Zinc – Essential Trace Mineral for Human Health
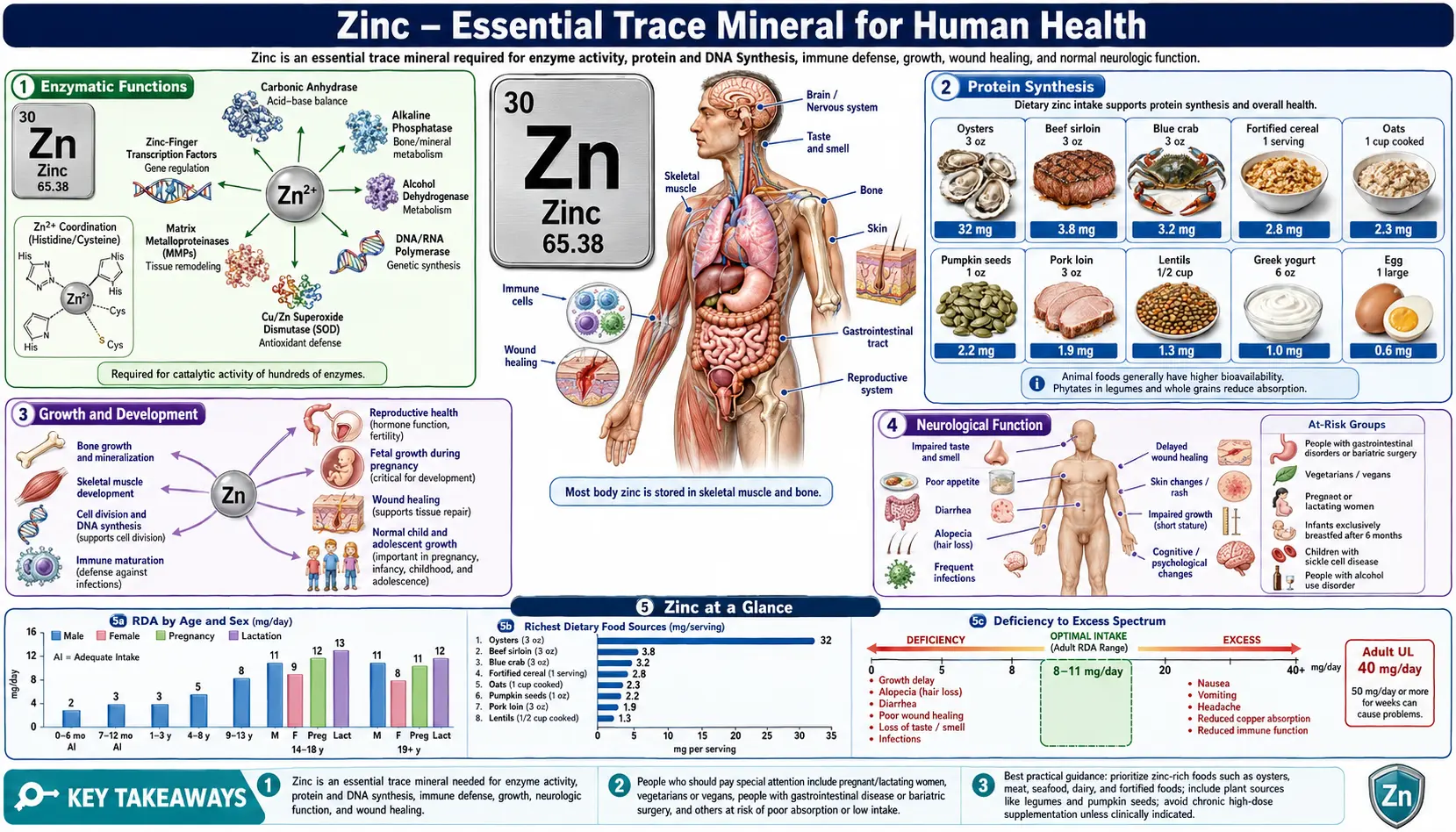
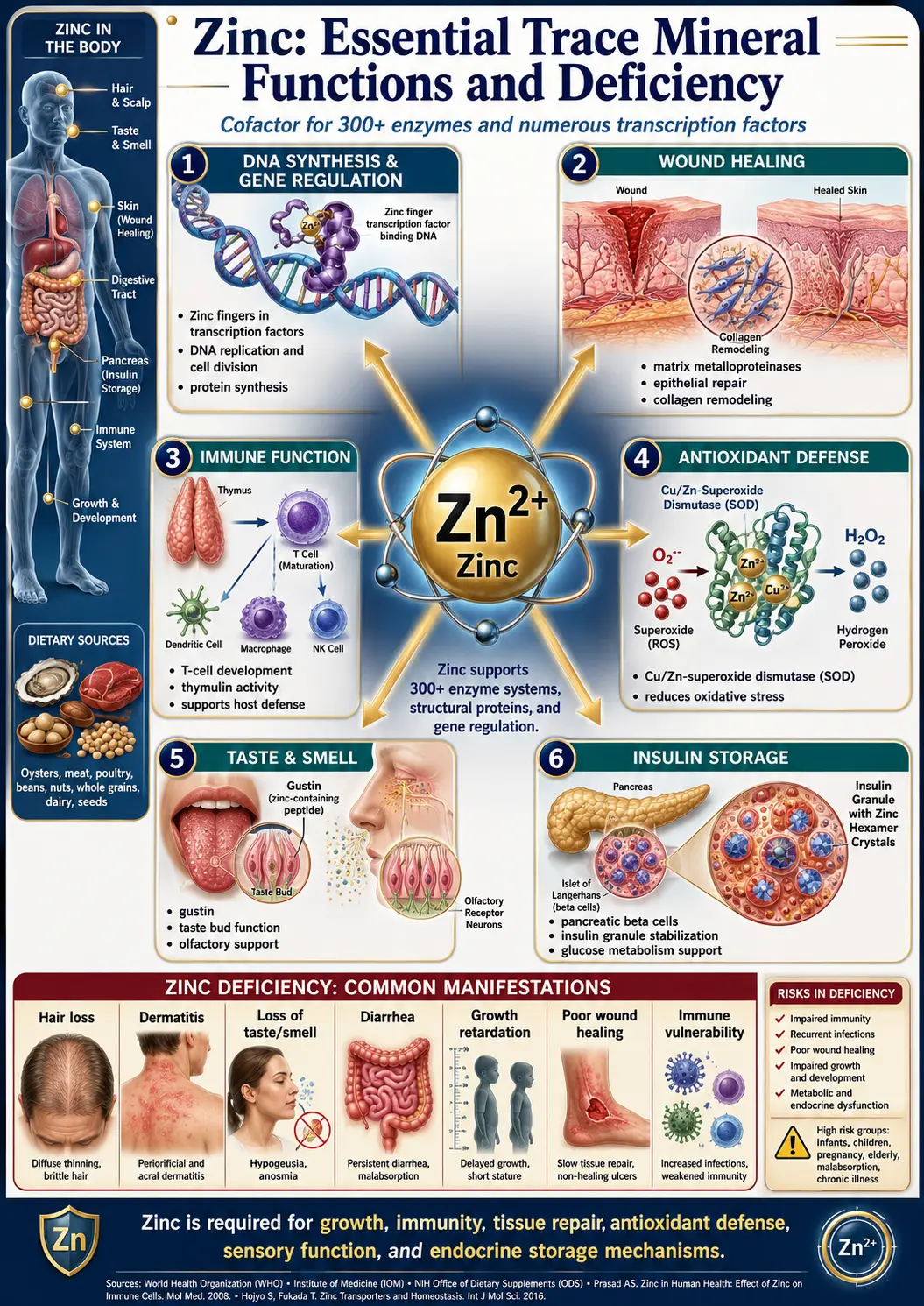
Table of Contents
- Enzymatic Functions
- Immune System Function
- Wound Healing
- Protein Synthesis
- DNA Synthesis and Cell Division
- Taste and Smell
- Reproductive Health
- Growth and Development
- Antioxidant Defense
- Skin Health
- Neurological Function
- Connections
- Featured Videos
Zinc is the second most abundant trace mineral in the human body after iron. Present in every cell, it is indispensable for a vast range of biological processes including enzymatic catalysis, immune defense, wound repair, growth, and neurological function. The adult body contains approximately 2–3 grams of zinc, distributed primarily in skeletal muscle, bone, skin, liver, and the prostate gland. Because the body lacks a dedicated zinc storage system, regular dietary intake is essential to maintain adequate levels.
Enzymatic Functions
Zinc serves as a structural or catalytic cofactor in more than 300 enzymes spanning all six classes of enzyme activity. Its role in metalloenzymes is fundamental to human biochemistry.
- Carbonic anhydrase – Zinc is essential for this enzyme, which catalyzes the reversible hydration of carbon dioxide. Carbonic anhydrase is critical for respiration, acid–base balance, and renal tubular reabsorption of bicarbonate.
- Alcohol dehydrogenase – A zinc-dependent enzyme in the liver responsible for the metabolism of ethanol and other alcohols, as well as the interconversion of retinol and retinal in vitamin A metabolism.
- Carboxypeptidases – Zinc-containing digestive enzymes that cleave peptide bonds at the carboxyl terminus of proteins, facilitating protein digestion in the small intestine.
- Alkaline phosphatase – Requires zinc for catalytic activity. This enzyme is involved in dephosphorylation reactions across many tissues including bone, liver, and kidney.
- RNA polymerase – Zinc is required for RNA polymerase function, linking the mineral directly to gene transcription and the production of messenger RNA.
- Superoxide dismutase (Cu/Zn-SOD) – The cytoplasmic form of this antioxidant enzyme depends on both copper and zinc for its structure and catalytic action.
- Matrix metalloproteinases (MMPs) – A family of zinc-dependent endopeptidases that degrade extracellular matrix components, playing roles in tissue remodeling, wound healing, and embryonic development.
- Zinc finger proteins – Zinc stabilizes the finger-like protein domains that bind DNA and regulate gene expression. The human genome encodes an estimated 2,500 zinc finger proteins, making zinc one of the most important elements in transcriptional regulation.
Immune System Function
Zinc is sometimes called the "gatekeeper of immune function" because of its broad influence on both innate and adaptive immunity. Deficiency even at marginal levels can impair immune surveillance and increase susceptibility to infection.
- T-cell development and function – Zinc is required for the maturation of T lymphocytes in the thymus. It activates thymulin, a thymic hormone that is necessary for T-cell differentiation. Without adequate zinc, the thymus atrophies and T-cell output declines, weakening cell-mediated immunity.
- Natural killer (NK) cell activity – NK cells provide a first line of defense against virally infected and malignant cells. Zinc deficiency reduces NK cell cytotoxicity, impairing the body's ability to eliminate abnormal cells.
- Neutrophil and macrophage function – Zinc supports phagocytosis, oxidative burst, and chemotaxis in neutrophils and macrophages. These innate immune cells depend on zinc-containing enzymes for their antimicrobial activity.
- Cytokine regulation – Zinc modulates the production of pro-inflammatory cytokines such as interleukin-1 beta (IL-1β), interleukin-6 (IL-6), and tumor necrosis factor alpha (TNF-α). Both deficiency and excess zinc can dysregulate cytokine balance.
- Barrier function – Zinc maintains the integrity of skin and mucosal membranes, which serve as physical barriers to pathogen entry.
For a detailed discussion, see Zinc for Immune Function.
Wound Healing
Zinc participates in every phase of wound repair, from initial hemostasis and inflammation through proliferation and tissue remodeling.
- Collagen synthesis – Zinc is a cofactor for enzymes involved in collagen cross-linking and maturation. Adequate zinc ensures that newly formed collagen has proper tensile strength.
- Cell proliferation – Zinc is required for DNA synthesis and mitosis, both of which are essential for the rapid cell division that occurs during wound closure. Keratinocyte and fibroblast proliferation depend on zinc availability.
- Inflammatory modulation – Zinc helps regulate the inflammatory phase so that it is effective against pathogens without causing excessive tissue damage. It influences neutrophil recruitment and the resolution of inflammation.
- Metalloproteinase activity – Zinc-dependent matrix metalloproteinases (MMPs) remodel the extracellular matrix during the proliferative and remodeling phases, enabling scar maturation and tissue restoration.
- Clinical significance – Zinc deficiency is associated with delayed wound healing, chronic ulcers, and impaired surgical recovery. Zinc supplementation has been shown to accelerate healing in zinc-deficient patients.
For a detailed discussion, see Zinc for Wound Healing.
Protein Synthesis
Zinc is integral to the machinery of protein synthesis at multiple levels.
- Ribosomal structure – Zinc ions are incorporated into ribosomal proteins and are necessary for the structural integrity of ribosomes, the cellular organelles where translation of mRNA into protein occurs.
- Transfer RNA (tRNA) metabolism – Zinc-dependent enzymes participate in tRNA processing and aminoacyl-tRNA synthetase activity, ensuring the accurate delivery of amino acids during translation.
- Transcription factor activity – Many transcription factors contain zinc finger motifs. These proteins regulate the expression of genes encoding structural proteins, enzymes, hormones, and receptors.
- Protein folding – Zinc ions serve as structural elements that stabilize the three-dimensional conformation of numerous proteins, ensuring their proper folding and biological activity.
DNA Synthesis and Cell Division
Zinc is essential for faithful DNA replication and orderly cell division, making it critically important during periods of rapid growth.
- DNA polymerase – This zinc-containing enzyme catalyzes the replication of DNA during the S phase of the cell cycle. Without sufficient zinc, DNA replication is impaired and cell division slows.
- Thymidine kinase – A zinc-dependent enzyme involved in the salvage pathway for thymidine, a nucleoside required for DNA synthesis.
- DNA repair – Zinc is a component of several DNA repair enzymes, including poly(ADP-ribose) polymerase (PARP), which detects and signals DNA strand breaks. Zinc deficiency increases susceptibility to DNA damage and genomic instability.
- Cell cycle regulation – Zinc influences the activity of cyclin-dependent kinases and other cell cycle regulators, ensuring that cells progress through division checkpoints in an orderly manner.
- Apoptosis – Zinc modulates programmed cell death. Both excessive and insufficient zinc levels can trigger aberrant apoptosis, with implications for cancer, immune function, and tissue homeostasis.
Taste and Smell
Zinc plays a specialized role in the chemical senses of taste (gustation) and smell (olfaction).
- Gustin (carbonic anhydrase VI) – This zinc-dependent protein is secreted by the parotid salivary glands and is essential for the growth and maintenance of taste bud papillae. Zinc deficiency leads to reduced gustin levels and impaired taste acuity, a condition known as hypogeusia.
- Olfactory receptor neurons – Zinc is concentrated in the olfactory epithelium and the olfactory bulb of the brain. It modulates neurotransmission in olfactory pathways, and deficiency can cause hyposmia (reduced sense of smell) or anosmia (complete loss of smell).
- Clinical relevance – Dysgeusia (distorted taste) and anosmia are recognized clinical signs of zinc deficiency. Restoration of zinc status through supplementation often leads to recovery of these senses.
Reproductive Health
Zinc is vital for reproductive function in both males and females.
Male Reproductive Function
- Spermatogenesis – Zinc is present at high concentrations in the prostate gland and seminal fluid. It is required for normal sperm development, motility, and membrane stability. Zinc deficiency is associated with reduced sperm count and impaired fertility.
- Testosterone metabolism – Zinc influences testosterone synthesis and metabolism. Deficiency can lead to hypogonadism and reduced serum testosterone levels, which may be partially reversible with zinc repletion.
- Prostate health – The prostate gland accumulates more zinc than any other soft tissue in the body. Adequate zinc levels are believed to support normal prostate cell function, and research has explored the relationship between zinc status and prostate pathology.
Female Reproductive Function
- Oocyte development – Zinc is essential for oocyte maturation and the meiotic cell divisions that produce viable eggs. At fertilization, a dramatic release of zinc ions known as the "zinc spark" occurs, which is necessary for egg activation and the block to polyspermy.
- Pregnancy – Zinc requirements increase during pregnancy to support placental development, fetal growth, and maternal tissue expansion. Severe zinc deficiency during pregnancy is associated with complications including preterm delivery, low birth weight, and congenital abnormalities.
- Lactation – Breast milk provides zinc to the nursing infant. Maternal zinc intake during lactation influences milk zinc concentration and the infant's zinc status during the critical early months of development.
Growth and Development
Zinc is indispensable for normal growth from conception through adolescence.
- Prenatal development – Zinc is required for embryogenesis, organogenesis, and fetal skeletal development. Zinc-dependent transcription factors regulate the genes that guide tissue differentiation during embryonic development.
- Childhood growth – Zinc deficiency is one of the most common causes of growth faltering in children worldwide. Supplementation trials in zinc-deficient populations have demonstrated significant improvements in linear growth and weight gain.
- Growth hormone signaling – Zinc influences the growth hormone–insulin-like growth factor 1 (GH-IGF-1) axis. Deficiency impairs growth hormone release and reduces IGF-1 levels, contributing to stunted growth.
- Bone development – Zinc stimulates osteoblast proliferation and differentiation while inhibiting osteoclast activity, contributing to bone formation and mineralization during growth.
- Pubertal development – Adequate zinc is necessary for the hormonal changes of puberty. Zinc deficiency in adolescence can delay sexual maturation and secondary sexual characteristic development.
Antioxidant Defense
Although zinc is not a classical antioxidant in the way that vitamins C and E are, it contributes significantly to the body's antioxidant defense systems.
- Cu/Zn superoxide dismutase (SOD1) – This cytoplasmic enzyme converts superoxide radicals into hydrogen peroxide and molecular oxygen, representing a critical first step in neutralizing reactive oxygen species (ROS). Zinc provides structural stability to the enzyme, while copper participates directly in catalysis.
- Metallothionein induction – Zinc induces the synthesis of metallothioneins, a family of cysteine-rich proteins that scavenge free radicals, sequester toxic metals such as cadmium and mercury, and serve as an intracellular zinc reservoir.
- Protection of sulfhydryl groups – Zinc binds to thiol groups on proteins, shielding them from oxidative damage. This protective effect helps preserve the function of enzymes and structural proteins that contain critical cysteine residues.
- Inhibition of NADPH oxidase – Zinc inhibits the enzyme NADPH oxidase, which generates superoxide radicals. By reducing NADPH oxidase activity, zinc limits the production of ROS in endothelial cells and other tissues.
- NF-kB modulation – Zinc regulates the nuclear factor kappa-B (NF-κB) signaling pathway, which controls the expression of genes involved in inflammation and oxidative stress. Adequate zinc levels help prevent excessive NF-κB activation.
Skin Health
The skin contains approximately 5% of the body's total zinc content, making it the third most zinc-rich tissue after muscle and bone.
- Keratinocyte differentiation – Zinc regulates the proliferation and differentiation of keratinocytes, the predominant cell type in the epidermis. Proper zinc status supports normal epidermal turnover and barrier function.
- Sebaceous gland function – Zinc influences sebum production and has anti-androgenic effects at the level of the sebaceous gland. This is one reason zinc supplementation is sometimes used as an adjunct in the management of acne vulgaris.
- Photoprotection – Zinc oxide is widely used in topical sunscreens as a physical UV filter. Internally, zinc's antioxidant properties help protect skin cells from ultraviolet radiation-induced oxidative damage.
- Dermatological conditions – Zinc deficiency causes acrodermatitis enteropathica, a genetic disorder characterized by severe dermatitis, diarrhea, and alopecia. Acquired zinc deficiency can produce similar skin lesions. Zinc is also used therapeutically in conditions such as diaper dermatitis, warts, and certain inflammatory skin diseases.
- Collagen and elastin maintenance – Zinc supports the enzymes responsible for collagen and elastin cross-linking, which maintain skin firmness and elasticity.
Neurological Function
Zinc is highly concentrated in the brain, particularly in the hippocampus, amygdala, and cerebral cortex, where it plays roles in neurotransmission, synaptic plasticity, and neuroprotection.
- Synaptic zinc signaling – A subset of glutamatergic neurons in the forebrain sequester free zinc in synaptic vesicles and release it during neurotransmission. This synaptically released zinc modulates the activity of NMDA receptors, GABA receptors, and glycine receptors, influencing excitatory and inhibitory signaling.
- Learning and memory – Zinc is involved in long-term potentiation (LTP), the synaptic mechanism underlying memory formation. Disruptions in brain zinc homeostasis have been linked to cognitive impairment and memory deficits.
- Neurodevelopment – Zinc is critical for brain development during fetal life and early childhood. Deficiency during these periods can lead to impaired cognitive development, behavioral abnormalities, and attention deficits.
- Neuroprotection – Through its antioxidant functions and its ability to regulate apoptotic pathways, zinc helps protect neurons from excitotoxicity and oxidative damage. However, excessive free zinc release during ischemic events can paradoxically contribute to neuronal death, highlighting the importance of tightly regulated zinc homeostasis in the brain.
- Mood regulation – Zinc modulates serotonergic and dopaminergic neurotransmission. Low serum zinc levels have been observed in individuals with depression, and zinc supplementation has shown potential as an adjunctive therapy in clinical studies of major depressive disorder.
- Neurodegenerative disease – Dysregulated zinc metabolism has been implicated in Alzheimer's disease, where zinc interacts with amyloid-beta peptides and may influence plaque formation. Research continues to explore the complex relationship between zinc homeostasis and neurodegeneration.
Connections
- Zinc Benefits Deep Dive
- Zinc for Immune Function
- Zinc for Wound Healing
- Zinc for Testosterone
- Zinc for Skin Health
- Copper
- Relationship Between Hemoglobin and Ceruloplasmin — why ceruloplasmin (the copper ferroxidase) is required to load iron into hemoglobin, and why "iron-deficiency anemia" is often functional copper deficiency.
- Selenium
- Iron
- Vitamin A
- Vitamin C
- Vitamin D3
- Collagen
- Testosterone Test
- Glycine
- Cysteine
- Acne
- Alopecia
- Depression
- Lead
- Copper-Iron Dysregulation
- Eggs