Endocarditis
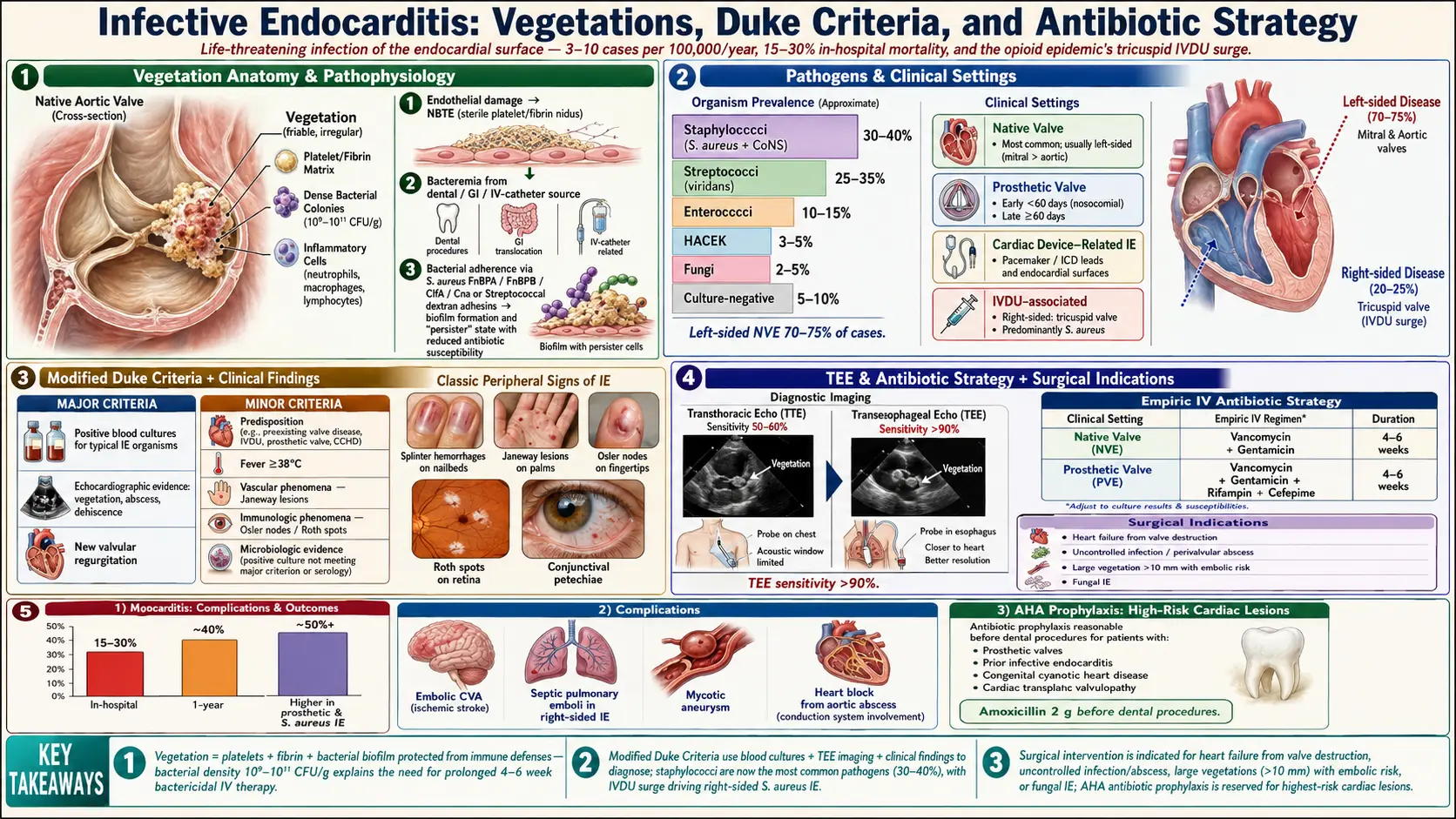
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- Research Papers
- Connections
- Featured Videos
1. Overview
Infective endocarditis (IE) is a life-threatening infection of the endocardial surface of the heart, most commonly affecting the cardiac valves (native or prosthetic), though it may also involve intracardiac devices, septal defects, or the mural endocardium. The hallmark pathologic lesion is the vegetation — an irregular, friable mass composed of platelets, fibrin, microorganisms, and inflammatory cells adherent to damaged endothelium.
IE is classified by the affected valve and setting:
- Native valve endocarditis (NVE): Affecting natural valves
- Prosthetic valve endocarditis (PVE): Early (<60 days post-surgery) or late (>60 days); early PVE is typically nosocomial, caused by staphylococci or Gram-negative organisms
- Cardiac device-related IE (CDRIE): Involving pacemaker leads, ICD leads, left ventricular assist devices
- Healthcare-associated IE: Nosocomial or non-nosocomial healthcare-associated exposure
- Intravenous drug use (IVDU)-associated IE: Predominately right-sided (tricuspid valve), caused by S. aureus
IE remains associated with high in-hospital mortality (15–30%) and high rates of cardiac and embolic complications, necessitating prompt diagnosis, multidisciplinary management, and often surgical intervention.
2. Epidemiology
The annual incidence of IE is approximately 3–10 cases per 100,000 population in high-income countries, with consistent increases observed over the past two decades. In the United States, incidence increased from 8.5 to 12.7 per 100,000 between 1998 and 2009, largely driven by the opioid epidemic and increased use of intravascular devices.
IE demonstrates clear demographic patterns:
- Male predominance (2:1 male-to-female ratio), though this gap narrows in elderly populations
- Median age at diagnosis 60–65 years in developed countries (shifting from rheumatic valve disease in young patients toward degenerative valve disease and healthcare-associated infection in older adults)
- IVDU-associated IE disproportionately affects younger patients (20–40 years); its incidence has more than tripled in rural and suburban US communities since 2007 due to the opioid crisis
- Left-sided NVE (mitral and aortic valves) accounts for 70–75% of cases; right-sided (predominantly tricuspid valve) accounts for 5–10% (predominantly IVDU-associated)
- Staphylococci now account for the most common etiologic agents in most developed countries (30–40%), followed by streptococci (25–35%)
- In-hospital mortality remains 15–30% despite advances in diagnostics and treatment; 1-year mortality approaches 40%
3. Pathophysiology
IE development requires the confluence of three key elements: endothelial damage or abnormality, bacteremia, and bacterial adherence and proliferation.
Step 1: Endothelial Damage and Non-Bacterial Thrombotic Endocarditis (NBTE)
Normal intact endothelium is resistant to bacterial colonization. Mechanical trauma (turbulent blood flow across abnormal valves, jet lesions from regurgitant streams, prosthetic material), immune complex deposition, or catheter-induced injury disrupts the endothelial surface, exposing subendothelial collagen and extracellular matrix. This triggers a thrombotic response — platelet and fibrin deposition — creating the sterile NBTE (or marantic endocarditis) lesion that serves as the nidus for bacterial seeding.
Step 2: Bacteremia
Transient bacteremia occurs during numerous daily activities (toothbrushing, dental procedures, gastrointestinal procedures, intravascular catheter insertion, surgery). In most individuals, circulating bacteria are rapidly cleared by immune defenses. In predisposed individuals, bacteria reach the NBTE lesion and adhere to the thrombotic matrix.
Step 3: Bacterial Adherence and Vegetation Formation
Bacterial adhesins mediate attachment to host matrix proteins on the NBTE lesion. Staphylococcus aureus expresses fibronectin-binding proteins (FnBPA, FnBPB), clumping factors (ClfA, ClfB), and collagen-binding protein (Cna), enabling adherence to damaged endothelium. Viridans streptococci produce extracellular polysaccharides (dextrans) that facilitate adhesion.
Once adherent, bacteria proliferate within the vegetation, protected from host immune defenses by the fibrin matrix. Colony-forming biofilms develop, with bacteria transitioning to a slow-growing "persister" state with markedly reduced antibiotic susceptibility. Platelets aggregate and fibrin is continuously deposited, enlarging the vegetation. Bacterial density within mature vegetations can reach 10⁹–10¹¹ colony-forming units per gram, explaining why prolonged bactericidal antibiotic therapy is required.
Inflammatory and Embolic Consequences
Cytokines (IL-1β, IL-6, TNF-α) drive systemic inflammatory response, fever, and leukocytosis. Vegetation fragments embolize to systemic (left-sided IE) or pulmonary (right-sided IE) circulations. Perivalvular tissue destruction — driven by bacterial proteases, matrix metalloproteinases, and local inflammatory response — leads to valve leaflet destruction, perforation, rupture of chordae tendineae, annular abscess formation, and fistulae, with resulting acute hemodynamic compromise.
4. Etiology and Risk Factors
Causative Organisms
- Staphylococcus aureus (30–40%): Most common cause overall; associated with acute, aggressive presentation, high rates of embolism, perivalvular abscess, and mortality; MRSA increasingly prevalent in healthcare-associated and IVDU cases
- Viridans group streptococci (20–30%): S. sanguinis, S. mutans, S. salivarius, S. bovis group; subacute presentation; associated with dental procedures and periodontal disease; S. bovis biotype I (S. gallolyticus) — colonic neoplasia association (mandatory colonoscopy on diagnosis)
- Enterococci (5–15%): E. faecalis (dominant), E. faecium; GI/GU tract source; elderly men; difficult to treat due to intrinsic aminoglycoside resistance and increasing ampicillin and vancomycin resistance (VRE)
- Coagulase-negative staphylococci (CoNS, 10–15%): Predominant in PVE (especially early PVE); S. epidermidis most common; healthcare-associated; biofilm formation
- HACEK organisms (<5%): Haemophilus spp., Aggregatibacter spp., Cardiobacterium hominis, Eikenella corrodens, Kingella spp.; culture-negative pattern; fastidious Gram-negative organisms; dental source; subacute presentation
- Fungal endocarditis (<2%): Candida spp. (most common), Aspergillus spp.; associated with prolonged IV access, total parenteral nutrition, immunosuppression; very high mortality; typically requires surgical treatment
- Culture-negative IE (5–10%): Prior antibiotic therapy (most common cause), fastidious organisms, intracellular organisms (Coxiella burnetii [Q fever], Bartonella spp., Brucella spp., Tropheryma whipplei); requires special serologies and PCR testing
Host Risk Factors
- Structural cardiac abnormalities: Degenerative valvular disease (mitral valve prolapse with MR, aortic stenosis), congenital heart disease (bicuspid aortic valve, VSD, patent ductus arteriosus), rheumatic heart disease, previous IE (10-fold increased risk)
- Prosthetic heart valves: Highest individual risk (400–1000x); mechanical and bioprosthetic valves are equally susceptible
- Intracardiac devices: Permanent pacemakers, ICDs, LVADs
- Intravenous drug use (IVDU): Highest population-attributable risk; needle sharing, skin flora inoculation
- Intravascular catheters: Central venous catheters, hemodialysis access, PICC lines
- Immunosuppression: HIV infection (CD4 <200/μL), organ transplant, prolonged steroid therapy, malignancy
- Hemodialysis: Vascular access site bacteremia; 50–100x higher IE incidence
- Poor dental hygiene and dental procedures: Bacteremia source (though routine dental procedures account for only a minority of IE cases)
5. Clinical Presentation
Acute vs. Subacute Presentations
Acute IE (typically S. aureus): Rapid onset (<2 weeks), high-grade fever, rigors, and rapid valve destruction with hemodynamic deterioration; high early mortality without prompt treatment.
Subacute IE (viridans streptococci, HACEK): Indolent course (>2 weeks), low-grade fever, weight loss, malaise, anemia; symptoms may be attributed to viral illness or non-specific malaise, delaying diagnosis.
Cardinal Features
- Fever: Present in 90% of patients; low-grade in subacute; high-grade (39–40°C) in acute
- New or changing cardiac murmur: Present in 85%; new regurgitant murmur (especially aortic regurgitation in acute aortic IE) is highly significant
- Constitutional symptoms: Fatigue, malaise, anorexia, weight loss, myalgias, arthralgias, night sweats
Embolic and Immunologic Phenomena (Peripheral Stigmata)
- Petechiae: Conjunctival, oral mucosa, skin; non-specific
- Splinter hemorrhages: Linear dark streaks beneath fingernails or toenails; non-specific (also seen in trauma)
- Janeway lesions: Painless, irregular erythematous or hemorrhagic macules on the palms and soles; embolic phenomenon; more common in acute IE (S. aureus)
- Osler nodes: Tender, raised, violaceous nodules on finger and toe pads; immune complex deposition; more common in subacute IE (viridans streptococci)
- Roth spots: Oval, pale-centered retinal hemorrhages on fundoscopy; immune-mediated; <5% of cases
- Splenomegaly: Present in 30–40%; more common in subacute IE
Right-Sided IE (IVDU)
Tricuspid valve IE presents with septic pulmonary emboli: pleuritic chest pain, dyspnea, hemoptysis, and multiple bilateral nodular pulmonary infiltrates on imaging. Fever is prominent. Left-sided murmur is absent or a tricuspid regurgitation murmur may be heard.
6. Diagnosis
Blood Cultures
Blood cultures are the most critical diagnostic test. Recommended protocol: at least 3 sets of blood cultures drawn from separate venipuncture sites over 24 hours (not from indwelling catheters), each set including one aerobic and one anaerobic bottle. Blood cultures are positive in 85–95% of IE cases if obtained before antibiotic administration. Volume of blood cultured is critical: 10 mL per bottle, 20 mL per set.
For culture-negative IE: additional cultures in specialized media; serology for Coxiella burnetii (anti-phase I IgG ≥1:800), Bartonella spp. (anti-B. henselae/quintana IgG), Brucella spp.; PCR of blood or valve tissue; broad-spectrum 16S ribosomal RNA PCR; metagenomic next-generation sequencing.
Echocardiography
Echocardiography is essential for diagnosis, risk stratification, and surgical planning:
- Transthoracic echocardiography (TTE): Initial study; sensitivity 40–63% for vegetations in NVE; reduced sensitivity in obesity, COPD, prosthetic valves
- Transesophageal echocardiography (TEE): Superior sensitivity (87–100%) and specificity; required if TTE non-diagnostic and IE suspected, all prosthetic valve IE, all cases with potential perivalvular extension (abscess, fistula), pre-surgical planning; identifies vegetations <3 mm
- Findings: Oscillating intracardiac mass (vegetation), perivalvular abscess or pseudoaneurysm, new valvular regurgitation, dehiscence of prosthetic valve (new rocking motion)
- 3D TEE: Superior anatomic detail for surgical planning; identifies involvement of specific leaflets, annulus, subvalvular apparatus
Modified Duke Criteria
The Modified Duke Criteria (Li et al., 2000) remain the diagnostic standard, classifying IE as definite, possible, or rejected:
Major criteria:
- Positive blood cultures: Typical IE organism in ≥2 separate cultures; persistently positive blood cultures ≥12 hours apart; or single positive culture for Coxiella burnetii or anti-phase I IgG ≥1:800
- Evidence of endocardial involvement: Positive echocardiogram (vegetation, abscess, new partial dehiscence of prosthetic valve); new valvular regurgitation
Minor criteria:
- Predisposing cardiac condition or IVDU
- Fever ≥38°C
- Vascular phenomena (arterial emboli, septic pulmonary infarcts, intracranial hemorrhage, conjunctival hemorrhages, Janeway lesions)
- Immunologic phenomena (glomerulonephritis, Osler nodes, Roth spots, positive rheumatoid factor)
- Positive blood culture not meeting major criterion or serologic evidence of infection with IE-compatible organism
Definite IE: 2 major, 1 major + 3 minor, or 5 minor criteria (or pathologic criteria: organisms/lesions from surgery or autopsy).
Additional Imaging
- CT angiography of the chest/abdomen/pelvis: Evaluates embolic complications (splenic infarcts, renal infarcts, mesenteric emboli, septic pulmonary emboli); characterizes perivalvular disease complementary to TEE
- Brain MRI: Mandatory pre-operatively (if urgent surgery not required) to detect silent cerebral emboli; presence of silent emboli is common (30–50%) and may influence anticoagulation and surgical timing decisions
- ¹⁸F-FDG PET/CT: Detects abnormal metabolic activity at valves and prostheses in culture-negative or prosthetic valve IE with inconclusive echocardiography; aids in detecting extracardiac foci; incorporated into ESC 2023 modified Duke Criteria as major criterion for PVE
- Radiolabeled white blood cell SPECT/CT: Alternative nuclear imaging modality for CDRIE and PVE
- Dental and ENT evaluation: Identifies portal of entry; mandatory in all confirmed IE cases
Laboratory Findings
- Elevated ESR, CRP, procalcitonin; leukocytosis (or leukopenia in severe sepsis)
- Normochromic normocytic anemia (chronic inflammation)
- Microscopic hematuria and proteinuria (immune complex glomerulonephritis)
- Elevated creatinine (renal emboli, glomerulonephritis, antibiotic nephrotoxicity)
- Elevated NT-proBNP and troponin (HF, myocardial involvement)
- Rheumatoid factor positive in 50% (subacute)
- Hypocomplementemia (C3, C4) in immune complex disease
7. Treatment
Antibiotic Therapy
Prolonged parenteral bactericidal antibiotic therapy is essential. Treatment duration is typically 4–6 weeks for most NVE and 6 weeks or longer for PVE. Antibiotic selection is pathogen-specific:
- Streptococcal NVE (penicillin-susceptible MIC ≤0.125 mg/L):
- Penicillin G 12–18 million units/day IV (divided every 4 hours) or ceftriaxone 2 g IV/IM once daily, 4 weeks
- Short-course: 2-week ceftriaxone + gentamicin regimen (OPAT-eligible uncomplicated cases)
- Penicillin allergy: Vancomycin 30 mg/kg/day IV in 2 divided doses
- Staphylococcal NVE (MSSA):
- Nafcillin or oxacillin 12 g/day IV in 6 divided doses × 6 weeks (standard)
- Cefazolin 2 g IV every 8 hours — non-inferior to nafcillin with improved tolerability (POET subanalysis); suitable alternative
- Rifampin adjunct for prosthetic valve involvement
- MRSA NVE:
- Vancomycin 25–30 mg/kg/day IV in 2–3 divided doses × 6 weeks; target AUC/MIC 400–600
- Daptomycin 8–10 mg/kg IV once daily — non-inferior to vancomycin for right-sided MRSA IE (S. aureus Endocarditis trial); pulmonary surfactant inactivates daptomycin — not for left-sided IE with active pneumonia
- Linezolid 600 mg IV/PO every 12 hours — reserved for VRE or refractory MRSA; bacteriostatic; limited duration due to myelosuppression
- Enterococcal IE:
- Ampicillin-susceptible: Ampicillin 12 g/day IV + ceftriaxone 2 g IV every 12 hours × 6 weeks; SADBE trial demonstrated combination superior to ampicillin + gentamicin for E. faecalis IE
- VRE: Daptomycin 8–10 mg/kg + ampicillin; linezolid; consult infectious disease
- Fungal IE: Liposomal amphotericin B ± flucytosine for Candida spp.; azole step-down suppressive therapy after surgical treatment; itraconazole or voriconazole for Aspergillus
- Culture-negative IE: Empiric coverage while serology/PCR pending; doxycycline 100 mg twice daily for Coxiella or Bartonella; hydroxychloroquine added for Q fever (18-month course)
Outpatient Parenteral Antibiotic Therapy (OPAT)
The POET trial (2019) demonstrated non-inferiority of oral step-down antibiotic therapy (following ≥10 days IV therapy) vs. continued IV therapy in stabilized patients with left-sided IE caused by streptococci, enterococci, S. aureus, or CoNS; published in NEJM. Requires: hemodynamic stability, no perivalvular complications, vegetation <20 mm, competent patient with follow-up availability.
Surgical Indications
Approximately 50% of IE patients require cardiac surgery. Indications include:
- Urgent/emergent (within 24 hours): Acute heart failure from severe valve regurgitation (cardiogenic shock or pulmonary edema); aortic or mitral valve IE with severe acute regurgitation causing hemodynamic instability
- Urgent (within days, same hospitalization): Perivalvular abscess or fistula or destructive perforating lesion; prosthetic valve dehiscence; vegetation >10 mm with ≥1 embolic episode; vegetation >10 mm with severe valve stenosis or regurgitation; large vegetation (>15 mm) with high embolic risk at sites with good surgical risk
- Elective (during hospitalization): Fungal IE; highly resistant organisms (MRSA, VRE); recurrent emboli despite antibiotic therapy; enlarging vegetation on therapy
The optimal timing of surgery in the setting of neurologic complications remains challenging: ischemic stroke is not a contraindication to urgent surgery if deficits are mild; hemorrhagic stroke requires deferral of 4 weeks if possible to avoid hemorrhagic transformation with cardiopulmonary bypass anticoagulation.
Supportive Measures
- Anticoagulation: VKA (warfarin) should be temporarily discontinued in NVE due to embolic stroke risk if neurologic complication present; DOACs generally avoided in active IE; anticoagulation may be continued cautiously in mechanical valve PVE after stroke risk assessment
- Embolic event management: Neurosurgical or interventional consultation for mycotic aneurysms; percutaneous/surgical drainage of metastatic abscesses
- Multidisciplinary endocarditis team: Cardiologist, cardiac surgeon, infectious disease specialist, neurologist, microbiologist — improves outcomes (in-hospital mortality 18.5% vs. 35.6% without endocarditis team management; GAMES cohort)
8. Complications
- Heart failure (30–40%): Most common complication; most frequent indication for surgery; caused by acute valve regurgitation, myocarditis, intracardiac fistulae, or valve obstruction
- Embolic events (25–50%): Cerebral embolism (ischemic stroke/TIA, 20–40%); splenic infarcts (30–40%); renal infarcts (15–30%); mesenteric infarcts; limb ischemia; silent emboli detected by MRI in 30–50% of cases; embolic risk is highest in the first 2 weeks of antibiotic therapy and with vegetation size >10 mm and highly mobile vegetations
- Perivalvular extension (15–40%): Abscess (most common in aortic IE, prosthetic valve IE); pseudoaneurysm; intracardiac fistula (aorto-mitral continuity disruption — "intervalvular fibrosa" destruction); often requires surgical repair
- Mycotic aneurysms (2–5%): Intracranial mycotic aneurysms — at risk for rupture causing intracranial hemorrhage; diagnosis by CT angiography or MR angiography; may require endovascular embolization or surgical clipping
- Neurologic complications (30–50%): Ischemic stroke, TIA, silent cerebral emboli (30%), intracranial hemorrhage (mycotic aneurysm rupture, hemorrhagic transformation), meningitis, cerebral abscess, encephalopathy
- Splenic complications: Splenic abscess (requires drainage/splenectomy) vs. uncomplicated splenic infarct
- Acute kidney injury: Embolic renal infarcts, immune complex glomerulonephritis, antibiotic nephrotoxicity (vancomycin, aminoglycosides)
- Conduction abnormalities: New PR prolongation or bundle branch block signaling perivalvular abscess extension to the conduction system; indicates urgent surgical evaluation
- Relapse and reinfection: Relapse (same organism, ≤6 months) in 2–6%; reinfection (different organism or >6 months) most common in IVDU; recurrent IE risk 15–22%
9. Prognosis
IE carries high mortality: in-hospital mortality 15–30%; 1-year mortality 20–40%. Prognosis varies significantly by patient, organism, and management factors.
- Poor prognostic indicators: Age (>65 years), diabetes mellitus, Charlson comorbidity score >3, S. aureus etiology, PVE, perivalvular complications, heart failure at presentation, embolic events, creatinine >1.5 mg/dL, immunocompromised state
- Organism-specific mortality: Fungal IE mortality >50%; MRSA IE hospital mortality 20–35%; viridans streptococcal IE mortality 5–10%
- Surgical impact: Appropriate and timely surgery significantly reduces mortality in complex IE; conversely, delayed or inappropriate surgery worsens outcomes
- IVDU-associated IE: In-hospital mortality 6–11% (lower than community-acquired IE due to younger, less-comorbid patients); but long-term mortality is very high (1-year mortality 27–30%) due to continued IVDU; recurrence rate 30–40% in active IVDU
- Risk scores: ICE-PCS score, PALSUSE score, and Euro-ENDO risk score assist in predicting in-hospital mortality and risk stratification
- Post-IE surveillance: Annual echocardiography to monitor valve function; dental hygiene and prophylaxis education; repeat blood cultures if febrile; rehabilitation and quality-of-life support
10. Prevention
Antibiotic Prophylaxis
Current guidelines (AHA 2007, ESC 2015/2023) significantly restrict IE prophylaxis recommendations based on evidence that most IE cases are not preceded by procedure-related bacteremia and that routine prophylaxis would need to be given to an enormous number of patients to prevent one case.
IE prophylaxis is recommended ONLY for patients at highest risk:
- Prosthetic cardiac valves (including transcatheter valves)
- Previous episode of IE
- Congenital heart disease (CHD): Unrepaired cyanotic CHD; CHD repaired with prosthetic material within 6 months; repaired CHD with residual defect adjacent to prosthetic material
- Cardiac transplant recipients with valvulopathy
Dental procedures warranting prophylaxis (in above high-risk patients): Procedures involving manipulation of gingival tissue, periapical region, or oral mucosa perforation.
Prophylaxis regimens (dental procedures):
- Amoxicillin 2 g PO (or ampicillin 2 g IV/IM) 30–60 minutes before procedure
- Penicillin allergy: Clindamycin 600 mg PO or cephalexin 2 g PO (avoid in PCN anaphylaxis history)
General Preventive Strategies
- Strict aseptic technique for all intravascular procedures and line insertions
- Prompt removal of unnecessary intravascular catheters
- Maintenance of good oral hygiene — most effective long-term strategy for reducing streptococcal bacteremia
- Harm reduction programs for IVDU patients (needle exchange, medication-assisted therapy with buprenorphine/methadone)
- Valve repair over replacement when technically feasible (reduces prosthetic valve risk)
- Skin and soft tissue infection management in IVDU patients
- Antibiotic stewardship to minimize selection of resistant organisms
11. Recent Research and Advances
- POET trial (2019): Partial oral step-down antibiotic therapy after ≥10 days IV therapy in stabilized IE is non-inferior to continued IV therapy for 6-month treatment failure. A landmark practice-changing study enabling early hospital discharge and OPAT in selected patients.
- Updated ESC 2023 IE Guidelines: Incorporated ¹⁸F-FDG PET/CT and radiolabeled WBC SPECT/CT as major diagnostic criteria for PVE; strengthened multidisciplinary endocarditis team recommendation; updated surgical timing criteria based on large registry data.
- Endocarditis Team (Heart Team) concept: Multiple observational studies and registry data (GAMES cohort, EURO-ENDO registry) demonstrate 40–50% mortality reduction with formal multidisciplinary endocarditis team management; increasingly adopted as standard of care.
- IVDU-associated IE epidemic: The opioid crisis has fundamentally changed IE epidemiology in the US, with rural and suburban communities experiencing epidemic increases; surgical outcomes in IVDU patients are complicated by high recurrence and ongoing substance use disorder; multidisciplinary programs integrating addiction medicine and harm reduction are emerging as critical components of IE care in IVDU populations.
- Transcatheter aortic valve implantation (TAVI)-related IE: TAVI-IE has unique features (higher incidence of S. aureus and enterococcal etiology; complex anatomy limiting echocardiographic assessment; prohibitive surgical re-intervention risk); PET/CT plays a critical role; management algorithms are being refined.
- Anti-virulence and anti-biofilm therapies: Preclinical and phase I/II trials of anti-biofilm strategies (dispersin B, DNase) and immunotherapy (exolysin inhibition, anti-toxin monoclonal antibodies) for adjunct IE treatment; clinical efficacy yet to be established.
- Metagenomic next-generation sequencing (mNGS): Pathogen-agnostic sequencing of blood or valve tissue; increases diagnostic yield in culture-negative IE; clinical validation studies ongoing.
- Mitral valve repair in IE: Increased adoption of valve repair over replacement in IE surgical series at high-volume centers; superior long-term outcomes; feasibility in selected cases even with active infection.
Research Papers
The following PubMed topic searches return current peer-reviewed literature relevant to this condition. Each link opens a live PubMed query.
- Infective endocarditis epidemiology
- Duke criteria endocarditis
- Infective endocarditis antibiotic treatment
- Infective endocarditis prophylaxis
- Endocarditis echocardiography
- Staphylococcus aureus endocarditis
- Streptococcus endocarditis
- Prosthetic valve endocarditis
- Endocarditis valve surgery
- Infective endocarditis guidelines
- Infective endocarditis blood cultures
- Infective endocarditis mortality
Connections
- Stroke
- Heart Failure
- Myocarditis
- Valvular Heart Disease
- Arrhythmia
- Complete Blood Count
- Glomerulonephritis
- Staphylococcus Aureus
- Anemia
- Sepsis
- Pneumonia
- Fatigue
- Chest Pain
- Diabetes
- Creatinine
- Atrial Fibrillation
- Lyme Carditis and Cardiac Complications
- Bacillus Subtilis