Diabetic Foot Care & Ulcer Prevention
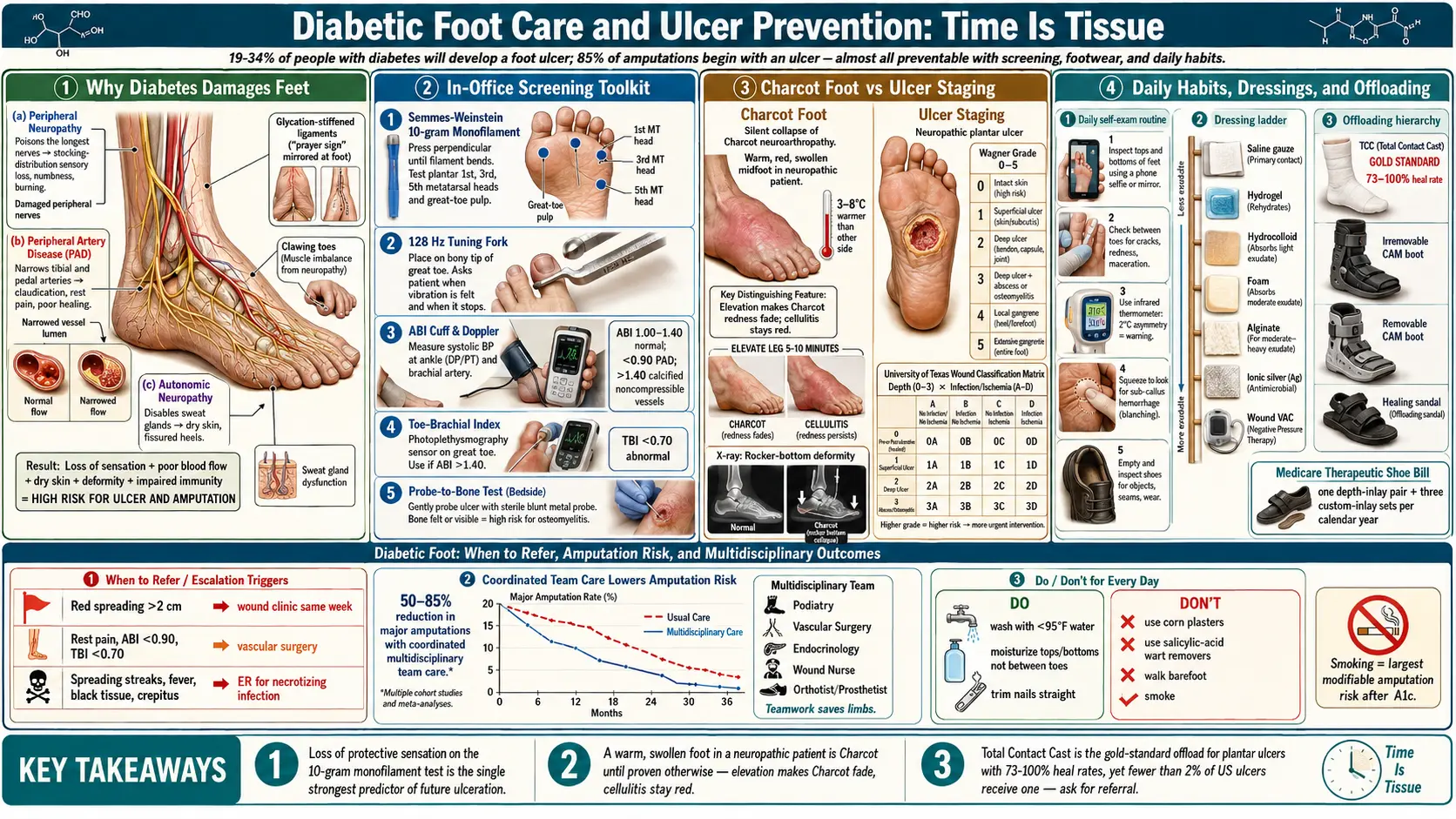
If you have diabetes, your feet deserve the same respect as your eyes, your kidneys, and your heart. About 19–34% of people with diabetes will develop a foot ulcer in their lifetime, and roughly 85% of diabetes-related amputations begin with an ulcer. Those numbers sound terrifying, but the other side of the statistic is the good news: the overwhelming majority of diabetic foot ulcers — and the amputations that follow — are preventable with consistent, unglamorous daily habits and the right screening visits.
Two problems drive the damage. First, peripheral neuropathy quietly erases the warning system. A pebble in your shoe, a too-tight seam, or a minor cut stops hurting long before it stops causing harm, so wounds develop silently. Second, peripheral artery disease (PAD) narrows the arteries that would normally flood a wound with oxygen, white cells, and antibiotics — so once a wound forms, it heals slowly or not at all. Put neuropathy and PAD together and a blister becomes an ulcer, an ulcer becomes an infection, and an infection threatens the limb.
This article walks through the whole chain of defense: how feet are screened, what to check at home every single day, which shoes and socks actually help, how Charcot foot hides in plain sight, how ulcers are staged and dressed, when to call podiatry versus the ER, and the specific habits that the IWGDF (International Working Group on the Diabetic Foot) 2023 guidelines say reduce amputations most.
Table of Contents
- Why Diabetes Damages Feet
- In-Office Screening: Monofilament, Vibration, ABI, TBI
- The Daily Self-Exam Routine
- Footwear, Orthotics, and Medicare Coverage
- Charcot Foot: The Silent Collapse
- Ulcer Staging: Wagner and University of Texas
- Dressings, Silver, and Offloading (Total Contact Casts)
- Infection Signs and When to Refer
- Nail and Skin Care: Dos and Don'ts
- Key Research Papers
- PubMed Topic Searches
- Connections
- Featured Videos
Why Diabetes Damages Feet
Three slow-motion processes converge below the ankle.
Peripheral neuropathy. High blood glucose, over years, poisons the longest nerves in the body — and the longest nerves are the ones running down to your toes. The damage is typically a stocking-distribution sensory loss: numbness, tingling, or burning starting at the toes and creeping up. By the time the numbness is obvious, small-fiber protective sensation is often already gone. That is the sensation that makes you yank your foot away from a hot bath, shift in your chair when a seam digs in, or notice a tack on the carpet. Without it, your foot takes injuries your brain never hears about. For the broader nerve story, see the peripheral neuropathy page.
Peripheral artery disease (PAD). The same high glucose, plus the cholesterol, blood pressure, and smoking patterns that cluster with diabetes, narrows and stiffens arteries. In the leg this shows up as PAD — reduced blood flow through the calf, ankle, and foot. Early PAD is silent; later you get calf pain with walking that stops with rest (claudication), cold feet, hair loss over the shins, and shiny skin. Advanced PAD causes rest pain and non-healing wounds. Blood flow is what delivers healing.
Autonomic neuropathy and skin changes. Diabetic nerves also control sweat glands and small vessels. When those fail, skin becomes dry, thick, and cracked, especially at the heels. Cracks are doorways for bacteria. Autonomic neuropathy also alters arteriovenous shunting in the foot, giving some patients a warm, dry, bounding-pulse foot that still somehow does not heal — blood is going through but not where it needs to go.
Add in reduced joint mobility from glycation of ligaments (the "prayer sign" at the hands mirrors a stiff ankle), abnormal pressure points from clawing toes, and immune dysfunction from chronic hyperglycemia, and the foot becomes a setup waiting for a trigger.
In-Office Screening: Monofilament, Vibration, ABI, TBI
Every person with diabetes should have a comprehensive foot exam at least once a year; more often if you already have neuropathy, PAD, foot deformity, or a prior ulcer. Four tools do most of the work.
Semmes-Weinstein 10-gram monofilament. A calibrated nylon filament that buckles at exactly 10 grams of force. The examiner presses it against specific sites on the sole (typically the plantar surface of the first, third, and fifth metatarsal heads and the pulp of the great toe) while your eyes are closed. If you cannot feel the filament at even one site, you have lost protective sensation (LOPS). This single test is one of the strongest predictors of future ulceration. It takes ninety seconds and requires no equipment more sophisticated than a piece of plastic — if your doctor is not doing it, ask for it.
128-Hz tuning fork (vibration testing). Large-fiber nerve check. The examiner strikes the tuning fork and touches the stem to the bony tip of your great toe; you report when the buzzing stops. Comparing to the examiner's own perception (at the wrist, for example) gives a rough gradient. Abnormal vibration sense is another early neuropathy marker.
Ankle-Brachial Index (ABI). A blood-pressure cuff is placed at the ankle and a handheld Doppler picks up the posterior tibial and dorsalis pedis arteries. The ankle systolic pressure is divided by the higher of the two arm pressures. Normal is 1.00–1.40. Below 0.90 indicates PAD; below 0.50 is severe and warrants vascular referral. A score above 1.40 is also abnormal — it usually means the arteries are calcified and noncompressible (common in long-standing diabetes), which makes the ABI unreliable.
Toe-Brachial Index (TBI). When the ABI is falsely high because of calcified vessels, a small cuff around the great toe bypasses the problem (toe arteries rarely calcify). A TBI below 0.70 is abnormal; below 0.40 signals critical limb ischemia. Any diabetic patient with a non-healing wound, ischemic-sounding rest pain, or ABI above 1.40 should have a TBI.
Additional imaging — arterial duplex ultrasound, CT angiography, or formal angiography — belongs to the vascular surgeon once screening is abnormal.
The Daily Self-Exam Routine
Ten seconds, every night, ideally after a shower when the skin is clean and dry. Attach it to an existing habit so you never forget.
- Look at the tops and bottoms. If you cannot reach or see your soles, use a handheld mirror or the inside of a dinner plate propped on the floor. A phone camera on selfie mode with the flash on works beautifully. You are looking for redness, blisters, cuts, cracks, calluses, corns, scrapes, ingrown nail edges, or changes in color or shape.
- Check between every toe. Interdigital spaces are where maceration, athlete's foot, and unnoticed cuts hide. Dry them thoroughly.
- Feel for temperature differences. A spot that is noticeably warmer than its mirror image on the other foot is an early sign of inflammation — brewing ulcer, Charcot foot, or infection. A 2°C (4°F) difference between matched spots predicts ulceration weeks ahead. Inexpensive infrared skin thermometers (under $30) make this quantitative; products such as SmartMat or Podimetrics do it automatically.
- Squeeze gently. A soft, boggy area under a callus may hide a sub-callus hemorrhage or an early ulcer.
- Empty your shoes before putting them on. Every time. A pebble, a bent insole, a stray thumbtack — your numb foot will not warn you.
If anything new shows up — a red spot that does not fade in 24 hours, a blister, a crack that bleeds, new warmth or swelling — call your primary care office or podiatrist the same week, not eventually.
Footwear, Orthotics, and Medicare Coverage
Your shoes are medical equipment. Treat them that way.
What a good diabetic shoe has: a wide, deep toe box (your toes should wiggle freely); soft, seamless uppers that do not dig in; a rocker or rigid rocker sole that reduces forefoot pressure; a firm heel counter; and removable insoles so a custom orthotic can be swapped in. Brands routinely prescribed include Dr. Comfort, Orthofeet, Apex, Drew, and Propet. Avoid anything pointed, anything with thin leather over a bony prominence, and anything you "break in" — if it does not fit on day one, it will ulcerate you by day ten.
Socks matter too. White or light-colored (so blood or drainage shows up), seamless, non-binding at the ankle, moisture-wicking. Change daily. Never wear darned or mended socks — the lump is a pressure point.
Custom orthotics redistribute pressure away from ulcer-prone spots (first and fifth metatarsal heads, the tip of a clawed toe, a previous ulcer scar). Total-contact custom inserts are molded from a cast or scan of your foot; prefabricated multi-density inserts are cheaper but less precise.
Medicare's Therapeutic Shoe Bill (the "diabetic shoe benefit") covers one pair of depth-inlay shoes plus three pairs of custom-molded multi-density inserts each calendar year for Medicare Part B enrollees with diabetes who have at least one of: previous amputation, history of foot ulcer, pre-ulcerative callus, peripheral neuropathy with callus formation, foot deformity, or poor circulation. Your primary-care physician must certify the diabetes diagnosis and need; a podiatrist, orthotist, or pedorthist dispenses the shoes. Most commercial insurers mirror this benefit. Ask — many eligible patients have no idea the benefit exists.
Charcot Foot: The Silent Collapse
Charcot neuroarthropathy is the sneakiest diabetic foot complication. Bones and joints in a numb foot weaken, fracture, and collapse — often without the person feeling anything more than mild warmth or swelling. The midfoot arch can cave downward ("rocker-bottom foot"), turning a flat plantar surface into a convex one that ulcerates under every step.
Classic early presentation: a warm, red, swollen foot after a minor injury or even no remembered injury, in someone with long-standing neuropathy. The temperature difference between the two feet is typically 3–8°C. It is frequently misdiagnosed as cellulitis, gout, or a sprain. The critical distinguishing feature: elevating the foot above heart level for several minutes makes cellulitis stay red but makes Charcot redness fade. X-rays may look normal for the first few weeks; MRI catches it earlier.
Treatment is immediate non-weight-bearing immobilization, usually in a total contact cast, for two to four months or until the skin-temperature difference normalizes. Walking on an acute Charcot foot is how a slightly swollen ankle becomes a permanent rocker-bottom deformity. If you have neuropathy and notice a hot, swollen foot, get it imaged and offloaded the same week.
Ulcer Staging: Wagner and University of Texas
Once an ulcer exists, clinicians classify it to guide treatment and predict outcome. Two systems dominate.
Wagner Classification (depth-based, the older system):
- Grade 0 — intact skin at risk (callus, deformity).
- Grade 1 — superficial ulcer, full skin thickness, no deeper involvement.
- Grade 2 — deep ulcer penetrating to tendon, capsule, or bone, without abscess or osteomyelitis.
- Grade 3 — deep ulcer with abscess, osteomyelitis, or septic joint.
- Grade 4 — localized gangrene (toes or forefoot).
- Grade 5 — gangrene of the entire foot.
University of Texas (UT) System adds two axes — depth (0–3) and the presence of infection and/or ischemia (A = neither, B = infected, C = ischemic, D = both). A UT 2-D wound (penetrating to tendon, infected, and ischemic) has dramatically worse outcomes than a UT 1-A (superficial, clean, well-perfused) and is treated far more aggressively.
The "probe to bone" test is a simple bedside exam: a sterile blunt probe gently inserted into the wound. If it touches bone, osteomyelitis is likely and an MRI plus bone biopsy is warranted.
Dressings, Silver, and Offloading (Total Contact Casts)
Two parallel jobs: keep the wound bed healthy and keep weight off it.
Dressings. The old "dry the wound out with hydrogen peroxide and leave it open to air" approach is obsolete; it kills new granulation tissue. Modern diabetic wound care keeps the wound moist but not wet.
- Saline-moistened gauze — cheapest, still appropriate for clean superficial wounds with minimal drainage; changed once or twice daily.
- Hydrogels (Curasol, Solosite, IntraSite Gel) — add moisture to dry wounds with scant drainage; useful on a dry, eschar-covered ulcer.
- Hydrocolloids (DuoDERM, Comfeel) — form a gel over the wound; for clean, low-drainage wounds. Avoid on infected wounds.
- Foams (Mepilex, Allevyn) — absorbent, useful for moderate-to-heavy drainage.
- Alginates (Kaltostat, Sorbsan) — highly absorbent, derived from seaweed, good for heavily draining wounds.
- Silver dressings (Acticoat, Aquacel Ag, Mepilex Ag) — ionic silver is broad-spectrum antimicrobial. Reasonable for locally infected or critically colonized wounds; evidence for routine use on clean wounds is weaker, and costs are higher. Stop once the wound is clean.
- Negative pressure wound therapy (wound VAC) — a sealed dressing under suction; accelerates granulation in deep or post-surgical wounds.
Offloading is often more important than what you put on the wound. You cannot heal a pressure wound while continuing to step on it.
- Total contact cast (TCC) — the gold standard for plantar forefoot ulcers. A fiberglass cast molded to the entire lower leg and foot redistributes pressure, immobilizes the joint, and forces adherence (you cannot take it off). Healing rates of 73–100% in trials; yet fewer than 2% of U.S. ulcers receive one, largely because casting takes time and skill. If you have a plantar ulcer and your clinic cannot cast, ask for a referral to one that can.
- Removable cast walker / CAM boot — similar biomechanics but the patient can remove it. Heal rates drop because people take it off. "Instant total contact casts" (a CAM boot wrapped with a layer of fiberglass so it cannot be removed) close that adherence gap.
- Healing sandals, felted foam, half-shoes — lesser options for patients who cannot tolerate casts.
Infection Signs and When to Refer
Any of the following means the ulcer needs an urgent clinician visit, not a watch-and-wait:
- Increasing pain in a previously painless wound.
- Redness spreading more than 2 cm beyond the wound edge.
- Warmth, swelling, or firmness around the wound.
- Foul odor or change in drainage color (cloudy, yellow-green, grey).
- Fever, chills, elevated blood sugars without another explanation — a jump in glucose is often the first systemic sign of foot infection.
- Exposed bone, tendon, or joint capsule visible in the wound.
Who to call:
- Podiatrist or wound-care clinic for any new ulcer, non-healing callus, or suspicious red spot. Same-week appointment.
- Vascular surgeon if ABI < 0.90, TBI < 0.70, rest pain, or non-healing wound despite offloading.
- Emergency department for spreading redness, fever, streaks running up the leg, severe pain, new black tissue, or crepitus (gas under the skin). Necrotizing infection is a surgical emergency, not a next-day appointment.
The IDSA/IWGDF diabetic foot infection classification grades infections mild (local, ≤ 2 cm surrounding cellulitis), moderate (> 2 cm, deeper structures), or severe (systemic signs). Mild infections can often be managed with oral antibiotics (typically covering gram-positive staph/strep); moderate and severe require cultures, possibly IV antibiotics, imaging for osteomyelitis, and frequently surgical debridement. A simple ulcer probed to bone has a high enough pre-test probability of osteomyelitis that empirical coverage of bone infection is often started.
Amputation prevention, in numbers. Multidisciplinary foot-care teams (podiatry + vascular + endocrine + wound nurse + orthotist) have reduced major amputation rates by 50–85% in published cohorts. The single biggest lever is speed: ulcers seen within a week of onset heal faster and amputate less than ulcers that wait a month. The mantra in the field — "time is tissue" — is literal.
Nail and Skin Care: Dos and Don'ts
Do:
- Wash feet daily with lukewarm (not hot) water. Test with an elbow or a thermometer — under 95°F / 35°C.
- Dry thoroughly, especially between toes.
- Apply a plain moisturizer (CeraVe, Eucerin, Aquaphor) to the tops and bottoms, but not between the toes — trapped moisture macerates skin.
- Trim nails straight across, not curved into the corners. File sharp edges with an emery board.
- Wear shoes and socks at all times, even indoors. Especially on hot sand, hot pavement, and hotel-room carpets where a dropped pin disappears.
- Keep your A1c in range, your blood pressure controlled, your lipids treated, and — above all else — stop smoking. Smoking is the single most modifiable amputation risk factor after glucose itself.
Don't:
- Don't use corn plasters, callus-removing pads, or salicylic acid wart removers. They contain acids that cause ulcers on neuropathic feet.
- Don't cut your own corns or calluses with a razor blade or pocket knife. See a podiatrist.
- Don't soak feet for long periods. Prolonged soaking macerates skin.
- Don't use heating pads, electric blankets, hot water bottles, or radiators to warm cold feet — neuropathic burns are a classic cause of ulcers.
- Don't walk barefoot, ever.
- Don't ignore a blister, cut, or red spot "for a few days to see." Call the same week.
- Don't wait for pain. Neuropathic ulcers are often painless even when deep.
Key Research Papers
- Armstrong DG, Boulton AJM, Bus SA. Diabetic foot ulcers and their recurrence. N Engl J Med. 2017;376:2367–2375.
- Bus SA, et al. IWGDF Guidelines on the prevention and management of diabetes-related foot disease. Diabetes Metab Res Rev. 2023 (IWGDF 2023 Guidelines).
- Lipsky BA, et al. Guidelines on the diagnosis and treatment of foot infection in persons with diabetes (IWGDF/IDSA 2020). Diabetes Metab Res Rev. 2020.
- Boulton AJM, et al. Comprehensive foot examination and risk assessment. Diabetes Care. 2008;31:1679–1685.
- Total contact casting for plantar diabetic foot ulcers — PubMed systematic reviews.
- Charcot neuroarthropathy diagnosis and management — PubMed.
PubMed Topic Searches
- Diabetic foot ulcer prevention
- Semmes-Weinstein monofilament screening
- Ankle-brachial index and diabetic PAD
- Toe-brachial index in diabetes
- Therapeutic footwear and Medicare coverage
- Charcot foot management
- Wagner and UT ulcer classifications
- Silver dressings in diabetic foot ulcers
- Total contact cast offloading
- Multidisciplinary foot teams and amputation reduction
Connections
- Diabetes Overview
- Complications: Retinopathy, Nephropathy, Neuropathy
- Cardio-Renal Protection: SGLT2 and GLP-1
- Hypoglycemia Awareness and Prevention
- Insulin Types and Dosing Strategies
- Remission and Reversal Strategies
- CGM and Time-in-Range
- A1c, Fructosamine, and Glycation Markers
- Type 1 vs Type 2 vs LADA and MODY
- Peripheral Neuropathy
- Peripheral Artery Disease
- Cellulitis
- Gout
- Honey