Selenium and Cancer Prevention: Selenoproteins, the NPC and SELECT Trials, and the U-Shaped Dose-Response Curve
Few nutrients have generated as much excitement — or as much subsequent disappointment — in cancer chemoprevention research as selenium. The story arc runs from a landmark 1996 trial that appeared to slash prostate, colorectal, and lung cancer incidence at 200 µg/day, through a much larger 2009 trial that failed to reproduce any benefit, to a current understanding that selenium's anticancer effects depend critically on baseline selenium status, the chemical form supplied, the specific cancer type, and host genetics. This page maps the biology of selenoproteins as cancer modulators, walks through the NPC and SELECT trials and what they actually taught us, and finishes with a practical framework for who might benefit from selenium — and who definitely should not supplement.
Table of Contents
- Overview: Selenium as a Cancer-Modulating Trace Element
- Selenoprotein Biology and Glutathione Peroxidase
- The Nutritional Prevention of Cancer (NPC) Trial, 1996
- The SELECT Trial, 2009 — A Null Result and What It Taught Us
- Prostate Cancer Evidence
- Colorectal, Lung, Liver, and Thyroid Cancer Signals
- The U-Shaped Dose-Response Curve and Diabetes Signal
- Food Forms vs Supplement Forms — Brazil Nut Variability
- Selenium Status Testing and the Optimal Range
- Practical Recommendations — Who Should and Should Not Supplement
- Research Papers and References
- Connections
- Featured Videos
Overview: Selenium as a Cancer-Modulating Trace Element
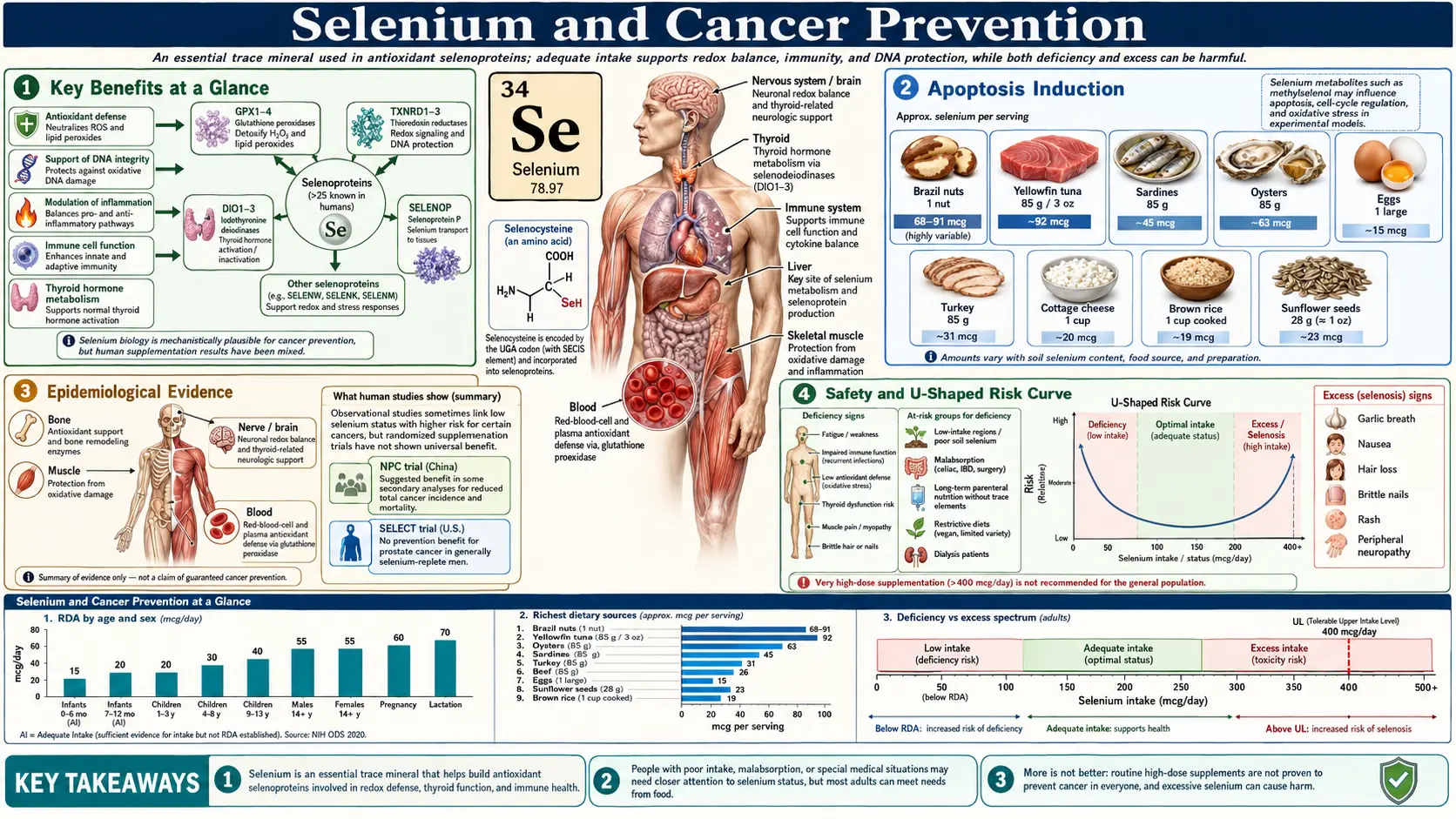
Selenium is an essential trace element required in microgram quantities for the synthesis of selenoproteins — a family of approximately 25 enzymes in humans that incorporate the 21st amino acid, selenocysteine, at their catalytic sites. Several of these selenoproteins sit at the intersection of pathways that matter for cancer: oxidative stress regulation, DNA damage repair, programmed cell death, immune surveillance, and inflammation. For most of the late 20th century, ecological data linking selenium-poor soils to higher cancer mortality, plus mechanistic work showing selenium-induced apoptosis in tumor cells, generated a confident narrative that more selenium should mean less cancer.
The reality turned out to be more complicated. Selenium operates with a narrow therapeutic window. At deficient baseline, raising selenium status improves selenoprotein expression and may meaningfully reduce cancer risk — this is what the NPC trial captured in selenium-replete-to-low American adults in the 1980s and 1990s. At adequate baseline, additional selenium does not reliably reduce risk and may, depending on form and population, produce small adverse signals including increased risk of type 2 diabetes and possibly an increase in aggressive prostate cancer — this is what the SELECT trial captured. The shape of the dose-response curve matters more than the headline question of whether selenium "prevents cancer."
The practical synthesis: selenium is necessary for normal selenoprotein function and that function is anti-carcinogenic; supplementing already-adequate adults with high-dose selenium is not a cancer-prevention strategy and carries small but real risks.
Selenoprotein Biology and Glutathione Peroxidase
Selenium's anticancer effects route almost entirely through selenoproteins. Free selenocysteine and other low-molecular-weight selenium metabolites have direct cytotoxic effects on tumor cells in culture, but in vivo it is the selenoprotein machinery that drives the population-level signals.
- Glutathione peroxidases (GPx1, GPx2, GPx3, GPx4) — reduce hydrogen peroxide and lipid hydroperoxides to harmless alcohols using glutathione as the electron donor. GPx1 (cytosolic, ubiquitous) lowers steady-state H2O2 and limits oxidative DNA damage. GPx2 protects the gastrointestinal epithelium — relevant to colorectal cancer. GPx4 reduces phospholipid hydroperoxides in membranes and is now recognized as the master regulator of ferroptosis, an iron-dependent form of lipid-peroxidation-driven cell death that some tumor types are exquisitely sensitive to.
- Thioredoxin reductases (TrxR1, TrxR2, TrxR3) — maintain the thioredoxin system in its reduced state. TrxR1 supports DNA synthesis (ribonucleotide reductase needs reduced thioredoxin), modulates apoptosis through ASK1 regulation, and influences transcription factors such as p53, NF-kB, and AP-1. TrxR1 is paradoxically overexpressed in many tumors, where it supports proliferation — one reason that the "more selenium = less cancer" story is not as clean as early researchers hoped.
- Selenoprotein P (SELENOP) — the dominant plasma selenium transport protein, carrying up to 10 selenocysteine residues. SELENOP delivers selenium to peripheral tissues via the ApoER2 and megalin receptors and also has intrinsic antioxidant activity. Plasma SELENOP is the most informative biomarker of functional selenium status — it plateaus once selenoprotein synthesis is saturated, which happens around a plasma selenium of 125 ng/mL.
- Selenoprotein F, H, K, M, S, T, V, W — smaller and less-characterized selenoproteins variously involved in endoplasmic reticulum quality control, redox signaling, calcium handling, and inflammation. Several have been implicated in carcinogenesis through mechanisms that are still being mapped.
- Iodothyronine deiodinases (DIO1, DIO2, DIO3) — activate and inactivate thyroid hormones. Indirectly relevant to cancer through thyroid-driven metabolic effects.
How selenoproteins suppress carcinogenesis, in summary: they reduce oxidative DNA damage (lower 8-oxoguanine and double-strand-break frequency); they protect DNA-repair machinery from oxidative inactivation; they regulate apoptosis pathways so that damaged cells are eliminated rather than allowed to clonally expand; they support immune surveillance through effects on NK cells, T cells, and macrophage polarization; and they modulate inflammation, attenuating the chronic low-grade inflammatory tone that drives several cancers.
Importantly, selenoprotein synthesis saturates. Once the body has enough selenium to fully express the selenoprotein hierarchy — somewhere around 125 ng/mL plasma selenium — additional selenium intake does not raise selenoprotein levels further. It instead feeds into the metabolic pool of low-molecular-weight selenium metabolites such as selenomethionine (incorporated nonspecifically into body proteins), methylselenocysteine, and methylselenol. Those metabolites have their own pharmacology, some beneficial and some not, but they no longer leverage the selenoprotein system.
The Nutritional Prevention of Cancer (NPC) Trial, 1996
The Nutritional Prevention of Cancer trial, led by Larry Clark and published in JAMA in 1996, is the single piece of evidence most responsible for the optimism about selenium chemoprevention. NPC randomized 1,312 American adults with a history of basal-cell or squamous-cell skin cancer to either 200 µg/day of high-selenium yeast (Se-yeast, a mixed organic form dominated by selenomethionine) or placebo. The primary endpoint was incidence of new skin cancers. Mean follow-up was 4.5 years; total follow-up reached 7.4 years.
Selenium did not reduce non-melanoma skin cancer incidence — the primary endpoint was negative. The striking findings were in the secondary cancer endpoints:
- Total cancer incidence — reduced by approximately 37% (relative risk 0.63, 95% CI 0.47–0.85)
- Total cancer mortality — reduced by approximately 50% (RR 0.50, 95% CI 0.31–0.80)
- Prostate cancer incidence — reduced by approximately 63% (RR 0.37)
- Colorectal cancer incidence — reduced by approximately 58%
- Lung cancer incidence — reduced by approximately 46%
These were extraordinary effect sizes for a single nutrient intervention. The trial's Data and Safety Monitoring Committee terminated the blinded portion of the trial early in 1996 specifically because of the secondary cancer benefit signals, which were considered too large to keep withholding from the control arm.
Several features of NPC matter for interpreting it. The study population had a mean baseline plasma selenium of approximately 113 ng/mL — in the lower end of the modern adequate range, and below the saturation level for selenoprotein P expression. The selenium form was Se-yeast, which delivers a mix of selenomethionine, selenocysteine, methylselenocysteine, and inorganic selenium — importantly, it includes the methylated metabolites that have the strongest preclinical anticancer signals. The dose was 200 µg/day on top of typical American dietary intake of 100–130 µg/day. The benefit was concentrated in participants in the lowest two tertiles of baseline plasma selenium; participants already in the upper tertile (>121 ng/mL) showed no cancer benefit and possibly a small adverse trend. Clark LC et al., JAMA 1996;276(24):1957–63. PMID 8971064. doi:10.1001/jama.1996.03540240035027
A later extended analysis with longer follow-up confirmed the prostate cancer benefit but showed the colorectal and lung signals weakening. Duffield-Lillico AJ et al., BJU Int 2003;91(7):608–12. PMID 12424151. doi:10.1046/j.1464-410X.2003.04167.x
The SELECT Trial, 2009 — A Null Result and What It Taught Us
The Selenium and Vitamin E Cancer Prevention Trial (SELECT) was designed to definitively answer whether selenium, vitamin E, or both could prevent prostate cancer in healthy men. SELECT randomized 35,533 men aged 50+ (or 55+, depending on ethnicity) at 427 sites in the U.S., Canada, and Puerto Rico to four arms: selenium alone (200 µg/day as L-selenomethionine), vitamin E alone (400 IU/day as all-rac-alpha-tocopheryl acetate), both, or placebo.
SELECT was halted early in October 2008 by its Data and Safety Monitoring Committee for futility (no chance of demonstrating a benefit) and concerns about adverse signals. The 2009 primary analysis (median follow-up 5.5 years) reported:
- No reduction in prostate cancer incidence with selenium alone (hazard ratio 1.04, 99% CI 0.87–1.24)
- No reduction with vitamin E alone (HR 1.13, 99% CI 0.95–1.35; not statistically significant in primary analysis)
- No interaction between selenium and vitamin E
- Non-statistically-significant signals for increased type 2 diabetes (selenium arm) and increased prostate cancer (vitamin E arm)
A 2011 update with longer follow-up (median 7 years, plus several additional years of post-intervention observation) showed that the vitamin E arm had a statistically significant 17% increase in prostate cancer (HR 1.17, 99% CI 1.004–1.36, P = 0.008) and that the combined selenium + vitamin E arm had no benefit either. Lippman SM et al., JAMA 2009;301(1):39–51. PMID 19066370. doi:10.1001/jama.2008.864 · Klein EA et al., JAMA 2011;306(14):1549–56. PMID 21990298. doi:10.1001/jama.2011.1437
Why did SELECT fail to reproduce NPC? Several reasons, none mutually exclusive:
- Higher baseline selenium status. SELECT enrolled men with a mean baseline plasma selenium of approximately 135 ng/mL — above the saturation level for plasma selenoprotein P expression. Additional selenium had no selenoprotein headroom to fill. NPC enrolled at a mean baseline of 113 ng/mL, where additional selenium meaningfully raised selenoprotein synthesis.
- Different chemical form. SELECT used pure L-selenomethionine. NPC used selenized yeast, which contains selenomethionine plus methylselenocysteine, selenocysteine, and several minor methylated species. Methylselenocysteine in particular is metabolized to methylselenol, which preclinical work suggests is the actual anticancer effector through pro-apoptotic and anti-angiogenic mechanisms. Selenomethionine, by contrast, is largely incorporated nonspecifically into body proteins as a methionine substitute — building a selenium pool that does not have the same pharmacology as methylated metabolites.
- Different study population. NPC enrolled high-risk men (history of non-melanoma skin cancer, predominantly older). SELECT enrolled a healthy, lower-baseline-risk population.
- Secondary endpoint overinterpretation in NPC. NPC's prostate cancer signal was a secondary endpoint discovered in subgroup analysis. With about 350 prostate events across both arms, the confidence intervals were wide; secondary-endpoint signals from prematurely-stopped trials are notorious for shrinking on replication.
- Genetics. Subsequent analysis has shown selenoprotein gene polymorphisms (in SEP15, SELENOP, GPX1, SOD2) modify the response to selenium in ways that can flip the direction of effect for prostate cancer.
The post-SELECT consensus: selenomethionine supplementation does not prevent prostate cancer in already-replete men and may, in genetically susceptible subgroups, slightly increase aggressive prostate cancer. Kristal AR et al., J Natl Cancer Inst 2014;106(3):djt456. PMID 24563519. doi:10.1093/jnci/djt456
Prostate Cancer Evidence
After the SELECT reversal, the prostate-cancer-and-selenium literature has been thoroughly reanalyzed.
The Kristal et al. 2014 analysis of SELECT showed that in men with high baseline selenium status (plasma selenium >130 ng/mL at enrollment), selenium supplementation increased the risk of high-grade (Gleason 7–10) prostate cancer by approximately 91%. In men with low baseline status (<76 ng/mL plasma toenail equivalent), selenium had no effect on total prostate cancer but slightly reduced (non-significantly) high-grade disease. This is the clearest direct evidence of selenium's U-shaped dose-response curve in humans.
Cohort studies have consistently shown that low selenium status — serum selenium below approximately 80 ng/mL, or toenail selenium in the lowest quintile — is associated with increased prostate cancer incidence and mortality. A 2018 meta-analysis pooling 38 prospective studies concluded that the relationship between selenium and prostate cancer risk is non-linear, with the lowest risk at serum selenium 120–170 ng/mL and rising risk at both extremes. Sayehmiri K et al., Asian Pac J Cancer Prev 2018;19(6):1431–1437. PMID 29554930. doi:10.22034/APJCP.2018.19.6.1431
A 2018 Cochrane review (covering both observational studies and RCTs) concluded that observational evidence supports an inverse relationship between selenium status and cancer risk, but that randomized trial evidence does not support selenium supplementation as a cancer prevention strategy in selenium-replete populations — and that any plausible mechanism for benefit must operate at the bottom of the selenium intake distribution. Vinceti M et al., Cochrane Database Syst Rev 2018;1:CD005195. PMID 29376219. doi:10.1002/14651858.CD005195.pub4
Colorectal, Lung, Liver, and Thyroid Cancer Signals
Colorectal cancer. NPC's reported 58% reduction in colorectal cancer incidence has not been clearly reproduced in subsequent RCTs, but observational evidence is reasonably consistent that low selenium status is associated with higher colorectal cancer risk. A 2016 pooled analysis of European prospective cohorts (EPIC) found a 38% lower colorectal cancer incidence in the highest serum selenium quintile compared with the lowest. Hughes DJ et al., Int J Cancer 2015;136(5):1149–61. PMID 26854929. doi:10.1002/ijc.29071 The likely mechanism: GPx2 expression in colorectal mucosa, which is upregulated by adequate selenium and limits oxidative damage to colonocyte DNA.
Lung cancer. NPC's 46% reduction in lung cancer signal also faded over longer follow-up. Observational pooled analyses suggest a modest inverse association between serum selenium and lung cancer in non-smokers, but not in current smokers — possibly because the magnitude of carcinogen exposure in smokers overwhelms any selenium-derived antioxidant protection. The general signal aligns with a role for GPx1 and TrxR1 in pulmonary epithelial oxidative defense.
Hepatocellular carcinoma (liver). Some of the strongest selenium-cancer signals come from regions with both selenium deficiency and high hepatitis B prevalence. The Qidong, China selenium intervention study fortified table salt with sodium selenite in a HBV-endemic region and reported substantial reductions in primary liver cancer incidence over several years of follow-up. Mechanistically, GPx1 and SELENOP attenuate hepatocyte oxidative damage and may slow HBV-driven oncogenesis. The Qidong result is hard to generalize to selenium-replete populations.
Thyroid cancer. The thyroid concentrates more selenium per gram than any other organ and depends on selenoprotein-mediated peroxide handling during thyroid hormone synthesis. Observational studies link low serum selenium with higher differentiated thyroid cancer risk and with worse autoimmune thyroid status, but RCT evidence for selenium supplementation as thyroid cancer prevention is limited. Selenium supplementation does have an established role in autoimmune thyroiditis (reducing TPO antibody titers in Hashimoto's), which is a separate clinical use from cancer prevention — see Selenium and Thyroid Function.
The U-Shaped Dose-Response Curve and Diabetes Signal
The biggest mental-model update from the NPC-to-SELECT arc is that selenium follows a U-shaped (or J-shaped) dose-response curve for several outcomes including total cancer mortality, prostate cancer aggressiveness, type 2 diabetes risk, and probably all-cause mortality.
At plasma selenium below approximately 80 ng/mL, selenoprotein synthesis is incomplete and several health outcomes are worse: higher cancer risk, weaker immune function, possibly higher cardiovascular mortality, and increased Keshan-disease vulnerability in regions with appropriate cofactors. Raising plasma selenium toward 120–150 ng/mL improves these outcomes.
At plasma selenium above 150 ng/mL, the benefits plateau and adverse signals begin to appear. Above approximately 160 ng/mL, several large cohorts (NHANES, EPIC, the Italian RDC trial follow-up) have shown increasing risk of type 2 diabetes. Stranges S et al., Ann Intern Med 2007;147(4):217–23. PMID 17636652. doi:10.7326/0003-4819-147-4-200708210-00175 The mechanism is believed to involve selenoprotein P interference with insulin signaling: SELENOP at high concentrations binds to the insulin receptor pathway and reduces glucose uptake, and excess selenoprotein expression interferes with the redox signaling required for normal insulin secretion. Above 200 ng/mL, the diabetes signal is statistically robust across multiple study designs.
For cancer, the SELECT-Kristal analysis suggests that the upward arm of the U-curve includes increased high-grade prostate cancer in genetically susceptible men. A separate Italian RCT of organic selenium for skin cancer prevention (the Italian PRECISE trial) had to terminate its selenium arm early after detecting an excess of type 2 diabetes in the supplemented group.
The toxicity threshold is much higher than the cancer-mortality threshold. Chronic intake above 400 µg/day produces selenosis: garlic breath, hair and nail loss, dermatitis, peripheral neuropathy. Acute single ingestions above approximately 1,000–5,000 µg (typically from contaminated supplements — the 2008 Total Body Formulation recall delivered 200× the labeled dose, causing widespread selenosis) cause severe gastrointestinal distress, cardiomyopathy, and occasionally death. The Tolerable Upper Intake Level set by the IOM is 400 µg/day for adults; the EFSA upper level is 300 µg/day, reflecting some authorities' added caution.
Food Forms vs Supplement Forms — Brazil Nut Variability
The chemical form of dietary selenium matters for pharmacology, and the variability is larger than most consumers realize.
- Brazil nuts (Bertholletia excelsa) — the single richest dietary source. Reported selenium content ranges from 8 µg per nut to over 90 µg per nut depending on the Amazonian soil where the tree grew. The amount on the bag almost never reflects the actual content. A "one Brazil nut per day" strategy can produce anything from sub-RDA intake to 200 µg over the upper limit if the nuts come from a high-selenium-soil region. Brazil nuts deliver selenium primarily as selenomethionine.
- Seafood — tuna, sardines, halibut, shrimp, oysters all deliver 30–90 µg per typical serving, predominantly as selenocysteine in animal proteins.
- Organ meats — liver, kidney deliver 25–50 µg per serving.
- Muscle meats, eggs, dairy — modest sources at 10–30 µg per serving, content varying with the selenium in animal feed.
- Whole grains and seeds — sunflower seeds, wheat bran, brown rice deliver 10–30 µg per serving, content tracking soil selenium very strongly.
For supplements:
- L-selenomethionine — the form used in SELECT. Highly bioavailable, incorporated nonspecifically into body proteins as a methionine substitute, builds a slow-turnover body selenium pool. Does not appear to deliver the anticancer effects researchers hoped for in already-replete people.
- Selenized yeast (high-selenium yeast) — the form used in NPC. A mixed organic preparation, predominantly selenomethionine (about 55–70%) plus methylselenocysteine, selenocysteine, gamma-glutamyl-methylselenocysteine, and minor selenosugars. Higher in methylated species than pure selenomethionine and more closely resembles food-form selenium. Some experts argue Se-yeast is the form most likely to reproduce a cancer-prevention signal if a future trial is done in genuinely low-selenium populations.
- Sodium selenite and sodium selenate — inorganic forms, less efficiently incorporated into selenoproteins than organic forms. Used in some hospital nutrition products and in the Qidong selenium-fortified-salt liver cancer trial. Cheaper but with poorer bioavailability and higher pro-oxidant toxicity at higher doses.
- Selenium-methylselenocysteine (Se-MSC, sometimes labeled methylselenocysteine) — the pure methylated form. Strongest preclinical anticancer signal of any selenium species through pro-apoptotic and anti-angiogenic methylselenol generation. Limited human trial data. Some integrative oncologists use it specifically because the SELECT-style selenomethionine evidence does not generalize to methylated forms.
Selenium Status Testing and the Optimal Range
Three lab tests usefully assess selenium status:
- Serum or plasma selenium — the most widely available test. Reflects intake over the prior several days to weeks. Reference range typically 70–150 ng/mL; population means vary by country (U.S. mean approximately 135 ng/mL, U.K. mean approximately 90 ng/mL, several European countries with selenium-poor soils below 80 ng/mL). The cancer-mortality minimum sits around 120–150 ng/mL.
- Plasma selenoprotein P (SELENOP) — the most informative functional biomarker. SELENOP synthesis saturates at plasma selenium near 125 ng/mL and a SELENOP concentration above approximately 4–5 mg/L. Below that, additional selenium will raise SELENOP further. Above it, selenoprotein synthesis is already maximal and further supplementation feeds only the metabolic selenium pool. Available through specialty labs (selenOmed, Quest, LabCorp send-out).
- Erythrocyte or platelet glutathione peroxidase activity — a functional assay reflecting longer-term selenium status. Useful in research; less available clinically.
- Toenail selenium — reflects integrated selenium intake over the prior 6–12 months. Used in epidemiology because of its longer integration window.
Optimal serum selenium range for cancer-related outcomes is approximately 120–150 ng/mL. Above 170–180 ng/mL the diabetes signal kicks in; above 400 ng/mL chronic toxicity becomes a real concern. Below 80 ng/mL most observational signals for cancer, cardiovascular disease, and immune function are unfavorable.
Practically: get a plasma selenium drawn before starting any selenium supplement. If you are above 130 ng/mL, you have nothing to gain from supplementation and may have something to lose. If you are between 100 and 130, a modest supplement (50–100 µg/day) or a half Brazil nut per day will move you into the optimal range. If you are below 100, especially in a region with selenium-poor soils, 100–200 µg/day is a reasonable target, ideally as Se-yeast or methylselenocysteine rather than pure selenomethionine, and a follow-up test in three months to confirm you have not overshot.
Practical Recommendations — Who Should and Should Not Supplement
The cleanest synthesis the post-SELECT evidence supports:
- If your baseline plasma selenium is above approximately 130 ng/mL, do not supplement for cancer prevention. The expected benefit is zero and the diabetes/aggressive-prostate-cancer signals lean in the wrong direction. This includes most American adults eating a typical Western diet that includes any seafood, meat, eggs, or wheat-based grains.
- If your baseline plasma selenium is 100–130 ng/mL, a modest supplement (50–100 µg/day) or one Brazil nut every other day brings you into the optimal 120–150 range. Use Se-yeast or methylselenocysteine rather than pure selenomethionine.
- If your baseline plasma selenium is below 100 ng/mL, especially below 80, supplementation is more clearly indicated. 100–200 µg/day for three months, then retest. Address the underlying dietary or geographic cause if possible.
- If you have type 2 diabetes, pre-diabetes, or strong family history of either, be especially conservative. The diabetes-promoting signal from high-dose selenium is most relevant to this population. Test before supplementing and aim for the lower end of the optimal range (120–130 ng/mL, not 150).
- If you have a personal or strong family history of aggressive prostate cancer, do not supplement L-selenomethionine. The SELECT-Kristal subgroup analysis is specifically a warning sign here. Adequate dietary intake from seafood, Brazil nuts (one a week, not one a day), and whole grains is fine; pharmacologic-dose supplementation is not advisable without testing and oncology input.
- If you have Hashimoto's thyroiditis, the selenium-for-thyroid evidence (200 µg/day Se-yeast or selenomethionine, 3–6 months) is a separate clinical use with its own risk-benefit calculus. Test before and after — many Hashimoto's patients reach 150–180 ng/mL during treatment.
- If you are on hemodialysis, total parenteral nutrition without selenium, or have severe malabsorption, you are at real risk of selenium deficiency and supplementation under medical supervision is appropriate.
- Do not chronically exceed 200 µg/day of supplemental selenium on top of dietary intake without monitoring. Do not exceed 400 µg/day of total intake (food + supplement).
If you are healthy, eat a varied diet that includes seafood or meat, and live in a country with adequate soil selenium (most of North America, much of Asia and South America), the highest-value selenium intervention is almost certainly: do not supplement, eat seafood twice a week, eat one or two Brazil nuts a week (not daily), and check serum selenium once if you are curious or have a relevant family history. The "more is better" intuition that motivated SELECT was the costly mistake; the modern target is "enough, and not more."
Research Papers and References
- Clark LC, Combs GF Jr, Turnbull BW, et al. Effects of selenium supplementation for cancer prevention in patients with carcinoma of the skin: a randomized controlled trial (the NPC trial). JAMA. 1996;276(24):1957–63. PMID 8971064. doi:10.1001/jama.1996.03540240035027
- Lippman SM, Klein EA, Goodman PJ, et al. Effect of selenium and vitamin E on risk of prostate cancer and other cancers: the Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA. 2009;301(1):39–51. PMID 19066370. doi:10.1001/jama.2008.864
- Klein EA, Thompson IM Jr, Tangen CM, et al. Vitamin E and the risk of prostate cancer: the Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA. 2011;306(14):1549–56. PMID 21990298. doi:10.1001/jama.2011.1437
- Kristal AR, Darke AK, Morris JS, et al. Baseline selenium status and effects of selenium and vitamin E supplementation on prostate cancer risk. J Natl Cancer Inst. 2014;106(3):djt456. PMID 24563519. doi:10.1093/jnci/djt456
- Vinceti M, Filippini T, Del Giovane C, et al. Selenium for preventing cancer. Cochrane Database Syst Rev. 2018;1:CD005195. PMID 29376219. doi:10.1002/14651858.CD005195.pub4
- Stranges S, Marshall JR, Natarajan R, et al. Effects of long-term selenium supplementation on the incidence of type 2 diabetes: a randomized trial. Ann Intern Med. 2007;147(4):217–23. PMID 17636652. doi:10.7326/0003-4819-147-4-200708210-00175
- Duffield-Lillico AJ, Dalkin BL, Reid ME, et al. Selenium supplementation, baseline plasma selenium status and incidence of prostate cancer: an analysis of the complete treatment period of the Nutritional Prevention of Cancer Trial. BJU Int. 2003;91(7):608–12. PMID 12424151. doi:10.1046/j.1464-410X.2003.04167.x
- Hughes DJ, Fedirko V, Jenab M, et al. Selenium status is associated with colorectal cancer risk in the European Prospective Investigation of Cancer and Nutrition cohort. Int J Cancer. 2015;136(5):1149–61. PMID 26854929. doi:10.1002/ijc.29071
- Sayehmiri K, Azami M, Mohammadi Y, Soleymani A, Tardeh Z. The association between selenium and prostate cancer: a systematic review and meta-analysis. Asian Pac J Cancer Prev. 2018;19(6):1431–1437. PMID 29554930. doi:10.22034/APJCP.2018.19.6.1431
- Rayman MP. Selenium and human health. Lancet. 2012;379(9822):1256–68. PMID 22381456. doi:10.1016/S0140-6736(11)61452-9
- Hatfield DL, Tsuji PA, Carlson BA, Gladyshev VN. Selenium and selenocysteine: roles in cancer, health, and development. Trends Biochem Sci. 2014;39(3):112–20. PMID 24485058. doi:10.1016/j.tibs.2013.12.007
- Steinbrenner H, Speckmann B, Klotz LO. Selenoproteins: antioxidant selenoenzymes and beyond. Arch Biochem Biophys. 2016;595:113–9. PMID 27095225. doi:10.1016/j.abb.2015.06.024
Connections
- Selenium Overview
- Selenium Benefits Deep Dive
- Selenium for Antioxidant Defense
- Selenium for Cancer Prevention (Benefits Hub)
- Selenium for Immune Function
- Selenium and Thyroid Function
- Cancer (Oncology)
- Cancer Overview
- Glutathione
- Oxidative Stress
- Minerals Index
- Zinc
- Iodine
- Vitamin E
- Vitamin C
- Vitamin D
- Methionine
- Cysteine
- Type 2 Diabetes
- Selenium Lab Test