Zinc and Immune System Function
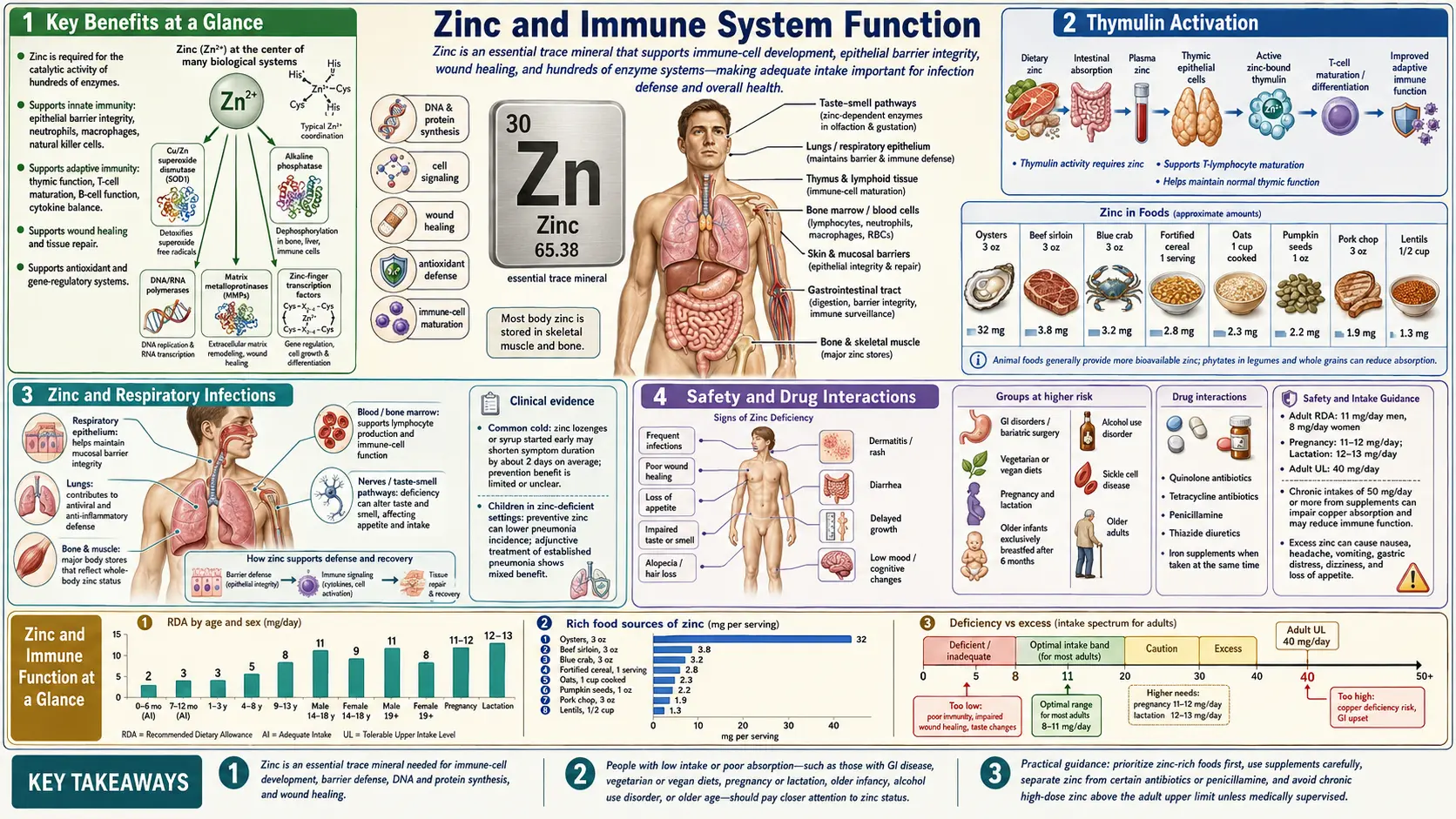
Zinc is one of the most important micronutrients for immune competence. It influences virtually every aspect of the immune response, from the physical barriers that prevent pathogen entry to the highly specialized adaptive immune cells that target and eliminate specific threats. An estimated two billion people worldwide have inadequate zinc intake, and even mild deficiency can significantly compromise immune function and increase vulnerability to infectious disease.
Table of Contents
- Key Benefits at a Glance
- Innate Immunity
- Adaptive Immunity
- Thymulin Activation
- Natural Killer (NK) Cell Activity
- Cytokine Production and Regulation
- Deficiency and Susceptibility to Infection
- Zinc and Respiratory Infections
- Clinical Evidence and Therapeutic Considerations
- Dosing and Dietary Sources
- Safety and Drug Interactions
- Research Papers
- Connections
- Featured Videos
Key Benefits at a Glance
- Reduces cold duration – Cochrane review: zinc lozenges within 24 hours shorten cold symptoms by ~1–2 days.
- Cuts diarrheal disease – WHO/UNICEF standard: zinc supplementation reduces childhood diarrhea duration by ~1 day.
- Supports thymus function – Required for thymulin activity and T-cell maturation.
- Promotes Th1 immunity – Balances Th1/Th2 response, critical against intracellular pathogens.
- Barrier integrity – Maintains epithelial tight junctions in skin, gut, and respiratory tract.
- NK and neutrophil function – Essential for chemotaxis, phagocytosis, and cytotoxic granule release.
- Reverses immunosenescence – Supplementation in deficient elderly restores thymulin and T-cell function.
Innate Immunity
The innate immune system provides the body's first line of defense against invading pathogens. Zinc supports multiple components of innate immunity.
- Epithelial barrier integrity – Zinc is essential for maintaining the structural integrity of the skin and mucosal surfaces that line the respiratory, gastrointestinal, and genitourinary tracts. These barriers physically prevent pathogen entry. Zinc deficiency leads to increased epithelial permeability, allowing bacteria and viruses to breach these protective surfaces more easily.
- Neutrophil function – Neutrophils are the most abundant white blood cells and are among the first responders to sites of infection. Zinc is required for neutrophil chemotaxis (directed migration toward pathogens), phagocytosis (engulfment of pathogens), and the oxidative burst (generation of reactive oxygen species that kill engulfed microorganisms). Zinc-deficient individuals demonstrate impaired neutrophil function across all of these activities.
- Macrophage activity – Macrophages perform phagocytosis, antigen presentation, and cytokine secretion. Zinc supports intracellular killing mechanisms within macrophages and influences their polarization toward pro-inflammatory (M1) or anti-inflammatory (M2) phenotypes. Zinc deficiency shifts macrophage function in ways that impair pathogen clearance.
- Complement system – Certain components of the complement cascade, which enhances the ability of antibodies and phagocytic cells to clear pathogens, are influenced by zinc status. Zinc deficiency can reduce complement-mediated opsonization and lysis of microbial targets.
- Dendritic cells – These antigen-presenting cells serve as a bridge between innate and adaptive immunity. Zinc affects dendritic cell maturation, antigen processing, and the ability to activate T cells. Altered zinc levels can skew the type of immune response that dendritic cells initiate.
Adaptive Immunity
The adaptive immune system mounts highly specific responses to particular pathogens and generates immunological memory. Zinc is essential for the development and function of adaptive immune cells.
- Thymus gland and T-cell maturation – The thymus is the primary site of T-cell development and maturation. Zinc is required for normal thymic architecture and function. In zinc deficiency, the thymus undergoes significant atrophy, reducing the output of mature, functional T cells. This thymic involution is one of the most well-characterized consequences of zinc deficiency and is reversible with zinc repletion.
- CD4+ helper T cells – These cells coordinate the immune response by secreting cytokines that direct other immune cells. Zinc deficiency reduces the number and function of CD4+ T cells, impairing the orchestration of both humoral and cell-mediated immunity.
- CD8+ cytotoxic T cells – Responsible for killing virus-infected and tumor cells, CD8+ T cells require zinc for their cytolytic activity. Zinc deficiency diminishes cytotoxic T-cell function, reducing the body's capacity to eliminate intracellular pathogens.
- Th1/Th2 balance – Zinc promotes a Th1-dominant immune response, which is critical for defense against intracellular pathogens such as viruses and certain bacteria. Zinc deficiency shifts the balance toward a Th2-dominant response, which is less effective against these threats and may predispose to allergic and autoimmune conditions.
- B cells and antibody production – While the effects of zinc on B-cell function are less pronounced than on T cells, zinc deficiency can impair antibody production, particularly the generation of new antibody responses to novel antigens.
- Regulatory T cells – Zinc supports the development of regulatory T cells (Tregs), which are critical for maintaining immune tolerance and preventing autoimmunity. Dysregulated Treg function due to zinc deficiency may contribute to chronic inflammatory conditions.
Thymulin Activation
Thymulin (formerly known as facteur thymique serique, or FTS) is a nonapeptide hormone secreted by thymic epithelial cells. It is unique among thymic hormones in that it requires zinc for biological activity.
- Zinc-thymulin complex – Thymulin circulates in the blood in a zinc-bound active form. The zinc ion is coordinated within the peptide structure and is essential for thymulin's ability to bind to high-affinity receptors on T cells. Without zinc, thymulin remains in an inactive form and cannot exert its immunomodulatory effects.
- T-cell differentiation – Active thymulin promotes the differentiation of immature thymocytes into mature T cells expressing the appropriate surface markers (CD4 or CD8). It also enhances T-cell cytotoxicity and the production of interleukin-2 (IL-2), a key T-cell growth factor.
- Biomarker of zinc status – Serum thymulin activity is one of the most sensitive functional indicators of zinc status. It declines early in zinc deficiency, often before other clinical signs appear, and is restored rapidly with zinc supplementation. This has made thymulin activity a valuable tool in research studies evaluating marginal zinc deficiency.
- Age-related decline – Thymulin levels decline with age, paralleling the involution of the thymus gland. This decline is accelerated by zinc deficiency and may contribute to the immunosenescence (age-related weakening of the immune system) observed in elderly populations. Zinc supplementation in older adults has been shown to partially restore thymulin activity.
Natural Killer (NK) Cell Activity
Natural killer cells are large granular lymphocytes of the innate immune system that provide rapid responses to virus-infected cells and tumor cells without requiring prior sensitization.
- Cytotoxic mechanism – NK cells kill target cells by releasing cytotoxic granules containing perforin and granzymes. Perforin creates pores in the target cell membrane, allowing granzymes to enter and trigger apoptosis. Zinc is required for the proper packaging, release, and function of these cytotoxic molecules.
- Impact of deficiency – Zinc deficiency reduces NK cell numbers and impairs their cytolytic activity. This diminished NK cell function contributes to increased susceptibility to viral infections and may impair immune surveillance against malignant cells. Studies in zinc-deficient human subjects have demonstrated reduced NK cell lytic activity that is correctable with zinc supplementation.
- NK cell receptor expression – Zinc influences the expression of activating and inhibitory receptors on the NK cell surface. The balance between these receptors determines whether an NK cell will attack or spare a given target cell. Zinc deficiency may alter this balance, reducing the efficiency of NK cell-mediated killing.
- Interferon production – NK cells are important producers of interferon-gamma (IFN-γ), a cytokine that activates macrophages and promotes antiviral defenses. Zinc supports IFN-γ production by NK cells, linking zinc status to broader antiviral immunity.
Cytokine Production and Regulation
Cytokines are signaling molecules that coordinate the immune response. Zinc has a complex and dose-dependent relationship with cytokine production.
- Pro-inflammatory cytokines – Zinc modulates the production of interleukin-1 beta (IL-1β), interleukin-6 (IL-6), and tumor necrosis factor alpha (TNF-α). In zinc deficiency, the production of these cytokines may be dysregulated, leading to either an inadequate inflammatory response to infection or, paradoxically, a chronic low-grade inflammatory state.
- Anti-inflammatory effects – Zinc induces the production of the anti-inflammatory cytokine interleukin-10 (IL-10) and inhibits the activation of NF-κB, a transcription factor that drives the expression of many pro-inflammatory genes. This dual action helps zinc maintain a balanced inflammatory response.
- Interleukin-2 (IL-2) – This cytokine is critical for T-cell proliferation and the expansion of antigen-specific T-cell clones. Zinc is required for optimal IL-2 production and IL-2 receptor expression, making it essential for effective adaptive immune responses.
- Cytokine storm prevention – By regulating NF-κB signaling and promoting anti-inflammatory pathways, adequate zinc status may help prevent the excessive cytokine release (cytokine storm) that can cause severe tissue damage during acute infections. This regulatory role has drawn particular attention in the context of severe respiratory infections.
Zinc Deficiency and Susceptibility to Infection
Zinc deficiency is one of the most common micronutrient deficiencies globally and has profound consequences for immune competence and infection risk.
- Prevalence – The World Health Organization estimates that zinc deficiency contributes to approximately 16% of lower respiratory infections, 18% of malaria cases, and 10% of diarrheal disease globally. It is most prevalent in low- and middle-income countries where diets are based heavily on cereals and legumes that contain phytates, which inhibit zinc absorption.
- Diarrheal disease – Zinc deficiency increases the duration and severity of diarrheal episodes in children. Meta-analyses of clinical trials have shown that zinc supplementation reduces the incidence of diarrhea by approximately 25% and the duration of acute diarrheal episodes by about one day. The World Health Organization and UNICEF recommend zinc supplementation as part of the standard treatment for childhood diarrhea.
- Pneumonia – Zinc-deficient children are at significantly greater risk of developing pneumonia. Supplementation trials have demonstrated reductions in the incidence of pneumonia ranging from 15% to 40% in zinc-deficient populations.
- Malaria – Studies in malaria-endemic regions have found that zinc supplementation can reduce the incidence of clinical malaria episodes, likely through enhanced immune function and improved epithelial barrier integrity.
- HIV/AIDS – Zinc deficiency is common in people living with HIV and is associated with faster disease progression, lower CD4+ T-cell counts, and increased mortality. Zinc supplementation in HIV-positive individuals has been shown to reduce the incidence of opportunistic infections and slow immunological decline in some studies.
- Vulnerable populations – The elderly, pregnant women, infants, and individuals with chronic diseases such as diabetes, chronic kidney disease, and inflammatory bowel disease are at heightened risk of zinc deficiency and its immunological consequences.
Zinc and Respiratory Infections
The relationship between zinc and respiratory tract infections has been extensively studied, with particular attention to the common cold and lower respiratory infections.
- Common cold – A substantial body of evidence suggests that zinc supplementation, particularly in the form of zinc lozenges or syrup taken within 24 hours of symptom onset, can reduce the duration and severity of common cold symptoms. A Cochrane systematic review found that zinc supplementation reduced the duration of cold symptoms by approximately one to two days when initiated early. The mechanism is thought to involve direct antiviral effects of ionic zinc on rhinovirus replication and enhanced local immune responses in the nasopharyngeal mucosa.
- Influenza – Preclinical studies have shown that zinc inhibits the replication of influenza viruses in cell culture. Clinical data on zinc supplementation for influenza prevention and treatment are more limited, but the mineral's broad immunomodulatory effects suggest a supportive role in host defense against influenza.
- Lower respiratory infections in children – Zinc supplementation has been shown to reduce the incidence and mortality of lower respiratory infections in children in developing countries. These effects are most pronounced in children who are zinc-deficient at baseline.
- Mechanism of action in respiratory defense – Zinc supports respiratory immunity through multiple pathways: maintaining airway epithelial barrier integrity, enhancing mucociliary clearance, supporting alveolar macrophage function, and modulating the inflammatory response within the lungs. Zinc also inhibits viral RNA-dependent RNA polymerase activity in several respiratory viruses.
Clinical Evidence and Therapeutic Considerations
The clinical evidence for zinc's role in immune function spans decades of observational studies, randomized controlled trials, and meta-analyses.
- Supplementation in the elderly – Randomized trials in elderly nursing home residents have shown that zinc supplementation (approximately 30–45 mg/day of elemental zinc) reduces the incidence of infections and improves T-cell function. These benefits are most evident in individuals with low baseline zinc status.
- Optimal dosing – The recommended dietary allowance (RDA) for zinc is 11 mg/day for adult men and 8 mg/day for adult women. Therapeutic doses for immune support typically range from 15 to 45 mg/day of elemental zinc. The tolerable upper intake level (UL) is 40 mg/day for adults; doses above this level taken chronically can cause copper deficiency, which itself impairs immune function.
- Forms of supplementation – Zinc is available in several supplemental forms including zinc gluconate, zinc acetate, zinc sulfate, zinc picolinate, and zinc citrate. Zinc gluconate and zinc acetate are the forms most commonly used in lozenge preparations for respiratory infections. Bioavailability varies among forms, with zinc picolinate and zinc citrate generally demonstrating good absorption.
- Risks of excess – While zinc deficiency impairs immunity, excessive zinc intake can also be immunosuppressive. High-dose zinc supplementation (150–300 mg/day) has been shown to reduce lymphocyte proliferation, impair neutrophil function, and lower HDL cholesterol. Chronic excess intake also depletes copper stores by inducing metallothionein in enterocytes, which preferentially binds copper and prevents its absorption.
- Drug interactions – Zinc supplements can interact with several medications, including tetracycline and quinolone antibiotics (reduced absorption of both the mineral and the antibiotic), penicillamine (used for Wilson's disease and rheumatoid arthritis), and thiazide diuretics (which increase urinary zinc excretion). Patients taking these medications should be counseled on appropriate timing of zinc supplementation.
Dosing and Dietary Sources
- RDA – Men 11 mg/day; women 8 mg/day; pregnancy 11 mg/day; lactation 12 mg/day.
- Immune support dose – 15–45 mg/day elemental zinc for short-term use.
- Cold lozenges – Zinc gluconate or acetate providing ~13–23 mg per lozenge, every 2–3 hours while awake, started within 24 hours of symptom onset.
- Oysters – Richest source (~32 mg per 3 oz).
- Meat and poultry – Beef, lamb, chicken thigh.
- Legumes, nuts, seeds – Chickpeas, pumpkin seeds, cashews; soak or sprout to reduce phytate chelation.
- Fortified cereals – Common source in Western diets.
Safety and Drug Interactions
- Tolerable Upper Intake Level (UL) – 40 mg/day for adults.
- Copper deficiency – Chronic doses > 40 mg/day induce metallothionein and block copper absorption; consider 1–2 mg copper supplementation if taking high-dose zinc long-term.
- GI upset – Nausea, cramping; take with food.
- Anosmia risk – Intranasal zinc gel products were linked to permanent smell loss and have been withdrawn; oral lozenges do not share this risk.
- Drug interactions – Separate from tetracyclines, quinolones, and penicillamine by 2 hours; thiazides increase zinc loss.
This content is provided for informational purposes only and does not constitute medical advice. Consult a qualified healthcare provider before starting high-dose or long-term zinc supplementation.
Research Papers
- Wessels I, Maywald M, Rink L. Zinc as a gatekeeper of immune function. Nutrients. 2017;9(12):1286.
- Prasad AS. Zinc in human health: effect of zinc on immune cells. Mol Med. 2008;14(5-6):353-357.
- Singh M, Das RR. Zinc for the common cold. Cochrane Database Syst Rev. 2013;(6):CD001364.
- Science M, Johnstone J, Roth DE, Guyatt G, Loeb M. Zinc for the treatment of the common cold: a systematic review and meta-analysis of randomized controlled trials. CMAJ. 2012;184(10):E551-E561.
- Lassi ZS, Moin A, Bhutta ZA. Zinc supplementation for the prevention of pneumonia in children aged 2 months to 59 months. Cochrane Database Syst Rev. 2016;(12):CD005978.
- te Velthuis AJ, van den Worm SH, Sims AC, Baric RS, Snijder EJ, van Hemert MJ. Zn2+ inhibits coronavirus and arterivirus RNA polymerase activity in vitro. PLoS Pathog. 2010;6(11):e1001176.
- Prasad AS, Beck FW, Bao B, et al. Zinc supplementation decreases incidence of infections in the elderly. Am J Clin Nutr. 2007;85(3):837-844.
- Haase H, Rink L. The immune system and the impact of zinc during aging. Immun Ageing. 2009;6:9.
- Fraker PJ, King LE. Reprogramming of the immune system during zinc deficiency. Annu Rev Nutr. 2004;24:277-298.
- PubMed — zinc supplementation + immune + randomized
- NIH Office of Dietary Supplements — Zinc Fact Sheet
- Linus Pauling Institute — Zinc
Connections
- Zinc
- Zinc and Wound Healing
- Copper
- Selenium
- Iron
- Vitamin C
- Vitamin D3
- Vitamin A
- Pneumonia
- Influenza
- Malaria
- Immune Boosting
- Copper-Iron Dysregulation
- Inflammatory Bowel Disease
- Hair Loss
- Acne
Featured Videos

The Surprising Benefits of Zinc Supplementation for the Common Cold

Think Zinc! Fighting Infection and Boosting Your Immune System

Importance of ZINC for Health & Immune System Function

Fix Zinc Deficiency with Food | Ayurvedic Tips for Immunity, Hair & Hormones

Your Body Needs Zinc (Zn)

Will Zinc Boost Your Immune System and Protect From Covid or a Cold?

Zinc Deficiency Signs & Symptoms (ex. Hair Loss, Acne, Infections) & Why They Occur

Zinc Benefits You Didn't Know About: Boost Immunity and Fix Deficiency Fast!

What You Actually Need to Know About Zinc Supplements