Valvular Heart Disease
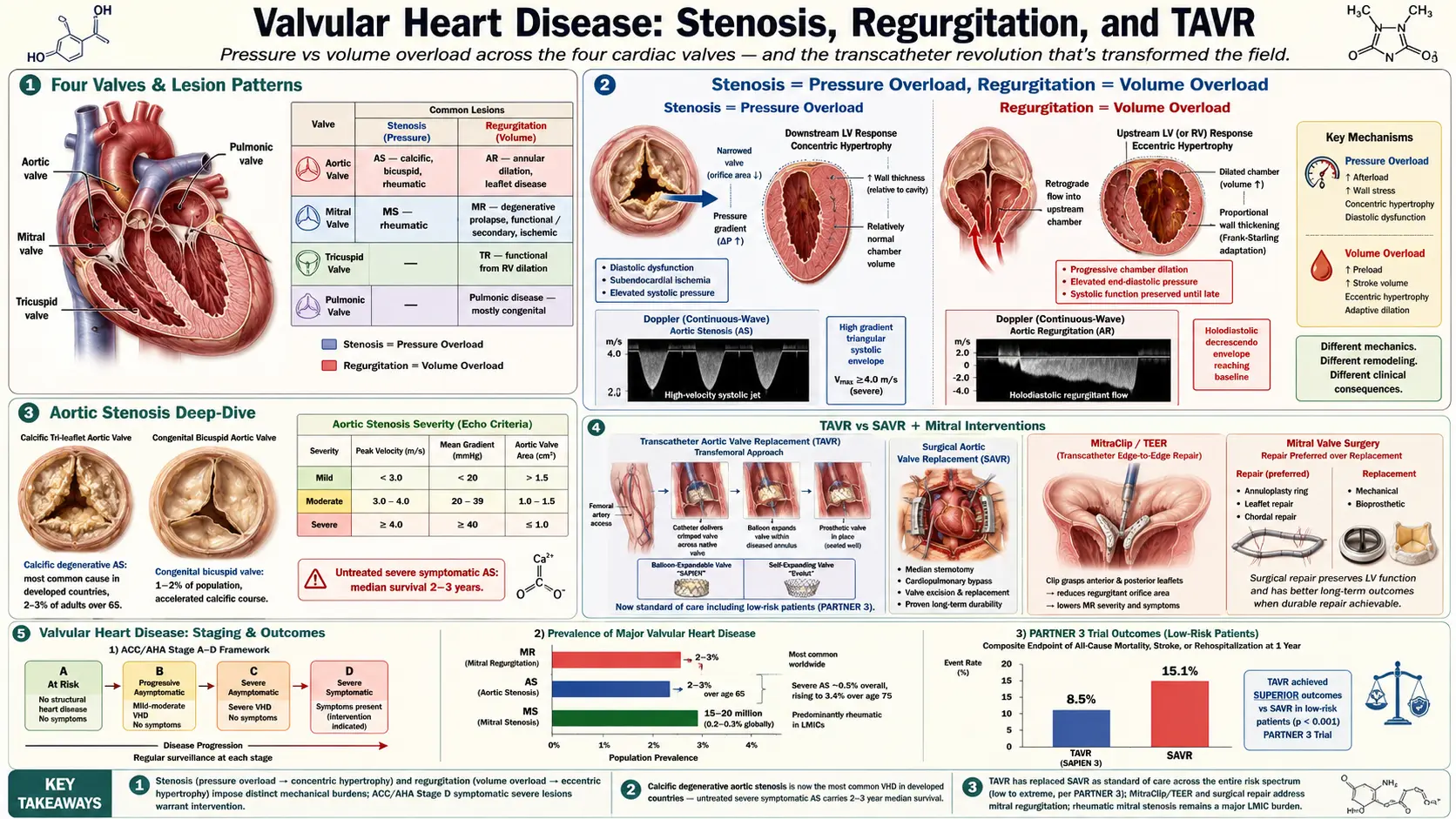
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- ACC/AHA Staging (A–D)
- Aortic Stenosis
- Aortic Regurgitation
- Mitral Stenosis
- Mitral Regurgitation
- Tricuspid and Pulmonic Valve Disease
- Treatment: Interventional and Surgical Indications
- Transcatheter Aortic Valve Replacement (TAVR)
- Complications
- Prognosis
- Prevention
- Recent Research
- Research Papers
- Connections
- Featured Videos
1. Overview
Valvular heart disease (VHD) encompasses a spectrum of structural and functional abnormalities of one or more of the four cardiac valves — the aortic, mitral, tricuspid, and pulmonic valves — that result in impaired cardiac hemodynamics. Lesions are broadly classified as stenosis (obstruction to forward flow due to restricted valve opening) or regurgitation (retrograde flow due to incomplete valve closure), though mixed lesions are common, particularly in rheumatic disease.
VHD imposes chronic hemodynamic burdens on the cardiac chambers, ultimately resulting in myocardial hypertrophy, dilation, dysfunction, and — if untreated — heart failure, arrhythmia, and death. The field has undergone a transformative revolution with the advent of transcatheter technologies, expanding therapeutic options to high-risk and previously inoperable patients. This article provides a comprehensive reference for cardiologists, cardiothoracic surgeons, and internists managing patients with valvular heart disease.
2. Epidemiology
VHD is common and its prevalence rises steeply with age. In population-based studies (Euro Heart Survey, Framingham Heart Study, Helsinki Aging Study), the overall prevalence of moderate or severe VHD is approximately 2.5% in the general adult population, increasing to over 10% in adults over age 75.
- Aortic stenosis (AS): Most common valve lesion in developed countries; affects approximately 2–3% of adults over 65. Calcific degenerative AS is the predominant etiology. Severe AS prevalence is approximately 0.5% overall but rises to 3.4% in those over 75.
- Mitral regurgitation (MR): Most common valve lesion worldwide; prevalence 2–3% in the general population, increasing with age. Degenerative (prolapse, flail leaflet) etiologies predominate in Western populations; functional (secondary) MR is the most common cause in patients with heart failure.
- Mitral stenosis (MS): Predominantly rheumatic in etiology; rare in high-income countries (0.1% prevalence) but highly prevalent in low- and middle-income countries where rheumatic fever remains endemic. Global burden estimated at 15–20 million cases.
- Aortic regurgitation (AR): Moderate or greater prevalence approximately 4.9% (men) and 8.5% (women) in older populations; severe AR prevalence approximately 0.5%.
- Tricuspid regurgitation (TR): Moderate or greater TR present in approximately 0.55% of the general population; severe TR is increasingly recognized as an independent predictor of mortality.
- Pulmonic valve disease: Predominantly congenital in adults; acquired pulmonic stenosis and regurgitation are rare in the absence of prior congenital intervention.
Global mortality from VHD is substantial. Untreated severe symptomatic AS carries a median survival of 2–3 years. VHD is responsible for approximately 25,000 deaths annually in the United States and contributes significantly to hospitalizations for heart failure and atrial fibrillation.
3. Pathophysiology
The pathophysiology of VHD centers on the hemodynamic consequences of abnormal valve function and the resulting compensatory and maladaptive responses of the myocardium.
Stenosis: Pressure Overload
Obstruction to forward flow creates a pressure gradient across the valve. The upstream chamber must generate higher pressure to maintain cardiac output, resulting in concentric hypertrophy (increased wall thickness without chamber dilation). While initially compensatory, sustained pressure overload leads to:
- Diastolic dysfunction from impaired myocardial relaxation
- Subendocardial ischemia (increased oxygen demand, reduced perfusion)
- Progressive systolic dysfunction (afterload mismatch)
- Elevated filling pressures, pulmonary hypertension, right heart failure
Regurgitation: Volume Overload
Incompetent valve closure allows regurgitant flow into the upstream chamber, increasing its volume load. The chamber responds with eccentric hypertrophy (dilation with proportional wall thickening). Compensation maintains forward stroke volume via the Frank-Starling mechanism. Progressive volume overload leads to:
- Chamber dilation and elevated end-diastolic pressure
- Myocardial fiber stretch and eventual systolic dysfunction
- Reduced effective forward cardiac output
- Pulmonary venous hypertension (left-sided lesions) or systemic venous congestion (right-sided lesions)
Neurohormonal Activation
Reduced cardiac output and elevated filling pressures activate the renin-angiotensin-aldosterone system (RAAS) and sympathetic nervous system, leading to sodium and water retention, vasoconstriction, and further cardiac remodeling. Over time, this neurohormonal milieu accelerates myocardial fibrosis and dysfunction, even after valve correction — underscoring the importance of timely intervention before irreversible remodeling occurs.
Pulmonary Hypertension
Chronic elevation of left atrial pressure (from mitral or aortic valve disease) is transmitted to the pulmonary venous and arterial systems, leading to reactive pulmonary hypertension. Initially reversible, pulmonary hypertension may become fixed with pulmonary vascular remodeling, contributing to right ventricular failure and significantly worsening prognosis.
4. Etiology and Risk Factors
Etiologies vary by valve and lesion type, and differ substantially between high-income and low-income countries.
Degenerative / Calcific
The most common cause of AS and AR in adults over 65. Calcific aortic stenosis shares risk factors with atherosclerosis: age, male sex, hypertension, hyperlipidemia, diabetes, smoking, and elevated Lp(a). Calcification begins at the base of the leaflets and progresses centrally, restricting opening. Mitral annular calcification (MAC) may contribute to functional MS and MR. Calcific degenerative changes also affect the mitral leaflets, causing mitral valve prolapse progression.
Rheumatic Heart Disease
Sequela of acute rheumatic fever (ARF) from group A Streptococcus pharyngitis, causing valve leaflet thickening, commissural fusion, and subvalvular apparatus fibrosis. The mitral valve is most commonly affected (MS, MR), followed by the aortic valve (combined AS/AR). Tricuspid involvement occurs in advanced rheumatic disease. Pulmonic valve is rarely affected. ARF primarily afflicts children in low-income countries; long-latency VHD presents in adulthood.
Congenital
- Bicuspid aortic valve (BAV): Present in 1–2% of the population; most common congenital cardiac anomaly. Associated with accelerated AS (1–2 decades earlier than tricuspid valve), aortic regurgitation, and aortopathy.
- Mitral valve prolapse (MVP): Myxomatous degeneration; affects 2–3% of the general population; most common cause of severe primary MR in Western countries.
- Ebstein anomaly: Apical displacement of the tricuspid valve, causing TR and right heart enlargement.
- Congenital pulmonic stenosis: Most common congenital pulmonic lesion; often treated in childhood but may persist or recur.
Infective Endocarditis
Microbial infection of valve leaflets or prosthetic valves causes acute or subacute valvular destruction, perforation, and regurgitation. Staphylococcus aureus, viridans Streptococci, and Enterococcus are most common. Embolic phenomena, abscess formation, and fistula are complications. Urgent surgery is often required for acute severe regurgitation or uncontrolled infection.
Functional (Secondary) Valve Disease
Functional MR arises from global or regional left ventricular remodeling (ischemic or non-ischemic cardiomyopathy) that displaces the papillary muscles, causing leaflet tethering and incomplete coaptation without intrinsic leaflet pathology. Functional TR results from right ventricular dilation and annular enlargement secondary to pulmonary hypertension or left heart disease.
Other Etiologies
- Radiation-induced VHD: Prior mediastinal radiation (e.g., for Hodgkin lymphoma, breast cancer) causes fibrotic thickening of valve leaflets and subvalvular apparatus; often affects multiple valves.
- Carcinoid syndrome: Serotonin-secreting tumors cause fibrotic plaque formation on right-sided valves (tricuspid and pulmonic stenosis/regurgitation).
- Systemic lupus erythematosus (Libman-Sacks endocarditis): Sterile vegetations, predominantly on the mitral and aortic valves.
- Drug-induced: Ergot alkaloids, fenfluramine, cabergoline (high-dose), and methysergide associated with fibrotic valve changes.
- Hypertrophic obstructive cardiomyopathy (HOCM): Dynamic LVOT obstruction and SAM (systolic anterior motion of the mitral valve) causing functional MR.
5. Clinical Presentation
The onset and nature of symptoms reflect the specific lesion, its severity, and the adaptive capacity of the myocardium. Long asymptomatic periods are typical for most chronic valve lesions.
Common Symptom Complexes
- Dyspnea: Exertional dyspnea progressing to rest dyspnea and orthopnea; reflects elevated left-sided filling pressures and pulmonary venous hypertension. Cardinal in AS, MS, MR, and AR.
- Angina: Particularly in AS (supply-demand mismatch from LVH and reduced diastolic perfusion); may occur without obstructive CAD.
- Syncope or presyncope: A hallmark symptom in severe AS (inability to augment cardiac output with exertion, vasodepressor reflex); also in HOCM and critical pulmonic stenosis.
- Palpitations: Atrial fibrillation is common in MS (chronically elevated LA pressure) and late stages of MR and AS.
- Heart failure symptoms: Fatigue, reduced exercise tolerance, lower extremity edema, abdominal fullness.
- Hemoptysis: In MS, elevated pulmonary venous pressure may cause rupture of bronchopulmonary anastomoses (Dieulafoy lesion) or pulmonary edema-related hemoptysis.
- Embolic stroke or TIA: Atrial fibrillation associated with MS or MR; infective endocarditis vegetations; calcific emboli from degenerative AS.
Physical Examination Findings
- Aortic stenosis: Harsh crescendo-decrescendo systolic ejection murmur, best at right upper sternal border, radiating to carotids; parvus et tardus pulse; sustained PMI; absent S2 (A2); S4 gallop.
- Aortic regurgitation: High-pitched blowing early diastolic murmur at left sternal border; wide pulse pressure; Austin Flint murmur; Corrigan's pulse (water-hammer); Quincke's pulse; de Musset's sign.
- Mitral stenosis: Loud S1; opening snap (OS); low-pitched mid-diastolic rumble at apex; the shorter S2-OS interval correlates with higher LA pressure; pre-systolic accentuation in sinus rhythm.
- Mitral regurgitation: Holosystolic murmur at apex radiating to axilla; soft S1; S3 gallop; hyperdynamic PMI displaced laterally.
- Tricuspid regurgitation: Holosystolic murmur at left lower sternal border, increasing with inspiration (Carvallo's sign); elevated JVP with prominent cv waves; hepatic pulsations; peripheral edema.
- Pulmonic stenosis: Crescendo-decrescendo systolic ejection murmur at left upper sternal border; ejection click; widely split S2; right ventricular heave.
6. Diagnosis
Echocardiography is the cornerstone of diagnosis and follow-up for all valve lesions. Additional modalities provide complementary information for complex cases.
Echocardiography
Transthoracic echocardiography (TTE) is the first-line test. It provides:
- Real-time morphologic assessment of leaflets, annulus, subvalvular apparatus, and chamber dimensions
- Doppler-derived hemodynamic parameters (peak/mean gradients, valve area, regurgitant volume, EROA, PISA analysis)
- Assessment of LV/RV size and systolic function (EF, GLS)
- Pulmonary artery pressure estimation (TR velocity method)
- Grading of lesion severity per ASE/EACVI guidelines
Transesophageal echocardiography (TEE) provides superior spatial resolution and is indicated for:
- Inadequate TTE windows
- Detailed pre-procedural planning (mitral valve repair, TAVR, MitraClip)
- Assessment of left atrial appendage thrombus
- Intraoperative monitoring during valve surgery
- Evaluation of prosthetic valve function and endocarditis
Three-dimensional echocardiography (3D-TEE/3D-TTE) enables precise volumetric assessment of valve anatomy, particularly for surgical and transcatheter planning of mitral and tricuspid interventions.
Cardiac Catheterization
Invasive hemodynamic assessment is indicated when noninvasive data are discordant or inconclusive. Direct measurement of intracardiac pressures, cardiac output (Fick or thermodilution), and calculation of valve areas (Gorlin formula) remains the reference standard. Coronary angiography is mandatory preoperatively for patients ≥40 years old (men) or ≥50 years old (women), or in those with known or suspected CAD, to assess concurrent coronary revascularization needs.
Cardiac CT (CCT)
CCT is essential for TAVR planning: annulus sizing, LVOT dimensions, access route assessment, coronary ostia height, and aortic root geometry. Aortic valve calcium scoring (AVC) by CT (Agatston units) provides an objective measure of calcification burden — a threshold of ≥2,000 AU in men and ≥1,200 AU in women reliably predicts severe AS, useful in low-gradient AS.
Cardiac MRI (CMR)
CMR provides accurate volumetric quantification of regurgitant volumes and fractions (particularly for AR and MR when echo data are suboptimal), assessment of myocardial fibrosis via late gadolinium enhancement (LGE; prognostically significant in MR and AS), and evaluation of right heart function. CMR is the reference standard for RV volumetric assessment in pulmonic and tricuspid valve disease.
Exercise Testing
Exercise stress testing is valuable for unmasking symptoms in ostensibly asymptomatic severe VHD (particularly AS and MR). In AS, an abnormal blood pressure response (<20 mmHg rise), ST depression, or symptoms during exercise indicate higher risk and may prompt earlier intervention. Stress echocardiography can reveal dynamic changes in valve area and gradient with exercise.
Natriuretic Peptides
Elevated BNP or NT-proBNP correlates with symptomatic status, severity of hemodynamic derangement, and adverse prognosis in AS and MR. Thresholds are being incorporated into guidelines as markers of clinical decompensation in asymptomatic patients, particularly when BNP exceeds three times the upper limit of normal in asymptomatic severe AS.
7. ACC/AHA Staging (A–D)
The 2014 and 2021 ACC/AHA Valve Guidelines introduced a universal staging system (A–D) applicable to all valve lesions, replacing the previous "mild/moderate/severe" with a framework that integrates anatomy, hemodynamics, symptoms, and response to medical therapy. This system guides management decisions and timing of intervention.
- Stage A — At Risk: Patients with risk factors for development of VHD (e.g., bicuspid aortic valve, prior rheumatic fever, history of endocarditis) but no structural valve changes, hemodynamic abnormalities, or symptoms. Annual cardiovascular assessment; lifestyle and risk factor modification.
- Stage B — Progressive: Mild to moderate valve disease (structural changes present) with hemodynamic consequences that are not yet severe; asymptomatic. Serial echocardiographic surveillance on schedule determined by lesion and severity; medical therapy as appropriate; patient education about symptoms.
- Stage C — Severe Asymptomatic: Severe VHD by hemodynamic criteria (e.g., AVA <1.0 cm², peak velocity ≥4.0 m/s for AS) without symptoms. Two substages:
- Stage C1: Normal LV (or RV) function; no hemodynamic consequence. Close surveillance every 6–12 months; consider intervention when rapid progression, very high gradient, or exercise test abnormalities present.
- Stage C2: Severe VHD with LV (or RV) dysfunction (EF <50–60% depending on lesion) despite absence of symptoms. Intervention recommended even without symptoms due to myocardial damage.
- Stage D — Severe Symptomatic: Severe VHD with symptoms attributable to the valvular lesion (dyspnea, angina, syncope, heart failure). Three substages in AS:
- Stage D1: High-gradient severe AS (mean gradient ≥40 mmHg, AVA <1.0 cm², peak velocity ≥4.0 m/s) with symptoms. AVR recommended (Class I).
- Stage D2: Low-flow, low-gradient severe AS with reduced EF (<50%); stroke volume index <35 mL/m². Dobutamine stress echo or CT calcium scoring to confirm true severity; AVR recommended if confirmed severe.
- Stage D3: Low-gradient severe AS with preserved EF (paradoxical low-flow, low-gradient AS); typically seen in small, hypertrophied ventricles with reduced stroke volume despite preserved EF. AVR may be considered if clinical, anatomic, and hemodynamic data confirm severity.
Analogous staging criteria apply to each valve lesion, with lesion-specific hemodynamic thresholds defining each stage.
8. Aortic Stenosis
Aortic stenosis is characterized by progressive calcification and restricted opening of the aortic valve leaflets, leading to fixed obstruction of left ventricular outflow. It is the most common severe VHD in adults in high-income countries.
Hemodynamic Criteria for Severity
- Mild AS: Peak velocity 2.0–2.9 m/s; mean gradient <20 mmHg; AVA >1.5 cm²
- Moderate AS: Peak velocity 3.0–3.9 m/s; mean gradient 20–39 mmHg; AVA 1.0–1.5 cm²
- Severe AS: Peak velocity ≥4.0 m/s; mean gradient ≥40 mmHg; AVA <1.0 cm² (indexed AVA <0.6 cm²/m²)
- Very severe AS: Peak velocity ≥5.0 m/s; mean gradient ≥60 mmHg
Natural History and Symptoms
AS progresses at a rate of approximately 0.1–0.3 cm²/year and 5–7 mmHg/year in mean gradient. The asymptomatic phase may last decades; once symptoms develop, prognosis worsens rapidly. The classic triad of symptoms — angina, syncope, and dyspnea/heart failure — carries median survivals of 5, 3, and 2 years respectively without intervention. Sudden cardiac death occurs in approximately 1% per year in asymptomatic patients, increasing to 8–34% per year once symptoms appear.
Indications for Aortic Valve Replacement (AVR)
- Stage D (symptomatic severe AS): AVR strongly recommended regardless of intervention modality (Class I).
- Stage C2 (asymptomatic severe AS with LV dysfunction, EF <50%): AVR recommended (Class I).
- Stage C1, very severe AS (peak velocity ≥5.0 m/s) with low surgical risk: AVR reasonable (Class IIa).
- Stage C1 with abnormal exercise test (symptoms, BP drop): AVR reasonable (Class IIa).
- Stage C1 with rapid progression (>0.3 m/s per year) and low risk: AVR may be considered (Class IIb).
- Elevated BNP (>3× ULN) in asymptomatic severe AS: AVR may be considered (Class IIb; 2021 guidelines).
- Concomitant AVR at time of other cardiac surgery: Class I for severe AS, Class IIa for moderate AS.
9. Aortic Regurgitation
Aortic regurgitation results from failure of the aortic valve leaflets to coapt during diastole, allowing retrograde blood flow from the aorta into the left ventricle. Etiologies include leaflet pathology (bicuspid valve, endocarditis, rheumatic disease, prolapse) and aortic root dilation (Marfan syndrome, Loeys-Dietz syndrome, aortitis, hypertensive aortopathy).
Acute vs. Chronic AR
Acute severe AR (from endocarditis, aortic dissection, or trauma) is hemodynamically catastrophic. The unprepared LV cannot accommodate the sudden volume overload; LV end-diastolic pressure rises markedly, causing pulmonary edema and cardiogenic shock. Emergency surgical intervention is required. Physical examination may reveal a soft murmur (equalization of aortic and LV diastolic pressure) and sinus tachycardia rather than the classic findings of chronic AR.
Chronic severe AR is well tolerated for long periods via eccentric hypertrophy and increased stroke volume. Symptoms (dyspnea, reduced exercise tolerance) and LV dysfunction may develop insidiously.
Hemodynamic Criteria for Severity
- Mild AR: Regurgitant fraction <30%; EROA <0.10 cm²; regurgitant volume <30 mL
- Moderate AR: Regurgitant fraction 30–49%; EROA 0.10–0.29 cm²; regurgitant volume 30–59 mL
- Severe AR: Regurgitant fraction ≥50%; EROA ≥0.30 cm²; regurgitant volume ≥60 mL; PHT <200 ms; holodiastolic aortic flow reversal in descending aorta
Indications for AVR in Chronic AR
- Stage D (symptomatic severe AR): AVR recommended regardless of LV function (Class I).
- Stage C2 (asymptomatic severe AR with LV dysfunction, EF <55%): AVR recommended (Class I).
- Stage C1 (asymptomatic severe AR, preserved EF) with other cardiac surgery planned: Concomitant AVR recommended (Class I).
- Asymptomatic severe AR with progressive LV dilation (LVESD >50 mm or LVEDD >65 mm) and low surgical risk: AVR reasonable (Class IIa).
- Ascending aorta ≥5.5 cm (or ≥5.0 cm in connective tissue disorder) with severe AR: AVR with concomitant aortic repair recommended.
10. Mitral Stenosis
Mitral stenosis is defined as obstruction to left ventricular inflow at the mitral valve level, creating a diastolic gradient between the left atrium and left ventricle. Rheumatic etiology causes commissural fusion, leaflet thickening, and chordal shortening with subvalvular fusion. Non-rheumatic MS may result from severe MAC, congenital parachute mitral valve, or radiation injury.
Hemodynamic Criteria for Severity
- Mild MS: MVA >1.5 cm²; mean gradient <5 mmHg; PASP <30 mmHg
- Moderate MS: MVA 1.0–1.5 cm²; mean gradient 5–10 mmHg; PASP 30–50 mmHg
- Severe MS: MVA <1.0 cm² (indexed <0.6 cm²/m²); mean gradient >10 mmHg; PASP >50 mmHg
Wilkins Score
The Wilkins echocardiographic score assesses leaflet mobility, subvalvular thickening, leaflet thickening, and calcification, each scored 1–4 (total 4–16). Score ≤8 predicts favorable response to percutaneous mitral balloon commissurotomy (PMBC); score >8 (particularly with calcification or significant MR) favors surgical repair or replacement.
Atrial Fibrillation in Mitral Stenosis
Chronic LA hypertension causes progressive LA dilation and fibrosis, predisposing to AF in up to 40–60% of significant MS cases. AF dramatically worsens hemodynamics by eliminating atrial kick and shortening diastolic filling time, often precipitating acute pulmonary edema. AF in rheumatic MS carries extremely high thromboembolic risk; anticoagulation with vitamin K antagonists (VKA, target INR 2–3) is the standard of care (DOACs are inferior in rheumatic MS, per INVICTUS trial).
Indications for Intervention in MS
- Symptomatic severe MS (Stage D), favorable morphology: PMBC recommended (Class I).
- Symptomatic severe MS, unfavorable morphology (Wilkins >8, significant MR, LA thrombus): Mitral valve surgery (repair or replacement) recommended (Class I).
- Asymptomatic very severe MS (MVA <1.0 cm²) with favorable morphology: PMBC reasonable (Class IIa).
- Symptomatic moderate MS (MVA 1.5 cm²) with elevated pressures (PCWP ≥25 mmHg with exercise): PMBC reasonable (Class IIa).
- New-onset AF in severe MS: PMBC or surgery reasonable to restore sinus rhythm (Class IIa).
11. Mitral Regurgitation
Mitral regurgitation is the most prevalent valve lesion in high-income countries. It is classified as primary (degenerative) — due to intrinsic leaflet pathology (prolapse, flail leaflet, rheumatic disease, endocarditis) — or secondary (functional) — due to LV remodeling with structurally normal leaflets (ischemic MR, dilated cardiomyopathy).
Hemodynamic Criteria for Severity
- Mild MR: Regurgitant volume <30 mL; regurgitant fraction <30%; EROA <0.20 cm²
- Moderate MR: Regurgitant volume 30–59 mL; regurgitant fraction 30–49%; EROA 0.20–0.39 cm²
- Severe MR: Regurgitant volume ≥60 mL; regurgitant fraction ≥50%; EROA ≥0.40 cm²
Primary vs. Secondary MR Management
Primary severe MR: Surgical mitral valve repair (preferred over replacement when feasible) or replacement. Repair is superior, providing lower operative mortality, better LV function preservation, and freedom from anticoagulation.
Indications for surgery in primary MR:
- Symptomatic severe primary MR with EF >30% (Class I)
- Asymptomatic severe primary MR with LV dysfunction (EF 30–60% or LVESD ≥40 mm) — Class I
- Asymptomatic severe primary MR, preserved EF, when durable repair is highly likely (>95%) at a Heart Valve Center of Excellence — Class IIa
- Asymptomatic severe primary MR with new-onset AF or PASP >50 mmHg — Class IIa
Secondary (functional) severe MR: Primarily treated with guideline-directed medical therapy (GDMT) for underlying heart failure (RAAS inhibitors, beta-blockers, device therapy including CRT). When MR persists and contributes to symptoms despite optimal GDMT:
- MitraClip (transcatheter edge-to-edge repair, TEER): For patients with HFrEF (EF 20–50%), moderate-to-severe or severe secondary MR, meeting COAPT criteria (primary MR excluded, severely enlarged annulus excluded, favorable anatomy), on GDMT with persistent symptoms (NHYA Class II–IVa) — reduces hospitalizations and mortality (COAPT trial).
- Surgical mitral repair or replacement in secondary MR: Considered for patients undergoing other cardiac surgery; isolated surgical intervention for secondary MR has not demonstrated mortality benefit over GDMT alone (MIST, RIME trials).
12. Tricuspid and Pulmonic Valve Disease
Tricuspid Regurgitation
Tricuspid regurgitation (TR) is the most common right-sided valve lesion. Isolated primary TR is rare; most cases are secondary (functional), caused by RV dilation and tricuspid annular enlargement from pulmonary hypertension, left heart disease, AF, or prior RV infarction. Primary TR etiologies include rheumatic disease, carcinoid syndrome, endocarditis (particularly in IV drug users), Ebstein anomaly, and pacemaker/ICD lead trauma.
Severe TR causes systemic venous hypertension, right heart failure, hepatic congestion, ascites, and peripheral edema. Neurohormonal activation and reduced effective cardiac output contribute to progressive deterioration. Severe TR is an independent predictor of mortality even after correction of the primary left-sided lesion.
Management:
- Diuresis for symptomatic fluid overload
- Treatment of underlying cause (pulmonary hypertension management, correction of left-sided VHD)
- Tricuspid valve repair (annuloplasty) at time of left-sided valve surgery strongly recommended for severe TR or significant annular dilation (≥40 mm or ≥21 mm/m²) — Class I
- Isolated tricuspid valve surgery for severe TR with persistent symptoms despite GDMT — Class IIa
- Emerging transcatheter tricuspid interventions (TRILUMINATE, CLASP TR, TRISCEND II) are under active investigation; FDA approval for select devices obtained or pending
Pulmonic Stenosis
Most commonly congenital (dome-shaped fusion of leaflets in typical pulmonic stenosis), pulmonic stenosis causes RV pressure overload, hypertrophy, and eventual RV failure. Mild PS is well tolerated throughout life; severe PS (peak gradient >64 mmHg) requires intervention. Balloon pulmonary valvuloplasty (BPV) is the treatment of choice for typical congenital pulmonic stenosis with peak gradient ≥50 mmHg regardless of symptoms (Class I) or 40–49 mmHg with symptoms (Class IIa). Surgical valvuloplasty or replacement is reserved for dysplastic valves or failed BPV.
Pulmonic Regurgitation
Most commonly encountered as a consequence of prior repair of congenital pulmonic stenosis or tetralogy of Fallot. Severe PR causes progressive RV dilation and dysfunction. Indications for pulmonary valve replacement (PVR) include symptoms, RV dilation (indexed RV end-diastolic volume >150 mL/m² by CMR), or RV systolic dysfunction (EF <40%). Transcatheter pulmonary valve replacement (Melody, Sapien valve in the pulmonary position) is preferred for patients with conduit or bioprosthesis in the pulmonary position.
13. Treatment: Interventional and Surgical Indications
Principles of Timing
The fundamental principle guiding intervention timing is to intervene before irreversible myocardial damage or clinical decompensation occurs, while avoiding unnecessary early intervention in asymptomatic patients with good LV function. The introduction of TAVR and other transcatheter therapies has shifted the risk-benefit calculation for high-risk surgical patients.
Surgical Aortic Valve Replacement (SAVR)
SAVR remains the gold standard for low- and intermediate-risk patients with severe AS or AR. Mechanical valves are preferred in patients under 50–60 years (lifelong anticoagulation required), while bioprosthetic valves are appropriate for older patients or those with contraindications to anticoagulation. The Ross procedure (native pulmonary autograft to aortic position) is an option in young patients at experienced centers. Valve-in-valve TAVR at time of bioprosthetic structural valve deterioration (SVD) is a key consideration influencing initial valve choice.
Mitral Valve Surgery
Mitral valve repair is strongly preferred over replacement for primary MR when feasible. Repair techniques include annuloplasty ring implantation, leaflet resection (P2 quadrangular resection), chordal transfer or neochordae (ePTFE sutures), and commissurotomy. Repair rates exceeding 95% for posterior leaflet prolapse should be achievable at experienced centers. Mitral valve replacement (mechanical or bioprosthetic, with preservation of subvalvular apparatus) is required when repair is not feasible or durable.
Mechanical vs. Bioprosthetic Valves
- Mechanical valves: Excellent long-term durability (decades); require lifelong anticoagulation (warfarin, target INR 2.5–3.5 for mitral, 2.0–3.0 for aortic); preferred under age 50 (aortic) or 35 (mitral) per ACC/AHA guidelines; higher risk of thromboembolic and bleeding complications.
- Bioprosthetic valves: No anticoagulation required long-term (aspirin only); SVD occurs at a rate of approximately 1–3%/year after 10–15 years; preferred in older patients or those unwilling/unable to manage anticoagulation; valve-in-valve TAVR/TMVR feasible at SVD.
- Patient preference, age, lifestyle, and comorbidities guide individualized choice after shared decision-making.
Antithrombotic Therapy After Valve Repair or Replacement
- Bioprosthetic SAVR: Aspirin 75–100 mg; VKA for 3–6 months post-implant (reasonable, Class IIb)
- Post-TAVR without AF: Aspirin 75–100 mg (low-risk) or aspirin + clopidogrel for 3–6 months (intermediate/high-risk) depending on individual risk
- Mechanical valve: VKA (warfarin) indefinitely; DOACs are contraindicated (RE-ALIGN trial demonstrated harm with dabigatran)
- Post-mitral repair: VKA for 3 months, then aspirin alone if no other indication for anticoagulation
14. Transcatheter Aortic Valve Replacement (TAVR)
Transcatheter aortic valve replacement represents the most significant paradigm shift in the treatment of aortic stenosis since the development of open surgical valve replacement. First performed by Alain Cribier in 2002, TAVR has evolved from a compassionate therapy for inoperable patients to the dominant treatment modality for severe AS across all surgical risk categories.
Device Platforms
Two major self-expanding and balloon-expandable valve platforms dominate clinical practice:
- Edwards SAPIEN series (balloon-expandable): SAPIEN 3, SAPIEN 3 Ultra — delivered via transfemoral, transapical, transaortic, or transaxillary routes; outer skirt reduces paravalvular leak.
- Medtronic CoreValve series (self-expanding): CoreValve Evolut R, Evolut Pro, Evolut Pro+ — higher radial force; supra-annular leaflet position (larger effective orifice area); repositionable; retrievable before full deployment.
- Emerging platforms: JenaValve (for AR), Tendyne (transcatheter mitral), and others.
Access Routes
- Transfemoral (TF): Preferred approach (>90% of cases); associated with lowest mortality; now predominantly performed percutaneously.
- Transaxillary/subclavian: Alternative when femoral access is inadequate.
- Transaortic: Direct aortic access via mini-sternotomy or right anterior thoracotomy.
- Transapical: Left ventricular apical puncture via mini-thoracotomy; reserved for rare cases; associated with higher morbidity.
- Transcaval: Investigational; percutaneous access through the IVC into the aorta.
Pivotal Trials and Expanding Indications
- PARTNER 1B (2010): Inoperable patients — TAVR reduced 1-year mortality by 46% vs. medical therapy; established TAVR for inoperable AS.
- PARTNER 1A (2011): High-risk patients — TAVR non-inferior to SAVR at 1 year (24.2% vs. 26.8%); stroke higher with TAVR.
- PARTNER 2A / SURTAVI (2016): Intermediate-risk — TAVR non-inferior to SAVR at 2 years; established TAVR for intermediate risk.
- PARTNER 3 / Evolut Low Risk (2019): Low-risk patients — TAVR superior or non-inferior to SAVR at 1–2 years; landmark trials enabling FDA approval for low-risk patients.
- 5-year PARTNER 3 data (2024): Non-inferiority maintained; valve durability reassuring though longer follow-up required for definitive durability data.
TAVR Complications
- Permanent pacemaker implantation (PPM): Most common complication; 5–25% depending on device (self-expanding higher); due to conduction system injury from valve deployment in calcified LVOT.
- Paravalvular leak (PVL): More than mild PVL associated with increased mortality; modern devices with outer skirts have reduced PVL rates to <5%.
- Stroke: Neurologic events 2–4%; cerebral embolic protection devices (Sentinel) reduce captured debris but have not demonstrated significant clinical stroke reduction in trials.
- Vascular access complications: Significantly reduced with percutaneous technique and improved sheath technology.
- Coronary obstruction: Rare (<1%); occurs when displaced native leaflet occludes coronary ostium; risk predicted by CT (HALT, BASILICA technique for prevention).
- Subclinical leaflet thrombosis (HALT): Hypoattenuating leaflet thickening detected on CT in 10–15%; treated with anticoagulation; clinical significance under investigation (ATLANTIS, AUREA trials).
- Structural valve deterioration (SVD): Rates at 5 years comparable to surgical bioprostheses; longer follow-up data needed for younger patients.
TAVR for Aortic Regurgitation
Dedicated TAVR devices for pure aortic regurgitation without calcification (which provides anchoring for standard TAVR) are under development (JenaValve). TAVR-in-failed SAVR bioprosthesis (valve-in-valve TAVR) is established and widely used.
Transcatheter Mitral and Tricuspid Interventions
- MitraClip / PASCAL (TEER): FDA-approved for primary MR (high surgical risk) and secondary MR (COAPT criteria). Reduces regurgitation via edge-to-edge leaflet approximation.
- Transcatheter mitral valve replacement (TMVR): Tendyne, Intrepid, and other devices in trials for primary and secondary MR.
- Transcatheter tricuspid valve repair/replacement: TRILUMINATE Pivotal trial (TriClip) FDA-approved for symptomatic severe TR in high surgical risk patients (2023); TRISCEND II (EVOQUE transcatheter tricuspid replacement) with pivotal trial results anticipated.
15. Complications
Complications arise from the natural history of untreated VHD or as consequences of intervention.
Disease-Related Complications
- Heart failure: The terminal pathway for all severe, untreated valve lesions; acute decompensation may be precipitated by AF, infection, pregnancy, or anemia.
- Atrial fibrillation: Extremely common in MS and MR; increases thromboembolic risk and worsens hemodynamics.
- Thromboembolic events: Stroke, TIA, peripheral embolism; related to AF, LA enlargement, and low-flow states.
- Pulmonary hypertension and right heart failure: Progressive and potentially irreversible if intervention is delayed.
- Infective endocarditis: Risk is higher in abnormal native valves and prosthetic valves; requires prolonged antibiotic therapy and often urgent surgery.
- Sudden cardiac death: Risk is highest in symptomatic AS and HOCM; rare in primary MR.
- Aortic dissection: In patients with BAV aortopathy or Marfan syndrome.
Post-Surgical Complications
- Prosthetic valve endocarditis (PVE): Early (<60 days, nosocomial organisms) vs. late (>60 days, community organisms); requires prolonged antibiotics and often surgical re-intervention.
- Prosthetic valve thrombosis: Acute or subacute; presents with hemodynamic deterioration, embolism, or both; treat with fibrinolysis or surgical thrombectomy.
- Structural valve deterioration (SVD): Calcification and leaflet tear of bioprosthetic valves; may be treated with valve-in-valve TAVR.
- Paravalvular leak: Technical complication of valve implantation; may require re-intervention.
- Patient-prosthesis mismatch (PPM): Prosthetic orifice area insufficient relative to patient body size; associated with worse outcomes; indexed EOA <0.85 cm²/m² (severe PPM <0.65 cm²/m²).
- Hemolytic anemia: From turbulent flow across a regurgitant prosthesis.
- Complete heart block requiring pacemaker.
- Aortic root disruption (rare, catastrophic): During SAVR in heavily calcified root.
16. Prognosis
Prognosis depends on valve lesion, severity, timing of intervention, LV function at time of repair, and comorbid conditions.
- Symptomatic severe AS: Without AVR, median survival 2–5 years (class-dependent); with SAVR or TAVR, survival approaches age-matched controls in low-risk patients.
- Severe primary MR after successful repair: Excellent long-term outcome; 10-year survival >85% at experienced centers; repair superior to replacement (lower mortality, better LV function preservation).
- Severe MR with preoperative LV dysfunction (EF <30–40%): Substantially worse outcomes even after successful repair; early referral before dysfunction develops is critical.
- Rheumatic MS after PMBC: Freedom from reintervention approximately 50–70% at 10 years; favorable in younger patients with low Wilkins scores.
- Chronic severe AR with preserved LV function: Annual rate of symptom development or LV dysfunction approximately 4–6%; prognosis good with timely AVR.
- Secondary MR after MitraClip (COAPT criteria): Significant reduction in heart failure hospitalizations (47%) and all-cause mortality (29%) vs. medical therapy at 2 years.
- Severe TR: Associated with 50% 5-year mortality even after adjustment for left-sided VHD and other comorbidities; isolated surgical TR repair carries high mortality, driving interest in transcatheter approaches.
- TAVR in low-risk patients (PARTNER 3, 5 years): Freedom from death, stroke, or rehospitalization approximately 72%; outcomes continue to be evaluated at longer follow-up to assess valve durability.
17. Prevention
Prevention strategies target modifiable risk factors for VHD development and progression, as well as specific prophylactic measures for high-risk conditions.
Primary Prevention of Rheumatic Heart Disease
- Prompt antibiotic treatment of group A streptococcal pharyngitis (penicillin or amoxicillin for 10 days) reduces risk of acute rheumatic fever by >80%.
- Secondary prophylaxis with long-acting benzathine penicillin (IM every 3–4 weeks) for all patients with documented ARF or rheumatic heart disease; duration varies from 10 years (no carditis, no residual RHD) to lifelong (severe RHD or recurrent ARF).
- Global access to antibiotic treatment and rheumatic fever registries remain key public health priorities.
Prevention of Calcific Aortic Stenosis Progression
- No pharmacologic therapy has demonstrated slowing of AS progression in randomized trials (SALTIRE, SEAS, ASTRONOMER trials of statins were negative).
- Aggressive cardiovascular risk factor management remains standard of care for coexistent conditions.
- Lp(a) reduction (LPACIFYIN trial underway) and RNA therapeutics targeting Lp(a) hold promise as future disease-modifying therapies for AS.
Infective Endocarditis Prophylaxis
Current ACC/AHA and ESC guidelines restrict antibiotic prophylaxis before dental procedures to the highest-risk groups:
- Prosthetic heart valves (mechanical or bioprosthetic)
- Prior infective endocarditis
- Congenital heart disease (unrepaired cyanotic CHD, completely repaired with prosthetic material within 6 months, repaired with residual defects)
- Cardiac transplant recipients with valvulopathy
Standard regimen: Amoxicillin 2 g orally 30–60 minutes before dental procedure; clindamycin or azithromycin for penicillin-allergic patients. Prophylaxis is not recommended for degenerative or rheumatic native valve disease in the absence of prior endocarditis.
Surveillance and Timely Referral
Regular echocardiographic surveillance of known valve disease, prompt referral to a cardiac surgeon or structural heart program at the appropriate stage, and shared decision-making at a Heart Valve Center of Excellence are essential elements of comprehensive VHD management to ensure optimal timing of intervention before irreversible harm occurs.
18. Recent Research
- TAVR for low-risk patients — Long-term durability: Five-year follow-up data from PARTNER 3 and Evolut Low Risk confirm sustained favorable outcomes; bioprosthetic SVD rates comparable to SAVR. Longer follow-up (10+ years) essential before universal TAVR adoption in young patients.
- Transcatheter tricuspid valve therapies: TRILUMINATE Pivotal trial (2023) led to FDA approval of the TriClip system for symptomatic severe TR in high surgical risk patients. TRISCEND II (EVOQUE replacement valve) pivotal trial results demonstrate promising improvement in TR severity and quality of life. Multiple devices (CLASP TR, PASCAL Ace, Intrepid Tricuspid) in active trials.
- AVATAR trial (2022): Randomized trial demonstrating benefit of early aortic valve replacement vs. watchful waiting in asymptomatic severe AS with normal EF and exercise tolerance — supporting a shift toward earlier intervention in select patients.
- Lp(a) and calcific AS: Mendelian randomization studies confirm causal role of Lp(a) in AS. LPACIFYIN trial (pelacarsen, an Lp(a)-lowering antisense oligonucleotide) is the first prospective randomized trial to test pharmacologic slowing of AS progression.
- CLASP IID/IIF trials (PASCAL TEER): Demonstrate safety and efficacy of the PASCAL edge-to-edge repair system for primary and secondary MR; FDA approved in 2022 for primary MR.
- INVICTUS trial (2022): Landmark trial demonstrating that rivaroxaban (DOAC) is inferior to VKA (warfarin) in patients with rheumatic MS and AF, confirming VKA as the standard of care for this population.
- Transcatheter mitral valve replacement (TMVR): Tendyne valve achieved 30-day CE mark approval data with acceptable outcomes; Intrepid pivotal trial ongoing; anticipated to address patients with complex mitral anatomy unsuitable for repair or TEER.
- Concomitant tricuspid repair at left-sided surgery: TRILUMINATE MVARC data and registry analyses confirm improved outcomes when TR is addressed at time of mitral or aortic surgery, supporting guideline recommendations for concomitant repair.
- Artificial intelligence in VHD detection: AI-enabled ECG and echocardiogram analysis achieves cardiologist-level accuracy in detecting moderate-to-severe AS and AR; machine learning models are being validated for automated grading and surveillance scheduling.
- Subclinical leaflet thrombosis after TAVR (HALT): ATLANTIS trial showed superiority of anticoagulation over antiplatelet therapy in reducing HALT on CT imaging; clinical outcomes data from ongoing trials will clarify optimal antithrombotic strategy post-TAVR.
Research Papers
The following PubMed topic searches return current peer-reviewed literature relevant to this condition. Each link opens a live PubMed query.
- Aortic stenosis
- Aortic regurgitation
- Mitral regurgitation
- Mitral stenosis
- Transcatheter aortic valve replacement (TAVR)
- Mitral valve repair
- Valvular heart disease guidelines
- Rheumatic heart disease
- Tricuspid regurgitation
- Echocardiography valvular heart disease
- Bicuspid aortic valve
- Valvular heart disease mortality
Connections
- Endocarditis
- Heart Failure
- Hypertension
- Atrial Fibrillation
- Cardiomyopathy
- Arrhythmia
- Stroke
- Pulmonary Hypertension
- Edema
- Fatigue
- Shortness of Breath
- Complete Blood Count
- Calcium
- Omega-3 Fatty Acids
- Magnesium
- Vitamin K
- Anemia
- Cardiovascular Disease