Thyroid Panel
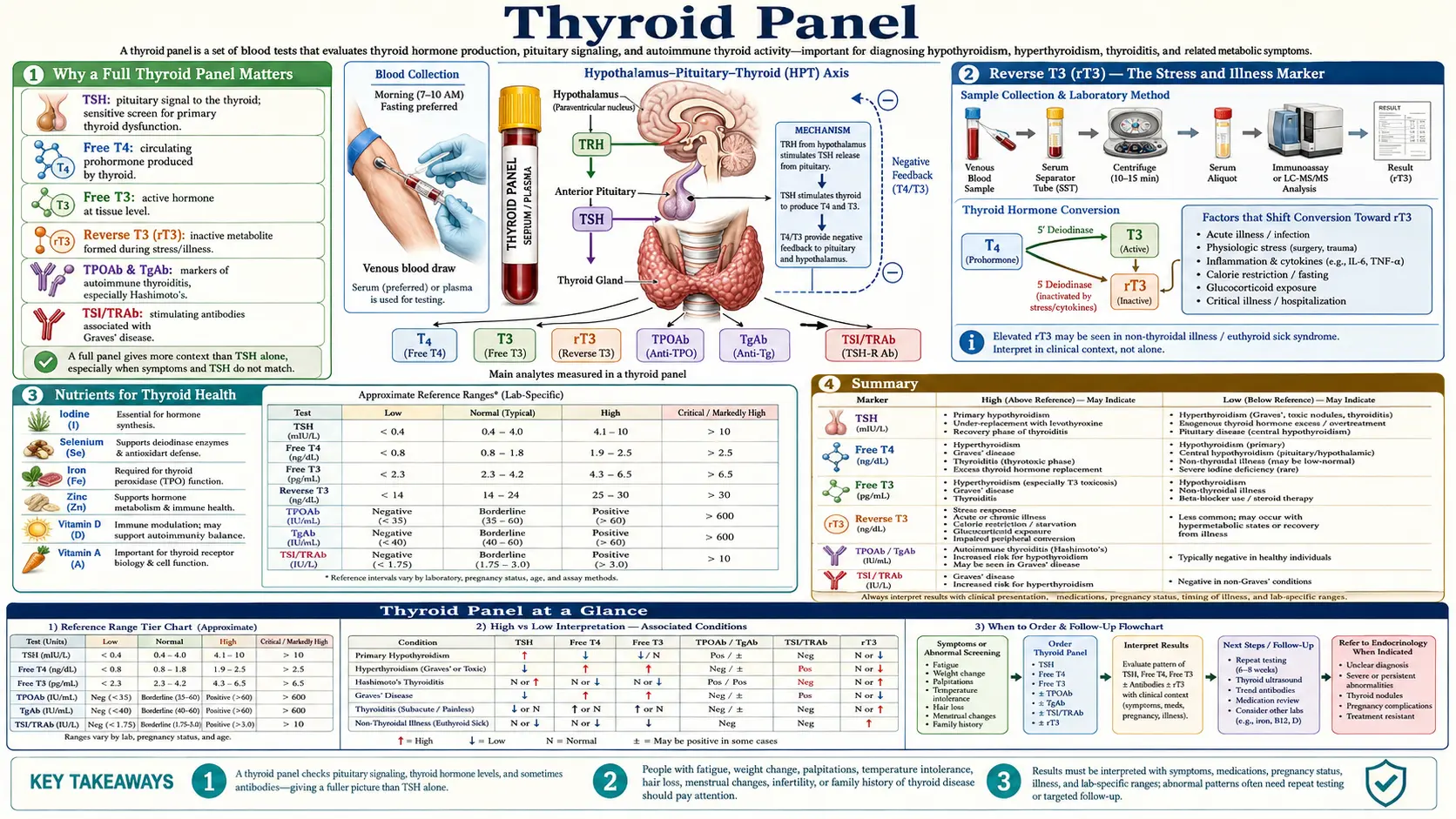
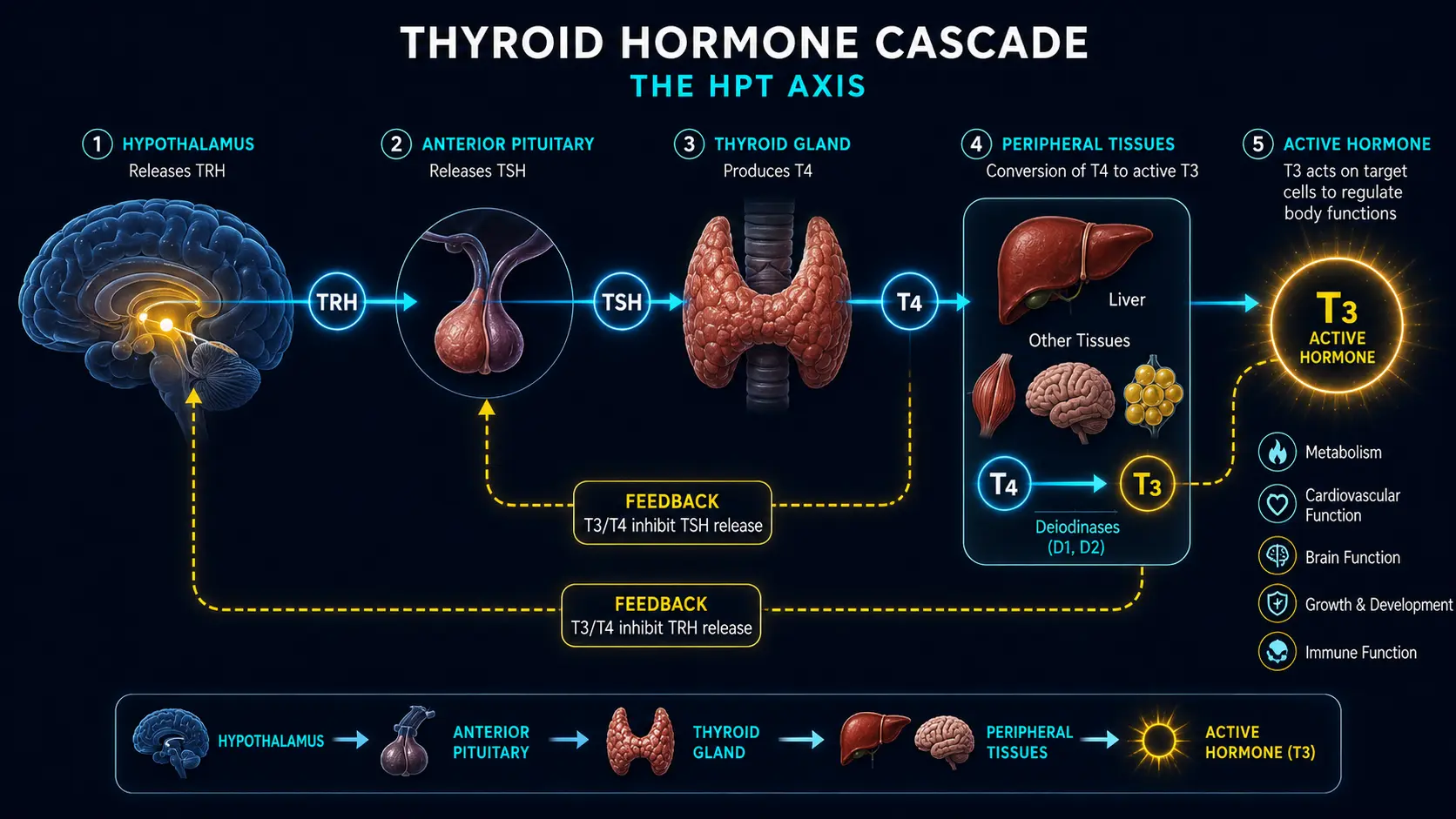
Table of Contents
- Why a Full Thyroid Panel Matters
- TSH (Thyroid Stimulating Hormone)
- Free T4 (Thyroxine) — The Storage Hormone
- Free T3 (Triiodothyronine) — The Active Hormone
- Reverse T3 (rT3) — The Stress and Illness Marker
- T3/rT3 Ratio
- Thyroid Antibodies — TPO, Thyroglobulin, and TSI
- Common Thyroid Dysfunction Patterns
- Symptoms of Thyroid Dysfunction
- Nutrients for Thyroid Health
- Gut Health and the Thyroid Connection
- When to Test — Timing and Preparation
- Cautions About Self-Treating
- Summary
- Featured Videos
Why a Full Thyroid Panel Matters
In conventional medicine, thyroid screening typically begins and ends with a single test: TSH. If TSH falls within the laboratory reference range, the patient is told their thyroid is "normal" and sent on their way. This approach is deeply flawed and misses a significant number of patients who are suffering from thyroid dysfunction.
A comprehensive thyroid panel includes TSH, Free T4, Free T3, Reverse T3, TPO antibodies, thyroglobulin antibodies, and in some cases TSI (thyroid-stimulating immunoglobulin). Each of these markers provides a unique piece of the puzzle. Testing only TSH is analogous to checking only the thermostat reading without verifying whether the furnace is actually producing heat or whether the vents are open.
From a functional medicine perspective, we recognize that thyroid physiology is a multi-step process. The hypothalamus signals the pituitary gland, which releases TSH to stimulate the thyroid gland. The thyroid produces primarily T4, which must be converted to the active hormone T3 in peripheral tissues such as the liver, gut, and kidneys. Any disruption along this cascade can produce symptoms, yet a single TSH test may appear entirely normal.
A full thyroid panel allows practitioners to identify:
- Autoimmune thyroid disease years before TSH becomes abnormal
- Poor conversion of T4 to T3
- Excessive production of Reverse T3, which blocks thyroid hormone action
- Subclinical hypothyroidism that conventional ranges miss
- The difference between a primary thyroid problem and a conversion or receptor problem
TSH (Thyroid Stimulating Hormone)
TSH is produced by the pituitary gland and acts as a messenger to the thyroid. When thyroid hormone levels drop, the pituitary releases more TSH to stimulate production. When thyroid hormones are sufficient, TSH decreases. It operates as a feedback loop.
Conventional reference range: Most laboratories report a TSH range of approximately 0.45 to 4.5 mIU/L. Some labs extend the upper limit to 5.0 or even higher. This wide range was established using population data that included individuals with undiagnosed thyroid disease, which artificially inflated the upper limit.
Functional optimal range: Many functional medicine and integrative practitioners consider an optimal TSH to be between 1.0 and 2.0 mIU/L. Research has shown that the majority of healthy individuals without thyroid disease cluster in this narrower range. A TSH above 2.5 mIU/L, while technically "normal" by conventional standards, may indicate early thyroid insufficiency, particularly when symptoms are present.
Why "normal" TSH can mask thyroid dysfunction:
- TSH is a pituitary hormone, not a direct measurement of thyroid hormone levels in the blood or tissues
- TSH can be suppressed by stress, cortisol dysregulation, inflammation, certain medications (including biotin supplements and dopamine agonists), and caloric restriction
- Pituitary function may be compromised in chronic illness, resulting in inappropriately normal or low TSH despite low thyroid hormone levels
- TSH follows a diurnal rhythm and can vary significantly throughout the day, peaking in the early morning hours
- A patient can have a TSH of 3.0 with markedly low Free T3 and still be told everything is fine
Important note: A very low TSH (below 0.4 mIU/L) should always be investigated, as it may indicate hyperthyroidism, Graves' disease, excessive thyroid medication dosing, or central hypothyroidism.
Free T4 (Thyroxine) — The Storage Hormone
T4 is the primary hormone produced by the thyroid gland and represents approximately 93% of thyroid hormone output. However, T4 is largely inactive in the body. It serves as a reservoir or storage form that must be converted into the active hormone T3 before cells can use it.
Conventional reference range: Approximately 0.82 to 1.77 ng/dL (varies by laboratory).
Functional optimal range: Ideally in the upper half of the reference range, roughly 1.1 to 1.5 ng/dL. A Free T4 in the lower quarter of the range may suggest the thyroid gland is underproducing, even if TSH has not yet risen out of range.
Key points about Free T4:
- We measure Free T4, not Total T4, because the free (unbound) fraction is the biologically available portion. The majority of T4 circulates bound to proteins such as thyroid-binding globulin (TBG)
- Estrogen increases TBG levels, which can raise Total T4 while Free T4 remains unchanged. This is particularly relevant for women on oral contraceptives or hormone replacement therapy
- A normal Free T4 with an elevated TSH suggests the thyroid is working harder to maintain output and may be in the early stages of failure
- A low Free T4 with a normal TSH may indicate central hypothyroidism (a pituitary or hypothalamic problem) rather than a primary thyroid issue
Free T3 (Triiodothyronine) — The Active Hormone
Free T3 is arguably the most important marker on the thyroid panel, yet it is the one most commonly omitted by conventional practitioners. T3 is the biologically active thyroid hormone that enters cells, binds to nuclear receptors, and drives metabolic processes including energy production, body temperature regulation, heart rate, cognitive function, and fat metabolism.
Conventional reference range: Approximately 2.0 to 4.4 pg/mL.
Functional optimal range: Ideally in the upper third of the reference range, roughly 3.0 to 4.0 pg/mL. Many symptomatic patients have a Free T3 below 3.0 pg/mL, which, while technically within range, is suboptimal for cellular function.
Why Free T3 is so critical:
- T3 is approximately four to five times more potent than T4 at the cellular level
- The conversion of T4 to T3 occurs primarily in the liver, gut, and kidneys via the enzyme 5'-deiodinase. This process requires adequate selenium, zinc, and iron
- Many patients have normal TSH and normal Free T4 but low Free T3, indicating a conversion problem rather than a production problem
- Chronic stress, inflammation, gut dysbiosis, caloric restriction, liver dysfunction, and certain medications (beta-blockers, amiodarone, lithium) can impair T4-to-T3 conversion
- Patients on levothyroxine (T4-only medication) may still feel unwell if they cannot efficiently convert T4 to T3
In functional medicine, we often say: "If you are not testing Free T3, you are not truly assessing thyroid function."
Reverse T3 (rT3) — The Stress and Illness Marker
Reverse T3 is an inactive metabolite of T4. Under normal circumstances, the body converts a small percentage of T4 into rT3 as part of healthy thyroid hormone metabolism. However, under conditions of physiological stress, the body preferentially shunts T4 into rT3 rather than active T3. This serves as a protective mechanism to slow metabolism during illness or famine, but in chronic stress it becomes problematic.
Conventional reference range: Approximately 9.2 to 24.1 ng/dL.
Functional optimal range: Below 15 ng/dL is generally considered optimal. Levels above 15 to 20 ng/dL, particularly when Free T3 is low, suggest excessive rT3 production.
What drives Reverse T3 elevation:
- Chronic psychological or physical stress and elevated cortisol
- Chronic illness, infection, or systemic inflammation
- Caloric restriction and extreme dieting
- Liver dysfunction, since the liver is a primary site of T4-to-T3 conversion
- Iron deficiency
- Selenium deficiency
- Certain medications, including high-dose T4-only thyroid medication
- Chronic exposure to environmental toxins and heavy metals
The problem with elevated rT3 is that it competes with T3 for receptor binding sites on cells. Even if Free T3 levels appear adequate on paper, high rT3 can block T3 from entering cells and performing its metabolic functions. This creates a state of cellular hypothyroidism that standard blood tests may not detect.
T3/rT3 Ratio
The ratio of Free T3 to Reverse T3 provides a more complete picture of thyroid hormone utilization at the tissue level than either marker alone. This ratio helps determine whether thyroid hormone is being effectively used by cells or being shunted into the inactive pathway.
How to calculate: Divide Free T3 (in pg/mL) by Reverse T3 (in ng/dL). Ensure units are consistent before calculating.
Optimal ratio: A ratio greater than 0.20 (some practitioners use a threshold of 0.20 to 0.27) is generally considered healthy. A ratio below 0.20 suggests that rT3 is disproportionately high relative to active T3, indicating impaired thyroid hormone utilization.
Clinical significance of a low T3/rT3 ratio:
- Even when TSH, Free T4, and Free T3 appear within range, a low ratio reveals that the body is favoring the inactive metabolic pathway
- This pattern is commonly seen in patients with chronic fatigue, fibromyalgia, depression, and unexplained weight gain who are told their thyroid is "fine"
- Addressing the root cause of elevated rT3 (stress, inflammation, nutrient deficiency) often improves the ratio and resolves symptoms
Thyroid Antibodies — TPO, Thyroglobulin, and TSI
Thyroid antibodies are markers of autoimmune thyroid disease and are among the most important yet frequently overlooked components of a comprehensive thyroid panel. Autoimmune thyroid disease, particularly Hashimoto's thyroiditis, is the leading cause of hypothyroidism in the developed world, accounting for an estimated 90% of cases.
TPO Antibodies (Thyroid Peroxidase Antibodies)
TPO is an enzyme essential for thyroid hormone synthesis. When the immune system produces antibodies against TPO, it progressively destroys thyroid tissue. Elevated TPO antibodies are the hallmark of Hashimoto's thyroiditis.
- Conventional reference range: Below 34 IU/mL (varies by laboratory)
- Functional optimal: As close to zero as possible. Any elevation indicates immune activation against the thyroid
- TPO antibodies can be elevated for years or even decades before TSH becomes abnormal. Testing antibodies early allows for intervention before significant thyroid damage occurs
- Elevated TPO antibodies are associated with increased risk of miscarriage, postpartum thyroiditis, and progression to overt hypothyroidism
Thyroglobulin Antibodies (TgAb)
Thyroglobulin is a protein produced by the thyroid gland that serves as a precursor for T4 and T3 synthesis. Antibodies against thyroglobulin also indicate autoimmune thyroid disease.
- Conventional reference range: Below 0.9 IU/mL (varies by laboratory)
- Some patients with Hashimoto's will have elevated thyroglobulin antibodies but normal TPO antibodies, so testing both provides a more complete picture
- Thyroglobulin antibodies are also relevant in thyroid cancer monitoring, as they can interfere with thyroglobulin measurements
TSI (Thyroid-Stimulating Immunoglobulin)
TSI is an antibody that mimics TSH and stimulates the thyroid to overproduce hormones. It is the primary marker for Graves' disease, an autoimmune condition causing hyperthyroidism.
- TSI binds to the TSH receptor and activates it, causing the thyroid to produce excessive T4 and T3
- Testing TSI is essential when hyperthyroidism is suspected (low TSH, elevated Free T4 and Free T3, symptoms such as rapid heart rate, weight loss, anxiety, tremor, and bulging eyes)
- Some patients can have both Hashimoto's and Graves' antibodies simultaneously, leading to fluctuating thyroid function
Common Thyroid Dysfunction Patterns
Subclinical Hypothyroidism
TSH is mildly elevated (typically 2.5 to 10 mIU/L) while Free T4 and Free T3 remain within range. Conventional medicine often takes a "watch and wait" approach, but functional medicine recognizes that many of these patients are symptomatic and benefit from intervention. This pattern may represent early thyroid failure, particularly if thyroid antibodies are present.
Hashimoto's Pattern
Elevated TPO and/or thyroglobulin antibodies with TSH that may be normal, borderline, or elevated. Free T4 and Free T3 may fluctuate. In the early stages ("Hashitoxicosis"), patients may experience alternating periods of hypothyroid and hyperthyroid symptoms as the immune system intermittently attacks and destroys thyroid tissue, releasing stored hormone into the bloodstream.
Poor T4-to-T3 Conversion
TSH may be normal. Free T4 is mid-range or even elevated (especially in patients on levothyroxine). Free T3 is low or in the lower third of the range. This pattern indicates that the body is not efficiently converting the storage hormone into the active form. Common causes include selenium deficiency, zinc deficiency, chronic stress, liver dysfunction, gut dysbiosis, and inflammation.
High Reverse T3 Pattern
TSH and Free T4 may appear normal. Reverse T3 is elevated, and the T3/rT3 ratio is low. This pattern is often seen in patients under chronic stress, those with chronic inflammatory conditions, and individuals on high-dose T4-only medication. The body is shunting T4 into the inactive rT3 pathway rather than the active T3 pathway, resulting in cellular hypothyroidism despite seemingly normal blood work.
Central Hypothyroidism
TSH is low or "normal" (often below 1.0), but Free T4 and Free T3 are also low. This uncommon pattern suggests a problem with the pituitary or hypothalamus rather than the thyroid gland itself. It can be caused by pituitary tumors, head trauma, chronic opioid use, or chronic illness (sometimes called "euthyroid sick syndrome" or "non-thyroidal illness syndrome").
Thyroid Hormone Resistance
All blood markers may appear within range, yet the patient has significant hypothyroid symptoms. This rare condition involves a genetic or acquired defect in thyroid hormone receptors, preventing T3 from effectively activating cellular processes. Diagnosis is challenging and often requires clinical correlation and genetic testing.
Symptoms of Thyroid Dysfunction
Thyroid hormones influence virtually every cell and organ system in the body. Dysfunction can manifest in a remarkably wide range of symptoms.
Hypothyroid Symptoms (Low Thyroid Function)
- Fatigue and low energy, especially in the morning
- Unexplained weight gain or difficulty losing weight
- Cold intolerance and low body temperature
- Dry skin, brittle nails, and thinning hair (especially the outer third of the eyebrows)
- Constipation and sluggish digestion
- Brain fog, poor memory, and difficulty concentrating
- Depression and low mood
- Muscle aches, joint pain, and stiffness
- Elevated cholesterol (particularly LDL)
- Menstrual irregularities, heavy periods, and fertility problems
- Fluid retention and puffiness, especially in the face and hands
- Slow heart rate (bradycardia)
- Hoarse voice
Hyperthyroid Symptoms (Excess Thyroid Function)
- Anxiety, nervousness, and irritability
- Rapid or irregular heartbeat (tachycardia, palpitations)
- Unexplained weight loss despite increased appetite
- Heat intolerance and excessive sweating
- Tremors, especially in the hands
- Insomnia and difficulty sleeping
- Frequent bowel movements or diarrhea
- Muscle weakness
- Eye changes (bulging, dryness, light sensitivity — particularly in Graves' disease)
- Menstrual irregularities and light periods
- Thinning skin and hair loss
Nutrients for Thyroid Health
The thyroid gland and the enzymes responsible for thyroid hormone conversion depend on several key nutrients. Deficiencies in any of these can impair thyroid function even when the gland itself is healthy.
Iodine
Iodine is the essential building block of thyroid hormones. T4 contains four iodine atoms and T3 contains three. Without adequate iodine, the thyroid cannot produce sufficient hormone. However, iodine supplementation must be approached with extreme caution in patients with autoimmune thyroid disease (Hashimoto's), as excessive iodine can exacerbate the autoimmune attack. Food sources include sea vegetables (kelp, nori, dulse), wild-caught fish, and iodized salt. Testing urinary iodine levels before supplementing is recommended.
Selenium
Selenium is critical for thyroid health on multiple levels. It is a cofactor for the deiodinase enzymes that convert T4 to T3 and is also required for glutathione peroxidase, which protects the thyroid gland from oxidative damage during hormone production. Studies have shown that selenium supplementation (typically 200 mcg daily) can reduce TPO antibodies in Hashimoto's patients. Food sources include Brazil nuts (one to two per day provides approximately 200 mcg), wild-caught fish, pasture-raised eggs, and sunflower seeds.
Zinc
Zinc is required for the synthesis of thyroid hormones, the conversion of T4 to T3, and the binding of T3 to its nuclear receptor. Zinc deficiency is associated with decreased thyroid hormone levels and impaired TSH synthesis. Food sources include oysters, grass-fed red meat, pumpkin seeds, and lentils. Typical supplemental doses range from 15 to 30 mg daily.
Iron
Iron is essential for thyroid peroxidase (TPO) enzyme activity, which catalyzes the first step in thyroid hormone synthesis. Iron deficiency, even without frank anemia, can impair thyroid function and reduce the effectiveness of thyroid medication. Ferritin (iron storage) should ideally be above 70 to 90 ng/mL for optimal thyroid function. Many women of reproductive age have suboptimal ferritin levels.
Tyrosine
Tyrosine is an amino acid that combines with iodine to form thyroid hormones. It is also a precursor for dopamine, norepinephrine, and epinephrine. Adequate protein intake generally provides sufficient tyrosine, but supplementation (500 to 1000 mg daily on an empty stomach) may be beneficial in some cases. Food sources include poultry, fish, eggs, dairy, almonds, and avocados.
Vitamin D
Vitamin D is technically a hormone and plays a significant role in immune regulation. Low vitamin D levels are strongly associated with autoimmune thyroid disease, including Hashimoto's thyroiditis. Optimal vitamin D levels (measured as 25-hydroxy vitamin D) should be between 50 and 80 ng/mL for immune modulation. Many patients with thyroid dysfunction are deficient and benefit from supplementation with vitamin D3 alongside vitamin K2.
Additional Supportive Nutrients
- Vitamin A: Required for thyroid hormone receptor activation and TSH regulation. Best obtained from animal sources (liver, egg yolks) as preformed retinol
- B vitamins: Particularly B12, which is commonly deficient in Hashimoto's patients, and B2 (riboflavin), which supports the flavin-dependent deiodinase pathway
- Magnesium: Supports T4-to-T3 conversion and is commonly depleted in modern diets
- Omega-3 fatty acids: Help reduce thyroid inflammation and support cell membrane fluidity for hormone receptor function
Gut Health and the Thyroid Connection
The relationship between gut health and thyroid function is one of the most important and underappreciated connections in medicine. Approximately 20% of T4-to-T3 conversion occurs in the gastrointestinal tract, making a healthy gut essential for optimal thyroid hormone activation.
Key connections between the gut and thyroid:
- Intestinal permeability ("leaky gut"): Damage to the intestinal lining allows large protein molecules to cross into the bloodstream, triggering immune activation. This is now recognized as a significant driver of autoimmune diseases, including Hashimoto's. Research by Dr. Alessio Fasano has demonstrated that intestinal permeability is a prerequisite for the development of autoimmunity
- Gut microbiome: Beneficial bacteria produce enzymes that assist in T4-to-T3 conversion and help recycle thyroid hormones through enterohepatic circulation. Dysbiosis (an imbalance in gut bacteria) impairs this process
- Gluten and molecular mimicry: The gliadin protein in gluten has a molecular structure similar to thyroid tissue. In susceptible individuals, the immune response to gliadin can cross-react with thyroid tissue, exacerbating autoimmune thyroid destruction. Many functional medicine practitioners recommend a gluten-free trial for patients with Hashimoto's
- Nutrient absorption: Gut inflammation and damage impair the absorption of thyroid-critical nutrients including iron, selenium, zinc, and B12
- LPS (lipopolysaccharide): Bacterial endotoxins from gram-negative bacteria can cross a permeable gut lining and trigger systemic inflammation, which increases Reverse T3 production and impairs thyroid function
- SIBO (Small Intestinal Bacterial Overgrowth): Hypothyroidism slows gut motility, which predisposes patients to SIBO. SIBO in turn impairs nutrient absorption and increases inflammation, creating a vicious cycle with thyroid dysfunction
Addressing gut health through dietary modifications, probiotic support, removal of food sensitivities, and healing the intestinal lining is often a foundational step in restoring thyroid function, particularly in autoimmune thyroid disease.
When to Test — Timing and Preparation
The accuracy of thyroid lab results can be significantly influenced by the time of day the blood is drawn and other factors. Following these guidelines ensures the most reliable and reproducible results.
Optimal Testing Conditions
- Time of day: TSH follows a circadian rhythm, peaking between approximately 2:00 AM and 4:00 AM, then declining throughout the day. Morning blood draws (before 10:00 AM) capture TSH closer to its peak and provide the most sensitive reading. An afternoon draw can yield a TSH value up to 50% lower than a morning draw, potentially masking early hypothyroidism
- Fasting: A fasting blood draw is recommended. Eating, particularly a meal containing carbohydrates, can transiently suppress TSH levels
- Thyroid medication: If taking thyroid hormone medication, do not take your dose the morning of the blood draw. Taking medication before the draw can artificially elevate Free T4 and Free T3 levels, giving an inaccurate picture of baseline hormone levels. Take the medication after the blood draw
- Biotin supplements: High-dose biotin (commonly found in hair, skin, and nail supplements) can interfere with thyroid immunoassays, causing falsely low TSH and falsely elevated Free T4 and Free T3. Discontinue biotin supplementation at least 48 to 72 hours before testing
- Consistency: For monitoring purposes, always draw labs at approximately the same time of day and under the same conditions to ensure accurate comparisons over time
How Often to Test
- Baseline: A full comprehensive thyroid panel when first evaluating thyroid function
- Follow-up: Retest 6 to 8 weeks after starting or adjusting thyroid medication
- Monitoring autoimmune disease: Check antibody levels every 3 to 6 months when actively working to reduce autoimmunity through dietary and lifestyle interventions
- Stable patients: Annual comprehensive panel for patients on stable thyroid treatment or those with known autoimmune thyroid disease
- Symptomatic changes: Retest whenever new or worsening symptoms develop
Cautions About Self-Treating
While understanding your thyroid labs is empowering, thyroid conditions require careful professional management. Self-treating thyroid dysfunction carries significant risks.
- Thyroid hormones are potent medications. Both T4 (levothyroxine) and T3 (liothyronine or compounded T3) are prescription medications for good reason. Incorrect dosing can cause or worsen heart arrhythmias, bone loss (osteoporosis), anxiety, insomnia, and adrenal stress
- Over-supplementing iodine is dangerous. Excessive iodine intake can worsen Hashimoto's thyroiditis, trigger thyroid storms in susceptible individuals, and cause paradoxical hypothyroidism (the Wolff-Chaikoff effect). Never take high-dose iodine supplements without testing and professional guidance
- Desiccated thyroid (NDT) products require supervision. While natural desiccated thyroid (such as Armour Thyroid or NP Thyroid) is favored by many functional medicine practitioners, it contains both T4 and T3 and requires careful dosing and monitoring
- Over-the-counter "thyroid support" supplements may contain undisclosed thyroid hormones derived from animal thyroid glands. These products are unregulated and can cause unpredictable fluctuations in thyroid hormone levels
- Adrenal function must be assessed alongside thyroid function. Correcting thyroid levels without addressing adrenal insufficiency or cortisol dysregulation can worsen symptoms. The adrenal glands and thyroid are intimately connected, and treating one without evaluating the other can lead to setbacks
- Interpreting labs in isolation is risky. Thyroid lab values must be interpreted in the context of symptoms, medical history, medications, other lab findings, and the whole patient. A single marker out of range does not necessarily warrant treatment
- Pregnancy considerations: Thyroid requirements change significantly during pregnancy. Inadequate thyroid hormone can cause miscarriage, developmental delays, and preeclampsia. Pregnant women with known thyroid conditions should be monitored closely by their healthcare team
Work with a knowledgeable practitioner — ideally one trained in functional or integrative medicine who will order a comprehensive panel and interpret results using optimal ranges rather than merely ruling out overt disease.
Summary
A comprehensive thyroid panel is an invaluable tool for uncovering the root cause of thyroid dysfunction. Testing TSH alone is insufficient and misses a significant number of patients whose thyroid issues lie in hormone conversion, autoimmunity, or cellular receptor resistance. By evaluating TSH, Free T4, Free T3, Reverse T3, TPO antibodies, thyroglobulin antibodies, and when indicated TSI, a complete picture of thyroid health emerges.
Key takeaways:
- Always request a full thyroid panel, not just TSH
- Functional optimal ranges are narrower than conventional reference ranges and often reveal dysfunction that standard screening misses
- Free T3 is the most important marker of active thyroid hormone status
- Reverse T3 reveals whether the body is shunting T4 away from the active pathway
- Thyroid antibodies can detect autoimmune thyroid disease years before other markers become abnormal
- Nutrient status — particularly selenium, zinc, iron, iodine, and vitamin D — profoundly influences thyroid function
- Gut health is foundational to thyroid hormone conversion and immune regulation
- Test in the morning, fasting, before taking thyroid medication, and without recent biotin supplementation
- Work with a qualified practitioner who understands the complexity of thyroid physiology
Key Research Papers
Foundational and widely-cited peer-reviewed publications behind thyroid panel interpretation, autoimmunity, treatment, and the controversies around TSH reference ranges and combination therapy. Each citation links to the full text via DOI.
- Hollowell JG, Staehling NW, Flanders WD, et al. Serum TSH, T₄, and Thyroid Antibodies in the United States Population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). Journal of Clinical Endocrinology & Metabolism. 2002;87(2):489–499.
- Surks MI, Goswami G, Daniels GH. The Thyrotropin Reference Range Should Remain Unchanged. Journal of Clinical Endocrinology & Metabolism. 2005;90(9):5489–5496.
- Wartofsky L, Dickey RA. The Evidence for a Narrower Thyrotropin Reference Range Is Compelling. Journal of Clinical Endocrinology & Metabolism. 2005;90(9):5483–5488.
- Rodondi N, den Elzen WP, Bauer DC, et al. Subclinical Hypothyroidism and the Risk of Coronary Heart Disease and Mortality. JAMA. 2010;304(12):1365–1374.
- Collet TH, Gussekloo J, Bauer DC, et al. Subclinical Hyperthyroidism and the Risk of Coronary Heart Disease and Mortality. Archives of Internal Medicine. 2012;172(10):799–809.
- Bianco AC, Salvatore D, Gereben B, Berry MJ, Larsen PR. Biochemistry, Cellular and Molecular Biology, and Physiological Roles of the Iodothyronine Selenodeiodinases. Endocrine Reviews. 2002;23(1):38–89.
- Peeters RP, Wouters PJ, Kaptein E, van Toor H, Visser TJ, Van den Berghe G. Reduced Activation and Increased Inactivation of Thyroid Hormone in Tissues of Critically Ill Patients. Journal of Clinical Endocrinology & Metabolism. 2003;88(7):3202–3211.
- Caturegli P, De Remigis A, Rose NR. Hashimoto Thyroiditis: Clinical and Diagnostic Criteria. Autoimmunity Reviews. 2014;13(4–5):391–397.
- Bunevicius R, Kazanavicius G, Zalinkevicius R, Prange AJ Jr. Effects of Thyroxine as Compared with Thyroxine plus Triiodothyronine in Patients with Hypothyroidism. New England Journal of Medicine. 1999;340(6):424–429.
- Jonklaas J, Bianco AC, Bauer AJ, et al. Guidelines for the Treatment of Hypothyroidism (American Thyroid Association Task Force). Thyroid. 2014;24(12):1670–1751.
- Hoang TD, Olsen CH, Mai VQ, Clyde PW, Shakir MK. Desiccated Thyroid Extract Compared with Levothyroxine in the Treatment of Hypothyroidism: A Randomized, Double-Blind, Crossover Study. Journal of Clinical Endocrinology & Metabolism. 2013;98(5):1982–1990.
- Zimmermann MB, Boelaert K. Iodine Deficiency and Thyroid Disorders. Lancet Diabetes & Endocrinology. 2015;3(4):286–295.
- Gärtner R, Gasnier BCH, Dietrich JW, Krebs B, Angstwurm MWA. Selenium Supplementation in Patients with Autoimmune Thyroiditis Decreases Thyroid Peroxidase Antibodies Concentrations. Journal of Clinical Endocrinology & Metabolism. 2002;87(4):1687–1691.
- Bahn RS, Burch HB, Cooper DS, et al. Hyperthyroidism and Other Causes of Thyrotoxicosis: Management Guidelines of the American Thyroid Association and American Association of Clinical Endocrinologists. Thyroid. 2011;21(6):593–646.
- Smith TJ, Hegedüs L. Graves' Disease. New England Journal of Medicine. 2016;375(16):1552–1565.
- Knezevic J, Starchl C, Tmava Berisha A, Amrein K. Thyroid–Gut-Axis: How Does the Microbiota Influence Thyroid Function? Nutrients. 2020;12(6):1769.
Live PubMed Searches
Live PubMed queries that update as new studies are indexed.
- PubMed: TSH reference range controversy
- PubMed: subclinical hypothyroidism & CV risk
- PubMed: Free T3 / reverse T3 ratio
- PubMed: Hashimoto & TPO antibodies
- PubMed: T4 vs T4+T3 combination therapy
- PubMed: iodine & thyroid function
- PubMed: selenium & thyroid autoimmunity
- PubMed: reverse T3 in non-thyroidal illness
- PubMed: Graves disease & TSI
- PubMed: thyroid hormone & gut microbiome
External Authoritative Resources
- MedlinePlus — Laboratory Tests Reference
- Lab Tests Online — Patient-Oriented Lab Test Reference (AACC)
- StatPearls / NCBI Bookshelf — Clinical Laboratory Reference
Connections
- Hashimoto's Thyroiditis
- Graves' Disease
- Thyroid Disorders
- Thyroid Cancer
- Iodine
- Selenium
- Zinc
- Iron
- Tyrosine
- Vitamin D3
- Depression
- Anxiety
- Osteoporosis
- Fatigue
- Brain Fog
- Full Body MRI
- Perimenopause
- Low Dose Naltrexone