Insulin Resistance: The Silent Upstream Driver of Chronic Disease
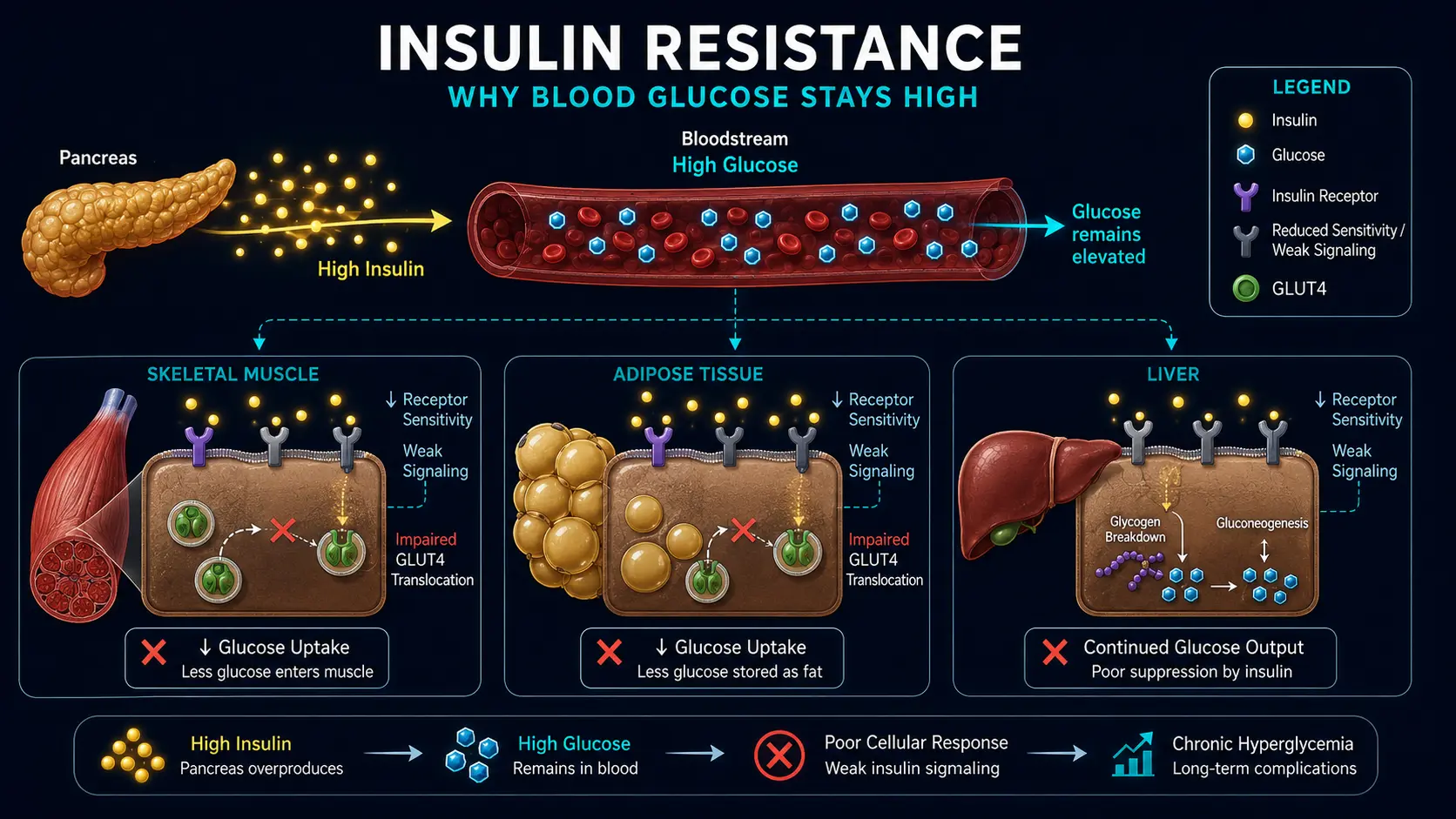
Insulin resistance is a state in which the body’s cells — particularly muscle, liver, and adipose tissue — respond less effectively to insulin, so the pancreas must release more insulin to achieve the same blood-glucose control. It is the central metabolic defect that precedes and drives type 2 diabetes, fatty liver disease, polycystic ovary syndrome, metabolic syndrome, much of cardiovascular disease, and a substantial fraction of Alzheimer’s disease (now called by some researchers “type 3 diabetes”). Perhaps most important, insulin resistance is usually present for 10 to 15 years before blood sugar rises into prediabetic or diabetic ranges, a long window in which it is reversible — but almost never measured in routine medical care.
Deep-Dive Articles
Fasting Insulin & HOMA-IR
Why fasting insulin catches problems 10+ years before fasting glucose moves. HOMA-IR formula, target ranges, OGTT with insulin, and what labs to ask for.
CGM for Non-Diabetics
Dexcom G7, Freestyle Libre, Levels/Nutrisense/Signos — what to actually learn from a 2-week CGM trial without pathologizing every spike.
Reversal: Diet & Fasting
The evidence for low-carb/ketogenic, Mediterranean, and protein-forward diets; time-restricted eating; DiRECT-trial 5%-weight-loss threshold; protein sparing modified fasts.
IR & PCOS
PCOS is insulin resistance in an ovary costume. Rotterdam criteria, androgen excess, metformin + inositol, GLP-1s, fertility impact.
Metabolic Syndrome & CV Risk
ATP-III criteria, waist circumference as the best single marker, triglyceride-to-HDL ratio, apoB, Lp(a), and why statins alone don't fix the metabolic engine.
NAFLD / MASLD Connection
Fatty liver (now renamed MASLD) as the hepatic arm of insulin resistance. FIB-4, FibroScan, why ALT "within normal range" still matters, and reversal.
Exercise & Muscle Sensitivity
Why resistance training + brisk walking beat cardio alone for insulin sensitivity. GLUT4 translocation, Zone 2, HIIT tradeoffs, muscle as the largest glucose sink.
Metformin, Berberine & GLP-1s
Metformin 500-2000 mg, berberine as the natural-medicine analog, GLP-1 receptor agonists (semaglutide, tirzepatide), and when each makes sense off-label for IR.
Sleep, Stress & Cortisol
Why sleep restriction, OSA, and chronic stress drive insulin resistance.
Table of Contents
- Deep-Dive Articles
- What Insulin Resistance Is
- Why It Matters — Downstream Consequences
- How to Test For It
- Clinical Signs
- Primary Drivers
- The Reversal Framework
- Pharmacologic Aids
- Evidence-Based Supplements
- Connections
- Featured Videos
What Insulin Resistance Is
Insulin is the anabolic hormone released by pancreatic beta cells in response to rising blood glucose and certain amino acids. It moves glucose from blood into cells via GLUT4 transporters in muscle and fat, suppresses hepatic glucose production, and promotes storage of glucose as glycogen and fat. In insulin-resistant tissue, the signaling cascade downstream of the insulin receptor is impaired. The pancreas compensates by secreting more insulin, maintaining glucose control at the cost of chronic hyperinsulinemia — which is itself a problem. Eventually, pancreatic beta cells fatigue and can no longer keep up, at which point fasting glucose rises and type 2 diabetes is diagnosed. By then the horse has been out of the barn for a decade.
Why It Matters — Downstream Consequences
- Type 2 diabetes
- Non-alcoholic fatty liver disease (MASLD/MASH)
- Polycystic ovary syndrome
- Metabolic syndrome — central adiposity, hypertension, dyslipidemia, hyperglycemia
- Cardiovascular disease — accelerated atherosclerosis, endothelial dysfunction
- Gout (hyperinsulinemia raises uric acid)
- Alzheimer’s disease (brain insulin resistance)
- Some cancers (breast, colon, endometrium, pancreatic)
- Acanthosis nigricans, skin tags, hirsutism in women
- Infertility and erectile dysfunction
How to Test For It
- Fasting insulin. Optimal <7 µIU/mL. 10–15 suggests insulin resistance even with normal fasting glucose. >15 is clearly abnormal.
- HOMA-IR. Calculated: (fasting insulin × fasting glucose) / 405. Values >2 suggest resistance; >2.5 strongly suggest it.
- Fasting triglyceride-to-HDL ratio. >2 (mg/dL) or >0.9 (mmol/L) suggests resistance in non-Black populations.
- Oral glucose tolerance test with insulin. The Kraft test — 2- or 3-hour paired glucose and insulin at 30-min intervals — is the most sensitive but less commonly available.
- Continuous glucose monitor. A 2-week CGM often reveals post-meal glucose excursions that are the earliest measurable clinical sign.
- Hemoglobin A1c rises late and catches only the tail end of the insulin-resistance trajectory.
- Waist-to-height ratio. >0.5 strongly suggests central adiposity driven by insulin resistance.
Clinical Signs
- Central adiposity — “apple” shape.
- Skin tags, particularly at neck and axilla.
- Acanthosis nigricans — dark velvety skin at neck, axilla, groin.
- Reactive hypoglycemia 2–4 hours after meals.
- Post-meal energy crashes and irresistible carb cravings.
- Hirsutism, irregular periods, acne in women.
Primary Drivers
- Diet dominated by ultra-processed foods, refined carbohydrates, and liquid calories.
- Chronic caloric surplus and excess adipose tissue, particularly visceral.
- Sedentary lifestyle.
- Chronic sleep restriction or fragmentation.
- Chronic stress and elevated cortisol.
- Genetics (modest individual contribution).
- Certain medications (glucocorticoids, atypical antipsychotics, some antivirals).
- Endocrine disruptors and persistent organic pollutants.
The Reversal Framework
- Reduce refined carbohydrate. Sugar, soft drinks, juice, ultra-processed snacks, and refined starch are the biggest levers.
- Increase protein and fiber. Both slow glucose absorption and improve satiety.
- Time-restricted eating. 10-, 12-, or 14-hour eating windows improve insulin sensitivity for many.
- Resistance training. Skeletal muscle mass is the largest glucose sink; building it is the single most effective sensitizer.
- Post-meal walking. 10–15 minutes after each meal cuts glucose excursion substantially.
- Sleep 7–9 hours. One night of 4-hour sleep impairs insulin sensitivity measurably.
- Stress management. Cortisol is a glucocorticoid — the name is literal.
- Weight loss of 5–10% body weight produces large improvements in insulin sensitivity and can reverse early type 2 diabetes.
Pharmacologic Aids
- Metformin — first-line for insulin-resistant states including prediabetes and PCOS.
- GLP-1 receptor agonists — powerful improvement in insulin sensitivity alongside weight loss. See GLP-1 Receptor Agonists.
- SGLT2 inhibitors — additional cardiovascular and renal benefits.
- Pioglitazone — insulin sensitizer with weight-gain trade-off.
Evidence-Based Supplements
- Berberine — 500 mg three times daily; comparable to metformin in some trials.
- Magnesium — deficiency impairs insulin action.
- Chromium picolinate — modest improvements.
- Alpha-lipoic acid, cinnamon, and fenugreek have supportive evidence.
- Omega-3 fatty acids — improve membrane fluidity and insulin signaling.
Connections
- Deep Dives: Fasting Insulin & HOMA-IR · CGM · Diet & Fasting · PCOS · MetS & CV Risk · NAFLD/MASLD · Exercise · Metformin, Berberine, GLP-1s · Sleep, Stress & Cortisol
- Diabetes — Types · A1c · Remission · SGLT2/GLP-1
- Metabolic Syndrome
- Obesity
- PCOS
- Fatty Liver Disease
- Fasting Insulin
- Hemoglobin A1C
- Continuous Glucose Monitor
- GLP-1 Agonists
- Berberine
- Blood Sugar
- Fasting
Research Papers
The following PubMed topic searches aggregate the current peer-reviewed literature. Each link opens a live PubMed query — results update as new studies are indexed.
- PubMed — insulin resistance
- PubMed — HOMA-IR
- PubMed — metabolic syndrome insulin resistance
- PubMed — visceral adiposity
- PubMed — low carbohydrate diet insulin resistance
- PubMed — time restricted eating insulin
- PubMed — exercise insulin sensitivity
- PubMed — metformin prediabetes
- PubMed — berberine insulin resistance
- PubMed — inositol PCOS insulin
- PubMed — hepatic insulin resistance NAFLD
- PubMed — magnesium insulin sensitivity
Featured Videos

How to Reverse Insulin Resistance

9 Signs of Insulin Resistance You Shouldn't Ignore

What is insulin resistance? Why does it happen? [Dr. Christopher Gardner]

Spot Insulin Resistance. No Labs Required

Signs of Insulin Resistance – Do You Have Them?

REVERSE--Insulin Resistance, Diabetes and Hypoglycemia

If You Do This Your Insulin Resistance Will Be Normal In 2 Weeks!

What is Insulin Resistance? | Jason Fung

337- Insulin resistance masterclass: The full body impact of metabolic dysfunction, treatment & more