Selenium and Thyroid Function
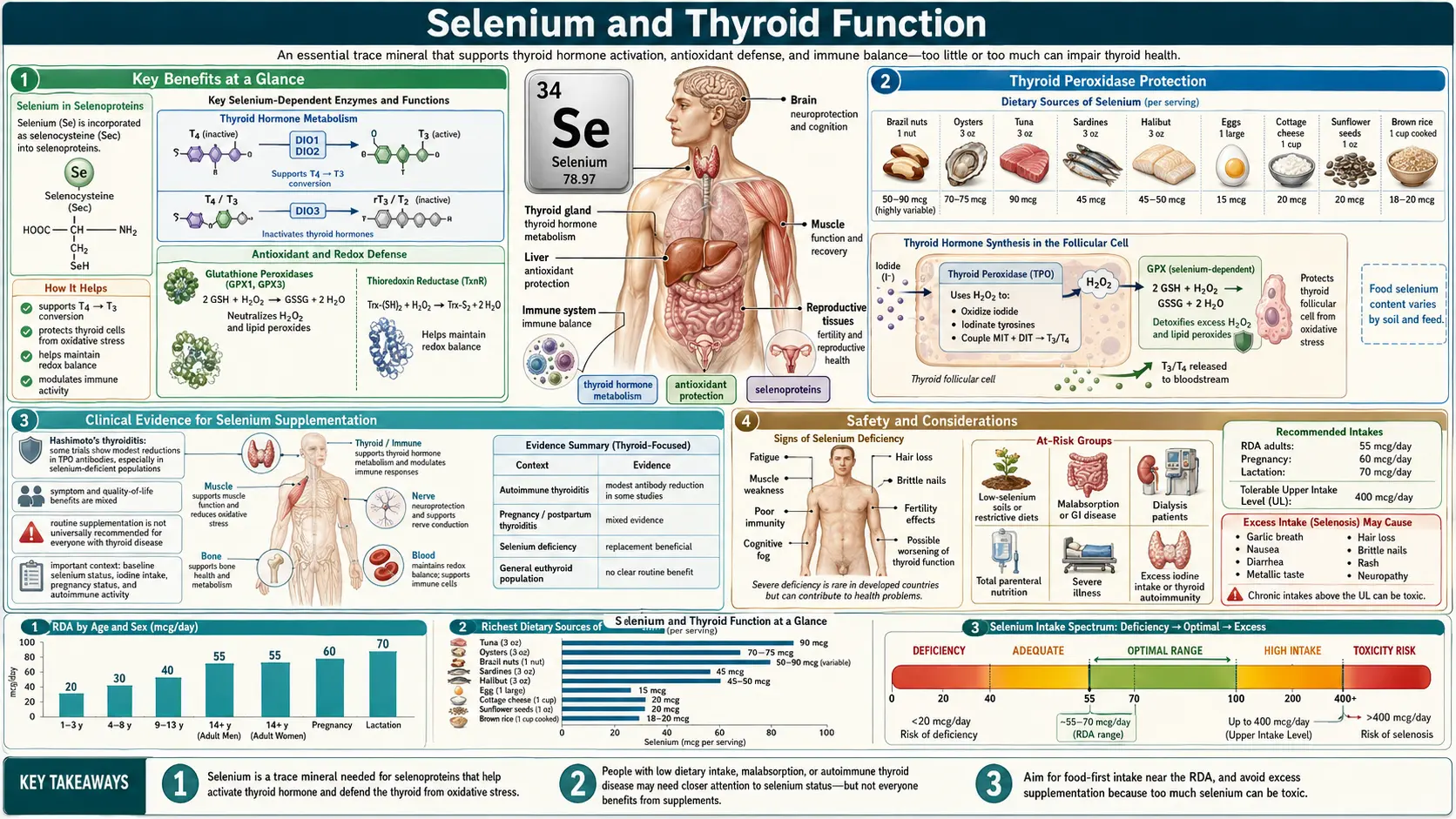
The thyroid gland contains more selenium per gram of tissue than any other organ in the human body. This remarkable concentration reflects the thyroid's profound dependence on selenoproteins for both hormone synthesis and self-protection. At least eleven selenoproteins are expressed in thyroid tissue, including the three iodothyronine deiodinases that control thyroid hormone activation and inactivation, the glutathione peroxidases that protect thyrocytes from oxidative damage, and the thioredoxin reductases that maintain cellular redox balance. Selenium deficiency impairs thyroid function at multiple levels, and the interplay between selenium and iodine status has significant clinical implications for thyroid health worldwide.
Table of Contents
- Key Benefits at a Glance
- Deiodinase Enzymes
- Type 1 Deiodinase (DIO1)
- Type 2 Deiodinase (DIO2)
- Type 3 Deiodinase (DIO3)
- Thyroid Peroxidase Protection
- Autoimmune Thyroid Disease
- Hashimoto’s Thyroiditis
- Graves’ Disease and Orbitopathy
- Selenium Deficiency and Thyroid Dysfunction
- Combined Selenium and Iodine Deficiency
- Clinical Evidence for Supplementation
- Dosing and Forms
- Safety and Considerations
- Research Papers
- Connections
- Featured Videos
Key Benefits at a Glance
- Activates thyroid hormone – Selenocysteine-containing deiodinases (DIO1, DIO2) catalyze the conversion of T4 into the biologically active T3.
- Protects thyrocytes – Glutathione peroxidases (GPx1, GPx3) and thioredoxin reductases neutralize the H2O2 generated during thyroid hormone synthesis.
- Lowers anti-TPO antibodies – Randomized trials show 200 mcg/day of selenomethionine or sodium selenite reduces anti-TPO titers by 20–40% in Hashimoto’s thyroiditis.
- First-line therapy for mild Graves’ orbitopathy – EUGOGO-guideline endorsed for improving eye outcomes and quality of life.
- Prevents postpartum thyroiditis – Supplementation during pregnancy reduces postpartum thyroid dysfunction in anti-TPO-positive women.
- Supports iodine utilization – Adequate selenium must accompany iodine repletion to avoid accelerated thyroid oxidative damage.
- Immune modulation – Enhances regulatory T cell function and dampens Th1/Th17 inflammatory responses in autoimmune thyroid disease.
Deiodinase Enzymes: Regulators of Thyroid Hormone Activation
The thyroid gland primarily secretes thyroxine (T4), a prohormone with relatively low biological activity. Conversion of T4 to the metabolically active triiodothyronine (T3) depends on selenium-containing deiodinase enzymes. These selenoenzymes catalyze the removal of specific iodine atoms from the thyronine ring structure, thereby activating or inactivating thyroid hormones in a tissue-specific manner.
Type 1 Deiodinase (DIO1)
DIO1 is a plasma membrane-associated selenoenzyme expressed predominantly in the liver, kidneys, and thyroid gland. It catalyzes both outer ring deiodination (converting T4 to T3) and inner ring deiodination (converting T4 to reverse T3, or rT3). DIO1 is the primary source of circulating T3 production and contributes significantly to the peripheral pool of active thyroid hormone available to target tissues. In the thyroid gland itself, DIO1 participates in local T3 generation and iodine salvage by deiodinating iodothyronine metabolites, allowing the released iodide to be recycled for new hormone synthesis.
DIO1 expression is positively regulated by T3 itself, creating a feedforward mechanism that increases T4-to-T3 conversion when thyroid hormone levels are adequate. In selenium deficiency, reduced DIO1 activity leads to elevated serum T4 and decreased T3, with a corresponding increase in rT3 levels, a pattern that can mimic nonthyroidal illness syndrome.
Type 2 Deiodinase (DIO2)
DIO2 is an endoplasmic reticulum-resident selenoenzyme responsible for local T3 production in tissues that are highly sensitive to thyroid hormone levels. It is expressed in the brain, anterior pituitary, thyroid, brown adipose tissue, skeletal muscle, and placenta. DIO2 exclusively catalyzes outer ring deiodination, converting T4 to T3 with high efficiency and low Km for its substrate.
In the brain, DIO2 activity in astrocytes and tanycytes generates the T3 that is critical for neuronal function, myelination, and neurotransmitter metabolism. In the anterior pituitary, DIO2-generated T3 mediates the negative feedback regulation of thyroid-stimulating hormone (TSH) secretion. A critical feature of DIO2 regulation is its rapid proteasomal degradation in response to its substrate T4, providing a homeostatic mechanism that adjusts local T3 production inversely to circulating T4 levels. Polymorphisms in the DIO2 gene (Thr92Ala) have been associated with altered thyroid hormone metabolism and clinical outcomes in hypothyroid patients on levothyroxine therapy.
Type 3 Deiodinase (DIO3)
DIO3 is the primary thyroid hormone-inactivating deiodinase. It catalyzes inner ring deiodination, converting T4 to rT3 and T3 to 3,3'-diiodothyronine (T2), both of which have minimal biological activity at thyroid hormone receptors. DIO3 is the most abundant deiodinase during fetal development, where it protects developing tissues from premature and excessive exposure to thyroid hormones. In the adult, DIO3 is expressed in the brain, skin, and placenta, and its expression can be markedly upregulated during critical illness, contributing to the low T3 state observed in nonthyroidal illness syndrome.
Coordinated Deiodinase Function
The three deiodinases work in concert to maintain precise thyroid hormone homeostasis at both the systemic and local tissue levels. In selenium deficiency, the hierarchy of selenoprotein expression ensures that DIO2 and DIO3 in the brain are relatively preserved at the expense of DIO1 in peripheral tissues. This prioritization reflects the critical importance of maintaining thyroid hormone homeostasis in the central nervous system. Nonetheless, prolonged or severe selenium deficiency can compromise all deiodinase activities, leading to widespread thyroid hormone dysregulation.
Thyroid Peroxidase Protection
Thyroid hormone synthesis is an oxidative process that generates substantial amounts of hydrogen peroxide (H2O2). The enzyme thyroid peroxidase (TPO) uses H2O2 as a co-substrate to oxidize iodide for incorporation into thyroglobulin (iodination) and to couple iodotyrosine residues to form T4 and T3 (coupling reaction). The dual oxidase enzymes DUOX1 and DUOX2 at the apical membrane of thyrocytes generate the H2O2 required for these reactions.
While H2O2 is essential for hormone synthesis, its excess poses a severe oxidative threat to thyrocytes. Uncontrolled H2O2 accumulation can damage cellular lipids, proteins, and DNA, leading to thyrocyte injury, apoptosis, and ultimately fibrosis. The thyroid gland relies heavily on selenoproteins to manage this oxidative burden.
- GPx3 (Extracellular Glutathione Peroxidase) – Highly expressed in the thyroid, GPx3 is secreted into the follicular lumen where it scavenges excess H2O2 that is not consumed during hormone synthesis. This extracellular defense is critical for preventing oxidative damage to thyroglobulin and the apical thyrocyte membrane.
- GPx1 (Cytoplasmic Glutathione Peroxidase) – Protects the intracellular compartment of thyrocytes from H2O2 that diffuses across the apical membrane. GPx1 serves as the primary cytoplasmic defense against hydrogen peroxide-induced oxidative stress.
- Thioredoxin Reductases (TrxR1 and TrxR2) – Maintain the thioredoxin and peroxiredoxin systems that provide an additional layer of H2O2 detoxification. TrxR2 in mitochondria protects against oxidative damage from the electron transport chain, which is particularly active in the metabolically demanding thyrocytes.
When selenium is deficient, the capacity to neutralize H2O2 is diminished. The resulting oxidative stress causes thyrocyte damage, promotes inflammatory infiltration, and may trigger autoimmune responses against damaged thyroid antigens. This mechanism is believed to contribute to the pathogenesis of autoimmune thyroid diseases, particularly in populations with combined selenium and iodine deficiency.
Autoimmune Thyroid Disease
Autoimmune thyroid diseases, including Hashimoto's thyroiditis and Graves' disease, are the most common organ-specific autoimmune disorders worldwide. A growing body of evidence implicates selenium deficiency as a contributing factor in their development and progression.
Hashimoto's Thyroiditis
Hashimoto's thyroiditis (chronic lymphocytic thyroiditis) is characterized by progressive autoimmune destruction of the thyroid gland, leading to hypothyroidism. The disease involves infiltration of the thyroid by autoreactive lymphocytes and the production of antibodies against thyroid peroxidase (anti-TPO antibodies) and thyroglobulin (anti-Tg antibodies).
Selenium deficiency may promote Hashimoto's thyroiditis through several mechanisms:
- Impaired Antioxidant Defense – Reduced selenoprotein activity allows H2O2 to accumulate in and around thyrocytes, causing oxidative damage that exposes intracellular antigens to the immune system and generates neoepitopes that break immune tolerance.
- Enhanced Inflammatory Signaling – Selenium deficiency promotes NF-kB activation and pro-inflammatory cytokine production (including TNF-alpha, IL-1beta, and IL-6), amplifying the inflammatory cascade within the thyroid.
- Altered T Cell Regulation – Selenium influences the balance between effector T cells and regulatory T cells (Tregs). Deficiency may impair Treg function, weakening peripheral immune tolerance and facilitating autoimmune attack.
- Increased Thyrocyte Apoptosis – Oxidative stress from inadequate selenoprotein protection promotes thyrocyte death, releasing thyroid antigens that perpetuate the autoimmune response.
Multiple clinical trials have investigated selenium supplementation in Hashimoto's thyroiditis. Several randomized controlled trials have demonstrated that supplementation with 200 micrograms of sodium selenite or selenomethionine daily for 3 to 12 months can significantly reduce anti-TPO antibody titers in patients with Hashimoto's thyroiditis, compared to placebo. Some trials have also reported improvements in thyroid ultrasound appearance (reduced echogenicity, indicating decreased inflammation) and improved quality of life measures. However, results have been heterogeneous across studies, and the degree of benefit may depend on baseline selenium status, with the greatest improvements observed in populations with lower initial selenium levels.
Graves' Disease
Graves' disease is caused by stimulating autoantibodies directed against the TSH receptor (TRAb), leading to hyperthyroidism, diffuse goiter, and in some cases, orbitopathy (Graves' ophthalmopathy). The role of selenium in Graves' disease is particularly notable in the context of Graves' orbitopathy.
- Graves' Orbitopathy – The European Group on Graves' Orbitopathy (EUGOGO) conducted a landmark randomized clinical trial demonstrating that selenium supplementation (100 micrograms of sodium selenite twice daily for 6 months) significantly improved quality of life, reduced ocular involvement, and slowed disease progression in patients with mild Graves' orbitopathy, compared to both placebo and pentoxifylline. These findings led to the recommendation of selenium supplementation as first-line treatment for mild Graves' orbitopathy in European Thyroid Association guidelines.
- Oxidative Stress in Orbital Tissue – Graves' orbitopathy involves oxidative stress and inflammation in the retro-orbital tissues. Selenoproteins may reduce oxidative damage and inflammatory cytokine production in orbital fibroblasts, mitigating tissue remodeling and edema.
- Immune Modulation – Selenium may help restore immune balance by promoting regulatory T cell activity and dampening the Th1/Th17-mediated autoimmune responses that drive orbital inflammation.
Selenium Deficiency and Thyroid Dysfunction
The consequences of selenium deficiency on thyroid function are determined by the severity and duration of deficiency, concurrent iodine status, and individual genetic factors.
Isolated Selenium Deficiency
In populations with adequate iodine intake, isolated selenium deficiency primarily affects peripheral thyroid hormone metabolism. Characteristic findings include:
- Elevated T4 and Reduced T3 – Impaired DIO1 activity in the liver and kidneys reduces the peripheral conversion of T4 to T3, resulting in elevated serum T4 with decreased or low-normal T3.
- Increased Reverse T3 – Reduced DIO1-mediated clearance of rT3 leads to elevated circulating levels of this inactive metabolite.
- Compensatory TSH Elevation – In more severe deficiency, reduced T3 production may trigger a mild compensatory increase in TSH secretion.
- Preserved Brain Thyroid Hormone Status – Due to the preferential preservation of DIO2 in the brain during selenium deficiency, central thyroid hormone levels are relatively maintained, ensuring that TSH feedback remains functional.
Combined Selenium and Iodine Deficiency
The interaction between selenium and iodine deficiency is clinically significant and geographically relevant, as regions with selenium-poor soils often also have iodine-deficient populations. Combined deficiency produces more severe thyroid dysfunction than either deficiency alone.
- Exacerbated Hypothyroidism – Iodine deficiency reduces thyroid hormone production, while concurrent selenium deficiency impairs the conversion of what little T4 is made to active T3, compounding the hypothyroid state.
- Increased Thyroid Vulnerability – Iodine deficiency stimulates TSH, which increases H2O2 production in the thyroid. When selenium-dependent GPx enzymes are simultaneously impaired, the thyroid sustains greater oxidative damage, promoting thyroid necrosis and fibrosis.
- Myxedematous Cretinism – In regions of combined severe iodine and selenium deficiency (such as parts of central Africa), children may develop myxedematous cretinism, characterized by thyroid fibrosis, severe hypothyroidism, growth retardation, and neurodevelopmental impairment. This contrasts with neurological cretinism seen in pure iodine deficiency regions.
- Iodine Supplementation Caution – Importantly, correcting iodine deficiency without addressing concurrent selenium deficiency can paradoxically worsen thyroid damage. Iodine supplementation increases thyroid H2O2 production for hormone synthesis, and without adequate selenoprotein-mediated antioxidant protection, the resulting oxidative stress can accelerate thyroid destruction. This has led to the recommendation that selenium status be assessed and corrected before or simultaneously with iodine supplementation programs in regions of dual deficiency.
Clinical Evidence for Selenium Supplementation
A substantial and growing body of clinical evidence supports the role of selenium supplementation in thyroid health, although important questions remain regarding optimal dose, form, duration, and patient selection.
Evidence in Autoimmune Thyroiditis
- Anti-TPO Antibody Reduction – Multiple randomized controlled trials and meta-analyses have demonstrated that selenium supplementation (typically 200 micrograms daily as selenomethionine or sodium selenite) reduces anti-TPO antibody concentrations by 20-40% over 3 to 12 months in patients with Hashimoto's thyroiditis. The clinical significance of antibody reduction alone remains debated, as it is not established whether lower antibody titers translate into preserved thyroid function over the long term.
- Thyroid Echogenicity – Some trials have shown improvements in thyroid ultrasound characteristics, with reduced hypoechogenicity suggesting decreased lymphocytic infiltration and inflammation in the thyroid parenchyma.
- Quality of Life – Several studies have reported improvements in patient-reported quality of life measures, including reductions in fatigue and improvements in mood, although these findings are not consistent across all trials.
- Thyroid Function Preservation – Limited evidence suggests that selenium supplementation may slow the progression from subclinical to overt hypothyroidism in patients with Hashimoto's thyroiditis, potentially reducing the need for or delaying the initiation of levothyroxine replacement therapy.
Evidence in Graves' Orbitopathy
- EUGOGO Trial – The pivotal randomized, double-blind, placebo-controlled trial conducted by the European Group on Graves' Orbitopathy demonstrated that 200 micrograms daily of sodium selenite for 6 months significantly improved overall eye assessment, quality of life (measured by the GO-QoL questionnaire), and clinical activity score in patients with mild Graves' orbitopathy.
- Guideline Recommendations – Based on this evidence, the European Thyroid Association and the American Thyroid Association have included selenium supplementation as a recommended intervention for mild active Graves' orbitopathy, representing one of the strongest guideline-level endorsements of selenium supplementation for any thyroid condition.
Evidence in Pregnancy
- Postpartum Thyroiditis Prevention – A notable Italian randomized controlled trial found that selenium supplementation (200 micrograms of selenomethionine daily) during pregnancy and the postpartum period significantly reduced the incidence of postpartum thyroiditis and permanent hypothyroidism in women positive for anti-TPO antibodies. These findings suggest that selenium may protect the vulnerable thyroid during the immune rebound that occurs after delivery.
- Preeclampsia Risk – Observational studies have associated low maternal selenium status with increased preeclampsia risk, and small interventional trials have suggested potential benefits of supplementation, although larger confirmatory trials are needed.
Considerations and Limitations
- Baseline Selenium Status – The benefits of supplementation appear most pronounced in populations with low baseline selenium levels. In selenium-replete populations, additional supplementation may provide little benefit and could potentially be harmful at high doses.
- Form of Selenium – Both organic (selenomethionine) and inorganic (sodium selenite) forms have been used in clinical trials. Selenomethionine has greater bioavailability and is incorporated into the general protein pool, while sodium selenite is more directly channeled into selenoprotein synthesis. The optimal form for thyroid-specific supplementation has not been definitively established.
- Duration of Supplementation – Most positive clinical trials have used supplementation periods of 6 to 12 months. The optimal duration of treatment and whether benefits persist after discontinuation remain unclear.
- Safety – At doses of 200 micrograms daily, selenium supplementation is generally well tolerated. However, long-term supplementation above 400 micrograms daily risks selenosis, and some epidemiological data have raised concerns about potential associations between high selenium status and type 2 diabetes risk, necessitating careful dose selection.
Dosing and Forms
- RDA (adults) – 55 mcg/day; pregnancy 60 mcg/day; lactation 70 mcg/day.
- Tolerable Upper Intake Level (UL) – 400 mcg/day from all sources combined.
- Clinical trial dose for thyroid conditions – Typically 200 mcg/day for 3–12 months.
- Selenomethionine – Organic form from yeast or plants; high bioavailability (~90%); incorporated non-specifically into body proteins, producing a gradual rise in tissue selenium.
- Sodium selenite / selenate – Inorganic forms used in the EUGOGO trial; directly channeled into selenoprotein synthesis.
- Selenium-rich foods – Brazil nuts (one nut can supply 68–91 mcg), tuna, sardines, halibut, pasture-raised eggs, sunflower seeds, organ meats.
Safety and Considerations
- Selenosis – Chronic intakes above 400 mcg/day can produce garlic breath, brittle nails, hair loss, neuropathy, and GI disturbance.
- Type 2 diabetes signal – Observational and NPC-trial data raise modest concerns about higher selenium exposure and type 2 diabetes risk in already-replete populations.
- Baseline status matters – Benefit is largest when baseline serum selenium is < 110 µg/L; supplementing a replete patient offers little benefit and potential harm.
- Drug interactions – May enhance anticoagulant effect; separate from levothyroxine by several hours to avoid absorption issues.
- Brazil nut caution – Content varies widely (5–100+ mcg per nut) by soil region; do not eat multiple nuts daily for extended periods.
This content is provided for informational purposes only and does not constitute medical advice. Consult a qualified healthcare provider before starting selenium supplementation, especially if you have thyroid disease or are taking levothyroxine.
Research Papers
- Rayman MP. Selenium and human health. Lancet. 2012;379(9822):1256-1268.
- Köhrle J. Selenium and the thyroid. Thyroid. 2010;20(10):1155-1160.
- Gartner R, Gasnier BC, Dietrich JW, et al. Selenium supplementation in patients with autoimmune thyroiditis decreases thyroid peroxidase antibodies concentrations. J Clin Endocrinol Metab. 2002;87(4):1687-1691.
- Marcocci C, Kahaly GJ, Krassas GE, et al. Selenium and the course of mild Graves’ orbitopathy. N Engl J Med. 2011;364(20):1920-1931.
- Negro R, Greco G, Mangieri T, Pezzarossa A, Dazzi D, Hassan H. The influence of selenium supplementation on postpartum thyroid status in pregnant women with thyroid peroxidase autoantibodies. J Clin Endocrinol Metab. 2007;92(4):1263-1268.
- van Zuuren EJ, Albusta AY, Fedorowicz Z, Carter B, Pijl H. Selenium supplementation for Hashimoto’s thyroiditis. Cochrane Database Syst Rev. 2013;(6):CD010223.
- Wichman J, Winther KH, Bonnema SJ, Hegedus L. Selenium supplementation significantly reduces thyroid autoantibody levels in patients with chronic autoimmune thyroiditis: a systematic review and meta-analysis. Thyroid. 2016;26(12):1681-1692.
- Duntas LH, Benvenga S. Selenium: an element for life. Endocrine. 2015;48(3):756-775.
- Schomburg L. Dietary selenium and human health. Nutrients. 2017;9(1):22.
- PubMed — selenium + Hashimoto + randomized trials
- PubMed — selenium + Graves’ orbitopathy
- NIH Office of Dietary Supplements — Selenium Fact Sheet for Health Professionals
- Linus Pauling Institute — Selenium
Connections
- Selenium
- Selenium and Cancer Prevention
- Iodine
- Hashimoto's Thyroiditis
- Graves' Disease
- Zinc
- Iron
- Oxidative Stress
- Glutathione
- Vitamin D3
- Hair Loss
- Fatigue
- Tuna
- Eggs
- Organ Meats
- Preeclampsia
Featured Videos

Selenium and Your Thyroid - A Summary of the Evidence
How Important is Selenium for Thyroid Function?

Selenium and The Thyroid- How Selenium Helps Patients with Graves, Hashimotos

Selenium in Thyroid Care: Clinical Insights and Applications

Selenium and your thyroid

Does Selenium Help Hashimoto's? Should you take it? (good news and a reality check)

Selenium: The BEST Treatment For Hashimoto's & Graves'

Selenium Deficiency and Your Thyroid Function | Dr. J Q & A

Selenium and Hashimoto's