: multiple studies since 2019 have shown that for each 10-percentage-point rise in TIR, rates of retinopathy progression fall measurably, microalbuminuria risk drops, and cardiovascular event rates trend down. A TIR of 70% corresponds roughly to an A1c of 7.0%, but with much better granularity on whether that 7.0% was achieved safely or on the back of frequent overnight lows.
Your TIR Targets by Situation
The Battelino consensus and its updates set the following targets. Your individual numbers should be negotiated with your endocrinologist, but these are the starting points.
- Most adults with T1D or T2D — TIR >70% (that is 16 hours 48 minutes per day in range), TBR <4% total and <1% for Level 2 (<54), TAR <25% total and <5% for >250.
- Older adults or high-risk patients (frailty, severe hypoglycemia history, short life expectancy) — TIR >50%, TBR <1%. Looser ceilings, tighter floors. Avoiding hypoglycemia matters more than chasing lower averages.
- Pregnancy with T1D — target range tightens to 63–140 mg/dL, with TIR >70%, TBR <4%, TAR <25%. This narrower band reflects tighter fetal-safety thresholds.
- Pregnancy with T2D or GDM on insulin — same 63–140 range is widely used though evidence is less complete.
- Glycemic variability — coefficient of variation <36% is the stability goal for everyone.
These targets are averages over at least 14 days of CGM wear — the minimum duration shown to represent a 90-day A1c window reliably.
GMI — Your CGM-Derived A1c
The Glucose Management Indicator (GMI) is a number your CGM report will show next to your TIR. GMI is calculated from your 14-day mean glucose using a formula validated by Roy Beck and colleagues in 2018. It estimates what your A1c would be if lab-measured right now, based on your CGM data.
GMI usually agrees with the lab A1c within a few tenths of a percent. When they disagree by more than 0.5%, the discordance is clinically meaningful and usually points to something altering the lifespan of your red blood cells or the glycation rate on hemoglobin — iron-deficiency anemia (falsely high A1c), recent blood transfusion, hemoglobinopathies (sickle trait, thalassemia), chronic kidney disease, or supplements and medications that affect red cell turnover. If your GMI reads 6.8% on a solid 14-day CGM record but the lab keeps returning A1c 8.5%, your lab A1c is the unreliable number, not the CGM. See the sibling article A1c, Fructosamine, and Glycation Markers for a full discussion of when each marker lies.
Reading the Ambulatory Glucose Profile
The Ambulatory Glucose Profile (AGP) is the one-page report most endocrinologists will hand you at every visit. It overlays your last 14 days of CGM readings into a single 24-hour panel, collapsing every day's data onto the same timeline. Five curves stack on top of each other: the median, the 25th–75th percentile band, and the 5th–95th percentile band. Wider bands mean more day-to-day variability at that hour.
Patterns worth learning to spot:
- Dawn phenomenon — the median line climbs between 3 and 7 a.m. even without eating. Driven by overnight cortisol and growth hormone. Fix: adjust basal insulin timing, add a small basal bump, or use an AID system with overnight auto-correction.
- Post-meal spikes — sharp peaks 60–90 minutes after meals. Fix: pre-bolus 15–20 minutes before eating, reduce rapid carbs, or increase the insulin-to-carb ratio.
- Overnight lows — median or lower-band dipping under 70 between midnight and 4 a.m. Fix: reduce basal, reduce evening correction doses, add a bedtime snack. Most dangerous pattern — treat first.
- Weekend vs weekday difference — look at the calendar heat-map page; many patients see markedly different curves Saturday/Sunday. Points to activity, meal timing, or alcohol patterns.
- Stress and sick-day arcs — broad upward drift in the whole band during illness, steroids, or emotional stress. Expected; the fix is a temporary basal increase and more frequent checks, not panic.
Read the AGP in this order every time: TIR percentage first, TBR second (any hypo is urgent), glycemic variability third, then walk the 24-hour panel looking for the four patterns above. Five minutes of structured reading beats an hour of staring at every daily trace.
Automated Insulin Delivery Systems
CGM becomes far more powerful when it controls an insulin pump directly. Four commercial Automated Insulin Delivery (AID) systems dominate the U.S. market in 2026, plus one thriving open-source community.
- Tandem Control-IQ (t:slim X2 pump + Dexcom G6/G7) — predicts glucose 30 minutes ahead and modulates basal delivery every five minutes. Delivers automatic correction boluses for projected highs. Sleep Mode tightens overnight targets. Basal-IQ is the simpler predecessor algorithm on the same pump that only suspends insulin to prevent lows — still available for users who prefer minimal automation.
- Medtronic 780G SmartGuard (780G pump + Guardian 4 / Simplera) — the most aggressive automation. Delivers auto-corrections every five minutes with an adjustable target as low as 100 mg/dL. Known for highest TIR in head-to-head studies but also the steepest learning curve.
- Omnipod 5 SmartAdjust (tubeless Omnipod patch pump + Dexcom G6/G7 or Libre 2 Plus) — the only tubeless AID. Algorithm runs on the pod itself. Very friendly for new pumpers and children. Slightly more conservative than 780G.
- Beta Bionics iLet Bionic Pancreas (iLet pump + Dexcom G6/G7) — the simplest AID in concept. No carb-counting required. The user announces meals qualitatively (usual, less, more) and the algorithm handles the rest. Initial programming requires only body weight. Best for patients who struggle with carb math or are burned out on traditional dosing.
- DIY loops — Loop (iPhone) and AndroidAPS — open-source community-built algorithms that drive compatible pumps. Not FDA-approved as systems (though the iAPS fork and Tidepool Loop have been submitted and cleared in parts). Active forums, steep setup learning curve, powerful customization. Many long-term T1D users prefer them to commercial systems.
For a deeper dive on insulin types, pharmacokinetics, and pre-bolus timing that underpins all of these systems, see Insulin Types and Dosing Strategies.
CGM in T2D Not on Insulin
The fastest-growing CGM population is T2D patients on metformin, GLP-1 agonists, or diet alone. The case for CGM in this group is motivational and diagnostic rather than dosing-driven.
The MOBILE trial (Martens 2021, JAMA) randomized T2D adults on basal insulin to CGM or fingersticks and found an A1c reduction of 0.4% favoring CGM at eight months — meaningful even with zero mealtime insulin to adjust. The Beck 2019 DIAMOND-T2D results showed similar effects. The mechanism appears to be behavior change: seeing a post-dinner spike of 260 mg/dL in real time changes what you eat at dinner tomorrow in a way that the abstract idea of "high blood sugar" does not.
Three practical options if you are T2D not on insulin:
- Professional diagnostic CGM — your endocrinologist places a 10- to 14-day sensor (often Libre Pro or equivalent) and reviews the data with you at a follow-up. Usually billed as a CPT procedure and covered even when personal-wear CGM is not. A single cycle can reveal dawn phenomenon, post-meal spikes to specific foods, and exercise patterns that reshape your eating for years.
- Personal CGM for motivation — if cash-pay is an option, 14 days of Libre 3 or a month of Dexcom Stelo (OTC, no prescription) costs roughly $50–$90. Worth running every six months even without insurance coverage.
- Remission-focused CGM — for patients attempting to reverse T2D with weight loss, low-carb nutrition, or bariatric surgery, CGM is the most direct feedback tool on which foods, sleep patterns, and activities move glucose. See Remission and Reversal Strategies.
Common Pitfalls and Artifacts
CGMs are not perfect, and knowing their failure modes prevents dangerous overreactions.
- Compression lows — lying on the sensor overnight compresses the interstitial fluid and causes falsely low readings, often triggering urgent-low alarms at 2 a.m. If the low vanishes as soon as you roll over and the reading is not corroborated by symptoms, it was likely compression. Fix: do not bolus a fast carb based on a single compression low — confirm with a fingerstick first if in doubt.
- Acetaminophen (Tylenol) interference — older Dexcom and Libre sensors read falsely high with acetaminophen. Dexcom G6, G7, and Libre 3 have largely eliminated this at standard doses, but high doses (>1 g in a single dose, or >4 g/day) can still skew readings upward on any sensor.
- Hydroxyurea — used in sickle cell disease and some cancers; causes falsely high CGM readings on all current sensors. Patients on hydroxyurea should continue fingersticks for dosing decisions.
- Vitamin C — doses above about 500 mg/day can elevate Libre sensor readings in some users; less effect on Dexcom. Worth knowing if you are on high-dose IV C or large oral supplementation.
- First 24 hours of a new sensor — accuracy improves after the first day. Take wide swings during hour 2 of a new sensor with a grain of salt.
- Rapid-change trend arrows during active dosing — do not stack correction doses on a rapidly rising arrow immediately after eating. The arrow reflects the carbs you already have on board; the insulin from your pre-meal bolus is still absorbing. Trust the plan; panic-dosing is a common cause of overnight hypoglycemia. See Hypoglycemia Awareness and Prevention.
- Interstitial vs blood glucose lag — CGM measures interstitial fluid, which trails blood glucose by roughly 5–10 minutes. During rapid changes (post-meal, post-exercise, treating a low) the CGM is behind reality. A treated low may still show 58 on the CGM while blood glucose has already climbed to 110.
Skin, Adhesive, and Site Rotation
Skin reactions are the most common reason people abandon CGM. The fix is rarely "give up"; it is usually a better adhesive strategy.
- Rotate sites every sensor — upper arm, opposite arm, abdomen, upper buttock. Never reinsert on an irritated or recently-used spot.
- Skin Tac — a tacky liquid applied before the sensor improves adhesion for swimmers, sweaty summers, and high-activity users.
- Flector patch or a hydrocortisone wipe — applied to the skin 10 minutes before sensor insertion, then wiped off, reduces contact dermatitis from the adhesive. Confirm with your dermatologist if you have recurrent rashes.
- Hypafix or Simpatch overpatches — precut adhesive frames that go over the sensor add a second anchor; essential if your sensor keeps peeling off mid-wear.
- Skin barrier wipes (Cavilon, Skin-Prep) — a thin barrier film between skin and adhesive can prevent the isobornyl acrylate allergy that causes most Libre and Dexcom rashes.
- Persistent allergy — if rashes recur with every site despite the above, ask about Eversense 365, which has no skin adhesive at all once the sensor is implanted.
Data Sharing and Remote Monitoring
CGM data can be streamed in real time to family, caregivers, and clinicians.
- Dexcom Follow — the wearer invites up to 10 followers; each follower sees live glucose, trend arrows, and configurable alerts. Essential for parents of T1D children and spouses of people with hypoglycemia unawareness.
- LibreLinkUp — Abbott's equivalent for Libre sensors. Similar functionality; up to 20 followers per account.
- Nightscout — an open-source, self-hosted CGM data platform. The user runs their own cloud instance and shares a URL. Integrates with nearly every AID and custom visualization. Popular in the T1D parent community.
- Clarity (Dexcom) and LibreView (Abbott) — clinic-facing portals; your endocrinologist can view AGP reports with your permission. Uploading before appointments makes visits dramatically more productive.
Insurance Tactics
If a claim is denied, it is almost never the final answer. A few practical levers:
- Pharmacy benefit first. Ask your prescriber to send the CGM prescription to your pharmacy, not a DME supplier. Pharmacy-billed CGM is faster, cheaper, and has lower out-of-pocket copays for most insured patients.
- Document hypoglycemia. A letter from your prescriber listing at least one hypoglycemic event (including measured or symptomatic lows, documented in the chart) is usually sufficient for prior authorization in borderline T2D cases.
- Appeal with the Battelino consensus. Insurance denials occasionally cite "not medically necessary." A short appeal letter citing the 2019 international consensus and the 2017 ADA standards usually succeeds on first appeal.
- Manufacturer copay programs. Dexcom, Abbott, and Medtronic all run commercial patient-assistance programs that can bring copays to $0–$50 per month for insured patients under a means threshold.
- OTC fallbacks. Dexcom Stelo and Abbott Lingo are over-the-counter and cash-pay; they are not FDA-indicated for insulin dosing but provide the same glucose data if cost is the only barrier.
- Medicare DME criteria — a diabetes diagnosis + any insulin prescription + quarterly visits with a prescriber managing insulin. Document the quarterly visits; they are the most commonly missed criterion.
Your Weekly Interpretation Workflow
A CGM without a review habit is just expensive wallpaper. The following 15-minute weekly ritual is what separates the patients who drop their A1c by a full point from the ones whose numbers stay flat.
- Open the 14-day AGP in Clarity, LibreView, or your pump app. Read TIR, TBR, TAR, and GMI first — in that order.
- Pick the single worst pattern. Overnight lows? Post-dinner spikes? 3 a.m. dawn rise? Only one. Fixing one thing at a time is how behavior sticks.
- Make exactly one adjustment. A small basal rate change, a meal pre-bolus timing tweak, a bedtime-snack removal, moving a medication dose. Not three changes; one.
- Tag the event in your CGM app. Most apps let you mark meals, exercise, stress, illness. Consistent tagging makes the next week's AGP tell a clearer story.
- Look at trend arrows in real time, but do not panic-dose. A double-up arrow 45 minutes after a pre-bolus meal is expected — the insulin has not fully kicked in yet. A double-up arrow two hours later after the bolus should have peaked is a real signal. The time since your last bolus matters as much as the arrow itself.
- Review with your endocrinologist quarterly, not just the A1c. Bring the AGP PDF; ask your provider to walk the 24-hour curve with you.
CGM is the most powerful tool diabetes has produced in a generation, but it rewards attention. Fifteen minutes a week of structured review consistently out-performs hours of staring at live numbers.
Key Research Papers
Foundational peer-reviewed work on continuous glucose monitoring, time-in-range targets, and the GMI-vs-A1c relationship. Each citation links to the full text via DOI.
- Battelino T, Danne T, Bergenstal RM, et al. Clinical Targets for Continuous Glucose Monitoring Data Interpretation: Recommendations from the International Consensus on Time in Range. Diabetes Care. 2019;42(8):1593–1603.
- Beck RW, Bergenstal RM, Riddlesworth TD, et al. Validation of Time in Range as an Outcome Measure for Diabetes Clinical Trials. Diabetes Care. 2019;42(3):400–405.
- Beck RW, Riddlesworth TD, Ruedy K, et al. Effect of Continuous Glucose Monitoring on Glycemic Control in Adults with Type 1 Diabetes Using Insulin Injections (DIAMOND Type 1). JAMA. 2017;317(4):371–378.
- Beck RW, Riddlesworth TD, Ruedy K, et al. Continuous Glucose Monitoring versus Usual Care in Patients with Type 2 Diabetes Receiving Multiple Daily Insulin Injections (DIAMOND Type 2). Annals of Internal Medicine. 2017;167(6):365–374.
- Bergenstal RM, Beck RW, Close KL, et al. Glucose Management Indicator (GMI): A New Term for Estimating A1C from Continuous Glucose Monitoring. Diabetes Care. 2018;41(11):2275–2280.
- Tamborlane WV, Beck RW, Bode BW, et al. Continuous Glucose Monitoring and Intensive Treatment of Type 1 Diabetes (JDRF CGM Trial). New England Journal of Medicine. 2008;359(14):1464–1476.
- The Diabetes Control and Complications Trial Research Group. The Effect of Intensive Treatment of Diabetes on the Development and Progression of Long-Term Complications in Insulin-Dependent Diabetes Mellitus (DCCT). New England Journal of Medicine. 1993;329(14):977–986.
- UK Prospective Diabetes Study (UKPDS) Group. Intensive Blood-Glucose Control with Sulphonylureas or Insulin Compared with Conventional Treatment and Risk of Complications in Patients with Type 2 Diabetes (UKPDS 33). Lancet. 1998;352(9131):837–853.
- American Diabetes Association Professional Practice Committee. Standards of Care in Diabetes—2024. Diabetes Care. 2024;47(Suppl 1):S1–S321.
- Brown SA, Kovatchev BP, Raghinaru D, et al. Six-Month Randomized, Multicenter Trial of Closed-Loop Control in Type 1 Diabetes (iDCL). New England Journal of Medicine. 2019;381(18):1707–1717.
- Bergenstal RM, Garg S, Weinzimer SA, et al. Safety of a Hybrid Closed-Loop Insulin Delivery System in Patients with Type 1 Diabetes. JAMA. 2016;316(13):1407–1408.
Live PubMed Searches
Each link opens a live PubMed query so results stay current as new papers are indexed.
- Continuous glucose monitoring and time-in-range outcomes
- Dexcom G7 accuracy and MARD
- Freestyle Libre 3 accuracy studies
- Automated insulin delivery and closed-loop systems
- GMI and lab A1c discordance
- Ambulatory glucose profile interpretation
- CGM in non-insulin-treated type 2 diabetes
- CGM and time-in-range in pregnancy
Connections
- Diabetes Overview
- Hypoglycemia Awareness and Prevention
- Remission and Reversal Strategies
- Insulin Types and Dosing Strategies
- A1c, Fructosamine, and Glycation Markers
- Cardio-Renal Protection: SGLT2 and GLP-1
- Complications: Retinopathy, Nephropathy, Neuropathy
- Type 1 vs Type 2 vs LADA and MODY
- Diabetic Foot Care and Ulcer Prevention
- Insulin Resistance
- Gestational Diabetes
- Continuous Glucose Monitor
- Hemoglobin A1C Test
- Blood Sugar
- Kidney Disease
- Metabolic Syndrome