Selenium: Essential Trace Mineral for Human Health
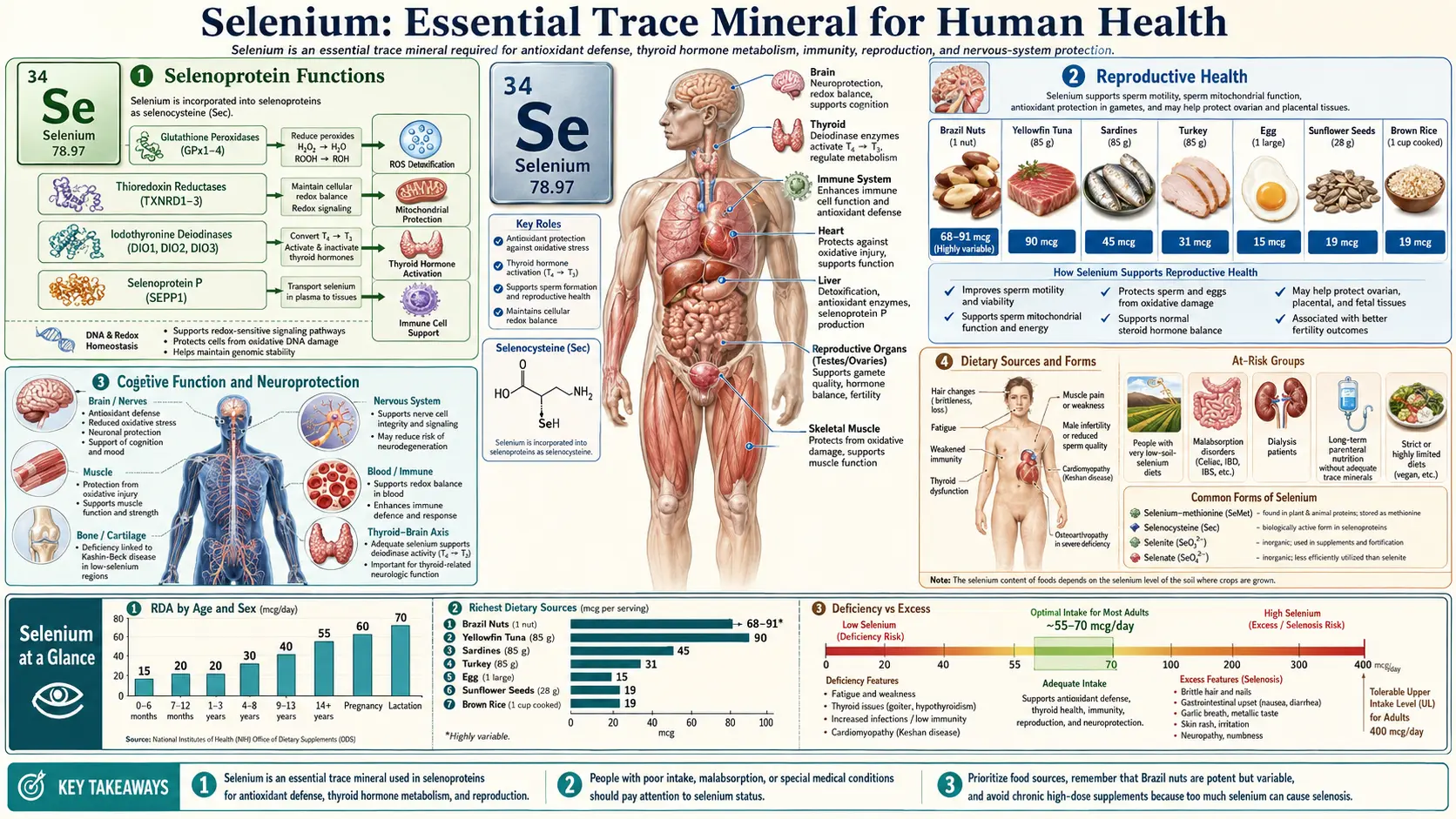
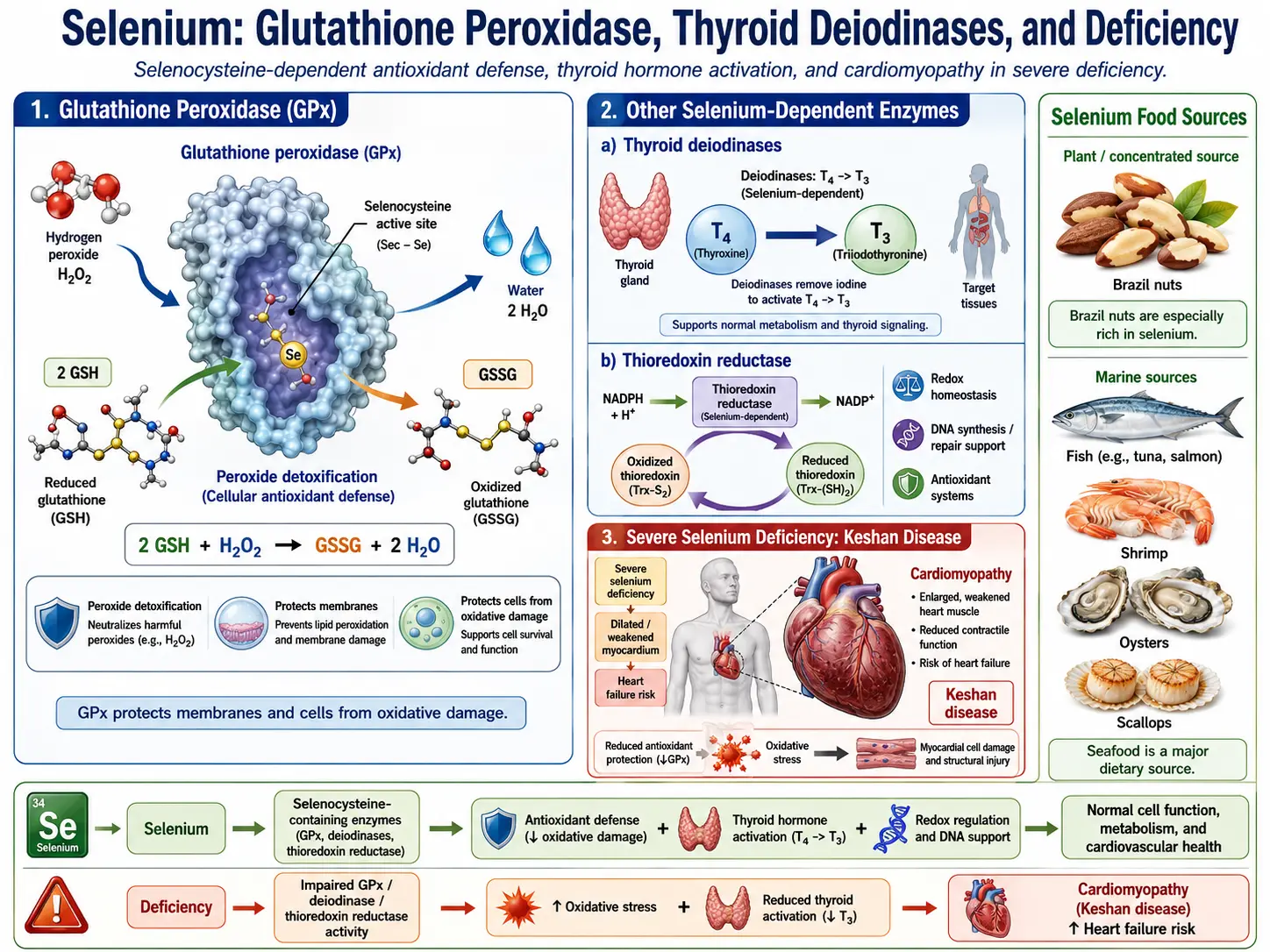
Table of Contents
- Selenoprotein Functions
- Antioxidant Defense
- Thyroid Hormone Metabolism
- Immune Function
- Reproductive Health
- DNA Synthesis and Repair
- Cancer-Protective Properties
- Cardiovascular Protection
- Cognitive Function and Neuroprotection
- Anti-Inflammatory Effects
- Selenium Deficiency
- Selenium Toxicity
- Dietary Sources and Forms
- Connections
- Featured Videos
Selenium is an essential trace mineral that plays a critical role in numerous physiological processes throughout the human body. Unlike many other minerals, selenium exerts its biological effects primarily through its incorporation into selenoproteins, a unique class of proteins that contain the amino acid selenocysteine at their active sites. The human genome encodes at least 25 selenoproteins, many of which serve as enzymes with vital antioxidant, metabolic, and regulatory functions. Although required only in small quantities, selenium is indispensable for optimal health, and both deficiency and excess can lead to significant clinical consequences.
Selenium is obtained from dietary sources including Brazil nuts, seafood, organ meats, muscle meats, grains, and dairy products. The selenium content of plant-based foods varies widely depending on the selenium concentration of the soil in which they were grown. The Recommended Dietary Allowance (RDA) for adults is 55 micrograms per day, with the Tolerable Upper Intake Level set at 400 micrograms per day.
Selenoprotein Functions
Selenoproteins are the molecular effectors through which selenium exerts its biological activity. These proteins contain selenocysteine (Sec), often referred to as the 21st amino acid, which is encoded by the UGA codon and requires specialized translational machinery including a selenocysteine insertion sequence (SECIS) element in the mRNA. The presence of selenocysteine at the catalytic site of these enzymes confers superior catalytic efficiency compared to cysteine-containing analogues.
- Glutathione Peroxidases (GPx1-GPx6) – A family of enzymes that reduce hydrogen peroxide and organic hydroperoxides using glutathione as the electron donor. GPx1 is the most abundant selenoprotein in the cytoplasm, while GPx4 (phospholipid hydroperoxide glutathione peroxidase) is uniquely capable of reducing lipid hydroperoxides within biological membranes.
- Thioredoxin Reductases (TrxR1-TrxR3) – These enzymes maintain the thioredoxin system in its reduced state, supporting cellular redox homeostasis, DNA synthesis, gene expression, and cell proliferation. TrxR1 is cytosolic, TrxR2 is mitochondrial, and TrxR3 is expressed predominantly in the testes.
- Iodothyronine Deiodinases (DIO1-DIO3) – These selenoenzymes catalyze the activation and inactivation of thyroid hormones through deiodination reactions, making them essential regulators of thyroid hormone metabolism.
- Selenoprotein P (SELENOP) – The primary selenium transport protein in plasma, containing up to 10 selenocysteine residues per molecule. SELENOP delivers selenium from the liver to peripheral tissues and also possesses antioxidant properties.
- Selenoprotein W (SELENOW) – Expressed primarily in skeletal and cardiac muscle, this selenoprotein is believed to play a role in muscle metabolism and protection against oxidative stress.
- Methionine Sulfoxide Reductase B1 (MSRB1) – Repairs oxidized methionine residues in proteins, restoring their normal function and protecting against oxidative damage to the proteome.
- Selenophosphate Synthetase 2 (SPS2) – Generates the selenium donor compound (selenophosphate) required for selenocysteine biosynthesis, thus playing a regulatory role in the entire selenoprotein synthesis pathway.
Antioxidant Defense
Selenium's most well-characterized biological role is its contribution to antioxidant defense systems. Reactive oxygen species (ROS) are produced continuously during normal cellular metabolism, particularly through mitochondrial electron transport, enzymatic reactions, and immune cell activation. While low levels of ROS serve important signaling functions, excessive accumulation leads to oxidative stress, which damages lipids, proteins, and nucleic acids, contributing to aging, chronic disease, and cellular death.
Glutathione Peroxidase System
The glutathione peroxidase (GPx) family represents the primary enzymatic antioxidant defense system dependent on selenium. These enzymes catalyze the reduction of potentially damaging peroxides to harmless alcohols and water, using reduced glutathione (GSH) as the electron donor. GPx1, the most abundant cytosolic form, efficiently scavenges hydrogen peroxide (H2O2) and soluble organic hydroperoxides. GPx4 occupies a unique niche in antioxidant defense because it can directly reduce phospholipid hydroperoxides, cholesterol hydroperoxides, and thymine hydroperoxides within biological membranes, thereby preventing lipid peroxidation chain reactions. GPx4 activity has also been identified as a critical regulator of ferroptosis, a form of regulated cell death driven by iron-dependent lipid peroxidation.
GPx2 is predominantly expressed in the gastrointestinal tract, where it protects the intestinal epithelium from oxidative damage caused by dietary pro-oxidants, gut microbiota, and inflammatory processes. GPx3 is the primary extracellular glutathione peroxidase, found in plasma, kidney, and the thyroid gland, where it provides antioxidant protection in the extracellular compartment.
Thioredoxin Reductase System
The thioredoxin reductase (TrxR) enzymes maintain the thioredoxin system, which serves as a parallel and complementary antioxidant defense to the glutathione system. TrxR reduces oxidized thioredoxin (Trx), which in turn reduces oxidized protein disulfides, peroxiredoxins, and other substrates. The thioredoxin system is essential for maintaining cellular redox balance, regulating transcription factors such as NF-kB and AP-1, supporting DNA synthesis through ribonucleotide reductase, and modulating apoptosis. Mitochondrial TrxR2 is particularly important for neutralizing ROS generated during oxidative phosphorylation.
Thyroid Hormone Metabolism
The thyroid gland has the highest concentration of selenium per gram of tissue of any organ in the human body, reflecting the critical importance of selenoproteins in thyroid function. Three iodothyronine deiodinase enzymes (DIO1, DIO2, DIO3) regulate thyroid hormone activation and inactivation throughout the body.
- Type 1 Deiodinase (DIO1) – Expressed primarily in the liver, kidney, and thyroid, DIO1 catalyzes the conversion of the prohormone thyroxine (T4) to the biologically active triiodothyronine (T3) through outer ring deiodination. It also contributes to the clearance of reverse T3 (rT3) and plays a role in iodine recycling.
- Type 2 Deiodinase (DIO2) – Found in the brain, pituitary, brown adipose tissue, thyroid, and skeletal muscle, DIO2 is the primary enzyme responsible for local T3 production in tissues that are highly sensitive to thyroid hormone levels. It serves as a critical regulator of intracellular T3 availability.
- Type 3 Deiodinase (DIO3) – The primary inactivating deiodinase, DIO3 converts T4 to reverse T3 (rT3) and T3 to diiodothyronine (T2), both of which are biologically inactive. DIO3 protects tissues from excessive thyroid hormone exposure and is particularly important during fetal development.
Selenium deficiency impairs deiodinase function, leading to altered thyroid hormone ratios, elevated T4 with reduced T3, and compensatory increases in thyroid-stimulating hormone (TSH). The thyroid also depends on glutathione peroxidases (especially GPx3) to neutralize the hydrogen peroxide generated during thyroid hormone synthesis by thyroid peroxidase. For more detailed information, see Selenium and Thyroid Function.
Immune Function
Selenium plays a multifaceted role in both innate and adaptive immune responses. Adequate selenium status is required for optimal immune cell function, appropriate inflammatory responses, and effective defense against viral, bacterial, and parasitic infections.
Innate Immunity
- Macrophage Function – Selenium enhances macrophage phagocytic activity, respiratory burst capacity, and cytokine production. Selenoproteins regulate the balance between pro-inflammatory and anti-inflammatory macrophage activation states (M1 vs. M2 polarization).
- Natural Killer (NK) Cell Activity – Selenium supplementation has been shown to increase NK cell cytotoxic activity, which is crucial for the early detection and elimination of virus-infected and transformed cells.
- Neutrophil Function – GPx enzymes protect neutrophils from self-inflicted oxidative damage during the respiratory burst, while selenoproteins modulate neutrophil migration and inflammatory mediator production.
Adaptive Immunity
- T Cell Proliferation and Differentiation – Selenium is required for optimal T cell receptor signaling, clonal expansion, and differentiation into effector and memory subsets. Selenium deficiency impairs T helper cell function and shifts the Th1/Th2 balance.
- B Cell and Antibody Responses – Adequate selenium supports immunoglobulin production and the generation of high-affinity antibodies.
- Viral Defense – Selenium deficiency has been linked to increased virulence and pathogenicity of certain viruses, including Coxsackievirus B3 (implicated in Keshan disease) and influenza. Studies have demonstrated that selenium-deficient hosts can harbor viral populations with increased mutation rates, potentially giving rise to more virulent strains.
Reproductive Health
Selenium is essential for normal reproductive function in both males and females. Its roles span gametogenesis, fertilization, embryonic development, and pregnancy maintenance.
Male Fertility
- Sperm Structure and Motility – GPx4 (phospholipid hydroperoxide glutathione peroxidase) plays a unique dual role in spermatogenesis. During sperm maturation, GPx4 functions as an active peroxidase protecting developing sperm cells from oxidative damage. In mature spermatozoa, enzymatically inactive GPx4 becomes a structural protein that crosslinks with other proteins to form the mitochondrial capsule of the sperm midpiece, which is essential for sperm motility.
- Testosterone Synthesis – Selenium supports Leydig cell function and testosterone production, partly through its antioxidant protection of testicular tissue from oxidative damage.
- Semen Quality – Multiple clinical studies have demonstrated positive correlations between selenium status and sperm concentration, morphology, and motility parameters.
Female Reproductive Health
- Ovarian Function – Selenoproteins protect oocytes and follicular cells from oxidative stress, supporting folliculogenesis and oocyte quality.
- Pregnancy – Low selenium status during pregnancy has been associated with increased risk of preeclampsia, gestational diabetes, preterm birth, and miscarriage. Selenium supplementation during pregnancy has shown promise in reducing the risk of preeclampsia in some clinical trials.
- Placental Function – Selenoproteins in the placenta provide antioxidant protection and contribute to normal placental development and function.
DNA Synthesis and Repair
Selenium contributes to DNA integrity through multiple mechanisms. The thioredoxin reductase system maintains thioredoxin in its reduced form, which is required by ribonucleotide reductase for the synthesis of deoxyribonucleotides, the building blocks of DNA. Without adequate TrxR activity, DNA synthesis and repair are compromised.
- Nucleotide Biosynthesis – Through the thioredoxin-ribonucleotide reductase pathway, selenium supports the de novo synthesis of deoxyribonucleotides necessary for DNA replication and repair.
- Protection of DNA from Oxidative Damage – Selenoproteins reduce oxidative stress that would otherwise cause base modifications (such as 8-oxoguanine), single-strand breaks, double-strand breaks, and DNA-protein crosslinks.
- DNA Repair Pathway Regulation – Selenium status influences the expression and activity of DNA repair proteins, including those involved in base excision repair (BER), nucleotide excision repair (NER), and mismatch repair (MMR) pathways.
- Epigenetic Regulation – Selenium influences DNA methylation patterns through its effects on one-carbon metabolism and the redox regulation of DNA methyltransferases, potentially influencing gene expression and genomic stability.
Cancer-Protective Properties
Selenium has attracted significant attention for its potential cancer-protective properties. The relationship between selenium and cancer risk is supported by epidemiological data, preclinical studies, and certain clinical trials, although the evidence is nuanced and depends on cancer type, selenium form, baseline selenium status, and genetic factors.
- Antioxidant Protection – By reducing oxidative DNA damage through GPx and TrxR enzyme systems, selenium helps prevent the mutagenic events that initiate carcinogenesis.
- Apoptosis Induction – Certain selenium metabolites, particularly methylselenol and selenodiglutathione, can trigger apoptosis in transformed cells through activation of caspase cascades and modulation of Bcl-2 family proteins, while sparing normal cells.
- Anti-Angiogenic Effects – Selenium compounds can inhibit the formation of new blood vessels that supply growing tumors by downregulating vascular endothelial growth factor (VEGF) expression.
- Cell Cycle Arrest – Selenium metabolites can induce cell cycle arrest at G1/S and G2/M checkpoints in cancer cells, preventing uncontrolled proliferation.
- Immune Surveillance – Selenium enhances the ability of NK cells and cytotoxic T lymphocytes to recognize and destroy transformed cells, strengthening the immune system's role as a tumor suppressor.
For a detailed discussion, see Selenium and Cancer Prevention.
Cardiovascular Protection
Selenium exerts several protective effects on the cardiovascular system through its antioxidant, anti-inflammatory, and metabolic regulatory functions.
- Prevention of LDL Oxidation – Selenoproteins, particularly GPx enzymes in the vascular endothelium and plasma, help prevent the oxidative modification of low-density lipoprotein (LDL) cholesterol. Oxidized LDL is a key driver of atherosclerotic plaque formation, as it triggers inflammatory responses in the arterial wall and promotes foam cell development.
- Endothelial Function – Selenium maintains endothelial health by reducing oxidative stress, preserving nitric oxide (NO) bioavailability, and attenuating endothelial activation. Adequate selenoprotein expression helps maintain the vasodilatory and anti-thrombotic properties of the endothelium.
- Platelet Aggregation – GPx3, the extracellular glutathione peroxidase expressed in plasma, modulates platelet activation by reducing hydroperoxides that promote thromboxane synthesis and platelet aggregation.
- Myocardial Protection – Selenoproteins protect cardiomyocytes from ischemia-reperfusion injury and oxidative damage. Severe selenium deficiency causes Keshan disease, an endemic dilated cardiomyopathy first described in selenium-poor regions of China.
- Anti-Atherogenic Effects – Selenium reduces the expression of adhesion molecules on endothelial cells and inhibits monocyte adhesion and migration into the subendothelial space, processes that are fundamental to atherosclerotic plaque initiation and progression.
Cognitive Function and Neuroprotection
The brain maintains selenium concentrations even during periods of systemic selenium depletion, reflecting the organ's high dependence on selenoprotein function. Selenoprotein P (SELENOP) is the primary vehicle for selenium delivery to the brain via the apolipoprotein E receptor 2 (ApoER2) at the blood-brain barrier.
- Neuronal Antioxidant Defense – Neurons are highly vulnerable to oxidative stress due to their high metabolic rate, abundant polyunsaturated fatty acid content, and limited regenerative capacity. GPx4 is particularly critical in the brain, where it prevents lipid peroxidation in neuronal membranes and protects against ferroptotic cell death.
- Neurodegenerative Disease – Low selenium status has been associated with cognitive decline and increased risk of neurodegenerative conditions including Alzheimer's disease and Parkinson's disease. Selenoproteins help mitigate the oxidative stress, mitochondrial dysfunction, and protein aggregation that characterize these disorders.
- Cerebrovascular Protection – By protecting the cerebrovascular endothelium and reducing neuroinflammation, selenium supports cerebral blood flow and blood-brain barrier integrity.
- Neurotransmitter Metabolism – Selenium influences the synthesis and metabolism of neurotransmitters including dopamine and serotonin, partly through its effects on thyroid hormone activation in the brain (via DIO2) and partly through redox regulation of biosynthetic enzymes.
Anti-Inflammatory Effects
Selenium modulates inflammatory processes through several interconnected mechanisms, positioning it as an important regulator of the inflammatory response.
- NF-kB Pathway Modulation – Selenoproteins, particularly thioredoxin reductases, influence the redox-sensitive transcription factor NF-kB, a master regulator of inflammatory gene expression. Adequate selenium status helps maintain appropriate NF-kB activation, preventing excessive pro-inflammatory cytokine production.
- Eicosanoid Metabolism – GPx enzymes regulate the availability of lipid hydroperoxides that serve as substrates for cyclooxygenase (COX) and lipoxygenase (LOX) pathways. By reducing hydroperoxide tone, selenium shifts eicosanoid production away from pro-inflammatory prostaglandins and leukotrienes toward anti-inflammatory and pro-resolving mediators.
- Inflammasome Regulation – Selenium influences NLRP3 inflammasome activation, which controls the maturation and release of the potent pro-inflammatory cytokines IL-1beta and IL-18. Selenoproteins help prevent inappropriate inflammasome activation driven by excessive ROS.
- Resolution of Inflammation – Selenium supports the active resolution of inflammation by promoting the switch from pro-inflammatory to pro-resolving macrophage phenotypes and enhancing the production of specialized pro-resolving mediators.
Selenium Deficiency
Selenium deficiency occurs primarily in regions with selenium-poor soils, including parts of China, Siberia, and certain areas of Europe and New Zealand. Populations at risk also include individuals receiving long-term parenteral nutrition without selenium supplementation, patients with malabsorptive conditions (such as Crohn's disease or short bowel syndrome), and those on highly restrictive diets.
- Keshan Disease – An endemic cardiomyopathy first identified in Keshan County, China, characterized by acute or chronic heart failure due to multifocal myocardial necrosis. While selenium deficiency is a necessary condition, Coxsackievirus B3 coinfection appears to act as a cofactor.
- Kashin-Beck Disease – An endemic osteoarthropathy characterized by degeneration and necrosis of joint cartilage and epiphyseal growth plates, leading to joint deformity and impaired growth. This condition is prevalent in selenium-deficient regions and may also involve iodine deficiency and mycotoxin exposure.
- Myxedematous Cretinism – Combined selenium and iodine deficiency can lead to a severe form of congenital hypothyroidism with neurodevelopmental impairment.
- Impaired Immunity – Selenium deficiency weakens immune function, increasing susceptibility to infections and potentially enhancing the virulence of certain pathogens.
Selenium Toxicity
Excessive selenium intake, known as selenosis, can occur from overconsumption of selenium supplements, consumption of foods grown in extremely selenium-rich soils, or occupational exposure. Acute selenosis can be life-threatening, while chronic excess leads to characteristic symptoms.
- Early Signs – Garlic-like breath odor (from dimethyl selenide exhalation), metallic taste, and gastrointestinal disturbances.
- Chronic Selenosis – Hair loss, nail brittleness and loss, skin rash, fatigue, irritability, and peripheral neuropathy.
- Severe Toxicity – Acute ingestion of large doses (greater than 1,000 micrograms) can cause severe gastrointestinal distress, cardiac abnormalities, renal failure, and in extreme cases, death.
Dietary Sources and Forms
- Brazil Nuts – The richest dietary source, with a single nut providing 70-90 micrograms, though content varies widely based on soil selenium levels.
- Seafood – Tuna, halibut, sardines, and shrimp provide 40-90 micrograms per serving.
- Organ Meats and Muscle Meats – Liver, kidney, beef, chicken, and pork provide 20-45 micrograms per serving.
- Grains and Cereals – Wheat, rice, and other grains contain variable amounts depending on soil conditions, typically 10-40 micrograms per serving.
- Dairy and Eggs – Moderate sources providing 10-20 micrograms per serving.
- Supplement Forms – Common supplemental forms include selenomethionine (organic, well-absorbed), sodium selenite (inorganic), sodium selenate (inorganic), and selenium-enriched yeast (mixed organic forms). Selenomethionine has the highest bioavailability and is nonspecifically incorporated into body proteins in place of methionine, creating a selenium reserve.
Connections
- Selenium and Cancer Prevention — selenoprotein biology, the NPC and SELECT trials, evidence across prostate/colorectal/lung cancers, and the U-shaped dose-response curve.
- Selenium Benefits Deep Dive
- Selenium for Antioxidant Defense
- Selenium for Cancer Prevention
- Selenium for Immune Function
- Selenium and Thyroid Function
- Glutathione
- Cancer
- Oxidative Stress
- Iodine
- Zinc
- Iron
- Cysteine
- Methionine
- Hair Loss
- Organ Meats
- Lead
- Vitamin E
- Vitamin C
- Cardiomyopathy
- Preeclampsia
- Testosterone Test