Preeclampsia
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment and Obstetric Management
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- References
1. Overview
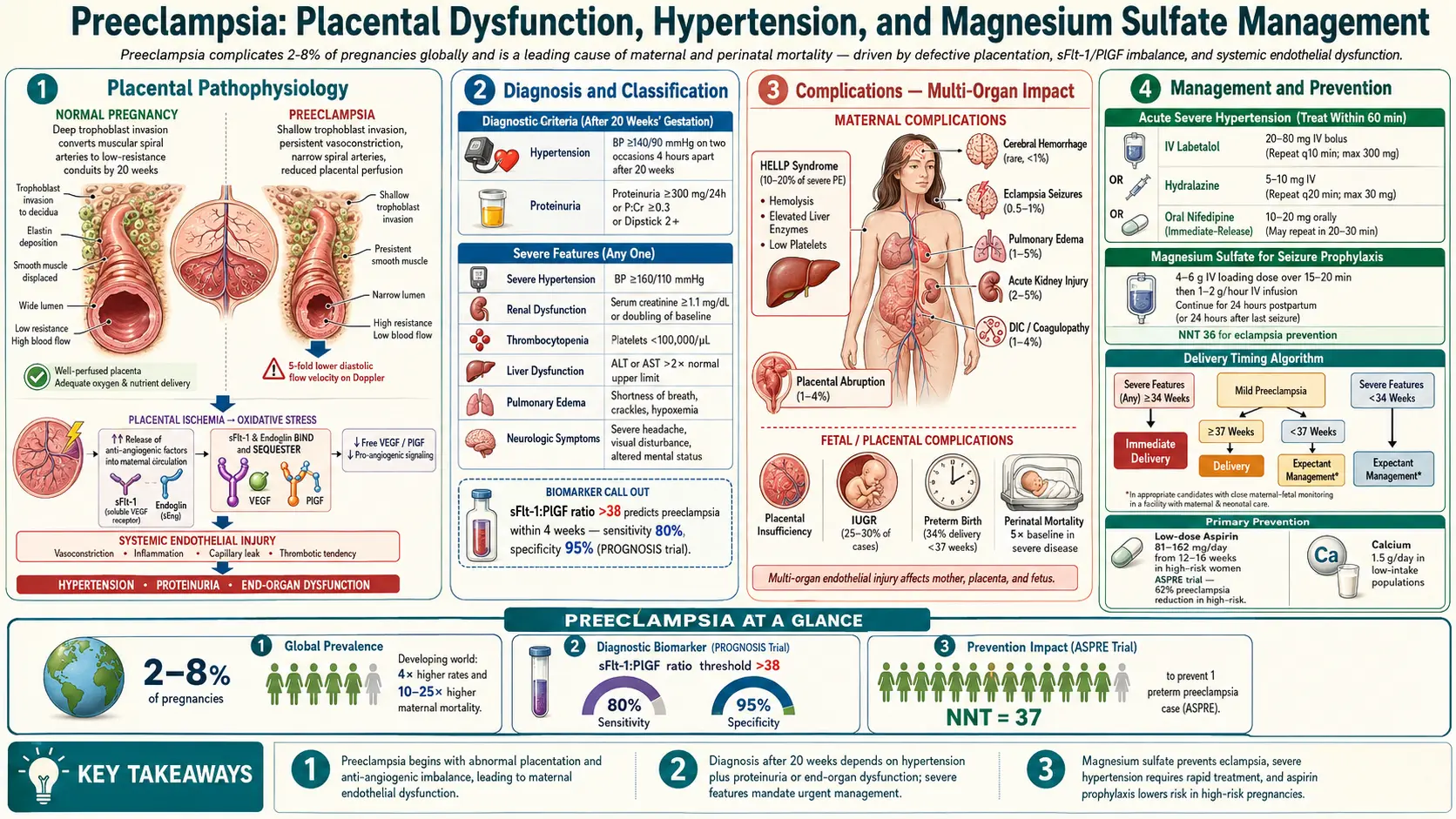
Preeclampsia is a multisystem hypertensive disorder of pregnancy defined by new-onset hypertension (systolic blood pressure of 140 mmHg or greater, or diastolic BP of 90 mmHg or greater) after 20 weeks of gestation with proteinuria or, in the absence of proteinuria, with severe features including thrombocytopenia, renal insufficiency, impaired liver function, pulmonary edema, or new-onset headache unresponsive to medication or visual disturbances.
Preeclampsia exists within a spectrum of hypertensive disorders of pregnancy that includes gestational hypertension, preeclampsia with and without severe features, HELLP syndrome (Hemolysis, Elevated Liver enzymes, Low Platelets), and eclampsia. It is a leading cause of maternal and perinatal morbidity and mortality globally, and its only definitive treatment remains delivery of the fetus and placenta.
2. Epidemiology
Preeclampsia complicates approximately 2–8% of pregnancies worldwide, accounting for an estimated 76,000 maternal deaths and 500,000 perinatal deaths annually. Incidence varies significantly by geographic region, population characteristics, and diagnostic criteria. Rates are highest in low- and middle-income countries where access to antenatal care and emergency obstetric services is limited.
In the United States, preeclampsia affects approximately 3–4% of pregnancies, but incidence has increased by approximately 25% over the past two decades, paralleling rising rates of obesity, hypertension, and assisted reproductive technology use. Black women are disproportionately affected, with incidence approximately 60% higher than in White women and case fatality rates three to four times higher, reflecting disparities in access to care, comorbidities, and potentially biological differences in placentation.
Early-onset preeclampsia (before 34 weeks gestation) accounts for approximately 10% of cases but carries disproportionately severe maternal and perinatal outcomes. Late-onset preeclampsia (after 34 weeks) is more common but generally less severe.
3. Pathophysiology
Preeclampsia is fundamentally a placental disease mediated by abnormal trophoblastic invasion of the maternal spiral arteries and subsequent systemic endothelial dysfunction.
Stage 1: Defective Trophoblast Invasion
In normal implantation, extravillous cytotrophoblasts invade the maternal decidua and myometrium, remodeling spiral arteries from high-resistance, low-flow muscular vessels into dilated, low-resistance conduits capable of supplying the exponentially increasing blood flow demands of the growing placenta. In preeclampsia, this physiological transformation is incomplete: spiral arteries retain their muscular media and remain narrow, high-resistance vessels. This defective remodeling produces placental ischemia, particularly under the metabolic demands of the second and third trimesters.
Stage 2: Systemic Endothelial Dysfunction
Ischemic and oxidatively stressed placental tissue releases an imbalanced repertoire of pro- and anti-angiogenic factors into the maternal circulation:
- sFlt-1 (soluble FMS-like tyrosine kinase-1): A circulating anti-angiogenic protein that acts as a decoy receptor, sequestering free VEGF (vascular endothelial growth factor) and PlGF (placental growth factor), preventing them from binding their endothelial receptors and maintaining normal endothelial function.
- sEng (soluble endoglin): A circulating anti-angiogenic factor that inhibits TGF-beta signaling in endothelium, contributing to vasoconstriction and vascular permeability.
Elevated sFlt-1 and sEng, combined with reduced free PlGF, produce generalized maternal endothelial dysfunction characterized by: (1) vasospasm and hypertension, (2) increased vascular permeability leading to edema and proteinuria, (3) activation of the coagulation cascade with microthrombus formation, and (4) end-organ ischemia (renal, hepatic, cerebral, uteroplacental).
Renal Pathology: Glomerular Endotheliosis
The characteristic renal lesion of preeclampsia is glomerular endotheliosis: swelling and vacuolization of glomerular endothelial cells obliterating the capillary lumen. This produces reduced GFR, sodium retention, hyperuricemia, and the characteristic proteinuria of 300 mg or more per 24 hours.
Inflammatory and Immunological Components
Exaggerated systemic maternal inflammatory response, characterized by neutrophil and monocyte activation, elevated TNF-alpha, IL-6, and IL-8, and reduced regulatory T-cell function, contributes to endothelial activation and vascular injury. Aberrant immune tolerance to paternal (fetal) alloantigens at the maternal-fetal interface may underlie the primigravid and partner-change associations with preeclampsia.
4. Etiology and Risk Factors
High-Risk Factors (single factor sufficient for aspirin prophylaxis per USPSTF)
- History of preeclampsia (recurrence risk 20–25%; higher in early-onset disease)
- Multifetal gestation (twin risk 3×, triplet risk 9× singleton)
- Chronic hypertension (superimposed preeclampsia risk 15–25%)
- Type 1 or Type 2 diabetes mellitus
- Renal disease
- Autoimmune conditions (SLE, antiphospholipid syndrome)
Moderate-Risk Factors (multiple factors needed to recommend aspirin)
- Nulliparity (two- to threefold increased risk)
- Obesity (BMI above 30): each unit increase in pre-pregnancy BMI associated with approximately 6% increased preeclampsia risk
- Age 35 or older at delivery
- Black race (biological and structural factors both implicated)
- Low socioeconomic status and food insecurity
- Family history of preeclampsia (mother or sister)
- Interpregnancy interval greater than 10 years
- Donor egg or donor sperm (reduced immune tolerance)
- Thrombophilias (factor V Leiden, prothrombin gene mutation)
- In vitro fertilization
Placental and Fetal Factors
- Molar pregnancy (exaggerated trophoblastic mass)
- Hydrops fetalis
- Triploidy
5. Clinical Presentation
Preeclampsia Without Severe Features
- BP 140/90 mmHg or greater on two occasions at least 4 hours apart after 20 weeks
- Proteinuria: 300 mg or greater on 24-hour collection, protein:creatinine ratio of 0.3 or greater, or 2+ on dipstick (least preferred)
- OR in absence of proteinuria: any severe feature (see below)
Preeclampsia With Severe Features (any of the following)
- Severe-range BP: systolic 160 mmHg or greater or diastolic 110 mmHg or greater on two occasions 4 hours apart (or immediately if both values are severe with clinical decision to treat)
- Thrombocytopenia: platelet count below 100,000/microliter
- Renal insufficiency: serum creatinine above 1.1 mg/dL or doubling of baseline creatinine
- Impaired liver function: liver transaminases twice normal upper limit; severe persistent right upper quadrant or epigastric pain unresponsive to medications
- Pulmonary edema
- New-onset headache unresponsive to medication and not accounted for by alternative diagnoses
- Visual disturbances (scotomata, photopsia, diplopia, blurring)
HELLP Syndrome
HELLP represents a severe variant with Hemolysis (microangiopathic hemolytic anemia: LDH above 600 IU/L, abnormal peripheral smear), Elevated Liver enzymes (AST/ALT twice upper normal limit), and Low Platelets (below 100,000/microliter). Presents in approximately 10–20% of preeclampsia with severe features; can occur without hypertension or proteinuria in up to 20% of cases. Epigastric pain, nausea, and malaise are typical presenting complaints.
Eclampsia
New-onset grand mal (generalized tonic-clonic) seizures in a woman with preeclampsia not attributable to other causes. May occur antepartum (50%), intrapartum (25%), or postpartum (25%), up to 6 weeks after delivery. Preceded by severe features in most but not all cases; up to 20% of eclampsia occurs without antecedent hypertension or proteinuria.
6. Diagnosis
Blood Pressure Measurement
Accurate BP measurement is foundational. The patient should be seated, resting for 5 minutes, with appropriate cuff size. Automated oscillometric devices validated for use in pregnancy (e.g., Omron M6) are preferred for consistency. Ambulatory blood pressure monitoring (ABPM) distinguishes white coat hypertension (20–30% of apparently hypertensive pregnancies) from sustained hypertension.
Urinary Protein Assessment
Preferred: spot protein:creatinine ratio (cutoff 0.3) or 24-hour urine collection (cutoff 300 mg). Dipstick proteinuria is unreliable and reserved for resource-limited settings only. Nephrotic-range proteinuria (above 3 g/24 hours) carries worse prognosis but does not by itself change management.
Laboratory Investigations
- Complete blood count with differential and platelet count
- Comprehensive metabolic panel (creatinine, BUN, LFTs, albumin)
- LDH, uric acid (hyperuricemia above 6 mg/dL correlates with disease severity)
- Peripheral blood smear (for hemolysis assessment in HELLP)
- Coagulation studies (PT, aPTT, fibrinogen): indicated if platelets below 100,000 or HELLP suspected
Fetal Assessment
- Non-stress test (NST) or biophysical profile (BPP)
- Umbilical artery Doppler velocimetry: absent or reversed end-diastolic flow in the umbilical artery identifies fetuses at highest risk for adverse outcomes
- Estimated fetal weight by ultrasound: fetal growth restriction present in approximately 30% of early-onset preeclampsia
Angiogenic Biomarkers
The sFlt-1:PlGF ratio has emerged as a powerful diagnostic and prognostic biomarker. A ratio below 38 has a negative predictive value of 99.3% for excluding preeclampsia within one week (PROGNOSIS trial). A ratio above 85 (or above 655 at 34 weeks or greater) is associated with a high probability of delivery within 2 weeks due to preeclampsia. The ratio is commercially available in Europe and is FDA-cleared for risk stratification in the US. PlGF-based testing (DELFIA, Elecsys) is increasingly integrated into early-onset preeclampsia screening protocols.
First-Trimester Preeclampsia Screening
The Fetal Medicine Foundation (FMF) combined first-trimester screening algorithm (11–13+6 weeks) integrates mean arterial pressure (MAP), uterine artery pulsatility index (UtA-PI), serum PlGF, and maternal history to predict early-onset preeclampsia with approximately 75% detection rate at a 10% false-positive rate. This approach, validated in the SPREE and ASPRE trials, enables targeted aspirin prophylaxis in the highest-risk women.
7. Treatment and Obstetric Management
Delivery: The Only Definitive Treatment
Delivery is the only definitive cure for preeclampsia. Timing is determined by gestational age, severity of maternal and fetal disease, and institutional capacity for neonatal care.
Gestational Age-Based Management Thresholds
- 37 weeks or greater: Delivery indicated for all preeclampsia (with or without severe features); induction of labor is preferred over expectant management (HYPITAT trial: 80% reduction in maternal complications with induction at 37 weeks).
- 34–36+6 weeks (late preterm): Delivery indicated for preeclampsia with severe features. Expectant management may be appropriate for carefully selected cases of preeclampsia without severe features in centers with adequate monitoring capability.
- Less than 34 weeks (severe features present): Immediate stabilization of maternal condition (antihypertensive therapy, magnesium sulfate seizure prophylaxis, corticosteroids for fetal lung maturity), then delivery after 48 hours if feasible. Expectant management beyond 34 weeks is not recommended for severe features. ACOG and SMFM recommend against expectant management at any gestational age for HELLP syndrome, eclampsia, uncontrolled severe-range BP, renal failure, or pulmonary edema.
- Less than 23–24 weeks (previable): Delivery is recommended for severe features; neonatal survival is generally not achievable; maternal health takes priority.
Antihypertensive Therapy
Severe-range BP (160/110 mmHg or greater) requires urgent treatment within 30–60 minutes to prevent maternal stroke. First-line agents:
- Labetalol IV: 20 mg bolus, repeat every 10 minutes (maximum 300 mg) or infusion 1–2 mg/min. Avoid in asthma.
- Hydralazine IV: 5–10 mg bolus every 20–30 minutes (maximum 20 mg). Reflex tachycardia and unpredictable response.
- Nifedipine oral (immediate-release): 10–20 mg; onset 15–30 minutes. Avoid sublingual route (precipitous hypotension). Concurrent use with magnesium sulfate was historically cautioned but is now considered safe.
- Oral antihypertensives for sustained control (non-severe BP): Labetalol, nifedipine XL, and methyldopa are used for non-severe-range BP in expectant management. ACE inhibitors and ARBs are contraindicated in pregnancy.
Magnesium Sulfate
The gold standard for eclampsia seizure prophylaxis and treatment. Loading dose: 4–6 g IV over 15–20 minutes; maintenance: 1–2 g/hour infusion. Continue for 24–48 hours postpartum. For eclamptic seizure: additional 2–4 g bolus; diazepam or lorazepam if seizure persists. Monitor for toxicity: loss of deep tendon reflexes (first sign, at 7–10 mEq/L), respiratory depression (above 10 mEq/L), cardiac arrest (above 15 mEq/L). Calcium gluconate 1 g IV is the antidote. Dose reduction required in renal insufficiency with urine output monitoring.
Corticosteroids
Betamethasone 12 mg IM every 24 hours for two doses (or dexamethasone 6 mg IM every 12 hours for four doses) to accelerate fetal lung maturity when delivery is anticipated before 36+6 weeks. In HELLP, high-dose dexamethasone (10 mg IV every 12 hours) may transiently improve platelet counts and transaminases, facilitating safe delivery, though does not alter maternal outcome.
Mode of Delivery
Vaginal delivery is preferred when maternal and fetal status permit. Cesarean delivery is indicated for obstetric indications (fetal malpresentation, prior classical uterine incision, unfavorable cervix with deteriorating maternal condition) rather than preeclampsia diagnosis alone. Spinal anesthesia is preferred over general anesthesia; epidural anesthesia is acceptable if platelet count is above 70,000–80,000/microliter.
Postpartum Management
Approximately 25% of eclamptic seizures occur postpartum, most within 48 hours but up to 6 weeks later. Continue magnesium sulfate for 24–48 hours postpartum. Continue antihypertensive therapy as needed; NSAID use in the puerperium may worsen hypertension and should be avoided in severe preeclampsia. Breastfeeding is encouraged; labetalol, nifedipine, and methyldopa are compatible with lactation.
8. Complications
Maternal Complications
- Eclampsia: Tonic-clonic seizures; risk of intracranial hemorrhage, aspiration pneumonia, placental abruption
- HELLP syndrome: Subcapsular liver hematoma with risk of hepatic rupture (catastrophic, mortality 50%); DIC
- Stroke and hypertensive encephalopathy
- Acute kidney injury: Acute tubular necrosis; cortical necrosis in severe prolonged cases
- Pulmonary edema: From increased capillary permeability and left ventricular dysfunction
- Placental abruption
- Disseminated intravascular coagulation (DIC)
- Long-term cardiovascular risk: Women with a history of preeclampsia have twofold increased lifetime risk of ischemic heart disease, stroke, and hypertension; fourfold increased risk of heart failure
Fetal and Neonatal Complications
- Fetal growth restriction (FGR)
- Placental insufficiency and non-reassuring fetal status
- Preterm birth (iatrogenic and spontaneous)
- Stillbirth (risk increased three- to fourfold)
- Neonatal complications of prematurity (respiratory distress syndrome, intraventricular hemorrhage, necrotizing enterocolitis)
- Long-term neurodevelopmental sequelae of extreme prematurity
9. Prognosis
The prognosis for mother and infant depends critically on gestational age at onset, severity of disease, quality of obstetric care, and interval from diagnosis to delivery. Term preeclampsia without severe features carries excellent short-term maternal prognosis with prompt delivery. Early-onset severe preeclampsia before 28 weeks carries significantly elevated maternal and perinatal mortality and morbidity.
Recurrence risk is approximately 20–25% overall; up to 55% for women with preeclampsia requiring delivery before 28 weeks. Long-term maternal cardiovascular risk is substantially elevated: women with prior preeclampsia should be counseled and monitored accordingly, with optimization of modifiable risk factors (hypertension, dyslipidemia, diabetes, obesity) throughout life.
10. Prevention
Low-Dose Aspirin
The cornerstone of preeclampsia prevention. Aspirin 81–162 mg daily (ACOG recommends 81 mg; NICE recommends 75–150 mg), initiated between 12 and 16 weeks gestation (preferably before 16 weeks) and continued until 36–37 weeks. The ASPRE trial demonstrated a 62% reduction in early-onset preeclampsia in high-risk women identified by first-trimester combined screening. Meta-analysis demonstrates approximately 17% overall reduction in preeclampsia with aspirin prophylaxis. USPSTF (2021) recommends aspirin prophylaxis for women with one or more high-risk factors or multiple moderate-risk factors (Grade B recommendation).
Calcium Supplementation
In populations with low dietary calcium intake (below 600 mg/day), calcium supplementation at 1.5–2 g elemental calcium daily reduces preeclampsia risk by approximately 55% (WHO recommendation). Benefit is reduced in populations with adequate calcium intake (i.e., most high-income countries).
Other Interventions
- Weight optimization before pregnancy (BMI 18.5–24.9 kg/m2)
- Tight glycemic control in pre-existing diabetes
- Antihypertensive therapy for chronic hypertension (though treating non-severe chronic hypertension to reduce preeclampsia risk itself is debated; the CHIPS trial supports treating to diastolic BP of 85 mmHg)
- Heparin prophylaxis for women with antiphospholipid syndrome (reduces superimposed preeclampsia and adverse pregnancy outcomes)
- Smoking cessation (paradoxically, smoking appears to reduce preeclampsia incidence but dramatically worsens fetal outcomes and overall health)
11. Recent Research and Advances
sFlt-1:PlGF ratio-guided management: The PROGNOSIS trial established the clinical utility of the sFlt-1:PlGF ratio for short-term prediction of preeclampsia onset. The ratio enables individualized decisions about hospitalization, surveillance intensity, and delivery timing. A ratio below 38 provides a one-week "safe window" to avoid unnecessary preterm delivery.
ASPRE trial and first-trimester screening: First-trimester combined screening with targeted aspirin prophylaxis reduces preterm preeclampsia by 62%, validating the FMF algorithm for widespread clinical implementation. Post-market effectiveness studies are confirming these benefits in real-world settings.
CHIPS trial (Control of Hypertension in Pregnancy Study): Demonstrated that "tight control" of maternal hypertension (diastolic BP target 85 mmHg) versus "less tight control" (100 mmHg) reduces severe maternal hypertension without increasing pregnancy loss or neonatal morbidity, establishing a new standard for antihypertensive management in pregnancy.
Anti-sFlt-1 therapy (aflibercept analog, recombinant PlGF): Investigational therapies targeting the sFlt-1/PlGF imbalance are in early clinical trials. Apheresis columns selectively removing sFlt-1 from maternal plasma (Fresenius Medical Care) demonstrated prolongation of pregnancy by approximately one week in early case series, with prospective trial data awaited.
Hydroxychloroquine: The PATCHES trial and other studies are evaluating hydroxychloroquine (400 mg daily from first trimester) in autoimmune preeclampsia (antiphospholipid syndrome, SLE) with preliminary positive data on pregnancy outcomes.
Postpartum cardiovascular risk management: Structured postpartum follow-up programs (cardiometabolic testing at 6 weeks, 6 months, and annually thereafter) are being implemented to translate the population-level epidemiological association between preeclampsia and long-term cardiovascular disease into actionable preventive care.
12. References
- ACOG Practice Bulletin No. 222: Gestational Hypertension and Preeclampsia. Obstet Gynecol. 2020;135(6):e237–e260.
- Magee LA, Brown MA, Hall DR, et al. The 2021 International Society for the Study of Hypertension in Pregnancy classification, diagnosis and management recommendations for international practice. Pregnancy Hypertens. 2022;27:148–169.
- Rolnik DL, Wright D, Poon LC, et al. Aspirin versus Placebo in Pregnancies at High Risk for Preterm Preeclampsia (ASPRE). N Engl J Med. 2017;377(7):613–622.
- Zeisler H, Llurba E, Chantraine F, et al. Predictive value of the sFlt-1:PlGF ratio in women with suspected preeclampsia (PROGNOSIS). N Engl J Med. 2016;374(1):13–22.
- Magee LA, von Dadelszen P, Rey E, et al. Less-tight versus tight control of hypertension in pregnancy (CHIPS). N Engl J Med. 2015;372(5):407–417.
- Mutter WP, Karumanchi SA. Molecular mechanisms of preeclampsia. Microvasc Res. 2008;75(1):1–8.
- Levine RJ, Maynard SE, Qian C, et al. Circulating angiogenic factors and the risk of preeclampsia. N Engl J Med. 2004;350(7):672–683.
- Redman CW, Sargent IL. Latest advances in understanding preeclampsia. Science. 2005;308(5728):1592–1594.
- Bellamy L, Casas JP, Hingorani AD, Williams DJ. Pre-eclampsia and risk of cardiovascular disease and cancer in later life: systematic review and meta-analysis. BMJ. 2007;335(7627):974.
- WHO recommendations for prevention and treatment of pre-eclampsia and eclampsia. Geneva: World Health Organization; 2011. ISBN 978 92 4 154833 5.
- Duley L, Meher S, Jones L. Drugs for treatment of very high blood pressure during pregnancy. Cochrane Database Syst Rev. 2013;(7):CD001449.
- Hofmeyr GJ, Lawrie TA, Atallah AN, Torloni MR. Calcium supplementation during pregnancy for preventing hypertensive disorders and related problems. Cochrane Database Syst Rev. 2018;10:CD001059.
- Karumanchi SA, Granger JP. Preeclampsia and pregnancy-related hypertensive disorders. Hypertension. 2016;67(2):238–242.
- Henderson JT, Vesco KK, Senger CA, et al. Aspirin use to prevent preeclampsia and related morbidity and mortality: updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2021;326(12):1192–1206.
- Phipps EA, Thadhani R, Benzing T, Karumanchi SA. Pre-eclampsia: pathogenesis, novel diagnostics and therapies. Nat Rev Nephrol. 2019;15(5):275–289.
- Featured Videos
Research Papers
The following PubMed topic searches aggregate the current peer-reviewed literature. Each link opens a live PubMed query — results update as new studies are indexed.
- PubMed — preeclampsia
- PubMed — severe preeclampsia management
- PubMed — HELLP syndrome
- PubMed — eclampsia magnesium sulfate
- PubMed — Magpie trial preeclampsia
- PubMed — low dose aspirin preeclampsia prevention
- PubMed — sFlt-1 PlGF ratio
- PubMed — chronic hypertension pregnancy
- PubMed — preeclampsia placental pathology
- PubMed — postpartum preeclampsia
- PubMed — preeclampsia cardiovascular risk
- PubMed — preeclampsia recurrence
Connections
- Hypertension
- Magnesium
- Calcium
- Gestational Diabetes
- Vitamin D3
- Pregnancy and Hashimoto's
- Hashimoto's Thyroiditis
- Stroke
- Edema
- Obesity
- Creatinine
- Diabetes
- Anemia
- Heart Failure
- Thrombocytopenia
- Cardiovascular Disease
- Acute Kidney Injury
- Disseminated Intravascular Coagulation