Type 1 vs Type 2 vs LADA vs MODY
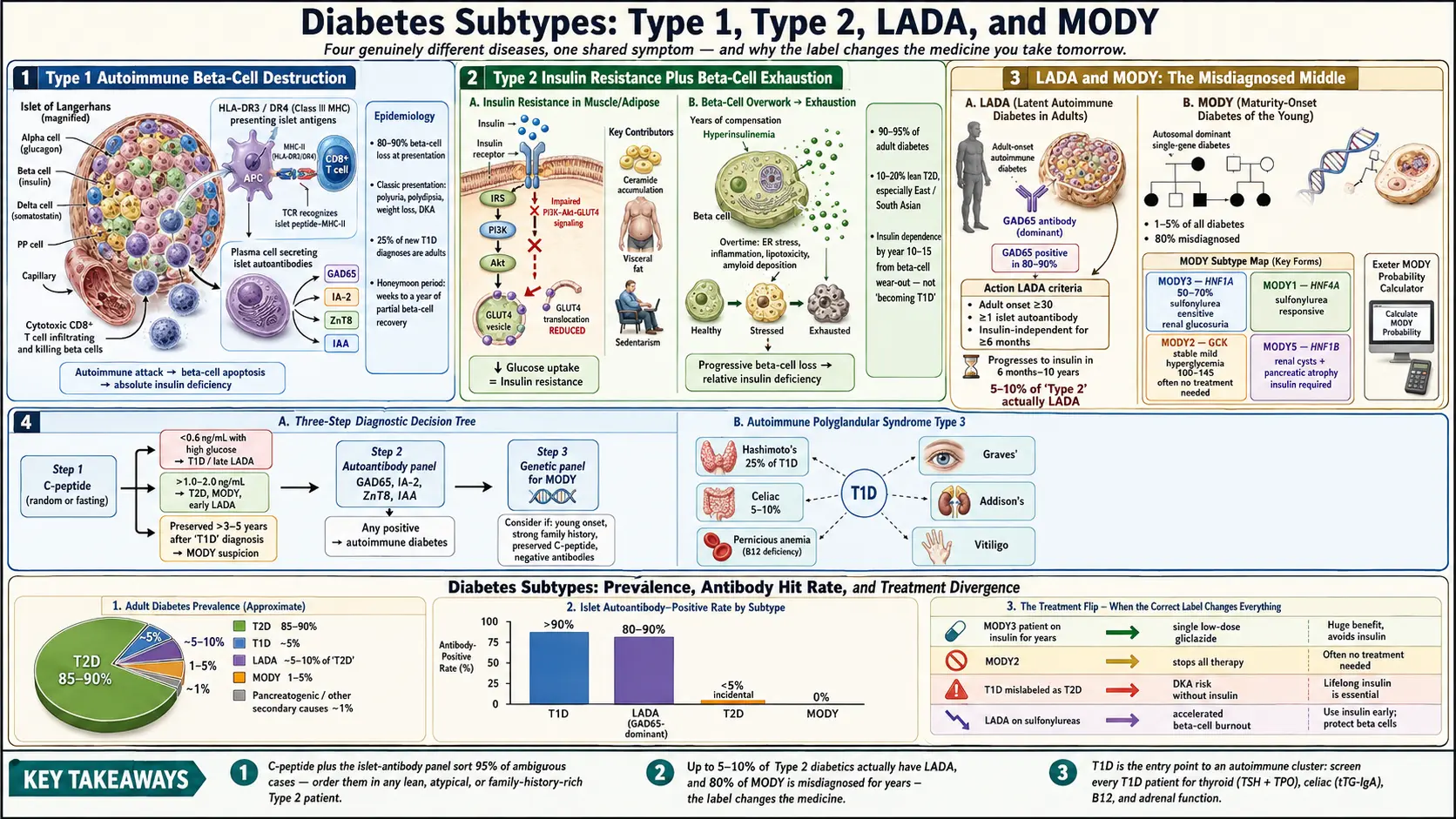
Table of Contents
- Why Getting the Subtype Right Matters
- Type 1 Diabetes — Autoimmune β-Cell Destruction
- Type 2 Diabetes — Insulin Resistance Plus β-Cell Failure
- LADA — The Slow-Burn Adult Type 1
- MODY — Monogenic Diabetes of the Young
- Diagnostic Workup — C-Peptide, Antibodies, Genes
- Why Misdiagnosis Hurts People
- Other Subtypes — Secondary and Gestational Diabetes
- T1D and the Polyglandular Autoimmune Cluster
- Screening Recommendations in T1D (ADA/ISPAD)
- Key Research Papers
- Connections
- Featured Videos
Why Getting the Subtype Right Matters
"Diabetes" is a single word for at least four genuinely different diseases. They share a final common symptom — high blood glucose — but the biology underneath is not the same, and neither is the correct treatment. A patient labeled "Type 2" who actually has slow-onset autoimmune diabetes (LADA) will be put on metformin and lifestyle advice, watch their A1c climb anyway, and eventually end up in the emergency room with diabetic ketoacidosis (DKA). A patient labeled "Type 1" who actually has MODY3 will spend decades on insulin injections when a single tiny sulfonylurea pill would control their glucose better and let them eat without calculating a carb ratio.
These mislabels are not rare. Studies suggest roughly 5–10% of adults diagnosed with "Type 2" diabetes actually have LADA, and up to 80% of MODY cases are misdiagnosed as Type 1 or Type 2 for years before genetic testing sorts them out. Getting the subtype right is not academic — it changes the medicine, the monitoring, the family counseling, and the long-term complication profile.
Type 1 Diabetes — Autoimmune β-Cell Destruction
Type 1 diabetes (T1D) is an autoimmune disease. Your own immune system, for reasons that involve a mix of genetics (mostly HLA class II genes like HLA-DR3 and HLA-DR4), viral triggers, and likely gut-microbiome and early-life exposures, sends T cells into the pancreatic islets of Langerhans and selectively destroys the insulin-producing β-cells. By the time symptoms appear, roughly 80–90% of β-cell mass is already gone. Without insulin injections, a T1D patient cannot survive more than a few weeks.
The classic picture is a thin child or teenager presenting over days to weeks with polyuria (frequent urination), polydipsia (intense thirst), unexplained weight loss, fatigue, and often DKA — a life-threatening emergency with fruity breath, abdominal pain, vomiting, dehydration, and deep rapid breathing. But the stereotype of T1D as a childhood disease is outdated. Roughly 25% of new T1D diagnoses are made in adults, including people in their 30s, 40s, and 50s.
Autoantibodies are the diagnostic fingerprint. The four commonly measured ones:
- GAD65 (glutamic acid decarboxylase 65) — the most durable antibody in adults; often the only one still positive years after diagnosis. GAD65 is also the signature antibody of LADA.
- IA-2 (islet antigen-2 / tyrosine phosphatase) — strongly associated with rapidly progressive T1D.
- ZnT8 (zinc transporter 8) — picks up roughly 26% of patients who are negative for the other three antibodies; particularly useful in fulminant-appearing cases.
- IAA (insulin autoantibodies) — most useful in young children, almost always the first antibody to appear. Must be drawn before exogenous insulin is started, because insulin therapy itself generates antibodies.
Having two or more positive antibodies confers a near-certain lifetime risk of clinical T1D. Having one is a warning flag. Having none does not fully rule out T1D, but it makes LADA or late-onset classical T1D less likely and shifts the differential toward Type 2, MODY, or secondary causes.
After diagnosis, many patients go through a "honeymoon period" lasting weeks to a year or more, during which residual β-cells recover enough function that insulin requirements fall dramatically. Some patients temporarily need no insulin at all. This is not remission — the autoimmune process continues, and insulin requirements will rise again — but it can confuse both patients and clinicians, and occasionally leads to dangerous insulin-stopping experiments.
Type 2 Diabetes — Insulin Resistance Plus β-Cell Failure
Type 2 diabetes (T2D) accounts for roughly 90–95% of adult diabetes. The biology is fundamentally different from T1D. The pancreas still makes insulin — often more insulin than a healthy person — but the muscle, liver, and fat cells have become resistant to its signal, so glucose stays elevated. Over years and decades, the overworked β-cells gradually exhaust themselves, insulin output falls, and glucose control worsens. By 10–15 years after diagnosis, many T2D patients need insulin not because they are "Type 1 now" but because the β-cells have simply worn out.
T2D is polygenic — dozens to hundreds of common gene variants each contribute a small amount of risk. Family history matters: if both parents have T2D, lifetime risk rises to 50–70%. Environmental contributors include excess visceral fat, sedentary lifestyle, sleep deprivation, chronic stress, and certain medications (glucocorticoids, atypical antipsychotics, some HIV protease inhibitors).
A common misconception: T2D does not require obesity. Roughly 10–20% of T2D patients are lean at diagnosis, particularly in East and South Asian populations where β-cell reserve is genetically smaller. Lean T2D often presents atypically and is the group most frequently misdiagnosed — some turn out to be LADA, some turn out to be MODY, and some are truly Type 2 despite normal BMI. See the sister article on Insulin Resistance for the mechanics of how visceral fat, hepatic de novo lipogenesis, and ceramide accumulation drive the resistance side of T2D.
The presentation is usually slow. Fatigue, blurred vision, recurrent yeast infections or urinary infections, slow-healing wounds, and tingling in the feet can precede diagnosis by years. A1c can sit in the 7–9% range for a long time before anyone checks it.
LADA — The Slow-Burn Adult Type 1
Latent Autoimmune Diabetes in Adults (LADA) is T1D in slow motion. The autoimmune process is the same — T cells destroying β-cells, detectable islet autoantibodies — but it unfolds over years rather than weeks. The patient is typically over 30, often normal-weight or modestly overweight, and presents looking enough like T2D that they are handed metformin and sent home.
The Action LADA consortium laid down three diagnostic criteria:
- Adult onset (age ≥30 at diagnosis)
- Presence of at least one positive islet autoantibody (GAD65 most often)
- Insulin independence for at least 6 months after diagnosis
GAD65 is the workhorse antibody here. It is present in roughly 80–90% of LADA cases and is often the only antibody detectable. A high GAD65 titer predicts faster progression to insulin dependence; a low titer is associated with a more indolent course.
The practical timeline: LADA patients usually progress to insulin dependence within 6 months to 10 years of diagnosis, far faster than classical T2D. Clinical red flags that should prompt GAD65 testing in anyone labeled "Type 2":
- Diagnosed before age 50, especially if lean (BMI <25)
- A1c climbing despite maximally titrated metformin and lifestyle changes
- Personal or family history of other autoimmune disease (Hashimoto's, Graves', celiac, vitiligo, pernicious anemia)
- Early insulin requirement (within 2–3 years of diagnosis)
- Lower C-peptide than expected for duration of disease
Getting the LADA label right matters because management diverges from T2D. Sulfonylureas — which squeeze the remaining β-cells harder — are generally avoided because they may accelerate β-cell exhaustion. Insulin is started earlier. Continuous glucose monitoring becomes worthwhile much sooner. And patients are screened for the other autoimmune diseases LADA clusters with.
MODY — Monogenic Diabetes of the Young
Maturity-Onset Diabetes of the Young (MODY) is not autoimmune and not insulin-resistant. It is a single-gene disease inherited in an autosomal dominant pattern — a child of an affected parent has a 50% chance of inheriting it. MODY accounts for roughly 1–5% of all diabetes, which sounds small until you realize that means hundreds of thousands of people in the United States alone, most of them misdiagnosed.
Fourteen MODY subtypes have been described, each caused by a mutation in a different gene. Four are clinically dominant:
- MODY3 (HNF1A) — the most common form, about 50–70% of MODY cases. Progressive hyperglycemia beginning in adolescence or young adulthood; renal glucosuria (sugar in the urine at relatively low blood glucose); dramatic sensitivity to low-dose sulfonylureas. Patients often mislabeled as T1D can be switched from multiple daily insulin injections to a single 20–40 mg gliclazide tablet with better control.
- MODY1 (HNF4A) — similar clinical picture to MODY3; also sulfonylurea-responsive. Babies of affected mothers are often born large (macrosomic) and may have transient neonatal hypoglycemia.
- MODY2 (GCK) — a glucokinase defect that resets the β-cell's glucose thermostat upward. Fasting glucose runs 100–145 mg/dL, A1c 5.6–7.6%, for life, essentially without progression. Usually requires no treatment at all. Complications are rare because the hyperglycemia is mild and stable. Patients are commonly and wrongly treated with insulin or oral agents that add risk without benefit.
- MODY5 (HNF1B) — diabetes plus renal cysts and other kidney malformations, sometimes genital-tract anomalies, pancreatic atrophy, and liver test abnormalities. Often presents with kidney disease before diabetes. Does not respond well to sulfonylureas; usually needs insulin.
When should MODY be suspected? The classic pattern is (1) diabetes diagnosed before age 25, (2) a strong family history spanning three or more consecutive generations, and (3) an atypical course — insulin-sensitive "T1D" with persistent C-peptide years after diagnosis, or lean "T2D" in a young non-obese patient with affected parents and grandparents. The Exeter MODY Probability Calculator (available free at diabetesgenes.org) takes a handful of inputs — age of diagnosis, BMI, current treatment, parental history, HbA1c — and returns a probability score. Scores above 25–40% generally justify genetic testing.
Genetic testing is a targeted panel (commonly HNF1A, HNF4A, GCK, HNF1B, plus rarer genes). Cost has fallen dramatically — many U.S. labs now run the panel for $300–$1500, and in the UK it is free through NHS referral when the probability score is high. A confirmed MODY diagnosis is one of the few diabetes diagnoses that genuinely changes what medication you take the next morning.
Diagnostic Workup — C-Peptide, Antibodies, Genes
When the subtype is not obvious, three tests usually sort it out:
C-peptide. C-peptide is released from the β-cell in equimolar amounts with endogenous insulin, but unlike insulin it is not consumed by the liver. A random or post-prandial C-peptide therefore reflects how much insulin you are still making. Interpretation:
- Low (<0.6 ng/mL) with high glucose — strong evidence of insulin deficiency, consistent with T1D or late-stage LADA.
- Preserved or high (>1.0–2.0 ng/mL) — endogenous insulin is intact; points toward T2D, MODY, or early LADA.
- Preserved C-peptide more than 3–5 years after a "T1D" diagnosis is a major red flag for MODY misdiagnosis.
Islet autoantibodies. A panel of GAD65, IA-2, ZnT8, and IAA distinguishes autoimmune diabetes (T1D, LADA) from non-autoimmune diabetes (T2D, MODY). Any single positive antibody, confirmed on a second draw, shifts the diagnosis toward autoimmune.
Genetic panel. Reserved for cases where the probability of MODY is high based on family history, age of onset, and atypical course. A positive finding is diagnostic; a negative panel does not entirely exclude monogenic diabetes because rarer genes may not be included.
Useful supporting labs: a fasting lipid panel (MODY3 often shows high HDL), urine albumin-to-creatinine ratio, fasting insulin for calculating HOMA-IR (high in T2D, low in T1D/late LADA), and glucose tolerance testing in ambiguous cases.
Why Misdiagnosis Hurts People
The subtype label is not paperwork — it drives real clinical decisions that can go badly wrong when the label is wrong.
- T1D labeled as T2D. Placed on metformin or diet alone, these patients continue to lose β-cells, their A1c climbs, and eventually they present in DKA. The fix is early antibody testing in any atypical-looking "T2D" patient.
- LADA labeled as T2D. Same pathway, slower timeline. Sulfonylureas may be prescribed, pushing stressed β-cells harder and potentially accelerating burnout. Insulin is often started years later than it should have been.
- MODY2 labeled as T1D or T2D. Treated with insulin or oral agents aimed at lowering a stable, benign mild hyperglycemia. Result: repeated hypoglycemia, hospital visits, and decades of unnecessary treatment for a condition that rarely causes complications.
- MODY3 labeled as T1D. Patient on a basal-bolus insulin regimen for decades when a daily sulfonylurea would give tighter control with less variability, no injections, and no carb counting. Switching often feels miraculous to patients who had assumed insulin was their fate.
- MODY5 missed. Kidney disease blamed on hypertension or idiopathic cysts when a single genetic test would explain both the diabetes and the renal findings and shape family screening.
Other Subtypes — Secondary and Gestational Diabetes
Not every diabetes is T1D, T2D, LADA, or MODY. The other categories worth knowing:
- Pancreatogenic (Type 3c) diabetes — loss of β-cells from structural pancreatic disease: chronic pancreatitis, hemochromatosis (iron overload damaging the pancreas), cystic fibrosis, pancreatic resection, or pancreatic adenocarcinoma. New-onset diabetes in an adult over 50 with weight loss should always prompt consideration of occult pancreatic cancer.
- Endocrinopathy-induced diabetes — Cushing's syndrome (cortisol excess), acromegaly (growth hormone excess), pheochromocytoma, glucagonoma, and thyrotoxicosis can all cause hyperglycemia that resolves when the underlying hormone excess is corrected.
- Drug-induced diabetes — systemic glucocorticoids are the classic cause, but atypical antipsychotics (olanzapine, clozapine), tacrolimus and cyclosporine, certain HIV protease inhibitors, and checkpoint-inhibitor immunotherapy (which can trigger a fulminant autoimmune diabetes) all contribute.
- Gestational diabetes mellitus (GDM) — glucose intolerance first detected during pregnancy, usually driven by placental hormones that induce insulin resistance. It resolves after delivery, but leaves behind a roughly 50% lifetime risk of progressing to T2D, highest in the 5–10 years post-partum. Every woman with a GDM history should have annual fasting glucose or A1c testing for life.
T1D and the Polyglandular Autoimmune Cluster
Autoimmunity rarely travels alone. T1D patients have a much higher lifetime risk than the general population of developing additional autoimmune conditions — a pattern called autoimmune polyglandular syndrome type 3 (APS-3) when two or more glands are affected. The common associations:
- Autoimmune thyroid disease — up to 25% of T1D patients develop Hashimoto's thyroiditis or, less commonly, Graves' disease. TPO and TgAb antibodies often appear years before clinical hypothyroidism (see TPO and TgAb Antibodies Explained).
- Celiac disease — 5–10% of T1D patients have celiac, often silent or minimally symptomatic. Untreated celiac worsens glycemic variability because of erratic carbohydrate absorption. See Celiac and Associated Autoimmune Conditions.
- Addison's disease (autoimmune adrenal insufficiency) — rare but serious. Presents with fatigue, unexplained hypoglycemia episodes, skin darkening, and salt craving. A T1D patient with rising insulin sensitivity and frequent lows should be screened.
- Pernicious anemia — autoimmune destruction of gastric parietal cells leading to B12 deficiency. Easily missed because fatigue and neuropathy overlap with diabetic complications.
- Vitiligo — autoimmune loss of skin melanocytes. Benign but a useful clinical clue to the polyglandular phenotype.
Screening Recommendations in T1D (ADA/ISPAD)
Because the autoimmune cluster is so common, the American Diabetes Association (ADA) and the International Society for Pediatric and Adolescent Diabetes (ISPAD) recommend routine screening of T1D patients for associated autoimmune conditions:
- Thyroid. TSH at diagnosis, then every 1–2 years, or sooner if symptoms or growth changes appear. TPO antibody at diagnosis to identify at-risk patients who warrant closer monitoring.
- Celiac. Tissue transglutaminase IgA (tTG-IgA) with total IgA at diagnosis, then every 1–2 years in children and at symptom onset in adults.
- Adrenal insufficiency. Not routine in asymptomatic patients, but 21-hydroxylase antibody or morning cortisol / ACTH should be checked if unexplained hypoglycemia, fatigue, hyperpigmentation, or salt craving appears.
- B12. Reasonable to check every few years, particularly in patients also on long-term metformin (which depletes B12).
- Lipids, kidney function, retinal exam, foot exam — the standard diabetes complication screens remain the backbone; see the complications article.
The principle is simple: one autoimmune disease earns a lifetime ticket to watchfulness for the others. The bloodwork is cheap, the diagnoses make a real difference when caught early, and patients who know to watch for the pattern catch problems months to years before a blinded clinician would.
Key Research Papers
Foundational peer-reviewed work on diabetes classification, autoimmune type 1, latent autoimmune diabetes in adults (LADA), and monogenic (MODY) diabetes. Each citation links to the full text via DOI.
- American Diabetes Association. 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes—2022. Diabetes Care. 2022;45(Suppl 1):S17–S38.
- Atkinson MA, Eisenbarth GS, Michels AW. Type 1 Diabetes. Lancet. 2014;383(9911):69–82.
- DeFronzo RA, Ferrannini E, Groop L, et al. Type 2 Diabetes Mellitus. Nature Reviews Disease Primers. 2015;1:15019.
- Buzzetti R, Tuomi T, Mauricio D, et al. Management of Latent Autoimmune Diabetes in Adults: A Consensus Statement from an International Expert Panel. Diabetes. 2020;69(10):2037–2047.
- Hattersley AT, Greeley SAW, Polak M, et al. ISPAD Clinical Practice Consensus Guidelines 2018: The Diagnosis and Management of Monogenic Diabetes in Children and Adolescents. Pediatric Diabetes. 2018;19(Suppl 27):47–63.
- Insel RA, Dunne JL, Atkinson MA, et al. Staging Presymptomatic Type 1 Diabetes: A Scientific Statement of JDRF, the Endocrine Society, and the American Diabetes Association. Diabetes Care. 2015;38(10):1964–1974.
- Shields BM, Shepherd M, Hudson M, et al. Population-Based Assessment of a Biomarker-Based Screening Pathway to Aid Diagnosis of Monogenic Diabetes in Young-Onset Patients. Diabetologia. 2017;60(11):2247–2255.
- Laugesen E, Østergaard JA, Leslie RDG. Latent Autoimmune Diabetes of the Adult: Current Knowledge and Uncertainty. Diabetologia. 2015;58(6):1205–1214.
- The Diabetes Control and Complications Trial Research Group. The Effect of Intensive Treatment of Diabetes on the Development and Progression of Long-Term Complications in Insulin-Dependent Diabetes Mellitus (DCCT). New England Journal of Medicine. 1993;329(14):977–986.
- UK Prospective Diabetes Study (UKPDS) Group. Intensive Blood-Glucose Control with Sulphonylureas or Insulin Compared with Conventional Treatment and Risk of Complications in Patients with Type 2 Diabetes (UKPDS 33). Lancet. 1998;352(9131):837–853.
Live PubMed Searches
Each link opens a live PubMed query so results stay current as new papers are indexed.
- Latent autoimmune diabetes in adults (LADA)
- MODY and monogenic diabetes (HNF1A, HNF4A, GCK, HNF1B)
- GAD65 antibody and type 1 diabetes
- C-peptide in diabetes classification
- Sulfonylurea response in HNF1A/MODY3
- Type 1 diabetes with celiac and autoimmune thyroid disease
- Autoimmune polyglandular syndrome type 3
- Gestational diabetes and progression to type 2
- Pancreatogenic (type 3c) diabetes
Connections
- Diabetes Overview
- Cardio-Renal Protection: SGLT2 and GLP-1
- Insulin Types and Dosing Strategies
- Hypoglycemia Awareness and Prevention
- Remission and Reversal Strategies
- CGM and Time-in-Range
- A1c, Fructosamine, and Glycation Markers
- Complications: Retinopathy, Nephropathy, Neuropathy
- Diabetic Foot Care and Ulcer Prevention
- Insulin Resistance
- Hashimoto's Thyroiditis
- Graves' Disease
- Celiac Disease
- Polycystic Ovary Syndrome
- Gestational Diabetes
- Obesity
- Fasting
- TPO and TgAb Antibodies