Iodine: The Master Mineral Your Body Cannot Live Without
A Comprehensive Clinical Guide
Presented for patients seeking deeper understanding of Iodine
Table of Contents
- Introduction — Why Iodine Deserves Your Attention
- Historical Context and the Iodine Crisis
- Biochemistry of Iodine in the Human Body
- Iodine and the Thyroid Gland — The Foundation of Metabolism
- Iodine Deficiency, Hypothyroidism, and Hashimoto's Thyroiditis
- Iodine and Breast Health
- Iodine and Reproductive Health — Fertility, Pregnancy, and Fetal Development
- Iodine and Brain Health — Cognition, IQ, and Neurological Development
- Iodine and Immune System Function
- Iodine as a Detoxification Agent — Halide Displacement and Heavy Metals
- Iodine and Cancer Prevention — Emerging Research
- Iodine and Skin, Hair, and Nail Health
- Iodine and Cardiovascular Health
- Iodine, Metabolic Syndrome, and Weight Management
- Iodine and Gastrointestinal Health
- Iodine and Eye Health
- Iodine as a Natural Antimicrobial Agent
- Recognizing Iodine Deficiency — Clinical Signs and Symptoms
- Testing for Iodine Status
- Dietary and Supplemental Sources of Iodine
- Clinical Dosing Strategies and Protocols
- Companion Nutrients — The Iodine Protocol
- Safety, Contraindications, and the Wolff-Chaikoff Debate
- Conclusion — Reclaiming the Master Mineral
- References and Recommended Reading
- Connections
- Featured Videos
1. Introduction — Why Iodine Deserves Your Attention
Among all of them, iodine occupies a uniquely vital position — one that modern medicine has, in many ways, tragically overlooked or underestimated. While most clinicians acknowledge iodine's role in thyroid function, the story of iodine extends far beyond the butterfly-shaped gland in your neck. Every single cell in the human body requires iodine. Every gland, every organ, every tissue uses this essential halide mineral. When iodine is deficient, the cascade of dysfunction that follows can touch virtually every system in the body.
The World Health Organization has identified iodine deficiency as the single most preventable cause of intellectual disability worldwide. Despite this, an estimated two billion people globally remain iodine-insufficient, and emerging research suggests that even in developed nations — including the United States, Canada, the United Kingdom, and Australia — iodine levels have been declining steadily for decades. The National Health and Nutrition Examination Survey (NHANES) data shows that median urinary iodine levels in the United States dropped by more than 50% between the 1970s and the early 2000s, and have not meaningfully recovered since.
This article is designed to serve as a comprehensive clinical resource. Whether you are a patient trying to understand your own health, or a practitioner seeking an integrative perspective on iodine's role in human physiology, the information that follows aims to be thorough, evidence-informed, and clinically practical. We will explore iodine's effects on thyroid function, brain health, breast tissue, reproductive health, immune defense, detoxification, cancer prevention, cardiovascular function, skin integrity, and more. We will also address the controversies, discuss testing methods, review dosing strategies, and outline the companion nutrients that make iodine supplementation safe and effective.
Let us begin with the story of how we got here.
2. Historical Context and the Iodine Crisis
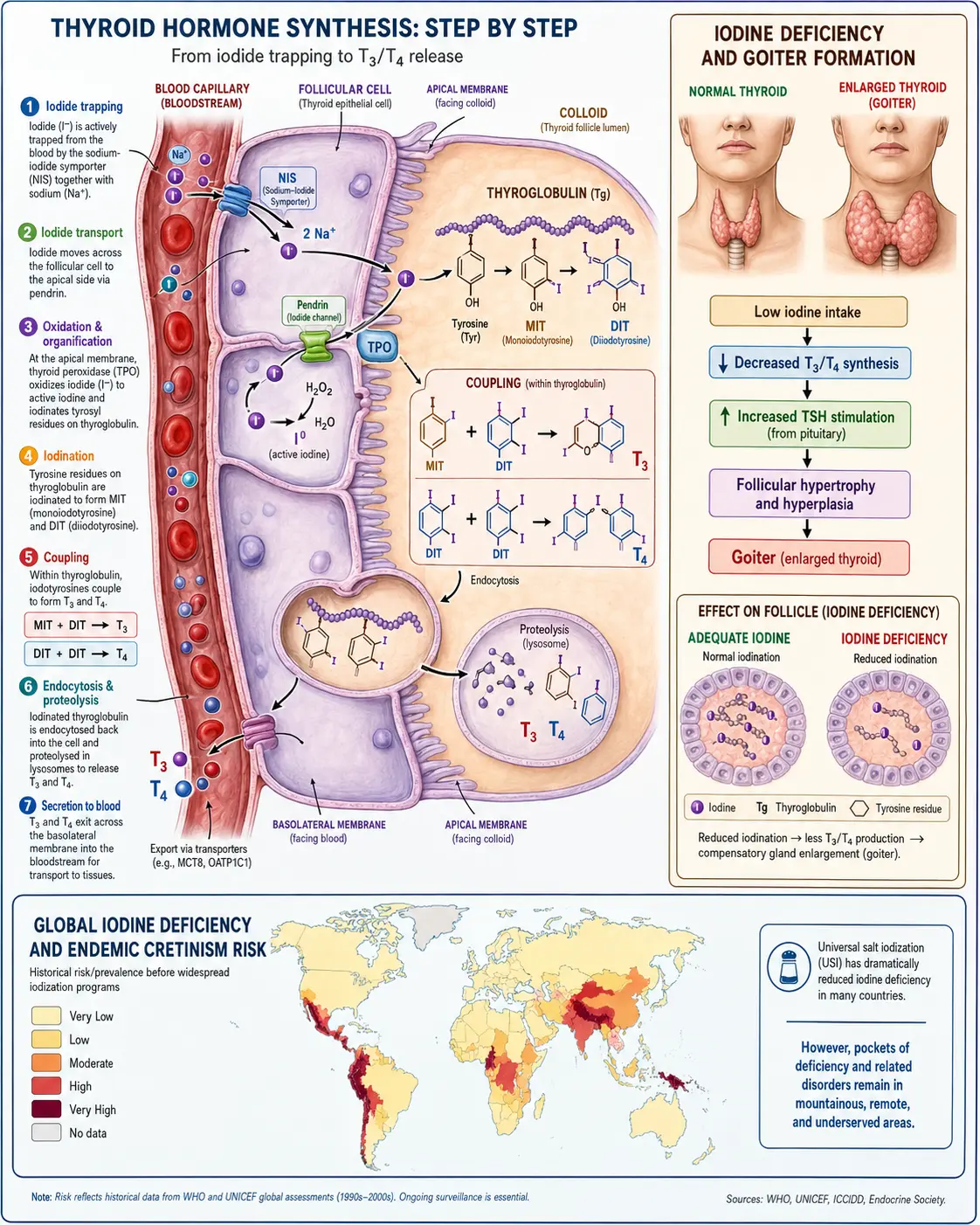
The element iodine was discovered by accident in 1811, when the French saltpetre maker Bernard Courtois saw a violet vapor rise from seaweed ash, and over the following century researchers traced its essential role in the thyroid and the goiter-fighting triumph of iodized salt (first sold in 1924). Yet even today iodine deficiency remains the leading preventable cause of intellectual disability worldwide, and intakes in several developed nations have quietly drifted downward as diets and salt habits change — the fuller story is told in the dedicated History article.
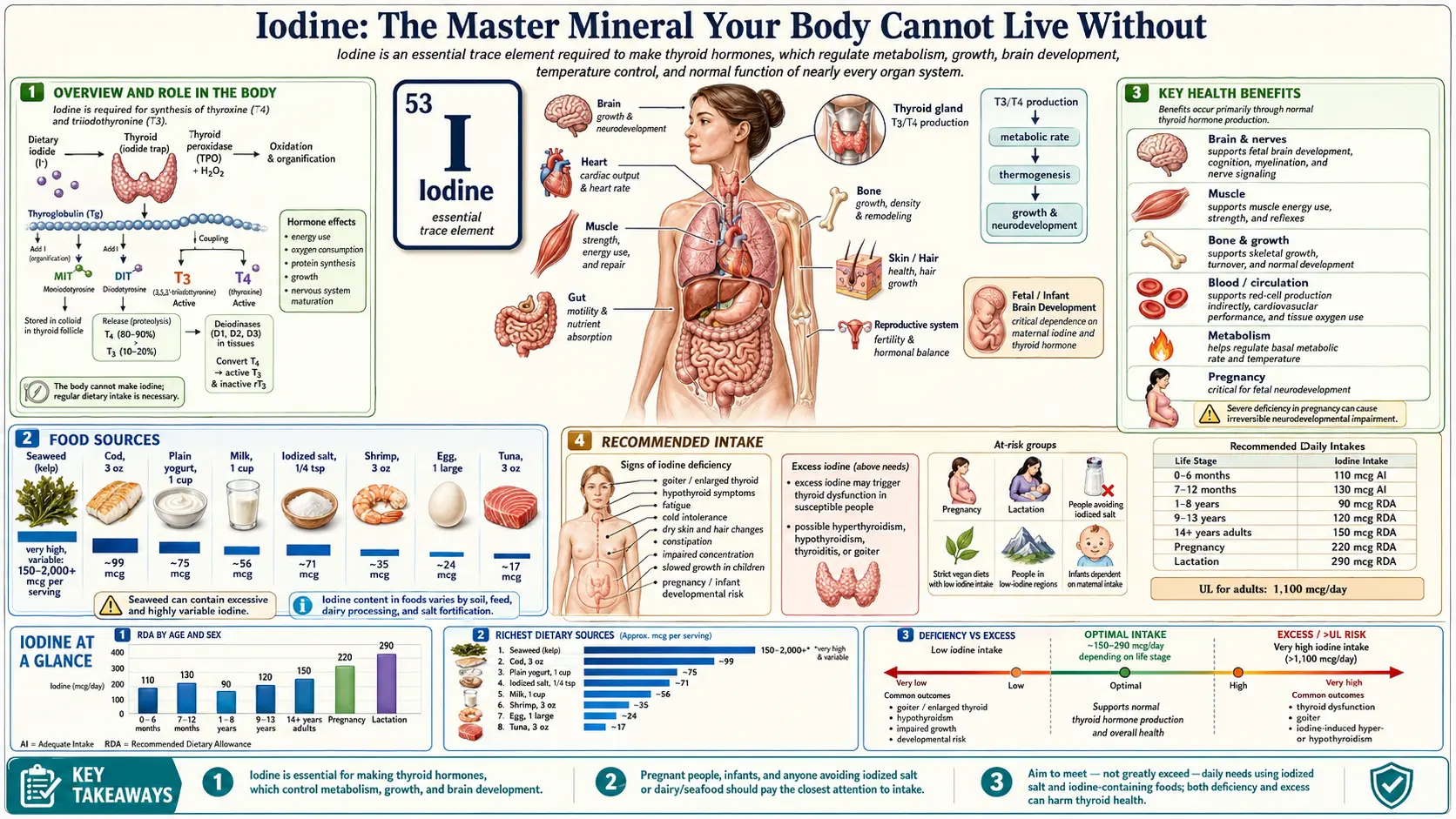
3. Biochemistry of Iodine in the Human Body
To understand why iodine is so essential, we must first understand how the body uses it at the biochemical level. Iodine exists in the body in two primary forms: iodide (I⁻), the reduced ionic form, and molecular iodine (I₂), the elemental form. These two forms are not interchangeable in their biological activity, and different tissues preferentially utilize different forms. The thyroid gland primarily uses iodide, which it actively concentrates through the sodium-iodide symporter (NIS) — a specialized transport protein embedded in the thyroid cell membrane. Breast tissue, the prostate, the gastric mucosa, and other extrathyroidal tissues appear to preferentially utilize molecular iodine (I₂), though they can also take up iodide.
The total body content of iodine in a sufficiently nourished individual is estimated to be between 25 and 50 milligrams, though some researchers, including Dr. Guy Abraham and Dr. David Brownstein, have argued that the optimal whole-body iodine sufficiency level may be significantly higher — possibly in the range of 1,500 milligrams (1.5 grams) when all tissues are fully saturated. This stands in stark contrast to the current Recommended Daily Allowance (RDA) of just 150 micrograms per day, which was established primarily to prevent goiter and does not address whole-body iodine sufficiency.
The distribution of iodine throughout the body is remarkable in its breadth. The thyroid gland contains the highest concentration, holding approximately 15 to 20 milligrams in a sufficiency state. But iodine is also concentrated in the salivary glands, the gastric mucosa, the mammary glands, the ovaries, the uterus, the cervix, the prostate, the choroid plexus of the brain, the ciliary body of the eyes, the skin, the lacrimal glands, and the thymus. This widespread distribution tells us something profoundly important: iodine is not merely a "thyroid nutrient." It is a whole-body nutrient with diverse and critical functions in virtually every organ system.
At the cellular level, iodine participates in several key processes. It is required for the synthesis of thyroid hormones (T4 and T3), which regulate basal metabolic rate, protein synthesis, and cellular oxygen consumption. It functions as an antioxidant, particularly in its molecular form (I₂), where it can directly scavenge reactive oxygen species and lipid peroxides. It is involved in the regulation of apoptosis — programmed cell death — a critical process for eliminating damaged, precancerous, or abnormal cells. It modulates immune function, influences gene expression through its effects on thyroid hormones and through direct genomic effects, and serves as a natural antimicrobial agent that the body uses as a first line of defense against pathogens in mucosal tissues.
4. Iodine and the Thyroid Gland — The Foundation of Metabolism
The thyroid gland's relationship with iodine is perhaps the most well-understood aspect of iodine physiology, and for good reason — the thyroid is the body's largest reservoir of iodine and the gland most immediately affected by iodine status. The thyroid produces two primary hormones: thyroxine (T4), which contains four iodine atoms, and triiodothyronine (T3), which contains three. T4 is the predominant circulating thyroid hormone, but T3 is the biologically active form that exerts effects at the cellular level. The conversion of T4 to T3 occurs primarily in the liver, kidneys, and peripheral tissues, and requires the selenium-dependent deiodinase enzymes.
The process of thyroid hormone synthesis begins when the sodium-iodide symporter (NIS) actively transports iodide from the bloodstream into the thyroid follicular cells, concentrating it to levels 20 to 40 times higher than in the plasma. This concentrated iodide is then transported to the apical surface of the cell, where the enzyme thyroid peroxidase (TPO) oxidizes it and incorporates it into tyrosine residues on the thyroglobulin protein. The coupling of iodinated tyrosine residues produces T4 and T3, which are stored in the colloid of the thyroid follicle and released into the bloodstream as needed.
When iodine is insufficient, the thyroid gland undergoes a series of adaptive responses that, over time, become pathological. Initially, the gland increases its avidity for iodide through upregulation of the NIS, and it shifts production from T4 toward T3 to conserve iodine (since T3 requires one fewer iodine atom). If deficiency persists, the thyroid begins to enlarge — a condition known as goiter — as the gland hypertrophies in an attempt to trap more iodine. Eventually, the gland may become nodular, developing autonomous areas that can produce thyroid hormone independent of TSH regulation, and in some cases, these nodules can become malignant.
From a clinical perspective, optimizing iodine status is foundational to supporting healthy thyroid function. In my practice, I consistently find that patients who present with symptoms of hypothyroidism — fatigue, weight gain, cold intolerance, constipation, dry skin, thinning hair, depression, brain fog, and menstrual irregularities — often have suboptimal iodine levels, even when standard thyroid panels (TSH, free T4) appear within the so-called "normal" range. Addressing the iodine deficit, along with supporting cofactors like selenium, zinc, and tyrosine, frequently produces meaningful clinical improvement.
5. Iodine Deficiency, Hypothyroidism, and Hashimoto's Thyroiditis
The relationship between iodine and autoimmune thyroid disease — particularly Hashimoto's thyroiditis — is one of the most controversial topics in iodine medicine, and it deserves careful, nuanced discussion. Hashimoto's thyroiditis is an autoimmune condition in which the immune system produces antibodies against thyroid peroxidase (TPO) and thyroglobulin (Tg), leading to chronic inflammation, gradual destruction of thyroid tissue, and eventual hypothyroidism. It is the most common cause of hypothyroidism in the developed world, and its prevalence has been rising steadily.
The conventional medical position has generally been that iodine supplementation should be avoided in Hashimoto's, based on epidemiological observations that thyroid autoantibody prevalence tends to increase in populations after iodine fortification programs are introduced. Several studies, particularly those from China and Sri Lanka, have shown that aggressive iodine supplementation in previously iodine-deficient populations is associated with an uptick in Hashimoto's diagnoses. This has led many endocrinologists to advise Hashimoto's patients to restrict iodine intake.
However, a growing body of integrative and naturopathic clinical experience, supported by research from clinicians like Dr. David Brownstein, Dr. Jorge Flechas, and Dr. Guy Abraham, suggests that this picture is incomplete. These practitioners argue that the observed increase in thyroid autoimmunity following iodine supplementation is not caused by iodine itself, but by the absence of adequate selenium and the failure to address the oxidative stress that iodine utilization generates. When iodine is incorporated into thyroglobulin by thyroid peroxidase, the process generates hydrogen peroxide (H₂O₂) as a necessary oxidant. If the body's antioxidant defenses — particularly the selenium-dependent glutathione peroxidase system — are insufficient, this hydrogen peroxide can damage thyroid cells and trigger an autoimmune response.
The clinical implication is that iodine and selenium should never be considered in isolation. Adequate selenium status is a prerequisite for safe iodine supplementation, particularly in individuals with existing thyroid autoimmunity. In practice, I typically ensure that patients are taking 200 to 400 micrograms of selenium (as selenomethionine or a mixed selenium supplement) before introducing or increasing iodine. I also monitor thyroid antibody levels and inflammatory markers closely. In most cases, when this protocol is followed, Hashimoto's patients not only tolerate iodine well — they often experience significant clinical improvement, including reductions in TPO antibodies over time.
It is also worth noting that iodine deficiency itself may be a contributing factor to thyroid autoimmunity. An iodine-starved thyroid is a stressed thyroid — it is working harder, generating more oxidative stress relative to its antioxidant capacity, and may be more vulnerable to immune dysregulation. The premise that iodine restriction is inherently protective in Hashimoto's has not been supported by prospective, randomized clinical trials, and the clinical evidence from practitioners who use iodine judiciously in this population suggests that thoughtful supplementation, with appropriate cofactors and monitoring, can be highly beneficial.
6. Iodine and Breast Health
One of the most important and underappreciated roles of iodine in the human body is its function in breast tissue. The mammary glands are among the body's most avid concentrators of iodine, second only to the thyroid gland, and this concentration increases dramatically during pregnancy and lactation. The breast tissue contains sodium-iodide symporters (NIS) and a separate transport mechanism called pendrin, and the lactating breast can concentrate iodine to levels sufficient to deliver approximately 75 to 200 micrograms per day to the nursing infant — indicating the critical importance of iodine for infant development and the evolutionary significance of breast tissue as an iodine delivery system.
Research dating back to the 1960s and 1970s by Dr. Bernard Eskin demonstrated that iodine deficiency in animal models produced histological changes in breast tissue that closely resemble the progression from fibrocystic changes to dysplasia to neoplasia in humans. Eskin's work showed that rats fed iodine-deficient diets developed fibrocystic breast disease, atypical ductal hyperplasia, and eventually mammary tumors — and that these changes could be reversed or prevented by iodine supplementation, particularly with molecular iodine (I₂).
In clinical practice, the relationship between iodine and fibrocystic breast disease (FBD) is one of the most consistently reproducible findings in iodine medicine. Fibrocystic breast disease — characterized by painful, lumpy breasts that worsen cyclically with menstruation — affects an estimated 50 to 90% of women at some point in their lives. Multiple clinical studies, including randomized controlled trials by Ghent et al. published in the Canadian Journal of Surgery (1993), have demonstrated that molecular iodine supplementation significantly reduces breast pain and nodularity in women with FBD. In the Ghent study, over 70% of women receiving molecular iodine experienced clinically significant improvement.
The mechanism by which iodine protects breast tissue appears to be multifaceted. Molecular iodine acts as a direct antioxidant in breast tissue, neutralizing lipid peroxidation products that can damage cellular DNA. It also promotes apoptosis — the orderly, programmed death of abnormal cells — through effects on the mitochondrial pathway and through modulation of peroxisome proliferator-activated receptors (PPARs). Additionally, iodine appears to have anti-estrogenic effects in breast tissue, reducing the expression of estrogen-responsive genes and modulating the estrogen receptor itself. This is particularly significant because estrogen dominance is a major driver of both fibrocystic changes and breast cancer risk.
For women's health practitioners, the message is clear: iodine is not optional for breast health. It is essential. Every woman presenting with breast pain, fibrocystic changes, or breast-related concerns should have her iodine status assessed, and iodine repletion should be a foundational component of any integrative breast health protocol.
7. Iodine and Reproductive Health — Fertility, Pregnancy, and Fetal Development
Iodine's role in reproductive health cannot be overstated. Adequate iodine is essential for fertility in both men and women, for the health of pregnancy, for fetal and neonatal development, and for successful lactation. The consequences of iodine deficiency during the reproductive years are among the most devastating of all iodine-related disorders, because they affect not only the individual but the next generation.
In women, iodine deficiency is associated with menstrual irregularities, anovulation, infertility, and increased risk of miscarriage and stillbirth. The ovaries contain the second-highest concentration of iodine after the thyroid, and iodine appears to play a role in follicular maturation, progesterone production, and the regulation of estrogen metabolism. Women with subclinical hypothyroidism due to iodine deficiency frequently experience luteal phase defects, which compromise embryo implantation and early pregnancy maintenance. In my clinical experience, addressing iodine insufficiency in women with unexplained infertility or recurrent pregnancy loss frequently results in successful conception and improved pregnancy outcomes.
In men, iodine deficiency can impair testosterone synthesis, reduce sperm count and motility, and contribute to erectile dysfunction. The prostate gland, like the breast, concentrates iodine through the sodium-iodide symporter, and there is emerging evidence that iodine plays a role in prostate health and the prevention of benign prostatic hyperplasia (BPH) and prostate cancer.
During pregnancy, iodine requirements increase by approximately 50%, from a baseline of 150 micrograms per day to at least 220 to 250 micrograms per day according to the American Thyroid Association and the WHO. This increase reflects the mother's need to maintain her own thyroid function while simultaneously supplying iodine to the fetus, which relies entirely on maternal iodine for thyroid hormone synthesis during the first trimester (before the fetal thyroid becomes functional at approximately 12 weeks of gestation). Maternal hypothyroidism and iodine deficiency during this critical window are associated with irreversible neurological damage in the offspring, including reduced IQ, impaired motor function, hearing deficits, and in severe cases, the constellation of physical and intellectual disabilities known as cretinism.
Alarmingly, recent studies suggest that a substantial proportion of pregnant women in developed countries — including the United States, the United Kingdom, and Australia — are iodine-insufficient. The Avon Longitudinal Study of Parents and Children (ALSPAC) in the UK found that children of mothers with urinary iodine levels below 150 micrograms per liter during pregnancy had significantly lower IQ scores at age 8, even in the absence of clinical hypothyroidism. These findings underscore the critical importance of ensuring adequate iodine intake before and during pregnancy, and during lactation.
I strongly recommend that all women of childbearing age optimize their iodine status as part of preconception care. Prenatal vitamins should contain at least 150 micrograms of iodine (as potassium iodide), and many integrative practitioners, including myself, recommend higher doses based on individual assessment and monitoring. Waiting until a pregnancy is confirmed to address iodine deficiency is too late — the most critical period of fetal brain development occurs in the first trimester, often before a woman even knows she is pregnant.
8. Iodine and Brain Health — Cognition, IQ, and Neurological Development
The relationship between iodine and brain function is one of the most well-established and most consequential in all of nutrition science. Iodine, through its essential role in thyroid hormone production, is a fundamental requirement for normal brain development, myelination of neural pathways, synaptic formation, neurotransmitter synthesis, and cognitive function throughout the lifespan.
In the developing fetus and young child, the impact of iodine deficiency on brain development is devastating and largely irreversible. Thyroid hormones, which require iodine for their synthesis, regulate the expression of hundreds of genes involved in neuronal migration, differentiation, and myelination during critical windows of brain development. Severe iodine deficiency during pregnancy results in cretinism — a condition characterized by profound intellectual disability, deaf-mutism, motor spasticity, and growth retardation. Moderate iodine deficiency, which is far more common, produces subtler but still significant effects: reduced IQ (estimated at 10 to 15 points on a population level), impaired memory and processing speed, attention deficits, and learning disabilities.
The WHO estimates that iodine deficiency results in a loss of approximately 10 to 15 IQ points on average in affected populations, making it the leading preventable cause of intellectual disability worldwide. A meta-analysis published in 2005 by Qian et al. analyzed 37 studies from China and found that children in iodine-sufficient areas had IQ scores averaging 12.45 points higher than children in iodine-deficient areas. These are enormous, population-level effects with profound implications for educational attainment, economic productivity, and societal wellbeing.
But iodine's importance for brain health does not end in childhood. In adults, suboptimal iodine status contributes to brain fog, poor concentration, memory impairment, slow processing speed, depression, and anxiety — symptoms that are often attributed to "stress" or "aging" but that may in fact reflect subclinical hypothyroidism secondary to iodine insufficiency. The choroid plexus, which produces cerebrospinal fluid, actively concentrates iodine, and there is evidence that iodine may have direct neuroprotective effects independent of thyroid hormone production, including antioxidant activity and modulation of inflammatory pathways in the central nervous system.
In the aging brain, thyroid hormone status — and by extension, iodine status — is increasingly recognized as a factor in cognitive decline and dementia risk. Several large prospective studies have found that both overt and subclinical hypothyroidism are associated with increased risk of Alzheimer's disease and other forms of dementia. While the relationship is complex and multifactorial, ensuring adequate iodine intake throughout life represents a foundational strategy for preserving cognitive function.
9. Iodine and Immune System Function
Iodine plays a multifaceted role in immune defense that extends well beyond its effects on thyroid hormone production. The immune system depends on adequate thyroid function for its baseline activity — thyroid hormones stimulate the production, maturation, and activity of immune cells, including T lymphocytes, natural killer (NK) cells, and macrophages. Hypothyroidism, whether overt or subclinical, is consistently associated with impaired immune function and increased susceptibility to infections.
But iodine also has direct, thyroid-independent effects on immune function. White blood cells — particularly neutrophils and monocytes — use iodine as a critical component of their antimicrobial arsenal. When neutrophils engulf a pathogen through phagocytosis, they generate a "respiratory burst" of reactive oxygen species to kill the invader. This process involves the enzyme myeloperoxidase (MPO), which uses iodide (along with chloride) to generate hypoiodous acid (HOI) — a potent antimicrobial oxidant that is more effective and less tissue-damaging than hypochlorous acid (HOCl, the chloride-derived oxidant). In other words, when iodine is available, the immune system's killing mechanism is both more effective and less inflammatory.
The thymus gland, which is the master gland of adaptive immunity and responsible for the maturation and education of T lymphocytes, is an iodine-concentrating tissue. Iodine deficiency may contribute to thymic atrophy and impaired T cell function — a connection that has significant implications for autoimmunity, chronic infection, and cancer immunosurveillance.
In mucosal tissues — the gastrointestinal tract, the respiratory tract, the salivary glands, and the genitourinary tract — iodine serves as a first-line antimicrobial defense. The salivary glands concentrate iodide and secrete it into saliva, where the enzyme salivary peroxidase converts it to hypoiodous acid, providing continuous antimicrobial protection in the oral cavity. The gastric mucosa similarly concentrates iodide and secretes it into gastric juice, contributing to the stomach's barrier function against ingested pathogens. The lactoperoxidase system in breast milk uses iodide to generate antimicrobial compounds that protect the nursing infant from gastrointestinal infections.
Clinically, I have observed that patients with recurrent infections — sinusitis, urinary tract infections, respiratory infections, and oral/dental infections — frequently have suboptimal iodine levels. Addressing iodine insufficiency in these patients, as part of a comprehensive immune support protocol, often leads to a meaningful reduction in the frequency and severity of infections.
10. Iodine as a Detoxification Agent — Halide Displacement and Heavy Metals
One of iodine's most important and clinically relevant functions is its role in detoxification — specifically, its ability to displace toxic halides and support the body's overall detoxification capacity. This aspect of iodine physiology has enormous implications in our modern, chemically saturated environment.
Iodine belongs to the halogen group on the periodic table, which also includes fluorine, chlorine, bromine, and astatine. These elements share similar chemical properties, and in the body, the toxic halides — fluoride, chloride, and particularly bromide — can compete with iodide for uptake by the sodium-iodide symporter (NIS) and other transport mechanisms. When iodine is deficient, bromide and fluoride accumulate in tissues that would normally concentrate iodine — the thyroid, the breast, the prostate, the ovaries, the skin — and interfere with iodine's normal biological functions.
Bromide exposure in the modern environment is pervasive. It is present in brominated vegetable oils (used in some soft drinks), brominated flame retardants (found in furniture, electronics, textiles, and automobile interiors), methyl bromide (a pesticide used in agriculture), and potassium bromate (a dough conditioner used in commercial baking). Bromide has no known essential biological function and is classified as a toxic substance. It competes directly with iodide for thyroid uptake, disrupts thyroid hormone synthesis, and has been associated with neurological symptoms, skin disorders, and increased cancer risk.
Fluoride, another toxic halide, is ubiquitous in public water supplies, dental products, certain medications (including some fluoroquinolone antibiotics and the antidepressant fluoxetine), and industrial emissions. Fluoride inhibits thyroid function, interferes with iodine uptake, and has been associated with neurodevelopmental toxicity, skeletal fluorosis, and endocrine disruption.
Iodine supplementation — particularly at doses above the minimal RDA — promotes the displacement and excretion of bromide and fluoride from the body through a process sometimes called "halide displacement" or "detox reaction." When patients begin iodine supplementation, they frequently experience temporary symptoms — skin rashes, acne, metallic taste, headache, irritability, sinus congestion, and gastrointestinal disturbance — that are attributed to the mobilization and excretion of stored bromide. These symptoms typically resolve within days to weeks and can be mitigated by adequate salt and water intake (which supports renal bromide excretion), vitamin C, and a gradual dose escalation approach.
Beyond halide displacement, iodine may also support the excretion of heavy metals, including mercury, lead, cadmium, and aluminum, though the evidence for this is less robust than for halide displacement. Some practitioners report that urinary heavy metal excretion increases following iodine loading, and there is mechanistic plausibility for iodine's role in chelation-like processes, but more research is needed to clarify these effects.
11. Iodine and Cancer Prevention — Emerging Research
The relationship between iodine and cancer is an area of active and fascinating research. Epidemiological, laboratory, and clinical evidence collectively suggest that iodine — particularly molecular iodine (I₂) — may play a significant role in the prevention and possibly the treatment of several types of cancer, most notably breast cancer, thyroid cancer, gastric cancer, and prostate cancer.
The epidemiological evidence is compelling. Japan, where dietary iodine intake is among the highest in the world (estimated at 1,000 to 13,000 micrograms per day from seaweed and seafood), has historically had among the lowest rates of breast cancer, prostate cancer, and endometrial cancer globally. When Japanese women migrate to Western countries and adopt Western diets with lower iodine content, their breast cancer rates rise to match those of the host country within one to two generations. While many dietary and lifestyle factors differ between Japan and Western nations, the iodine connection is biologically plausible and epidemiologically consistent.
At the cellular level, iodine's anticancer mechanisms are increasingly well characterized. Molecular iodine (I₂) has been shown to induce apoptosis in human breast cancer cell lines (MCF-7 and MDA-MB-231) through mitochondria-mediated pathways. It inhibits proliferation and induces differentiation of cancer cells. It reduces the expression of vascular endothelial growth factor (VEGF), thereby inhibiting the angiogenesis that tumors require for growth and metastasis. Iodine also modulates the expression of estrogen receptor-alpha and reduces estrogen-responsive gene transcription, which is particularly relevant in hormone-receptor-positive breast cancers.
Research by Aceves, Anguiano, and Delgado at the National Autonomous University of Mexico (UNAM) has been particularly illuminating. Their work has demonstrated that molecular iodine, administered as a dietary supplement, has antineoplastic effects in animal models of breast cancer and in human breast cancer cell cultures. They have identified iodolactones — iodinated lipid metabolites formed when iodine interacts with arachidonic acid — as key mediators of iodine's anticancer effects. These iodolactones activate PPAR-gamma receptors, promote apoptosis, and inhibit proliferation in a mechanism that is independent of thyroid function.
Thyroid cancer rates have been increasing dramatically worldwide, and while some of this increase is attributed to improved detection (through widespread ultrasonography), there is growing concern that genuine increases in incidence are also occurring. The role of iodine deficiency in thyroid cancer is complex: severe iodine deficiency is associated with increased risk of follicular thyroid cancer (the more aggressive subtype), while iodine sufficiency shifts the distribution toward papillary thyroid cancer (which has a more favorable prognosis). Some researchers have suggested that the pattern of iodine exposure — cycling between deficiency and excess, or rapid repletion after prolonged deficiency — may itself be a risk factor, further arguing for stable, consistent iodine adequacy throughout life.
Gastric cancer, which remains one of the leading causes of cancer death worldwide, is also associated with iodine deficiency. The gastric mucosa concentrates iodine through the NIS, and iodine appears to protect gastric epithelial cells through antioxidant, anti-inflammatory, and antimicrobial mechanisms (including activity against Helicobacter pylori). Regions with low soil iodine consistently show higher rates of gastric cancer.
12. Iodine and Skin, Hair, and Nail Health
The skin is the body's largest organ and one of its most visible indicators of overall health. Iodine plays an important role in maintaining skin integrity, promoting wound healing, and supporting the health of hair and nails. The skin concentrates iodine and contains both the NIS transporter and the enzyme iodothyronine deiodinase, indicating that it is both an iodine-utilizing and thyroid-hormone-responsive tissue.
Iodine deficiency is associated with dry, rough, and flaky skin — a classic symptom of hypothyroidism. Thyroid hormones stimulate the production and turnover of skin cells, regulate sebaceous gland function, and maintain the skin's moisture barrier. When thyroid function is compromised by iodine deficiency, the skin becomes dry, thick, and waxy — a condition known as myxedema in its severe form. Hair becomes brittle, thin, and prone to excessive shedding. Nails become ridged, brittle, and slow-growing. The outer third of the eyebrows may thin or disappear entirely — a classic clinical sign of hypothyroidism known as the "Queen Anne sign."
Beyond its thyroid-mediated effects, iodine has direct benefits for skin health. Topical iodine has been used for centuries as an antiseptic and wound-healing agent. Povidone-iodine (Betadine) remains one of the most widely used surgical antiseptics worldwide. In naturopathic practice, dilute Lugol's solution or povidone-iodine can be applied topically to support wound healing, treat fungal infections, and manage certain inflammatory skin conditions.
Interestingly, some patients experience a transient worsening of skin symptoms — including acne, rashes, and pustular eruptions — when they first begin iodine supplementation. This is frequently interpreted as a "bromide detoxification reaction" or "bromaderma," as the mobilization and excretion of stored bromide through the skin can produce inflammatory skin lesions. These reactions are typically self-limiting and resolve as bromide stores are depleted and iodine status normalizes.
13. Iodine and Cardiovascular Health
The cardiovascular system is exquisitely sensitive to thyroid hormone status, and by extension, to iodine status. Thyroid hormones directly regulate heart rate, cardiac contractility, systemic vascular resistance, and cholesterol metabolism. Both overt and subclinical hypothyroidism are well-established risk factors for cardiovascular disease, and iodine deficiency is a common underlying cause of subclinical hypothyroidism.
Hypothyroidism — even when mild or subclinical — is associated with elevated LDL cholesterol, elevated triglycerides, increased homocysteine levels, increased C-reactive protein (a marker of systemic inflammation), endothelial dysfunction, increased arterial stiffness, and diastolic hypertension. Over time, these metabolic disturbances contribute to accelerated atherosclerosis and increased risk of coronary artery disease, heart failure, and stroke. Large population studies, including the Rotterdam Study and the Whickham Survey follow-up, have demonstrated increased cardiovascular morbidity and mortality in individuals with subclinical hypothyroidism.
Iodine also appears to have direct cardiovascular effects that may be independent of thyroid function. Iodide has been shown to relax vascular smooth muscle and reduce blood pressure in animal models. Some clinical practitioners report that iodine supplementation improves blood pressure control in their patients, particularly those with resistant hypertension that has been refractory to standard pharmaceutical interventions. While these observations require further investigation in controlled clinical trials, they are consistent with iodine's known vascular and anti-inflammatory effects.
Additionally, iodine's role in displacing bromide may have indirect cardiovascular benefits. Bromide, which accumulates in iodine-deficient individuals, has been associated with cardiac arrhythmias and neurological symptoms. By promoting bromide excretion, iodine supplementation may reduce the toxic cardiovascular burden of this environmental halide.
14. Iodine, Metabolic Syndrome, and Weight Management
Thyroid hormones are the master regulators of basal metabolic rate — the rate at which the body burns calories at rest. When iodine deficiency compromises thyroid function, metabolic rate declines, and the body shifts into an energy-conservation mode that promotes fat storage, fluid retention, and weight gain. This is one of the most frustrating and demoralizing consequences of iodine deficiency for patients, who often find that despite diligent dieting and exercise, they cannot lose weight or maintain weight loss.
Subclinical hypothyroidism secondary to iodine deficiency is associated with many features of metabolic syndrome, including central obesity, insulin resistance, dyslipidemia, and elevated inflammatory markers. Addressing the underlying iodine deficit and restoring optimal thyroid function can produce meaningful improvements in metabolic parameters — weight loss, improved insulin sensitivity, reduced cholesterol and triglycerides, and enhanced energy and exercise tolerance.
It is important to emphasize that iodine is not a "weight loss supplement" in the conventional sense. It will not produce weight loss in individuals who are already iodine-sufficient and euthyroid. However, for the substantial proportion of the population that is iodine-insufficient and subclinically hypothyroid, iodine repletion can remove a major metabolic obstacle and allow the body to respond normally to appropriate diet and exercise interventions. In my practice, I consistently find that optimizing iodine and thyroid status is one of the most important early steps in any comprehensive metabolic rehabilitation program.
15. Iodine and Gastrointestinal Health
The gastrointestinal tract is a significant iodine-concentrating tissue, and iodine plays several important roles in digestive health. The gastric mucosa actively transports iodide through the NIS and secretes it into gastric juice, where it contributes to the antimicrobial barrier of the stomach. This iodide, in combination with gastric peroxidase, generates antimicrobial iodinated compounds that help prevent infection by ingested pathogens, including bacteria, viruses, and parasites.
Iodine deficiency may compromise the stomach's barrier function and increase susceptibility to Helicobacter pylori infection — the primary cause of chronic gastritis, peptic ulcer disease, and gastric cancer. As mentioned earlier, regions with low soil iodine consistently show higher rates of gastric cancer, and iodine's antimicrobial and antioxidant effects in the gastric mucosa likely contribute to this epidemiological pattern.
Thyroid hormones, which require iodine for their synthesis, are also critical regulators of gastrointestinal motility. Hypothyroidism is one of the most common causes of chronic constipation, and many patients with iodine-deficiency-related hypothyroidism experience sluggish digestion, bloating, abdominal discomfort, and irregular bowel habits. Restoring adequate iodine and thyroid hormone levels typically normalizes gastrointestinal motility and resolves these symptoms.
The salivary glands also concentrate iodine and secrete it into saliva, where salivary peroxidase uses it to generate hypoiodous acid — a potent antimicrobial agent that protects the oral cavity from pathogenic bacteria and fungi. Iodine insufficiency may therefore contribute to increased susceptibility to dental caries, periodontal disease, and oral candidiasis. This is another often-overlooked connection that underscores the breadth of iodine's influence on human health.
16. Iodine and Eye Health
The eyes contain specialized structures that concentrate iodine, particularly the ciliary body and the lacrimal (tear) glands. The ciliary body, which produces aqueous humor and controls the shape of the lens, contains the NIS transporter and actively concentrates iodide. The lacrimal glands secrete iodide into tears, where it contributes to the antimicrobial defense of the ocular surface.
Iodine deficiency may contribute to dry eye syndrome, a condition characterized by inadequate tear production or poor tear quality, leading to ocular irritation, redness, and visual disturbance. While dry eye has many causes (including autoimmune disease, medication side effects, and environmental factors), the role of iodine deficiency is increasingly recognized, and some practitioners report improvement in dry eye symptoms following iodine supplementation.
Hypothyroidism, a common consequence of iodine deficiency, is associated with several eye-related conditions, including periorbital edema (puffiness around the eyes), blepharitis (eyelid inflammation), and corneal changes. While the most dramatic ocular manifestation of thyroid disease — Graves' ophthalmopathy — occurs in the context of hyperthyroidism and Graves' disease, maintaining optimal iodine and thyroid status is important for overall eye health and function.
There is also preliminary research suggesting that iodine may play a role in the prevention of cataracts, through its antioxidant effects in the lens. The lens is highly susceptible to oxidative damage, and the accumulation of oxidized proteins is a primary mechanism of cataract formation. Iodine's ability to scavenge reactive oxygen species and reduce lipid peroxidation may contribute to lens protection, though more research is needed in this area.
17. Iodine as a Natural Antimicrobial Agent
Iodine is one of the oldest and most effective antimicrobial agents known to medicine. It possesses broad-spectrum activity against bacteria (both gram-positive and gram-negative), viruses (both enveloped and non-enveloped), fungi, protozoa, mycobacteria, and even bacterial spores. Remarkably, in over 150 years of clinical use, no significant microbial resistance to iodine has been documented — a stark contrast to the growing crisis of antibiotic resistance that threatens modern medicine.
The mechanism of iodine's antimicrobial action is fundamentally different from that of antibiotics. Iodine acts as a powerful oxidant that disrupts microbial cell membranes, denatures proteins, and inactivates enzymes and nucleic acids. Because it attacks multiple targets simultaneously, rather than a single metabolic pathway, the development of resistance is extremely unlikely. This makes iodine an invaluable tool in an era of increasing antimicrobial resistance.
Externally, iodine-based antiseptics — including povidone-iodine (Betadine), Lugol's solution, and cadexomer iodine — are used in wound care, surgical preparation, burn treatment, and the management of chronic and infected wounds. Povidone-iodine has been shown to be effective against methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant Enterococcus (VRE), and other multidrug-resistant organisms.
Internally, iodine's antimicrobial role is mediated through the peroxidase systems described earlier — particularly the salivary peroxidase system (protecting the oral cavity), the gastric peroxidase system (protecting the stomach), the lactoperoxidase system (protecting the nursing infant), and the myeloperoxidase system (used by white blood cells to kill phagocytosed pathogens). Ensuring adequate iodine availability for these systems is a foundational strategy for innate immune defense.
In the context of respiratory infections, including influenza and other viral illnesses, there has been considerable interest in the use of povidone-iodine nasal sprays and throat gargles as prophylactic and therapeutic measures. Laboratory studies have demonstrated potent virucidal activity of povidone-iodine against a wide range of respiratory viruses, and clinical trials exploring its use in infection prevention are ongoing.
18. Recognizing Iodine Deficiency — Clinical Signs and Symptoms
Iodine deficiency presents along a spectrum of severity, from subtle, subclinical manifestations to severe, overt disease. In clinical practice, the vast majority of iodine-insufficient patients present with nonspecific symptoms that can be easily attributed to other causes — which is precisely why iodine deficiency is so frequently overlooked. Recognizing the pattern is key to clinical detection.
The most common signs and symptoms of iodine deficiency include: persistent fatigue and low energy that is not explained by sleep deprivation or other obvious causes; unexplained weight gain or inability to lose weight despite appropriate diet and exercise; cold intolerance and a tendency to feel cold when others are comfortable; dry, rough, or flaky skin; thinning, brittle hair with excessive shedding; hair loss from the outer third of the eyebrows; brittle, ridged, or slow-growing nails; constipation and sluggish digestion; brain fog, poor concentration, and memory impairment; depression, anxiety, or mood instability; menstrual irregularities (heavy periods, irregular cycles, or anovulation); infertility or difficulty conceiving; fibrocystic breast changes (painful, lumpy breasts); swelling or fullness in the neck (goiter); puffy face, hands, or feet (myxedema); hoarse or deep voice; slow heart rate (bradycardia); elevated cholesterol; recurrent infections; and muscle weakness or joint pain.
In children and adolescents, iodine deficiency may present as poor academic performance, difficulty concentrating, behavioral problems, delayed growth and development, and delayed puberty. In infants and young children, severe deficiency produces developmental delay, intellectual disability, and in extreme cases, cretinism.
In clinical practice, I find it helpful to think of iodine deficiency not as a single condition but as a "whole-body insufficiency syndrome" that can manifest differently depending on which tissues are most affected and which compensatory mechanisms the body has engaged. A patient may present primarily with thyroid symptoms, while another presents primarily with breast symptoms, and another primarily with cognitive or immune symptoms. Maintaining a high clinical index of suspicion and routinely assessing iodine status is essential for identifying these patients.
19. Testing for Iodine Status
Accurate assessment of iodine status is essential for guiding clinical interventions. Several methods are available, each with its own strengths and limitations.
Urinary Iodine Concentration (Spot Urine): This is the most widely used method for assessing iodine status in epidemiological studies and population surveys. It measures the concentration of iodine in a single urine sample. Because more than 90% of ingested iodine is eventually excreted in the urine, urinary iodine reflects recent dietary intake. However, spot urine values fluctuate significantly based on hydration status and recent dietary intake, so a single measurement may not accurately represent an individual's long-term iodine status. The WHO considers a median urinary iodine concentration of 100 to 199 micrograms per liter as adequate for a population (but not necessarily for an individual).
24-Hour Urinary Iodine Excretion: This is a more reliable measure of individual iodine status than spot urine, as it captures the total amount of iodine excreted over a full 24-hour period, reducing the variability associated with hydration and timing. It provides a more accurate reflection of habitual dietary iodine intake and is useful for monitoring individual patients in clinical practice.
Iodine Loading Test: Developed by Dr. Guy Abraham, this test involves administering a standardized dose of iodine (typically 50 mg of iodine/iodide as Iodoral) and collecting urine for 24 hours to measure the percentage of the dose that is excreted. The premise is that an iodine-sufficient body will excrete approximately 90% or more of the loading dose, while an iodine-deficient body will retain a larger proportion to replenish depleted tissue stores. An excretion rate below 90% suggests whole-body iodine deficiency. This test has been widely used in integrative and naturopathic practice, though it has not been widely adopted in conventional medicine and has some methodological limitations that should be considered.
Serum Iodine: Blood levels of iodine can be measured but are less commonly used in clinical practice because they primarily reflect very recent intake and do not reliably indicate tissue stores or whole-body iodine status.
Thyroglobulin (Tg): Serum thyroglobulin is increasingly recognized as a sensitive biomarker of iodine status. Thyroglobulin levels rise in response to iodine deficiency (as the thyroid gland increases its production and turnover of thyroglobulin in an attempt to capture more iodine) and fall when iodine status is normalized. A thyroglobulin level below 10 micrograms per liter is generally considered indicative of iodine sufficiency.
In my clinical practice, I typically use a combination of spot or 24-hour urinary iodine, thyroid function tests (TSH, free T4, free T3), thyroid antibodies (TPO, Tg antibodies), and clinical symptom assessment to evaluate iodine status and guide supplementation. The iodine loading test can provide additional information in complex cases or when there is a clinical suspicion of significant whole-body depletion.
20. Dietary and Supplemental Sources of Iodine
Iodine is found in a limited number of foods, and the iodine content of most foods varies widely depending on the soil and water iodine content of the region where they are produced. This geographic variability is a major reason why iodine deficiency has been such a persistent global problem.
Sea Vegetables (Seaweed): These are by far the richest dietary sources of iodine. Kelp (kombu) contains the highest concentrations, typically ranging from 500 to 8,000 micrograms per gram of dry weight. Wakame, nori, arame, hijiki, and dulse are other commonly consumed seaweeds with significant iodine content, though generally lower than kelp. The high seaweed consumption in Japan is a primary reason for that population's high iodine intake. It is worth noting that the iodine content of seaweed can vary considerably between species, harvest locations, and preparation methods, so testing or careful sourcing is important for individuals who rely on seaweed as their primary iodine source.
Fish and Seafood: Cod, tuna, shrimp, and other seafood contain moderate amounts of iodine, typically in the range of 20 to 120 micrograms per serving. Regular consumption of seafood contributes meaningfully to iodine intake, though it is rarely sufficient on its own to achieve optimal iodine status.
Dairy Products: Milk, yogurt, and cheese are significant sources of iodine in Western diets, primarily due to the use of iodine-containing feed supplements in dairy cattle and the use of iodophor sanitizers in dairy processing. A single cup of milk typically contains 50 to 80 micrograms of iodine. However, iodine levels in dairy products have been declining in some regions as iodophor sanitizers are replaced by non-iodine alternatives.
Eggs: One large egg contains approximately 25 micrograms of iodine, primarily in the yolk.
Iodized Salt: In the United States and many other countries, table salt is fortified with potassium iodide at a concentration designed to provide approximately 45 micrograms of iodine per half-teaspoon serving. Iodized salt remains one of the most reliable and consistent dietary sources of iodine, though its contribution to intake is declining as more people reduce salt consumption or switch to non-iodized specialty salts (sea salt, Himalayan pink salt, kosher salt) that typically contain very little iodine.
Supplemental Sources: For patients who are unable to meet their iodine needs through diet alone — which, in my clinical experience, is the majority of patients in Western countries — several supplemental forms are available. Potassium iodide (KI) provides iodine in the iodide form. Lugol's solution is a combination of elemental iodine (I₂) and potassium iodide (KI) in water, providing both molecular iodine and iodide. Iodoral is a tablet form of Lugol's solution. Nascent iodine (atomic iodine) is a preparation that claims to provide iodine in a highly bioavailable atomic state. Kelp-based iodine supplements provide iodine from whole-food seaweed sources but may have variable iodine content and may also contain other seaweed constituents that could affect thyroid function.
21. Clinical Dosing Strategies and Protocols
The question of optimal iodine dosing is one of the most contentious in nutritional medicine, with a dramatic divide between the conventional medical establishment and the integrative/naturopathic community. Understanding this divide is essential for both patients and practitioners.
The Conventional Position: The RDA for iodine in the United States is 150 micrograms per day for adults, 220 micrograms per day during pregnancy, and 290 micrograms per day during lactation. The Tolerable Upper Intake Level (UL) is set at 1,100 micrograms (1.1 mg) per day for adults. Conventional endocrinologists generally consider intakes above the UL to be potentially harmful, citing the risk of thyroid dysfunction, thyroiditis, and hyperthyroidism.
The Integrative Position: Practitioners like Dr. Guy Abraham, Dr. David Brownstein, Dr. Jorge Flechas, and Dr. Donald Miller have argued that the RDA represents a minimum to prevent goiter, not an optimum for health, and that milligram-level dosing (typically 12.5 to 50 mg per day) is safe and potentially therapeutic for most patients. They point to the Japanese population, which consumes an estimated average of 1,000 to 3,000 micrograms (1 to 3 mg) per day with some individuals consuming 10 to 13 mg or more, with no apparent increase in thyroid disease incidence. They also cite the historical use of potassium iodide in gram-level doses for conditions like asthma and syphilis without widespread adverse effects.
In my practice, I take a middle path guided by clinical assessment, testing, and patient response. For general health maintenance in an individual with no known thyroid disease, I typically recommend a starting dose in the range of 200 to 1,000 micrograms (0.2 to 1 mg) per day, combined with appropriate cofactors (selenium, vitamin C, magnesium, and B vitamins). For patients with documented iodine deficiency, fibrocystic breast disease, or other conditions where higher doses may be indicated, I may gradually increase the dose to 3 to 12.5 mg per day or higher, with close monitoring of thyroid function, symptoms, and detoxification reactions. Doses above 12.5 mg per day are reserved for specific clinical situations and are always supervised with regular testing.
The principle of "start low, go slow" is critical in iodine supplementation. Rapid introduction of high-dose iodine in a depleted individual can provoke intense bromide detoxification reactions, transient thyroid dysfunction, and uncomfortable symptoms. A gradual dose escalation over weeks to months, with attention to cofactor support and detoxification management, produces the best outcomes and the highest patient compliance.
22. Companion Nutrients — The Iodine Protocol
Iodine does not work in isolation. Its metabolism, utilization, and safety are intimately dependent on the presence of several key companion nutrients. Failing to address these cofactors is one of the most common reasons for adverse reactions to iodine supplementation, and it is a critical oversight that can undermine the entire therapeutic process.
Selenium (200–400 mcg/day): Selenium is arguably the single most important cofactor for iodine supplementation. The selenium-dependent glutathione peroxidase enzymes protect the thyroid gland from the oxidative damage generated during thyroid hormone synthesis. The selenium-dependent deiodinase enzymes are responsible for converting T4 to the active T3 hormone. Selenium deficiency in the context of iodine supplementation is associated with increased risk of thyroid autoimmunity (Hashimoto's thyroiditis). Always ensure adequate selenium status before introducing iodine.
Vitamin C (1,000–3,000 mg/day): Vitamin C supports iodine absorption in the gut by reducing iodate (a common form in supplements and food) to iodide, which is the form absorbed by the intestinal epithelium. It also serves as a general antioxidant that supports detoxification and reduces the severity of bromide detox reactions. Some practitioners recommend taking vitamin C alongside iodine to enhance absorption and reduce side effects.
Magnesium (400–600 mg/day): Magnesium is required for over 300 enzymatic reactions in the body, including many involved in thyroid hormone synthesis and energy metabolism. Magnesium deficiency is extremely common and can exacerbate the symptoms of hypothyroidism and iodine deficiency. Supplementation with magnesium glycinate, citrate, or malate is generally well-tolerated and clinically beneficial.
B Vitamins (B2 and B3 in particular): Riboflavin (B2) and niacin (B3) are required cofactors for the NADPH oxidase system that generates the hydrogen peroxide needed for thyroid hormone synthesis. Deficiency of these vitamins can impair the thyroid's ability to utilize iodine, even when iodine is adequately supplied. A high-quality B-complex supplement is recommended for all patients on an iodine protocol.
Zinc (15–30 mg/day): Zinc is required for the conversion of T4 to T3, for the binding of T3 to its nuclear receptor, and for the synthesis of thyroid-releasing hormone (TRH) in the hypothalamus. Zinc deficiency impairs thyroid function and can blunt the response to iodine supplementation.
Unrefined Salt (Celtic sea salt or Himalayan salt, ½ tsp/day): The chloride in unrefined salt supports the renal excretion of bromide. When patients begin iodine supplementation and mobilize stored bromide, adequate salt and water intake is essential for facilitating bromide clearance through the kidneys. This is one of the simplest and most effective interventions for managing bromide detox symptoms.
Tyrosine: The amino acid L-tyrosine is the backbone of thyroid hormone molecules. T4 is formed from two tyrosine residues plus four iodine atoms. In patients with poor dietary protein intake or impaired amino acid absorption, tyrosine supplementation (500–1,000 mg per day) can support optimal thyroid hormone production.
23. Safety, Contraindications, and the Wolff-Chaikoff Debate
Any serious discussion of iodine supplementation must address the question of safety. Iodine has been surrounded by more fear, misinformation, and controversy than perhaps any other nutrient, and much of this fear is rooted in a misunderstanding of the so-called Wolff-Chaikoff effect.
In 1948, researchers Jan Wolff and Israel Chaikoff published a study showing that when rats were injected with large amounts of iodide, their thyroid glands temporarily reduced thyroid hormone production — a phenomenon they termed "the acute Wolff-Chaikoff effect." This finding was subsequently extrapolated to humans and used to justify extreme caution around iodine supplementation, with the argument that excess iodine could suppress thyroid function and cause hypothyroidism. This interpretation has profoundly shaped medical thinking about iodine for over 75 years.
However, as Dr. Guy Abraham and others have pointed out, the Wolff-Chaikoff paper has several significant limitations. The study was conducted in rats, not humans. The iodide was administered by injection, not orally. The thyroid function suppression was transient, resolving within 24 to 48 hours as the thyroid adapted (the so-called "escape from the Wolff-Chaikoff effect"). And the doses used were extreme. Despite these limitations, the Wolff-Chaikoff effect has been cited for decades as the primary rationale for restricting iodine intake and has contributed to what Abraham terms "medical iodophobia" — an irrational fear of iodine that has had profound negative consequences for public health.
That said, iodine is not without contraindications, and responsible supplementation requires clinical awareness and monitoring:
- Autonomous thyroid nodules: Patients with long-standing multinodular goiter may develop autonomously functioning nodules that can produce excess thyroid hormone when iodine is suddenly provided — a condition known as iodine-induced hyperthyroidism or Jod-Basedow phenomenon. These patients require careful evaluation and gradual iodine introduction under close medical supervision.
- Graves' disease (active, uncontrolled): Patients with active, untreated Graves' disease (autoimmune hyperthyroidism) should generally avoid iodine supplementation until their condition is controlled, as iodine can serve as additional substrate for excess thyroid hormone production.
- Dermatitis herpetiformis: This skin condition, associated with celiac disease, involves a specific sensitivity to iodine that can trigger flare-ups. Patients with dermatitis herpetiformis should approach iodine supplementation with caution and under medical guidance.
- Known iodine allergy: True iodine allergy is extremely rare, and what is commonly referred to as "iodine allergy" (reactions to iodine-containing contrast dyes, shellfish, or topical antiseptics) is typically a reaction to the carrier substance rather than to iodine itself. Nevertheless, patients with a history of severe reactions to iodine-containing substances should be evaluated carefully before initiating supplementation.
For the vast majority of people, iodine supplementation at therapeutic doses, when combined with appropriate cofactors and introduced gradually, is safe and well-tolerated. The risks of iodine deficiency — which include thyroid disease, breast disease, cognitive impairment, infertility, immune dysfunction, and increased cancer risk — far outweigh the risks of thoughtful, monitored supplementation in most clinical situations.
24. Conclusion — Reclaiming the Master Mineral
We have journeyed through the extraordinary breadth of iodine's influence on human health — from the thyroid to the brain, from the breast to the immune system, from the gut to the cardiovascular system, from fertility to cancer prevention, from detoxification to antimicrobial defense. The evidence is overwhelming: iodine is not merely a trace mineral of limited importance. It is a foundational nutrient — a master mineral — that every cell, every tissue, and every organ in the body requires for optimal function.
The tragedy of our current iodine crisis is that it is entirely preventable. The environmental, dietary, and policy factors that have driven iodine levels down are identifiable and addressable. The knowledge and tools to assess and correct iodine deficiency are available. The companion nutrients that make iodine supplementation safe and effective are well-characterized and readily accessible. What has been lacking is awareness — awareness among patients that their symptoms may be rooted in iodine insufficiency, and awareness among practitioners that iodine deserves a central place in clinical evaluation and treatment.
It is to advocate for informed, evidence-based, clinically monitored optimization of iodine status as a foundational component of health care. I encourage every patient reading this to discuss iodine with their healthcare provider and to request appropriate testing. I encourage every practitioner to incorporate iodine assessment into their clinical practice and to approach this nutrient with the respect and nuance it deserves.
The human body is extraordinarily resilient and self-healing when given the raw materials it needs. Iodine is one of those essential raw materials. Let us reclaim it.
25. References and Recommended Reading
Books:
- Brownstein, D. Iodine: Why You Need It, Why You Can't Live Without It. 5th edition. Medical Alternatives Press.
- Abraham, G.E. The Original Internist — collected iodine research publications.
- Flechas, J.D. Clinical publications on iodine and breast disease, thyroid disease, and diabetes.
- Eskin, B.A. Iodine in breast disease — collected research published in various peer-reviewed journals.
Key Research Articles and Reviews:
- Ghent, W.R., et al. "Iodine replacement in fibrocystic disease of the breast." Canadian Journal of Surgery, 1993;36(5):453-460.
- Aceves, C., Anguiano, B., Delgado, G. "The extrathyroidal actions of iodine as antioxidant, apoptotic, and differentiation factor in various tissues." Thyroid, 2013;23(8):938-946.
- Qian, M., et al. "The effects of iodine on intelligence in children: a meta-analysis of studies conducted in China." Asia Pacific Journal of Clinical Nutrition, 2005;14(1):32-42.
- Bath, S.C., et al. "Effect of inadequate iodine status in UK pregnant women on cognitive outcomes in their children: results from the Avon Longitudinal Study of Parents and Children (ALSPAC)." The Lancet, 2013;382(9889):331-337.
- Venturi, S., et al. "Role of iodine in evolution and carcinogenesis of thyroid, breast, and stomach." Advances in Clinical Chemistry, 2000;34:1-55.
- Abraham, G.E. "The safe and effective implementation of orthoiodosupplementation in medical practice." The Original Internist, 2004;11:17-36.
- Zimmermann, M.B. "Iodine deficiency." Endocrine Reviews, 2009;30(4):376-408.
- World Health Organization. "Assessment of iodine deficiency disorders and monitoring their elimination." 3rd edition. Geneva: WHO, 2007.
Professional Organizations and Resources:
- World Health Organization — Iodine Deficiency Disorders Program
- American Thyroid Association — Iodine Guidelines
- Iodine Global Network (IGN)
- The Iodine Project (iodineresearch.com)
Disclaimer: This article is intended for educational and informational purposes only. It does not constitute medical advice, diagnosis, or treatment. Always consult a qualified healthcare provider before making any changes to your health regimen, including the use of iodine supplements. Individual needs vary, and what is appropriate for one person may not be appropriate for another. The information presented here reflects the clinical perspective of a naturopathic physician and incorporates both conventional and integrative research.
Connections
- Iodine Benefits Deep Dive
- Lugol's Iodine — the concentrated iodine preparation: antiseptic, Graves' prep, and thyroid radiation blockade
- Selenium
- Hashimoto's Thyroiditis
- Thyroid Disorders
- Thyroid Cancer
- Tyrosine
- Zinc
- Magnesium
- Vitamin C
- Fluoride
- Heavy Metals
- Cancer
- Infertility
- Fatigue
- Depression
- Milk
- Brain Fog
- Metabolic Syndrome
- Eggs