Vitamin K: The Essential Vitamin for Blood Clotting, Bone Health, and Cardiovascular Protection
Vitamin K is a fat-soluble vitamin whose name derives from the German word "Koagulationsvitamin" — the coagulation vitamin — reflecting its original discovery in 1929 by Danish biochemist Henrik Dam as a dietary factor essential for blood clotting. Dam observed that chickens fed a fat-free diet developed a hemorrhagic disease that could be reversed by a fat-soluble substance he designated "Vitamin K." This discovery, along with Edward Doisy's elucidation of Vitamin K's chemical structure, earned both scientists the Nobel Prize in Physiology or Medicine in 1943. While Vitamin K's role in blood coagulation remains its most widely recognized function, research over the past several decades has revealed that this vitamin is far more versatile than originally understood. Vitamin K serves as an essential cofactor for a unique post-translational modification — gamma-carboxylation — that activates a family of proteins involved not only in coagulation but also in bone metabolism, cardiovascular protection, brain health, inflammation regulation, cell growth control, and potentially cancer prevention. The recognition that Vitamin K2 (menaquinones) has distinct and critical functions beyond those of Vitamin K1 (phylloquinone) has fundamentally expanded our understanding of this vitamin's importance in human health.
Table of Contents
- Chemical Forms — K1, K2, and the Menaquinone Family
- Gamma-Carboxylation — The Unique Biochemical Mechanism
- Blood Coagulation — The Classical Function
- Bone Health — Osteocalcin Activation and Calcium Direction
- Cardiovascular Protection — Matrix Gla Protein and Arterial Calcification Prevention
- Brain Health — Emerging Neuroprotective Roles
- Anti-Inflammatory Properties
- Cancer Research — Preliminary Evidence
- Insulin Sensitivity and Metabolic Health
- Dietary Sources — K1 in Greens, K2 in Fermented Foods
- Deficiency, Drug Interactions, and Newborn Prophylaxis
- Recommended Daily Allowance (RDA) and Supplementation
- Vitamin K and Dental Health
- Vitamin K in Specific Clinical Populations
- Historical Discovery and Nobel Prize Legacy
- Absorption, Bioavailability, and Tissue Distribution
- Vitamin K Status Assessment and Laboratory Testing
- Emerging Research and Future Directions
- Connections
- Featured Videos
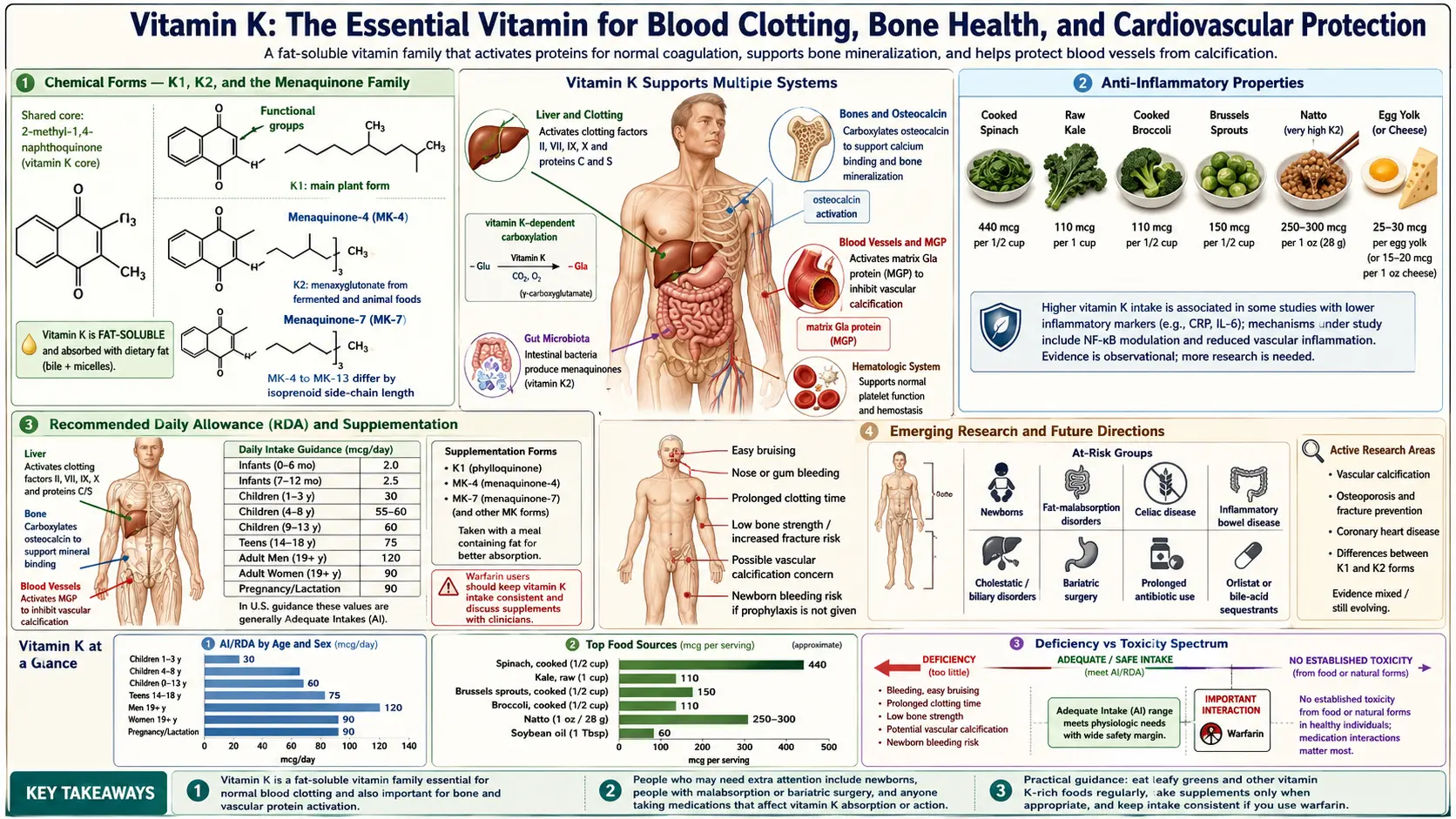
1. Chemical Forms — K1, K2, and the Menaquinone Family
Vitamin K exists in several naturally occurring forms with distinct sources, tissue distributions, biological half-lives, and health effects.
- Vitamin K1 (Phylloquinone): The primary dietary form of Vitamin K, synthesized by plants as part of the photosynthetic apparatus (it is a component of photosystem I). Phylloquinone is found in highest concentrations in green leafy vegetables — kale, spinach, collard greens, broccoli, Brussels sprouts, and lettuce. K1 is efficiently absorbed from the diet (particularly when consumed with dietary fat), transported to the liver, and preferentially used for the hepatic synthesis of coagulation factors. The liver retains K1 tightly and uses it primarily for blood clotting functions.
- Vitamin K2 (Menaquinones): A family of compounds designated MK-4 through MK-13 (menaquinone-4 through menaquinone-13), differing in the length of their isoprenoid side chains. Unlike K1, the menaquinones have longer biological half-lives, more extensive extrahepatic (non-liver) tissue distribution, and may play more prominent roles in bone health, cardiovascular protection, and other functions beyond coagulation. The two most studied and clinically relevant forms are MK-4 and MK-7.
- MK-4 (Menaquinone-4): A short-chain menaquinone that is unique among K2 vitamins because it is synthesized in human tissues from K1 by the enzyme UBIAD1 (UbiA prenyltransferase domain-containing protein 1). MK-4 is found in animal-derived foods (meat, eggs, dairy) and is concentrated in the brain, kidneys, pancreas, and arterial walls. It has a short biological half-life (hours) and is the form used in high-dose Japanese osteoporosis treatment protocols (45 mg/day).
- MK-7 (Menaquinone-7): A long-chain menaquinone produced by bacterial fermentation, most notably by Bacillus subtilis in the production of the Japanese fermented soybean food natto. MK-7 has a significantly longer biological half-life (approximately 72 hours) compared to K1 (1-2 hours) and MK-4 (hours), allowing it to accumulate to higher steady-state levels with daily supplementation. This longer half-life enables MK-7 to reach extrahepatic tissues more effectively, making it particularly relevant for bone and cardiovascular health.
- MK-8 through MK-13: Longer-chain menaquinones produced by various bacteria, found in fermented cheeses and other fermented foods. These forms have very long half-lives and may contribute to K2 status, though they are less well studied than MK-4 and MK-7.
- Vitamin K3 (Menadione): A synthetic form of Vitamin K without a side chain. Menadione is not used in human supplements due to toxicity concerns (it can cause hemolytic anemia and liver damage) but is used in some animal feed applications. It can be converted to MK-4 in tissues.
2. Gamma-Carboxylation — The Unique Biochemical Mechanism
Vitamin K's biological function centers on its role as an essential cofactor for the enzyme gamma-glutamyl carboxylase, which catalyzes a unique post-translational modification of specific proteins.
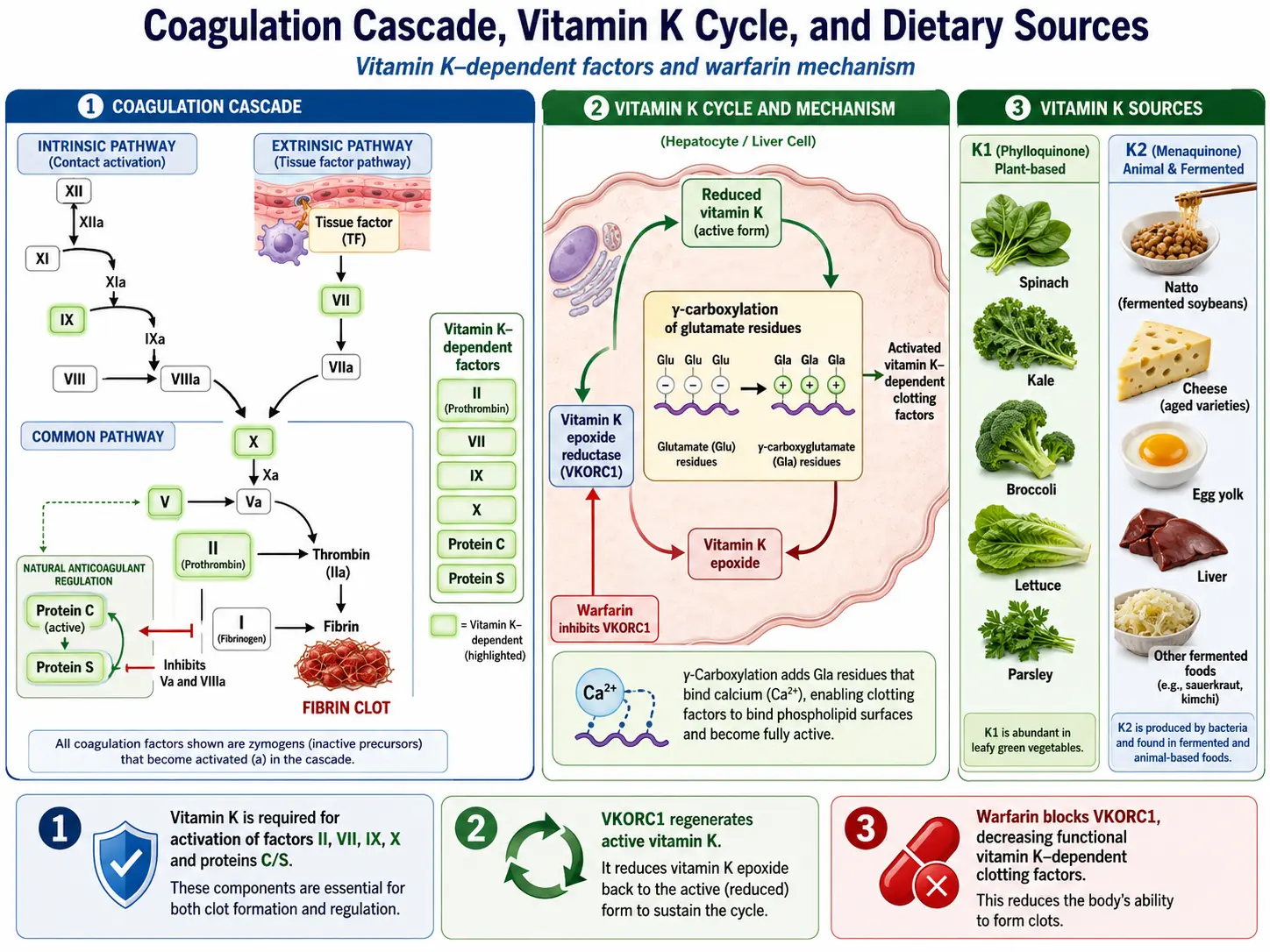
- The Carboxylation Reaction: Gamma-glutamyl carboxylase converts specific glutamic acid (Glu) residues in target proteins to gamma-carboxyglutamic acid (Gla) residues. This carboxylation adds an extra carboxyl group (-COOH) to the gamma carbon of glutamic acid, creating a dual-carboxyl amino acid with a powerful affinity for calcium ions.
- Calcium Binding: The Gla residues created by Vitamin K-dependent carboxylation bind calcium ions with high affinity. This calcium-binding capacity is essential for the biological function of all Vitamin K-dependent proteins — it enables coagulation factors to anchor to phospholipid membrane surfaces at sites of vascular injury, allows osteocalcin to bind to the calcium in bone mineral (hydroxyapatite), and enables Matrix Gla Protein to inhibit calcium deposition in arterial walls.
- The Vitamin K Cycle: During the carboxylation reaction, Vitamin K hydroquinone (the reduced, active form) is oxidized to Vitamin K epoxide. The enzyme Vitamin K epoxide reductase (VKORC1) then recycles Vitamin K epoxide back to the quinone form, and a second reductase converts the quinone to the active hydroquinone. This recycling — the Vitamin K cycle — is extraordinarily efficient, allowing a small pool of Vitamin K to participate in thousands of carboxylation reactions. Warfarin exerts its anticoagulant effect by inhibiting VKORC1, blocking this recycling and effectively creating a functional Vitamin K deficiency.
- Vitamin K-Dependent Proteins (VKDPs): At least 17 human proteins require Vitamin K-dependent gamma-carboxylation for their biological activity. These include coagulation factors (II, VII, IX, X), anticoagulant proteins (C, S, Z), bone proteins (osteocalcin, matrix Gla protein), growth regulation proteins (Gas6, protein S), and others with emerging functions.
3. Blood Coagulation — The Classical Function
Vitamin K's essential role in blood coagulation was its first discovered function and remains critically important for hemostasis.
- Procoagulant Factors: Vitamin K is required for the gamma-carboxylation and activation of four coagulation factors: Factor II (prothrombin), Factor VII, Factor IX, and Factor X. Without carboxylation, these factors cannot bind calcium ions and cannot anchor to the phospholipid membrane surfaces where the coagulation cascade assembles — rendering them functionally inactive.
- Anticoagulant Proteins: Vitamin K is equally essential for the activation of the natural anticoagulant proteins Protein C, Protein S, and Protein Z. Protein C, when activated by the thrombin-thrombomodulin complex, inactivates Factors Va and VIIIa, providing a critical negative feedback mechanism that prevents excessive clotting. Protein S serves as a cofactor for activated Protein C. This dual requirement for both procoagulant and anticoagulant factors means that Vitamin K is essential for balanced hemostasis — not simply for promoting clotting, but for maintaining the equilibrium between clot formation and clot prevention.
- Coagulation Cascade Assembly: The Gla residues on Vitamin K-dependent coagulation factors bind calcium ions that bridge the factors to negatively charged phospholipid membranes (exposed phosphatidylserine on activated platelets). This membrane localization concentrates the coagulation factors at the site of vascular injury, enabling the rapid, amplified cascade that generates thrombin and ultimately a fibrin clot.
- PIVKA (Proteins Induced by Vitamin K Absence): When Vitamin K is insufficient, undercarboxylated (inactive) forms of Vitamin K-dependent proteins — called PIVKAs — are released into the circulation. PIVKA-II (undercarboxylated prothrombin) is a sensitive biomarker of Vitamin K insufficiency and is also used as a tumor marker for hepatocellular carcinoma.
4. Bone Health — Osteocalcin Activation and Calcium Direction
Vitamin K plays a critical role in bone metabolism through the activation of osteocalcin, the most abundant non-collagen protein in bone.
- Osteocalcin Carboxylation: Osteocalcin is produced by osteoblasts (bone-building cells) and requires Vitamin K-dependent gamma-carboxylation of three glutamic acid residues to become fully active. Carboxylated osteocalcin binds calcium ions with high affinity and incorporates them into the hydroxyapatite crystal lattice of bone mineral, directly contributing to bone mineralization and strength.
- Undercarboxylated Osteocalcin (ucOC): When Vitamin K status is insufficient, a significant proportion of osteocalcin is released into the blood in its undercarboxylated (inactive) form (ucOC). Elevated ucOC is a sensitive marker of functional Vitamin K deficiency in bone and is associated with reduced bone mineral density and increased fracture risk. The ratio of ucOC to total osteocalcin (the ucOC/OC ratio) is used as a functional biomarker of Vitamin K status for bone health.
- Fracture Risk Reduction: Multiple epidemiological studies and clinical trials have demonstrated that higher Vitamin K intake (both K1 and K2) is associated with reduced fracture risk, particularly hip fracture. Japanese studies of MK-4 at pharmacological doses (45 mg/day) show significant fracture reduction, and MK-7 supplementation has been shown to reduce bone loss in postmenopausal women.
- Synergy with Vitamin D3 and Calcium: Vitamin K, Vitamin D3, and calcium work synergistically in bone metabolism. Vitamin D3 stimulates the synthesis of osteocalcin, while Vitamin K activates the osteocalcin through carboxylation. Calcium provides the mineral substrate that carboxylated osteocalcin binds and deposits into bone. This triad — Vitamin D3 to produce the protein, Vitamin K to activate it, and calcium to mineralize the bone — represents the optimal nutritional approach to bone health.
- K2 vs. K1 for Bone Health: While K1 can support osteocalcin carboxylation, K2 (particularly MK-4 and MK-7) appears to be more effective for bone health due to greater extrahepatic tissue distribution, longer half-lives (MK-7), and direct effects on osteoblast and osteoclast function that are independent of gamma-carboxylation.
5. Cardiovascular Protection — Matrix Gla Protein and Arterial Calcification Prevention
One of the most significant advances in Vitamin K research has been the recognition of its role in preventing arterial calcification — a major cardiovascular risk factor.
- Matrix Gla Protein (MGP): Matrix Gla Protein is the most potent natural inhibitor of vascular calcification in the human body. Produced by vascular smooth muscle cells and chondrocytes, MGP requires Vitamin K-dependent gamma-carboxylation to become active. Carboxylated MGP binds calcium ions in the arterial wall and prevents their deposition as hydroxyapatite crystite, which would otherwise stiffen and narrow the arteries.
- The Calcium Paradox: The concept of the "calcium paradox" describes the clinical observation that calcium can simultaneously be deficient in bones (osteoporosis) and excessive in arteries (vascular calcification). Vitamin K2 resolves this paradox by activating both osteocalcin (which directs calcium into bones) and Matrix Gla Protein (which prevents calcium deposition in arteries). Without adequate Vitamin K2, calcium is misdirected — leaving bones and depositing in arteries.
- The Rotterdam Study: This landmark Dutch population study of 4,807 subjects demonstrated that the highest dietary intake of Vitamin K2 (primarily MK-4 from cheese and meat) was associated with a 57% reduction in cardiovascular mortality, a 52% reduction in coronary heart disease, and a 26% reduction in all-cause mortality compared to the lowest intake group. Importantly, Vitamin K1 intake showed no significant association with cardiovascular outcomes, suggesting that the cardiovascular benefits are specific to K2.
- Dephospho-Uncarboxylated MGP (dp-ucMGP): Dp-ucMGP — the inactive form of MGP that is both uncarboxylated and unphosphorylated — has emerged as a biomarker of functional Vitamin K deficiency in the vasculature. Elevated dp-ucMGP levels are associated with increased arterial stiffness, vascular calcification, and cardiovascular mortality. This biomarker allows clinicians to assess whether an individual has adequate Vitamin K for vascular protection.
- Warfarin and Arterial Calcification: Long-term warfarin therapy, which inhibits the Vitamin K cycle, has been associated with accelerated arterial and valvular calcification. This is a direct consequence of MGP remaining in its inactive (undercarboxylated) form, unable to inhibit vascular calcium deposition. This observation has prompted research into whether Vitamin K2 supplementation might reverse warfarin-induced vascular calcification.
6. Brain Health — Emerging Neuroprotective Roles
Research has revealed previously unrecognized roles for Vitamin K in brain function and neuroprotection.
- Sphingolipid Synthesis: Vitamin K, particularly MK-4 (the predominant form in the brain), is involved in the synthesis of sphingolipids — a class of lipids that are critical components of neuronal cell membranes and myelin sheaths. Sphingolipids play essential roles in cell signaling, membrane structure, and cell-to-cell recognition in the nervous system. Alterations in sphingolipid metabolism are implicated in neurodegenerative diseases including Alzheimer's disease and Parkinson's disease.
- Gas6 Protein: Growth arrest-specific protein 6 (Gas6) is a Vitamin K-dependent protein expressed in the brain that acts as a ligand for the TAM receptor family (Tyro3, Axl, Mer). Gas6/TAM signaling supports neuronal survival, promotes myelination, protects against apoptosis, and modulates neuroinflammation. Adequate Vitamin K is essential for Gas6 carboxylation and function.
- Anti-Inflammatory Effects in the Brain: Vitamin K has been shown to suppress neuroinflammation, reduce the production of pro-inflammatory cytokines by microglia, and protect neurons from inflammatory damage. These anti-neuroinflammatory effects may be relevant to the prevention and management of neurodegenerative diseases.
- Cognitive Function in Aging: Epidemiological studies have found that higher Vitamin K intake is associated with better cognitive performance and reduced risk of cognitive decline in elderly individuals. Conversely, warfarin use (which inhibits Vitamin K function) has been associated with poorer cognitive performance in some studies, though the evidence is not entirely consistent.
- MK-4 Concentration in Brain: MK-4 is the predominant form of Vitamin K in the brain, present at concentrations significantly higher than K1. The brain actively converts K1 to MK-4 through the UBIAD1 enzyme, suggesting that MK-4 has specific and important functions in neural tissue.
7. Anti-Inflammatory Properties
Vitamin K possesses direct anti-inflammatory effects that extend beyond its antioxidant and gamma-carboxylation functions.
- NF-kB Pathway Inhibition: Vitamin K, particularly K2, has been shown to inhibit the NF-kB inflammatory signaling pathway, reducing the production of pro-inflammatory cytokines including TNF-alpha, IL-1beta, IL-6, and IL-8. This NF-kB inhibition appears to be independent of Vitamin K's gamma-carboxylation function.
- Inflammatory Marker Reduction: Clinical studies have demonstrated that Vitamin K supplementation (particularly K2) reduces circulating levels of inflammatory markers including C-reactive protein, interleukin-6, and TNF-alpha in various populations.
- 12-Lipoxygenase Inhibition: MK-4 has been shown to inhibit 12-lipoxygenase, reducing the production of pro-inflammatory eicosanoids. This anti-inflammatory mechanism is similar to that described for alpha-tocotrienol (a form of Vitamin E).
- Chronic Disease Inflammation: Given that chronic low-grade inflammation (inflammaging) is a common feature of age-related diseases — cardiovascular disease, osteoporosis, neurodegeneration, diabetes, and cancer — Vitamin K's anti-inflammatory properties may contribute to its protective effects across multiple organ systems.
8. Cancer Research — Preliminary Evidence
Emerging research suggests potential anticancer properties of Vitamin K, particularly Vitamin K2, though this evidence is still in relatively early stages.
- Hepatocellular Carcinoma: Multiple studies from Japan have reported that Vitamin K2 (MK-4) can suppress the recurrence of hepatocellular carcinoma (HCC) after treatment, reduce the progression of viral hepatitis to HCC, and induce apoptosis and cell cycle arrest in HCC cell lines. The mechanism may involve activation of hepatic Vitamin K-dependent proteins and direct transcriptional effects.
- Prostate Cancer: The European Prospective Investigation into Cancer and Nutrition (EPIC-Heidelberg) found that higher dietary intake of Vitamin K2 (but not K1) was associated with a 35% reduction in prostate cancer risk, particularly advanced prostate cancer.
- Lung Cancer: Some epidemiological studies have found inverse associations between Vitamin K2 intake and lung cancer risk, though the evidence is limited and requires confirmation.
- Mechanisms: Proposed anticancer mechanisms of Vitamin K2 include induction of apoptosis, cell cycle arrest, inhibition of NF-kB and other pro-survival signaling pathways, and modulation of Gas6/TAM receptor signaling. The prenyl side chain of menaquinones may also directly inhibit cancer cell growth through effects on the mevalonate pathway.
9. Insulin Sensitivity and Metabolic Health
Growing evidence links Vitamin K status to glucose metabolism, insulin sensitivity, and metabolic syndrome risk.
- Osteocalcin and Insulin Sensitivity: Undercarboxylated osteocalcin (ucOC), somewhat paradoxically, has been identified as a hormone that enhances insulin secretion from pancreatic beta cells and improves insulin sensitivity in peripheral tissues. This hormonal function of ucOC was discovered in animal models and is an active area of investigation in humans. The relationship between Vitamin K status (which affects the carboxylation state of osteocalcin) and metabolic health is complex.
- Diabetes Risk: Several epidemiological studies have found that higher Vitamin K intake is associated with reduced risk of type 2 diabetes. The mechanisms may include anti-inflammatory effects (since inflammation drives insulin resistance), direct effects on pancreatic beta cell function, and modulation of osteocalcin's hormonal activities.
- Inflammatory Markers and Insulin Resistance: Vitamin K2's anti-inflammatory effects (reduction of TNF-alpha, IL-6, CRP) may indirectly improve insulin sensitivity by reducing the chronic inflammatory state that drives insulin resistance in metabolic syndrome and type 2 diabetes.
- Adipose Tissue: Vitamin K-dependent proteins (including Gas6) are expressed in adipose tissue and may influence adipocyte differentiation, inflammation, and metabolic function. Research into these adipose tissue effects is ongoing.
10. Dietary Sources — K1 in Greens, K2 in Fermented Foods
The dietary sources of Vitamin K1 and K2 are distinctly different, reflecting their different biological origins.
- Vitamin K1 Sources (Phylloquinone): Green leafy vegetables are by far the richest sources: kale (~817 mcg per cup cooked), spinach (~888 mcg per cup cooked), collard greens (~1059 mcg per cup cooked), turnip greens, Swiss chard, broccoli (~220 mcg per cup cooked), Brussels sprouts, green leaf lettuce, and parsley. Vegetable oils (soybean, canola, olive) also contain significant K1. Absorption of K1 from vegetables is enhanced by consuming them with dietary fat.
- Natto: The Japanese fermented soybean food natto is the single richest dietary source of Vitamin K2, specifically MK-7, providing approximately 1,000-1,100 mcg of MK-7 per 100-gram serving. Natto consumption is strongly associated with reduced fracture risk and cardiovascular mortality in Japanese epidemiological studies.
- Fermented Cheeses: Hard, aged cheeses (Gouda, Brie, Edam, cheddar) contain significant amounts of Vitamin K2, primarily in the longer-chain MK-8 and MK-9 forms produced by the bacterial cultures used in cheese making. Gouda and Brie are among the richest cheese sources.
- Animal Products (MK-4): Chicken liver, egg yolks, butter, and dark meat poultry contain MK-4, which is synthesized from K1 in animal tissues. The MK-4 content varies with animal diet — pastured animals fed on K1-rich grasses produce higher MK-4 content in their products.
- Fermented Vegetables: Sauerkraut and other fermented vegetables contain small amounts of various menaquinones produced by the fermenting bacteria, though generally far less than natto.
- Gut Bacteria Synthesis: Bacteria in the human large intestine (primarily Bacteroides and Escherichia coli) produce menaquinones (MK-10 through MK-13). However, the contribution of bacterially synthesized K2 to systemic Vitamin K status is debated — much of it may not be efficiently absorbed from the colon. The gut microbiome's contribution to K2 status may be most significant in the context of the local intestinal environment.
11. Deficiency, Drug Interactions, and Newborn Prophylaxis
Vitamin K deficiency has serious clinical consequences, and several important drug interactions and clinical scenarios warrant attention.
- Deficiency Symptoms: Vitamin K deficiency manifests primarily as bleeding tendency — easy bruising, bleeding gums, nosebleeds, heavy menstrual bleeding, blood in urine or stool, and in severe cases, life-threatening hemorrhage. Laboratory findings include prolonged prothrombin time (PT) and elevated International Normalized Ratio (INR).
- Subclinical Deficiency: A significant proportion of the population may have Vitamin K levels that are adequate for coagulation but insufficient for full activation of extrahepatic Vitamin K-dependent proteins (osteocalcin, MGP). This "subclinical" deficiency may contribute to osteoporosis and arterial calcification without causing any bleeding symptoms.
- Warfarin Interaction: Warfarin (Coumadin) is the most important drug interaction with Vitamin K. Warfarin inhibits Vitamin K epoxide reductase (VKORC1), blocking the Vitamin K cycle and reducing the production of functional coagulation factors. Patients on warfarin must maintain consistent Vitamin K intake to ensure stable anticoagulation. Large fluctuations in Vitamin K intake can cause dangerous over- or under-anticoagulation. Patients should not avoid Vitamin K-rich foods but should consume them in consistent daily amounts.
- Antibiotic Effects: Broad-spectrum antibiotics can reduce gut bacterial synthesis of Vitamin K2 and, when combined with inadequate dietary intake, may precipitate Vitamin K deficiency. This is particularly relevant in critically ill patients on prolonged antibiotic therapy with limited oral intake.
- Fat Malabsorption: Because Vitamin K is fat-soluble, conditions that impair fat absorption — celiac disease, cystic fibrosis, bile duct obstruction, chronic pancreatitis, inflammatory bowel disease — can cause Vitamin K deficiency even with adequate dietary intake.
- Newborn Vitamin K Prophylaxis: Newborns are at risk for Vitamin K deficiency bleeding (VKDB, formerly called hemorrhagic disease of the newborn) because: (1) Vitamin K does not efficiently cross the placenta, (2) newborns have sterile intestines with no K2-producing bacteria, (3) breast milk is low in Vitamin K, and (4) newborn liver stores are minimal. A single intramuscular injection of Vitamin K1 (0.5-1 mg) at birth effectively prevents VKDB and is recommended by pediatric medical organizations worldwide. Without prophylaxis, early VKDB (first 24 hours), classical VKDB (days 2-7), and late VKDB (weeks 2-12, potentially causing fatal intracranial hemorrhage) can occur.
- Cholestyramine and Orlistat: These medications can impair Vitamin K absorption — cholestyramine by binding bile acids needed for fat absorption, and orlistat by inhibiting lipase and reducing fat (and fat-soluble vitamin) absorption.
12. Recommended Daily Allowance (RDA) and Supplementation
Understanding Vitamin K intake recommendations, supplementation forms, and optimal dosing is important for clinical practice.
- Adequate Intake (AI): No RDA has been established for Vitamin K; instead, an Adequate Intake (AI) is set at 120 mcg/day for adult men and 90 mcg/day for adult women. These values are based on the intake level that maintains normal blood clotting and are relatively easily achieved with one serving of green leafy vegetables daily.
- AI May Be Insufficient for Extrahepatic Functions: Growing evidence suggests that the current AI values, while adequate for coagulation, may be insufficient for optimal activation of extrahepatic Vitamin K-dependent proteins (osteocalcin, MGP). Higher intakes may be needed for full bone and cardiovascular benefit.
- No Established Upper Limit: No Tolerable Upper Intake Level (UL) has been established for Vitamin K1 or K2, as no adverse effects of high intake have been identified in individuals not taking anticoagulant medications. Vitamin K has remarkably low toxicity.
- K2 Supplementation — MK-7: MK-7 is the most popular supplemental form of Vitamin K2 due to its long half-life, superior bioavailability, and evidence for bone and cardiovascular benefits. Common supplemental doses range from 45 mcg to 200 mcg per day. MK-7 at 180-200 mcg/day has been shown to significantly reduce dp-ucMGP (a marker of vascular K deficiency) and improve osteocalcin carboxylation.
- K2 Supplementation — MK-4: MK-4 is used at much higher doses (typically 45 mg/day, divided into three doses) for osteoporosis treatment, particularly in Japan where it is an approved pharmaceutical for fracture prevention. At nutritional doses, MK-4 has a very short half-life and does not accumulate to significant steady-state levels.
- Vitamin D3 + K2 Combination: The combination of Vitamin D3 and K2 has become one of the most popular supplement combinations, based on their synergistic roles in calcium metabolism. Vitamin D3 increases calcium absorption and stimulates osteocalcin and MGP production, while K2 activates these proteins to direct calcium into bones and away from arteries. Many clinicians recommend taking K2 with D3 supplementation to ensure proper calcium distribution.
- Dental Health: Vitamin K2, through its activation of osteocalcin and other calcium-regulating proteins in dental tissues, may support dental mineralization and tooth health. Some research suggests that K2 deficiency may contribute to dental caries and poor jaw bone density, though clinical evidence in this area is still developing.
- Warfarin Patients: Patients on warfarin should not take Vitamin K supplements without medical supervision. However, some clinicians advocate for low-dose, consistent daily K1 supplementation (100 mcg/day) in warfarin patients to stabilize INR fluctuations caused by variable dietary K1 intake. This approach requires close INR monitoring and dose adjustment.
13. Vitamin K and Dental Health
Vitamin K2's role in calcium metabolism extends to dental health, where it supports tooth mineralization and jaw bone integrity.
- Dental Mineralization: Teeth, like bones, contain a mineralized matrix composed primarily of hydroxyapatite. Osteocalcin and other Vitamin K-dependent proteins are expressed in odontoblasts (the cells that form dentin) and play a role in directing calcium phosphate mineral deposition within tooth structure. Adequate Vitamin K2 supports proper dentin mineralization during tooth development.
- Alveolar Bone Health: The alveolar bone — the jaw bone that supports and anchors the teeth — is a metabolically active bone tissue that undergoes continuous remodeling. Vitamin K2's bone-protective effects (osteocalcin activation, osteoblast support, osteoclast inhibition) apply to alveolar bone, potentially supporting tooth retention and preventing periodontal bone loss.
- Weston Price's Observations: The early 20th-century dentist Weston Price observed that traditional diets rich in what he called "Activator X" (now believed to be Vitamin K2, particularly MK-4 from grass-fed animal products) were associated with excellent dental health, broad dental arches, and resistance to tooth decay. While Price's work predated the identification of Vitamin K2, his observations are consistent with K2's role in dental mineralization.
- Dental Caries Prevention: Some researchers have proposed that Vitamin K2 may influence dental caries susceptibility through effects on saliva composition, dentin remineralization, and the immune response to oral bacteria. However, clinical evidence for K2's role in caries prevention is preliminary and requires further investigation.
- Orthodontic and Implant Considerations: Adequate Vitamin K2 status may support bone healing and integration following dental implant placement and orthodontic tooth movement, given its role in osteocalcin activation and bone remodeling regulation. These applications are subjects of emerging clinical interest.
14. Vitamin K in Specific Clinical Populations
Certain patient populations have unique Vitamin K considerations that warrant clinical attention.
- Chronic Kidney Disease (CKD): CKD patients have dramatically accelerated vascular calcification, extremely elevated dp-ucMGP levels, and significantly impaired Vitamin K status. The combination of dietary restrictions (limiting K1-rich vegetables), impaired K2 metabolism, and uremic toxin effects creates severe functional Vitamin K deficiency in the vasculature. Vitamin K2 supplementation in CKD patients is an active area of clinical investigation with several ongoing randomized trials.
- Hemodialysis Patients: Patients on hemodialysis have among the highest rates of vascular calcification and cardiovascular mortality of any clinical population. Studies consistently show that hemodialysis patients have profoundly elevated dp-ucMGP and ucOC levels. MK-7 supplementation effectively reduces these biomarkers, though clinical endpoint trials are still needed.
- Post-Menopausal Women: Postmenopausal women are at particular risk for both osteoporosis and arterial calcification due to estrogen decline. Vitamin K2 supplementation addresses both risks simultaneously — activating osteocalcin for bone protection and MGP for arterial protection — making it a particularly rational supplement for this population.
- Patients on Proton Pump Inhibitors (PPIs): Long-term PPI use is associated with increased fracture risk. While the mechanism is debated (reduced calcium absorption, altered bone remodeling), some researchers suggest that PPI-treated patients may benefit from K2 supplementation to optimize bone health.
- Liver Disease Patients: Patients with chronic liver disease may have impaired Vitamin K metabolism, reduced coagulation factor synthesis, and altered Vitamin K-dependent protein carboxylation. The liver disease context complicates Vitamin K status assessment, as coagulopathy may reflect impaired hepatic synthesis rather than Vitamin K deficiency per se.
- Patients on Antibiotic Therapy: Prolonged broad-spectrum antibiotic therapy can deplete gut bacterial sources of Vitamin K2 and, when combined with poor dietary intake (common in hospitalized patients), can precipitate clinically significant Vitamin K deficiency. Monitoring and supplementation should be considered in patients on extended antibiotic courses with limited oral intake.
- Bariatric Surgery Patients: Patients who have undergone bariatric surgery (particularly malabsorptive procedures such as Roux-en-Y gastric bypass and biliopancreatic diversion) are at risk for fat-soluble vitamin deficiency, including Vitamin K. Lifelong monitoring and supplementation are recommended for these patients.
15. Historical Discovery and Nobel Prize Legacy
Vitamin K was discovered around 1929 by the Danish biochemist Henrik Dam, who noticed that chicks on a fat-stripped diet developed unexplained hemorrhages and named the missing factor the "coagulation vitamin" (the K is for the German and Scandinavian Koagulation). Dam shared the 1943 Nobel Prize in Physiology or Medicine with the American biochemist Edward Adelbert Doisy, whose laboratory worked out the vitamin's chemical structure and synthesized it. From there the trail runs through the sweet-clover cattle disease that gave us the drug warfarin, the 1974 discovery of gamma-carboxyglutamic acid that finally explained how the vitamin works, and the modern recognition of vitamin K2's roles in bone and arteries — the fuller story is told in the dedicated History & Discovery article.
16. Absorption, Bioavailability, and Tissue Distribution
Understanding the absorption, transport, and tissue distribution of different Vitamin K forms is critical for optimizing nutritional status and selecting appropriate supplementation strategies.
- K1 Absorption from Foods: Vitamin K1 from green leafy vegetables is tightly bound to chloroplast membranes, resulting in relatively low bioavailability (approximately 5-10% from raw vegetables). Cooking, chopping, and adding dietary fat significantly improve K1 absorption. K1 from oils and supplements is more bioavailable (approximately 80%) because it is already in a lipid-soluble form.
- K2 Absorption: Vitamin K2 forms (MK-4, MK-7) are generally well absorbed from both food and supplement sources, particularly when consumed with dietary fat. MK-7 from natto or supplements has excellent bioavailability, with absorption estimated at 80-90%. The long isoprenoid side chain of MK-7 contributes to its extended circulation time.
- Lipoprotein Transport: After intestinal absorption, Vitamin K (both K1 and K2) is incorporated into chylomicrons and transported to the liver via the lymphatic system. K1 is preferentially retained by the liver for coagulation factor synthesis. K2 forms, particularly MK-7 and longer-chain menaquinones, are redistributed from the liver in LDL and HDL particles, providing more effective delivery to extrahepatic tissues including bone, arterial walls, and the brain.
- Half-Life Comparison: The biological half-lives of different Vitamin K forms vary dramatically: K1 has a half-life of approximately 1-2 hours, MK-4 has a half-life of a few hours, while MK-7 has a half-life of approximately 72 hours. This 36-fold difference in half-life between K1 and MK-7 means that MK-7 accumulates to much higher steady-state plasma concentrations with daily dosing, providing more consistent and sustained tissue availability.
- Tissue-Specific Accumulation: K1 is concentrated primarily in the liver. MK-4 is found in the brain, kidneys, pancreas, salivary glands, and arterial walls — suggesting important tissue-specific functions. MK-7 distributes broadly to extrahepatic tissues and, due to its long half-life, achieves higher steady-state concentrations in bone and vascular tissue than K1 at equivalent dietary intakes.
- K1 to MK-4 Conversion: The enzyme UBIAD1 (UbiA prenyltransferase domain-containing protein 1) converts Vitamin K1 to MK-4 in various tissues, including the brain, kidneys, and testes. This tissue-specific conversion suggests that MK-4 has important local functions that the body ensures through endogenous synthesis, independent of dietary MK-4 intake. The side chain of K1 is removed and replaced with a geranylgeranyl side chain to produce MK-4.
- Factors Affecting Absorption: Vitamin K absorption requires bile salts (impaired in biliary obstruction and cholestatic liver disease), pancreatic lipase (impaired in pancreatic insufficiency), and healthy intestinal mucosa (impaired in celiac disease, Crohn's disease, and short bowel syndrome). Fat-soluble vitamin malabsorption syndromes affect Vitamin K along with Vitamins A, D, and E.
- Genetic Variations: Polymorphisms in genes encoding VKORC1, CYP4F2, and apolipoprotein E (ApoE) can affect individual Vitamin K metabolism, tissue levels, and response to supplementation. The VKORC1 -1639G>A polymorphism, which affects warfarin sensitivity, also influences baseline Vitamin K status and may affect the optimal K2 supplementation dose.
17. Vitamin K Status Assessment and Laboratory Testing
Assessing Vitamin K status is more complex than for many other vitamins due to the existence of multiple forms, tissue-specific distribution, and the difference between coagulation adequacy and optimal extrahepatic function.
- Plasma Phylloquinone (K1): Plasma K1 concentration reflects recent dietary intake and has a short half-life, making it a poor indicator of tissue stores. Fasting values are typically 0.3-2.6 nmol/L. Plasma K1 is useful for detecting acute deficiency but does not reflect long-term Vitamin K status or extrahepatic tissue availability.
- Prothrombin Time (PT) and INR: PT and INR are the traditional clinical tests for Vitamin K status but are insensitive — they only become abnormal when coagulation factor levels fall below approximately 50% of normal, representing severe deficiency. Normal PT/INR does not exclude subclinical Vitamin K insufficiency for bone and vascular health.
- PIVKA-II (des-gamma-carboxy prothrombin): A more sensitive marker of hepatic Vitamin K status than PT/INR. Elevated PIVKA-II indicates that the liver's Vitamin K supply is insufficient for complete prothrombin carboxylation, even when PT remains within the normal range. PIVKA-II is also used as a tumor marker for hepatocellular carcinoma.
- Undercarboxylated Osteocalcin (ucOC): The most widely used biomarker of Vitamin K status for bone health. Elevated ucOC (or a high ucOC/total OC ratio) indicates insufficient Vitamin K for complete osteocalcin carboxylation in bone. This marker is responsive to both K1 and K2 supplementation and has been validated as a predictor of fracture risk.
- dp-ucMGP: The emerging gold standard biomarker for vascular Vitamin K status. Elevated dp-ucMGP indicates insufficient Vitamin K2 for complete MGP carboxylation in the vascular wall and is associated with arterial calcification and cardiovascular mortality. This marker is particularly useful for guiding K2 supplementation for cardiovascular protection.
- Triage Theory: The concept of Vitamin K triage, proposed by Bruce Ames, suggests that when Vitamin K supply is limited, the body prioritizes coagulation (essential for immediate survival) over bone and vascular health (important for long-term health). This explains why subclinical deficiency in bone and arteries can exist alongside normal coagulation tests — a finding with profound implications for public health Vitamin K recommendations.
18. Emerging Research and Future Directions
Vitamin K research is advancing rapidly across multiple fronts, with significant potential for expanding clinical applications.
- Vascular Calcification Reversal: Perhaps the most exciting area of current research is whether Vitamin K2 supplementation can not only prevent but actually reverse established arterial calcification. Animal studies have shown calcification regression with K2 supplementation, and human trials (including the VitaK-CAC trial) are investigating whether MK-7 can slow or reverse coronary artery calcification progression. Positive results would have enormous clinical implications.
- Chronic Kidney Disease Outcomes: Multiple randomized trials are examining whether K2 supplementation improves cardiovascular outcomes, vascular calcification, and bone health in CKD patients — a population with some of the highest cardiovascular mortality rates and most severe Vitamin K insufficiency. These trials may establish K2 as a standard component of CKD management.
- Direct Oral Anticoagulant (DOAC) Era: The increasing replacement of warfarin with DOACs (rivaroxaban, apixaban, dabigatran, edoxaban) for anticoagulation may inadvertently improve Vitamin K-dependent extrahepatic protein function, since DOACs do not inhibit the Vitamin K cycle. Research is examining whether DOAC-treated patients have better bone and vascular outcomes than warfarin-treated patients due to preserved Vitamin K function.
- Vitamin K and Inflammation: Emerging research is exploring how matrix Gla protein (MGP) activation status, measured via dp-ucMGP, relates to systemic inflammation, endothelial dysfunction, and outcomes in acute respiratory illnesses generally. These studies are broadening the traditional view of Vitamin K beyond coagulation into vascular and immune biology.
- Microbiome and K2 Production: Advances in microbiome research are improving our understanding of how gut bacteria contribute to Vitamin K2 status. The composition of the gut microbiome varies dramatically between individuals, and this variation may influence endogenous K2 production and, consequently, bone and cardiovascular health. Probiotic and prebiotic strategies to enhance gut K2 production are being explored.
- Precision Nutrition: Genetic polymorphisms in VKORC1, CYP4F2, UBIAD1, and ApoE affect individual Vitamin K metabolism and may eventually guide personalized Vitamin K supplementation recommendations based on genotype. This pharmacogenomic approach could optimize the dose and form of Vitamin K supplementation for each individual's genetic profile.
- Combination Supplement Optimization: Research continues into the optimal ratios and doses of Vitamin K2 in combination with Vitamin D3, calcium, and magnesium for bone and cardiovascular health. The development of evidence-based combination formulations is a practical outcome of the growing understanding of nutrient synergies in calcium metabolism.
- Neonatal Applications: Research is refining optimal Vitamin K prophylaxis protocols for newborns, including investigations into oral prophylaxis regimens that may achieve comparable effectiveness to intramuscular injection while addressing parental concerns. The development of novel formulations (such as mixed micellar preparations) may improve oral K1 bioavailability in neonates.
- Cancer Prevention: The preliminary epidemiological associations between Vitamin K2 intake and reduced risk of certain cancers (hepatocellular carcinoma, prostate cancer) are being explored in mechanistic studies and early-phase clinical trials. Understanding the molecular pathways by which K2 influences cell growth, apoptosis, and immune surveillance may open new avenues for cancer prevention research.
- Skin and Wound Healing: Emerging evidence suggests that Vitamin K may play roles in skin health, wound healing, and reduction of bruising and dark under-eye circles. Topical Vitamin K formulations are being developed for dermatological applications, though clinical evidence remains limited and this represents an early-stage area of investigation.
Connections
- Vitamin K Benefits Hub
- K2 and Arterial Calcification
- K2 and Bone Health
- MK-7 vs MK-4
- K2 and Insulin Sensitivity
- Vitamin K and Blood Clotting
- Calcium
- Calcium and Muscle Function — excitation-contraction coupling, cramps, tetany, and the calcium-magnesium balance for cardiac and skeletal muscle.
- Osteoporosis
- Vitamin D3
- Cancer
- Natto
- Fermented Foods
- Broccoli
- Kale
- Spinach
- Cardiovascular Disease
- Liver Disease
- Magnesium
- Vitamin E
- Insulin Resistance
- Glutamic Acid