Pulmonary Hypertension
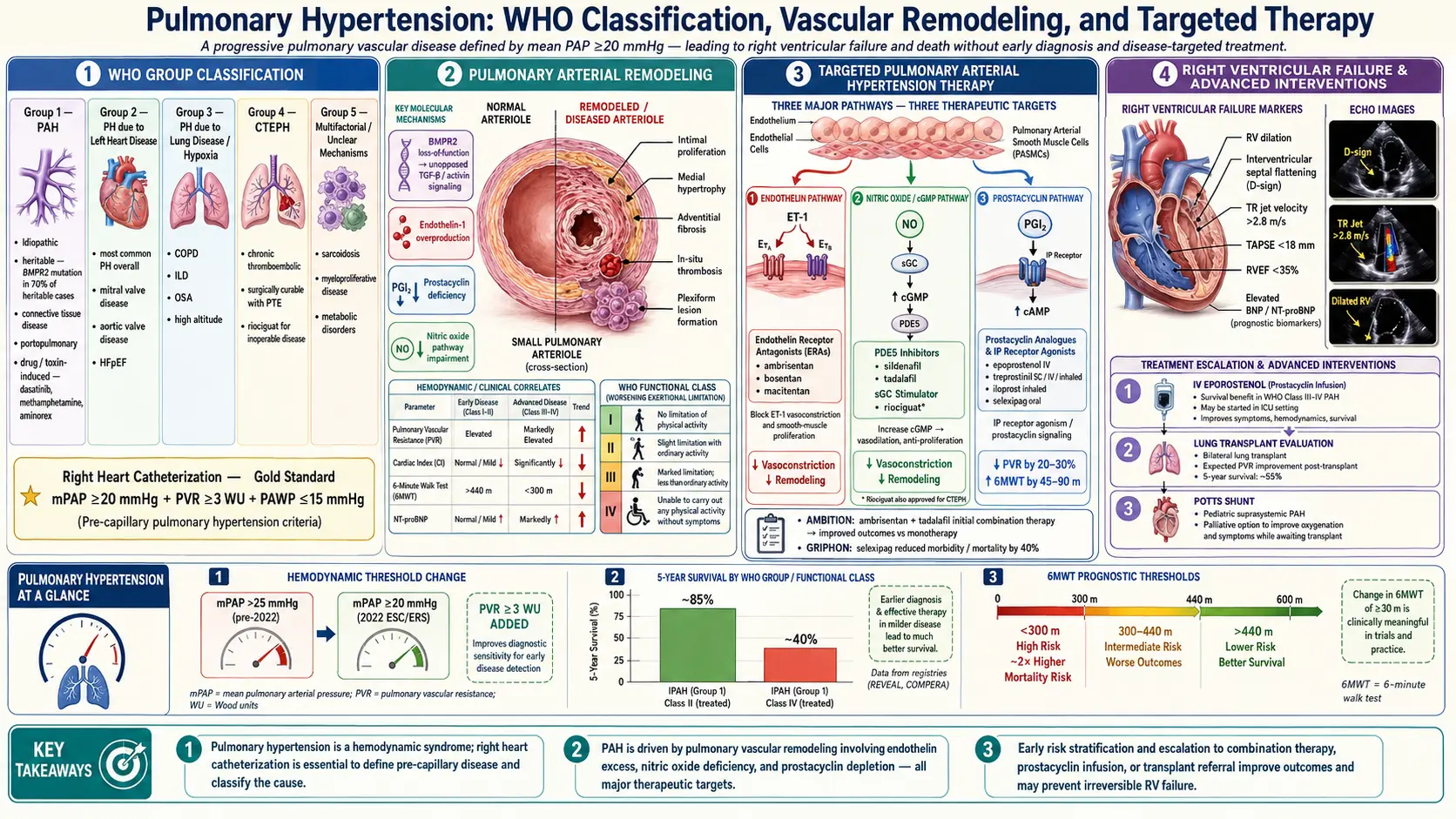
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors — WHO Classification
- Clinical Presentation — WHO Functional Class
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- Research Papers
- Connections
- Featured Videos
1. Overview
Pulmonary hypertension (PH) is a hemodynamic and pathophysiological condition defined as a mean pulmonary arterial pressure (mPAP) >20 mmHg at rest, measured by right heart catheterization. It is a common endpoint of diverse diseases affecting the pulmonary vasculature, cardiac chambers, lung parenchyma, or ventilatory control. Left untreated, progressive pulmonary vascular resistance leads to right ventricular failure and death. Pulmonary arterial hypertension (PAH), the most extensively studied subgroup, is defined additionally by a pulmonary arterial wedge pressure ≤15 mmHg and pulmonary vascular resistance (PVR) >2 Wood units (WU), indicating a pre-capillary etiology. The 2022 ESC/ERS guidelines updated the mPAP threshold from 25 to 20 mmHg, recognizing that even mildly elevated pressures confer increased morbidity and mortality.
2. Epidemiology
PH affects an estimated 1% of the global population, rising to 10% among those over 65 years of age, primarily driven by left heart disease and lung disease (WHO Groups 2 and 3). Idiopathic PAH (IPAH), the most studied form, has an estimated incidence of 2–5 cases per million per year and a prevalence of 15–50 per million. Women are disproportionately affected by PAH at a ratio of approximately 2–4:1 over men, though males tend to have worse RV adaptation and prognosis at diagnosis.
Connective tissue disease-associated PAH (CTD-PAH), particularly systemic sclerosis (SSc), is one of the most prevalent forms of Group 1 PAH, affecting 8–12% of SSc patients and carrying a 3-year mortality of ~50%. Heritable PAH (HPAH) accounts for ~15–20% of apparently idiopathic cases, with BMPR2 mutations identified in ~80% of HPAH and ~20% of IPAH patients. The French National Registry documented a median survival from diagnosis of 2.8 years before the modern treatment era, substantially improved with contemporary targeted therapies.
3. Pathophysiology
The pathobiology of PAH involves a complex interplay of vasoconstriction, vascular remodeling, inflammation, and thrombosis in situ. Three principal pathways are dysregulated:
- Nitric oxide (NO) pathway: Reduced endothelial NO synthase (eNOS) activity and decreased NO bioavailability impair vasorelaxation. Phosphodiesterase type 5 (PDE5) degrades cyclic GMP (cGMP); PDE5 inhibitors restore this pathway.
- Prostacyclin pathway: Decreased prostacyclin synthase expression reduces PGI2 production, tipping the balance toward vasoconstriction and platelet aggregation (excess thromboxane A2).
- Endothelin-1 (ET-1) pathway: ET-1 is markedly upregulated, acting on ETA receptors (vasoconstriction, smooth muscle proliferation) and ETB receptors (vasodilation, clearance). Endothelin receptor antagonists target this pathway.
Histologically, PAH is characterized by plexiform lesions — disordered angioproliferative lesions arising from monoclonal expansion of endothelial cells, medial hypertrophy (smooth muscle cell proliferation), intimal fibrosis, adventitial thickening, and in situ thrombosis. The net effect is progressive elevation of PVR.
The right ventricle adapts initially via concentric hypertrophy (adaptive remodeling), maintaining cardiac output. As PVR continues to rise, the RV dilates (maladaptive remodeling), tricuspid regurgitation develops, interventricular septal deviation compresses the LV, and cardiac output falls — the final common pathway of right heart failure.
In Group 2 PH (left heart disease), elevated left atrial pressure is transmitted retrogradely; initially passive (postcapillary PH), it may trigger reactive pulmonary vasoconstriction and remodeling (combined pre- and postcapillary PH, CpcPH).
4. Etiology and Risk Factors — WHO Classification
The updated WHO Clinical Classification (Nice 2018/ESC 2022) organizes PH into five groups:
Group 1 — Pulmonary Arterial Hypertension (PAH)
- Idiopathic PAH (IPAH)
- Heritable PAH: BMPR2 (most common), ALK1, ENG, SMAD9, CAV1, KCNK3 mutations
- Drug- and toxin-induced: anorexigens (aminorex, fenfluramine), methamphetamine, dasatinib, interferon
- Associated with CTD (SSc, SLE, MCTD, RA), HIV infection, portal hypertension, congenital heart disease, schistosomiasis
Group 2 — PH Due to Left Heart Disease
- Heart failure with preserved ejection fraction (HFpEF) — most prevalent cause of PH overall
- Heart failure with reduced ejection fraction (HFrEF)
- Valvular heart disease (mitral stenosis/regurgitation, aortic stenosis)
Group 3 — PH Due to Lung Disease and/or Hypoxia
- COPD, interstitial lung disease, combined pulmonary fibrosis and emphysema
- Sleep-disordered breathing, alveolar hypoventilation disorders
- Chronic exposure to high altitude
Group 4 — Chronic Thromboembolic Pulmonary Hypertension (CTEPH)
- Organized, non-resolving thromboemboli causing mechanical obstruction and secondary vasculopathy
- Affects ~4% of PE survivors
Group 5 — PH with Unclear or Multifactorial Mechanisms
- Hematologic disorders (chronic hemolytic anemia, myeloproliferative neoplasms, splenectomy)
- Systemic and metabolic disorders (sarcoidosis, pulmonary Langerhans cell histiocytosis, fibrosing mediastinitis, metabolic syndrome)
- Complex congenital heart disease
5. Clinical Presentation — WHO Functional Class
Symptoms develop insidiously; the diagnosis is often delayed by 2–3 years. Early PH is clinically silent or mimics more common conditions.
Symptoms:
- Exertional dyspnea (most common presenting symptom; ~60%)
- Fatigue, weakness
- Exertional syncope or pre-syncope (poor prognostic sign)
- Chest pain (RV ischemia, PA distension)
- Palpitations
- Peripheral edema, ascites (advanced RV failure)
- Hemoptysis (pulmonary artery aneurysm rupture or in situ thrombosis)
Signs:
- Loud, palpable P2 (pulmonic component of S2)
- Right-sided S4 (RV hypertrophy), S3 (RV failure)
- Holosystolic murmur at left sternal border (tricuspid regurgitation)
- Elevated JVP with prominent v-wave
- Left parasternal heave (RV hypertrophy)
- Peripheral cyanosis, clubbing (in advanced disease or CHD-PAH)
The WHO Functional Classification (modified from NYHA) stratifies symptom burden:
- Class I: No limitation; ordinary activity causes no dyspnea, fatigue, chest pain, or near syncope.
- Class II: Slight limitation; comfortable at rest; ordinary physical activity causes symptoms.
- Class III: Marked limitation; comfortable at rest; less-than-ordinary activity causes symptoms.
- Class IV: Inability to perform any activity without symptoms; symptoms at rest; signs of right heart failure.
6. Diagnosis
Diagnosis requires a systematic evaluation to confirm PH, characterize severity, and classify etiology.
Non-invasive Evaluation
- Echocardiography: First-line screening tool. Tricuspid regurgitation jet velocity (TRV) estimates RVSP: TRV >3.4 m/s = high probability PH. Also assesses RV size/function, LV morphology, pericardial effusion. V/Q mismatch pattern suggests CTEPH. Limitations: RVSP estimation inaccurate in up to 48% of cases.
- Electrocardiography: RV strain pattern (right axis deviation, RBBB, S1Q3T3 pattern, P-pulmonale); sensitivity limited (<55%).
- Chest X-ray: Enlarged main pulmonary arteries, right heart enlargement, pruning of peripheral pulmonary vasculature.
- Pulmonary function tests (PFTs): Typically show reduced DLCO (often first abnormality in PAH); mild restrictive pattern. Obstructive pattern suggests Group 3 PH.
- HRCT chest: Evaluate for ILD, emphysema, mosaic attenuation (CTEPH); ground-glass opacities in PAH may indicate pulmonary veno-occlusive disease (PVOD).
- V/Q lung scan: Screening test of choice for CTEPH — high sensitivity (~97%) for identifying segmental or larger perfusion defects.
- CT pulmonary angiography: Defines CTEPH anatomy for surgical planning; less sensitive than V/Q for distal disease.
- Six-minute walk distance (6MWD): Functional capacity assessment and treatment response monitoring.
- Cardiopulmonary exercise testing (CPET): Distinguishes pulmonary vascular from cardiac and deconditioning causes of dyspnea.
- Biomarkers: NT-proBNP/BNP reflect RV stress; prognostic value validated in multiple registries. Uric acid and troponin provide additional prognostic information.
Right Heart Catheterization (RHC)
The gold standard for definitive diagnosis. Mandatory before initiating PAH-specific therapy. Measures:
- mPAP (>20 mmHg defines PH)
- Pulmonary arterial wedge pressure (PAWP ≤15 mmHg confirms pre-capillary PH)
- Cardiac output/index (Fick or thermodilution method)
- PVR = (mPAP − PAWP) / CO; >2 WU in PAH
- Mixed venous oxygen saturation
Vasoreactivity testing: Performed during RHC using inhaled NO (40 ppm), IV adenosine, or inhaled iloprost. A positive response (mPAP decrease ≥10 mmHg to absolute value ≤40 mmHg, with maintained or increased CO) identifies approximately 10% of IPAH patients eligible for calcium channel blocker therapy.
7. Treatment
Management is stratified by WHO group and risk category. PAH-specific vasodilator therapy is used only in Group 1 (and CTEPH with residual disease). Groups 2 and 3 require treatment of the underlying condition; PAH therapies may be harmful in Group 2.
General Measures
- Supervised exercise rehabilitation (evidence base from randomized trials)
- Supplemental oxygen to maintain SpO2 >92%
- Diuretics for right heart failure/fluid overload (careful to avoid preload reduction)
- Avoid pregnancy (high maternal mortality; contraception counseling mandatory)
- Anticoagulation with warfarin (controversial; may benefit IPAH; avoid in CTD-PAH due to bleeding risk)
Calcium Channel Blockers (CCBs)
Reserved for vasoreactive-positive IPAH patients (∼10%). Nifedipine, diltiazem, or amlodipine at high doses. Annual reassessment of sustained response required.
Targeted PAH Therapies — Three Approved Pathways
Endothelin Receptor Antagonists (ERAs)
- Ambrisentan (ETA-selective): 5–10 mg daily
- Bosentan (dual ETA/ETB): 125 mg twice daily; monthly LFTs required
- Macitentan (dual, tissue-penetrating): 10 mg daily; SERAPHIN trial showed 45% reduction in morbidity/mortality
PDE5 Inhibitors and sGC Stimulators
- Sildenafil: 20 mg three times daily (approved dose for PAH)
- Tadalafil: 40 mg once daily; PHIRST trial demonstrated functional improvement
- Riociguat (sGC stimulator): 0.5–2.5 mg three times daily; approved for PAH and CTEPH; PATENT-1 and CHEST-1 trials
Prostacyclin Pathway Agents
- Epoprostenol (IV): Continuous IV infusion via central catheter; most potent agent; first to demonstrate mortality benefit; dose titrated upward (starting ~2 ng/kg/min)
- Treprostinil: IV, SC, inhaled, or oral; flexible administration routes; FREEDOM trials validated oral form
- Iloprost: Inhaled 6–9 times daily; 2.5–5 mcg per inhalation
- Selexipag (IP receptor agonist): 200–1600 mcg twice daily; GRIPHON trial showed 40% reduction in morbidity/mortality composite
Combination Therapy
Initial combination therapy with ERA + PDE5i is now preferred over sequential add-on therapy for most treatment-naive Group 1 PH patients based on the AMBITION trial (ambrisentan + tadalafil vs. monotherapy). Triple combination therapy (ERA + PDE5i + prostacyclin pathway agent) is used for high-risk or deteriorating patients.
CTEPH-Specific Treatment
- Pulmonary endarterectomy (PEA): Potentially curative surgical procedure; treatment of choice for operable CTEPH; 5-year survival >80%
- Balloon pulmonary angioplasty (BPA): Percutaneous treatment for inoperable CTEPH; multiple sessions; CTEPH-BPA and RACE trials
- Riociguat: Only approved medical therapy for CTEPH (residual/inoperable disease)
Lung Transplantation
Reserved for WHO FC III–IV patients failing maximal medical therapy. Bilateral lung transplant preferred. Median survival post-transplant ~5–6 years.
8. Complications
- Right heart failure: Principal cause of death; characterized by progressive RV dilation, reduced cardiac output, and end-organ hypoperfusion
- Arrhythmias: Atrial flutter/fibrillation common in advanced disease; poorly tolerated due to RV dependence on atrial kick
- Hemoptysis: May be massive; due to bronchial artery anastomoses, PA aneurysm rupture, or in situ thrombosis
- Syncope: Exercise-induced; reflects inability to augment cardiac output; poor prognostic sign
- Hepatic congestion and cardiac cirrhosis: From chronic venous hypertension
- Pulmonary veno-occlusive disease (PVOD): Aggressive variant of Group 1; may worsen with vasodilator therapy; diagnosis by HRCT (ground-glass opacities, septal lines)
- Pregnancy: Maternal mortality 30–56%; absolute contraindication in PAH
- Paradoxical embolism: Via right-to-left shunting through patent foramen ovale or ASD
9. Prognosis
Prognosis varies markedly by WHO group and individual patient factors. For PAH, the REVEAL 2.0 risk calculator and ESC/ERS low-/intermediate-/high-risk stratification guide treatment escalation:
- Low risk: WHO FC I–II, 6MWD >440 m, NT-proBNP <300 ng/L, no RV dysfunction, estimated 1-year mortality <5%
- Intermediate risk: WHO FC II–III, 6MWD 165–440 m, NT-proBNP 300–1,400 ng/L, mild RV dysfunction, 1-year mortality 5–20%
- High risk: WHO FC IV, 6MWD <165 m, NT-proBNP >1,400 ng/L, significant RV dysfunction/failure, 1-year mortality >20%
With modern combination therapy, 5-year survival for IPAH has improved from ~34% in the pre-treatment era to >60–70%. SSc-PAH continues to carry a worse prognosis (~50% 3-year mortality) due to underlying disease complexity. CTEPH treated with PEA has 5-year survival exceeding 80%.
10. Prevention
- Genetic counseling and screening for first-degree relatives of HPAH patients harboring BMPR2 or other disease-causing mutations
- Echocardiographic screening in high-risk populations: systemic sclerosis (annually), HIV-infected patients, portal hypertension candidates for liver transplant
- Avoidance of anorexigenic drugs, methamphetamine, and dasatinib in susceptible individuals
- Early recognition and treatment of PE to prevent CTEPH development; lifelong anticoagulation after unprovoked PE
- Management of underlying left heart disease (HFpEF, valvular disease) to prevent Group 2 PH progression
- Smoking cessation to prevent COPD-associated Group 3 PH
- Optimization of CPAP adherence in OSA to reduce PH risk
11. Recent Research and Advances
Sotatercept (ACE-011): An activin signaling inhibitor that rebalances the BMPR2/activin receptor signaling axis — the fundamental molecular defect in PAH. The PULSAR and STELLAR trials demonstrated significant reductions in PVR, improvements in 6MWD, and reductions in NT-proBNP. FDA approved in 2024 for WHO Group 1 PAH; represents the first approved therapy targeting the underlying vascular remodeling process rather than just vasoconstriction.
Ralinepag and Other IP Receptor Agonists: Next-generation prostacyclin receptor agonists with longer half-lives and improved tolerability are in late-stage development.
TYVASO DPI (inhaled treprostinil dry powder): FDA approved for PAH and PH-ILD (Group 3); the INCREASE trial demonstrated improvement in 6MWD and reduction in clinical worsening in PH-ILD — the first approval for Group 3 PH.
Genetic and Molecular Advances: Next-generation sequencing has identified novel causal genes beyond BMPR2 (GDF2/BMP9, ATP13A3, AQP1, SOX17), broadening understanding of the genetic architecture of PAH. iPSC-derived models from BMPR2-mutant patients are advancing drug discovery.
HFpEF-PH Phenotyping: Recognizing Group 2 PH in HFpEF requires hemodynamic exercise testing; passive vs. reactive PH subtypes have different prognoses and treatment implications. Trials of ERA and PDE5i in HFpEF-PH have yielded negative or mixed results, reinforcing the importance of correct group classification.
12. References
- Humbert M, et al. 2022 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension. Eur Heart J. 2022;43(38):3618–3731.
- Simonneau G, et al. Updated clinical classification of pulmonary hypertension. J Am Coll Cardiol. 2013;62(25 Suppl):D34–41.
- Tuder RM, et al. Relevant issues in the pathology and pathobiology of pulmonary hypertension. J Am Coll Cardiol. 2013;62(25 Suppl):D4–12.
- Galiè N, et al. (AMBITION Investigators). Initial Use of Ambrisentan plus Tadalafil in Pulmonary Arterial Hypertension. N Engl J Med. 2015;373(9):834–844.
- Pulido T, et al. (SERAPHIN Investigators). Macitentan and morbidity and mortality in pulmonary arterial hypertension. N Engl J Med. 2013;369(9):809–818.
- Sitbon O, et al. (GRIPHON Investigators). Selexipag for the treatment of pulmonary arterial hypertension. N Engl J Med. 2015;373(26):2522–2533.
- Ghofrani HA, et al. (PATENT-1). Riociguat for the treatment of pulmonary arterial hypertension. N Engl J Med. 2013;369(4):330–340.
- Ghofrani HA, et al. (CHEST-1). Riociguat for the treatment of chronic thromboembolic pulmonary hypertension. N Engl J Med. 2013;369(4):319–329.
- Hoeper MM, et al. A global view of pulmonary arterial hypertension. Lancet Respir Med. 2016;4(4):306–322.
- Badesch DB, et al. Diagnosis and assessment of pulmonary arterial hypertension. J Am Coll Cardiol. 2009;54(1 Suppl):S55–66.
- Rosenzweig EB, et al. Sotatercept for the treatment of pulmonary arterial hypertension (STELLAR). N Engl J Med. 2023;388(16):1478–1490.
- Channick RN, et al. Effects of the dual endothelin-receptor antagonist bosentan in patients with pulmonary hypertension: a randomised placebo-controlled study. Lancet. 2001;358(9288):1119–1123.
- Barberà JA, et al. Pulmonary hypertension in COPD. Eur Respir J. 2003;21(5):892–905.
- Olsson KM, et al. REVEAL 2.0: Updated risk score for pulmonary arterial hypertension. Chest. 2019;156(2):323–337.
- Waxman A, et al. (INCREASE Trial). Inhaled treprostinil in pulmonary hypertension due to interstitial lung disease. N Engl J Med. 2021;384(4):325–334.
- Delcroix M, et al. Long-term outcome of patients with chronic thromboembolic pulmonary hypertension. Circulation. 2016;133(9):859–871.
Research Papers
The following PubMed topic searches surface the current peer-reviewed literature on Pulmonary Hypertension. Each link opens a live PubMed query; results update as new papers are indexed.
- PubMed search: pulmonary hypertension
- PubMed search: pulmonary arterial hypertension
- PubMed search: pulmonary hypertension right heart catheterization
- PubMed search: pulmonary hypertension endothelin receptor antagonist
- PubMed search: pulmonary hypertension phosphodiesterase 5 inhibitor
- PubMed search: pulmonary hypertension prostacyclin epoprostenol
- PubMed search: chronic thromboembolic pulmonary hypertension
- PubMed search: pulmonary hypertension connective tissue disease
- PubMed search: pulmonary hypertension WHO classification
- PubMed search: pulmonary hypertension sotatercept
- PubMed search: pulmonary hypertension lung transplantation
- PubMed search: pulmonary hypertension echocardiography
Connections
- COPD
- Interstitial Lung Disease
- Heart Failure
- Obstructive Sleep Apnea
- Pulmonary Embolism
- Magnesium
- Hypertension
- Sarcoidosis
- Fatigue
- Anemia
- Chest Pain
- Calcium
- Valvular Heart Disease
- Edema
- Shortness of Breath
- Deep Vein Thrombosis
- Atrial Fibrillation
- Vitamin D3