Metformin, Berberine, and GLP-1 Agonists for Insulin Resistance
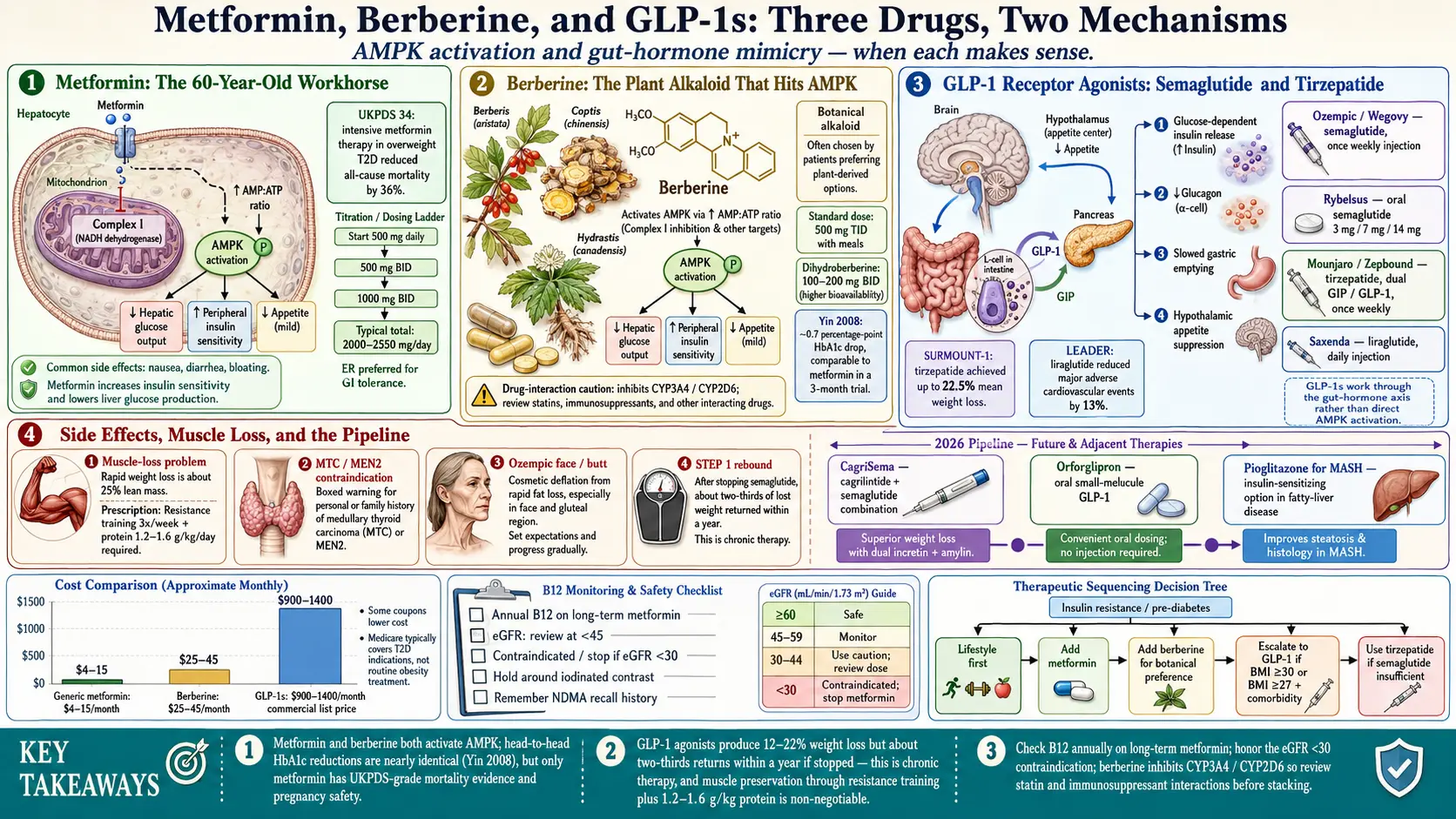
Table of Contents
- Why These Three Drug Classes Matter
- Metformin — The 60-Year-Old Workhorse
- Metformin Dosing, Forms, and GI Tolerance
- Metformin Risks: B12, Lactic Acidosis, NDMA
- Metformin Off-Label: PCOS, Prediabetes, Longevity
- Berberine — The "Natural-Medicine Metformin"
- When Berberine Is NOT Equivalent
- GLP-1 Receptor Agonists — Semaglutide and Friends
- Tirzepatide — The Dual GIP/GLP-1 Agonist
- GLP-1 Side Effects and the Muscle-Loss Problem
- Cost, Coverage, and Compounded Semaglutide
- The Pipeline: Cagrilintide, Orforglipron, Pioglitazone
- Practical Sequencing — Which Drug, When
- Monitoring on Therapy
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
Why These Three Drug Classes Matter
Insulin resistance does not always yield to diet and exercise alone. For many patients — especially those with a strong genetic push, PCOS, or decades of accumulated metabolic damage — pharmacology is part of the answer. Three drug classes dominate the conversation in 2026: metformin (cheap, old, remarkably safe), berberine (the plant alkaloid that hits the same molecular switch), and the GLP-1 receptor agonists (semaglutide, tirzepatide, and their relatives) that have rewritten what is possible for weight-driven metabolic disease.
None of these replaces the dietary strategy or the muscle-building work discussed in the sibling articles — they multiply their effect. This page is the practical guide: what each drug does, how to dose it, what it costs, what it breaks, and how to sequence them.
Metformin — The 60-Year-Old Workhorse
Metformin is a biguanide derived from the French lilac (Galega officinalis), in clinical use since 1957 in Europe and since 1994 in the United States. It remains the first-line drug for type 2 diabetes in essentially every international guideline, and it is the most-prescribed medication for insulin resistance short of full diabetes.
Its primary target is AMP-activated protein kinase (AMPK), the cellular "low-energy" switch. Metformin inhibits Complex I of the mitochondrial electron transport chain, which raises the AMP:ATP ratio and activates AMPK. Once AMPK is on, three useful things happen:
- Hepatic glucose production drops. The liver stops dumping glucose into the bloodstream overnight and between meals. This is the largest single effect — fasting blood sugar falls by 20–30% in most responders.
- Peripheral insulin sensitivity improves. Muscle and fat cells take up glucose more readily in response to the same amount of insulin.
- Appetite and weight modestly decrease. Metformin is weight-neutral to mildly weight-reducing (typically 2–5 lb over 6–12 months), unlike sulfonylureas and insulin, which drive weight up.
The landmark evidence is UKPDS 34, the 1998 arm of the UK Prospective Diabetes Study in overweight type 2 diabetics. Metformin reduced all-cause mortality by 36% and diabetes-related death by 42% versus conventional therapy — a mortality benefit no other oral diabetes drug has duplicated.
Metformin Dosing, Forms, and GI Tolerance
The classic mistake with metformin is starting too high. The drug's reputation for causing diarrhea and nausea is earned, but almost entirely avoidable with a slow titration.
- Start: 500 mg once daily with dinner (the largest meal).
- Week 2: 500 mg twice daily (breakfast and dinner).
- Week 3–4: 1000 mg twice daily.
- Maximum: 2550 mg/day (immediate-release) or 2000 mg/day (extended-release).
Always take metformin with food — mid-meal, not before. Taking it on an empty stomach roughly triples the rate of GI side effects.
Extended-release (Glucophage XR, metformin ER) is meaningfully better-tolerated than the immediate-release tablet. The drug is released over 8–10 hours rather than peaking in 2–3, which reduces the intestinal-lumen concentration that drives diarrhea. If you failed immediate-release, ask to try ER before giving up on the class. A once-daily dose of metformin ER 1500–2000 mg with dinner is equally effective for most patients.
Generic metformin is one of the cheapest drugs in American pharmacy: $4–15 per month without insurance at Walmart, Costco, and most chains. Mark Cuban's Cost Plus Drugs offers it at roughly $4/month for a 90-day supply.
Metformin Risks: B12, Lactic Acidosis, NDMA
Metformin is among the safest prescription drugs in routine use, but three issues deserve explicit mention because patients are rarely warned about them.
Vitamin B12 depletion. Long-term metformin use (generally beyond 4–5 years, sometimes sooner) interferes with calcium-dependent B12 absorption in the terminal ileum. Estimates suggest 10–30% of long-term users develop measurable deficiency, which presents as fatigue, peripheral neuropathy (tingling feet, which is easily mistaken for diabetic neuropathy), anemia, and cognitive fog. Check a serum B12 annually once you cross the one-year mark. If the level is below ~400 pg/mL, supplement with methylcobalamin 1000 mcg/day sublingual or, if deficiency is established, intramuscular injections.
Lactic acidosis. A rare but serious complication in which lactate accumulates to dangerous levels. Risk is essentially zero at normal kidney function but rises sharply below an eGFR of 30 mL/min/1.73 m². Current FDA guidance:
- eGFR ≥ 45: no dose change.
- eGFR 30–44: do not start; if already on, consider dose reduction (generally ≤ 1000 mg/day).
- eGFR < 30: stop metformin.
- Hold before iodinated IV contrast (CT scans with contrast, cardiac catheterization) in anyone with eGFR < 60 or at risk of acute kidney injury — stop on the day of the procedure and resume 48 hours later if kidney function is stable.
- Hold during acute illness with dehydration, sepsis, or severe GI illness.
NDMA impurity recalls. In 2020, the FDA recalled several extended-release metformin lots after finding N-nitrosodimethylamine, a probable human carcinogen, above acceptable limits. The recalls prompted real worry. The good news: manufacturing controls tightened substantially, and metformin lots released since late 2021 have been consistently clean. If you want certainty, ask your pharmacist for lot numbers and check FDA's recall database, but routine concern is no longer warranted.
Metformin Off-Label: PCOS, Prediabetes, Longevity
Metformin's FDA label is type 2 diabetes. Its real-world use is broader:
- PCOS. The strongest off-label use. Metformin improves ovulation, restores menstrual regularity, reduces androgen levels, and modestly improves acne and hirsutism. Standard dosing is 1500–2000 mg/day. Often combined with inositol and a low-carbohydrate approach. See the PCOS and insulin resistance article and the main PCOS page for the full protocol.
- Prediabetes. The Diabetes Prevention Program showed metformin reduced progression to type 2 diabetes by 31%, though lifestyle change performed nearly twice as well (58%). Reasonable to add for BMI ≥ 30, HbA1c ≥ 6.0%, or prior gestational diabetes.
- Weight maintenance. Modest, real, and mostly useful for preventing regain rather than driving loss.
- Longevity research. The TAME trial (Targeting Aging with Metformin), championed by Nir Barzilai, aims to test whether metformin delays age-related disease clusters in non-diabetic older adults. Mechanistic rationale is strong (AMPK activation, mTOR inhibition, mitochondrial effects). Definitive human evidence is still pending. Taking metformin specifically for longevity remains a reasonable but unproven choice to make with your physician.
Berberine — The "Natural-Medicine Metformin"
Berberine is a bright-yellow isoquinoline alkaloid extracted from Berberis (barberry), Coptis chinensis (goldthread), and Hydrastis canadensis (goldenseal). It has a two-thousand-year history in Chinese and Ayurvedic medicine for diarrhea, dysentery, and what we would now call metabolic sluggishness.
Its headline mechanism is remarkable: berberine activates the same AMPK pathway as metformin. Yin and colleagues published the first head-to-head human trial in 2008 (Metabolism), showing berberine 500 mg three times daily reduced HbA1c by roughly 0.7 percentage points over three months in newly diagnosed type 2 diabetics — essentially identical to metformin in the same trial. Fasting glucose, insulin, and triglycerides all moved favorably.
Practical dosing:
- 500 mg three times daily with meals. Total 1500 mg/day.
- Split dosing is non-negotiable — berberine has a short half-life and poor oral bioavailability (~5%), so once-daily dosing does not work.
- GI tolerance is often better than metformin, though some users get loose stools or cramping in the first 1–2 weeks. Take with food.
- Antimicrobial bonus. Berberine is a well-established botanical antimicrobial effective against many gram-negative gut bacteria. It is a mainstay of herbal SIBO protocols — see the herbal antimicrobials deep dive for context.
Drug interactions matter. Berberine is a moderate inhibitor of CYP3A4 and CYP2D6, which means it can raise blood levels of many prescription drugs — including statins (simvastatin, atorvastatin), cyclosporine, tacrolimus, many calcium-channel blockers, certain antidepressants, and some chemotherapeutics. Do not combine with immunosuppressants without pharmacist review. With statins, watch for muscle aches; dose reduction of the statin may be needed.
Quality sourcing. Supplements are unregulated and berberine capsules vary. Brands with third-party testing and consistent potency include Thorne Berberine-500, Integrative Therapeutics Berberine, NOW Foods Berberine Glucose Support, and Pure Encapsulations Berberine UltraPure. Expect $25–45 per month at standard dosing.
Dihydroberberine is a reduced form with substantially higher bioavailability — roughly 5× — which allows lower doses (100–200 mg twice daily) with less GI burden. It is more expensive and has a smaller evidence base, but is a reasonable choice for patients who cannot tolerate the full three-times-daily schedule.
When Berberine Is NOT Equivalent
Berberine gets marketed as "nature's Ozempic" and "metformin in a capsule." It is neither of those things at the margin. Know the limits:
- Severe or long-standing type 2 diabetes. Patients with HbA1c above ~8.5%, or with insulin deficiency, need prescription drugs. Berberine is adjunctive at best in that setting.
- Pregnancy and breastfeeding. Avoid. Berberine crosses the placenta and has been linked to kernicterus (displacement of bilirubin in newborns). Metformin, by contrast, is well-studied and generally safe in pregnancy at physician discretion.
- Children. Avoid outside of specialist supervision.
- Polypharmacy. If you are on three or more prescription drugs metabolized by CYP3A4, the interaction risk outweighs the benefit.
For mild insulin resistance, prediabetes, PCOS, or patients who firmly prefer a botanical approach, berberine is a legitimate trial. For anyone who has actually crossed into type 2 diabetes or who has significant cardiovascular risk, metformin remains the evidence-anchored choice.
GLP-1 Receptor Agonists — Semaglutide and Friends
Glucagon-like peptide-1 is a gut hormone released after meals. It stimulates insulin release in a glucose-dependent way (meaning it works only when blood sugar is high, which is why GLP-1 drugs rarely cause hypoglycemia on their own), suppresses glucagon, slows gastric emptying, and reduces appetite through the hypothalamus. The class of drugs that mimic GLP-1 has transformed obesity and diabetes care since 2005.
The current agents, roughly in order of clinical weight:
- Semaglutide. Injectable weekly as Ozempic (diabetes label) or Wegovy (obesity label); oral daily as Rybelsus at 3, 7, or 14 mg. Typical A1c reduction 1.5–1.8%, typical weight loss 12–15% of body weight over a year at full dose.
- Liraglutide. Injectable daily as Victoza (diabetes) or Saxenda (obesity). Older, shorter-acting, less weight loss than semaglutide, but covered by some insurance plans that balk at the weekly agents. The LEADER trial (Marso et al., NEJM 2016) showed liraglutide reduced major adverse cardiovascular events by 13% in high-risk T2D — the first clear CV-protective evidence for the class.
- Dulaglutide (Trulicity). Weekly injectable. Efficacy between liraglutide and semaglutide. Easier pen.
- Exenatide (Byetta, Bydureon). The original GLP-1, now largely supplanted.
For insulin resistance specifically (pre-diabetes, metabolic syndrome without a formal T2D diagnosis), GLP-1 use is off-label and cash-pay. Insurance covers them for T2D, or for obesity if you meet BMI criteria (BMI ≥ 30, or ≥ 27 with a comorbidity like hypertension).
Tirzepatide — The Dual GIP/GLP-1 Agonist
Tirzepatide (Eli Lilly's Mounjaro for diabetes, Zepbound for obesity) adds a second hormone receptor — GIP (glucose-dependent insulinotropic polypeptide) — to the GLP-1 activity. The dual mechanism produces the largest weight reductions any diabetes/obesity drug has achieved.
The SURPASS trials (T2D) showed A1c reductions of 2.0–2.5 percentage points and weight loss of 11–15% at the 15 mg dose — consistently superior to semaglutide head-to-head. The SURMOUNT-1 trial (Jastreboff et al., NEJM 2022) in non-diabetic obesity showed a mean weight reduction of 22.5% at 15 mg over 72 weeks — closer to bariatric surgery than to any prior drug.
Dosing starts at 2.5 mg weekly for four weeks, titrating monthly through 5, 7.5, 10, 12.5, and 15 mg. Most patients plateau at 10–15 mg.
GLP-1 Side Effects and the Muscle-Loss Problem
The class has real downsides that get glossed over in marketing.
- Nausea, vomiting, diarrhea, constipation. Common, dose-dependent, worst during titration. Slow titration is the single biggest predictor of tolerability. Smaller meals, lower fat, extra fluids, and avoiding alcohol all help.
- Pancreatitis. Rare but real. Discontinue for persistent severe upper-abdominal pain.
- Gallbladder disease. Rapid weight loss increases gallstone risk regardless of mechanism; GLP-1s appear to add a small independent risk.
- Thyroid C-cell tumors. Observed in rodents. The human risk is uncertain, but the drugs are contraindicated in personal or family history of medullary thyroid carcinoma or MEN2 syndrome.
- "Ozempic face" and "Ozempic butt." Rapid fat loss, especially in older patients, unmasks underlying tissue thinning — the face and buttocks look hollowed. This is cosmetic, not dangerous, but it is real.
- Muscle loss. The most underappreciated issue. Weight loss from any source is roughly 25% lean mass unless defended. At the GLP-1 rate of loss, an untrained patient can shed 10–15 lb of muscle in a year — catastrophic for long-term metabolic health, because muscle is the body's largest insulin-sensitive tissue. Resistance training 3× per week and protein intake of 1.2–1.6 g/kg body weight are not optional on a GLP-1. See the exercise and muscle article.
- Rebound weight regain. The STEP-1 extension trial showed that stopping semaglutide returned two-thirds of lost weight within a year. GLP-1s are chronic therapy or nothing.
Cost, Coverage, and Compounded Semaglutide
Commercial list prices in early 2026 run $900–1,400 per month for Ozempic, Wegovy, Mounjaro, and Zepbound. With commercial insurance and a manufacturer savings coupon, eligible patients can pay as little as $25/month for T2D; coverage for the obesity indications (Wegovy, Zepbound) is patchier but improving.
Medicare Part D covers GLP-1s only for a T2D indication — an anti-obesity indication is statutorily excluded unless Congress changes the law. Medicaid coverage varies widely by state.
Compounded semaglutide. During the 2022–2024 shortage, the FDA allowed 503A and 503B compounding pharmacies to produce semaglutide for cash-pay patients at $150–350/month. When the FDA removed semaglutide from its shortage list in Q1 2024 and tirzepatide in late 2024, the legal basis for large-scale compounding evaporated. The FDA gave 503A pharmacies until mid-2024 and 503B facilities until Q3 2024 to wind down. Some compounders continue to operate in gray areas, often by adding small amounts of B12 or amino acids and calling the product "non-FDA-approved." Patients should know: quality varies, the drug may not be pure semaglutide, and the legal standing is shaky. Buyer beware.
The Pipeline: Cagrilintide, Orforglipron, Pioglitazone
- CagriSema — a fixed combination of cagrilintide (an amylin analog) plus semaglutide. Phase 3 data show weight loss approaching 20–23%, with better appetite control and potentially less nausea than semaglutide alone. Likely FDA filing in late 2026.
- Orforglipron — Eli Lilly's oral small-molecule GLP-1 agonist. Unlike Rybelsus (which is peptide semaglutide with an absorption enhancer and rigid dosing rules), orforglipron is a true small molecule taken as a normal once-daily pill with food or without. Phase 3 results in 2025 showed weight loss of ~14% and A1c reductions comparable to injectable GLP-1s. Approval expected 2026–2027; if priced reasonably, it could reshape the market.
- Pioglitazone. An older thiazolidinedione that remains a genuinely powerful insulin sensitizer by activating PPAR-γ. Now FDA-approved for type 2 diabetes and MASH (metabolic dysfunction-associated steatohepatitis). Downsides are why it is rarely first-line: weight gain, fluid retention, increased heart failure risk, decreased bone density, and a disputed bladder cancer signal. Reasonable second- or third-line agent in patients with severe insulin resistance and fatty liver.
Practical Sequencing — Which Drug, When
A reasonable decision framework, to discuss with your physician:
- Start with lifestyle. Diet, resistance training, sleep, and stress reduction produce real HOMA-IR and A1c improvements. Give this 3–6 months before adding a drug unless numbers are severe.
- Metformin first. For anyone crossing into prediabetes or type 2 diabetes, or with PCOS, metformin is cheap, well-studied, and has mortality evidence going back to 1998. Titrate slowly; use ER if IR causes GI issues.
- Berberine is a reasonable alternative for mild cases, patients who refuse prescriptions, or patients with co-existing SIBO. It is not a replacement for metformin in established diabetes.
- Add or switch to a GLP-1 when weight is the dominant driver, when metformin is insufficient, or when the patient is metformin-intolerant. Semaglutide for most; tirzepatide when the weight-loss goal is > 15% or when semaglutide plateaus.
- Pioglitazone as a specialist's add-on for severe insulin resistance with fatty liver, after weighing heart failure and bone risks.
Whatever drug combination you land on, the diet, resistance training, and sleep work all continue. Drugs are force multipliers, not substitutes.
Monitoring on Therapy
- HbA1c every 3 months until stable, then every 6 months.
- HOMA-IR (fasting glucose and fasting insulin) every 6–12 months to track insulin resistance directly. See the lab testing article.
- Fasting lipid panel yearly (triglycerides fall meaningfully on metformin and GLP-1s).
- Comprehensive metabolic panel with eGFR yearly — more often if starting metformin or changing dose.
- Vitamin B12 yearly on metformin after year one.
- Weight, waist circumference, body composition. DEXA or bioimpedance every 6–12 months on a GLP-1 to catch muscle loss early.
- Blood pressure and resting heart rate at every visit (GLP-1s raise heart rate by 2–4 bpm on average).
- Amylase/lipase only if symptoms suggest pancreatitis — routine screening is not recommended.
Key Research Papers
- UK Prospective Diabetes Study (UKPDS) Group. Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). Lancet. 1998.
- Yin J, Xing H, Ye J. Efficacy of berberine in patients with type 2 diabetes mellitus. Metabolism. 2008.
- Marso SP, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes (LEADER). N Engl J Med. 2016.
- Jastreboff AM, et al. Tirzepatide once weekly for the treatment of obesity (SURMOUNT-1). N Engl J Med. 2022.
Research Papers
Current peer-reviewed work on these agents and insulin resistance:
- Metformin, AMPK, and insulin resistance
- Metformin and vitamin B12 deficiency
- Metformin in polycystic ovary syndrome
- Berberine for type 2 diabetes and HbA1c
- Berberine and AMPK mechanism
- Semaglutide and weight loss (STEP trials)
- Tirzepatide SURPASS and SURMOUNT trials
- GLP-1 agonists and muscle loss
- Pioglitazone, insulin sensitivity, and MASH
- Orforglipron and oral GLP-1 agonists
Connections
- Polycystic Ovary Syndrome
- Reversal Protocol: Diet and Fasting
- Fasting Insulin and HOMA-IR Testing
- Insulin Resistance and PCOS
- Exercise and Muscle Insulin Sensitivity
- Insulin Resistance
- Diabetes
- Obesity
- Berberine
- SIBO
- CGM for Non-Diabetics
- GLP-1 Receptor Agonists
- Metabolic Syndrome
- eGFR
- Vitamin B12
- Hypertension
- Herbal Antimicrobials
- NAFLD MASLD Connection