Erectile Dysfunction
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- References
- Research Papers
- Connections
- Featured Videos
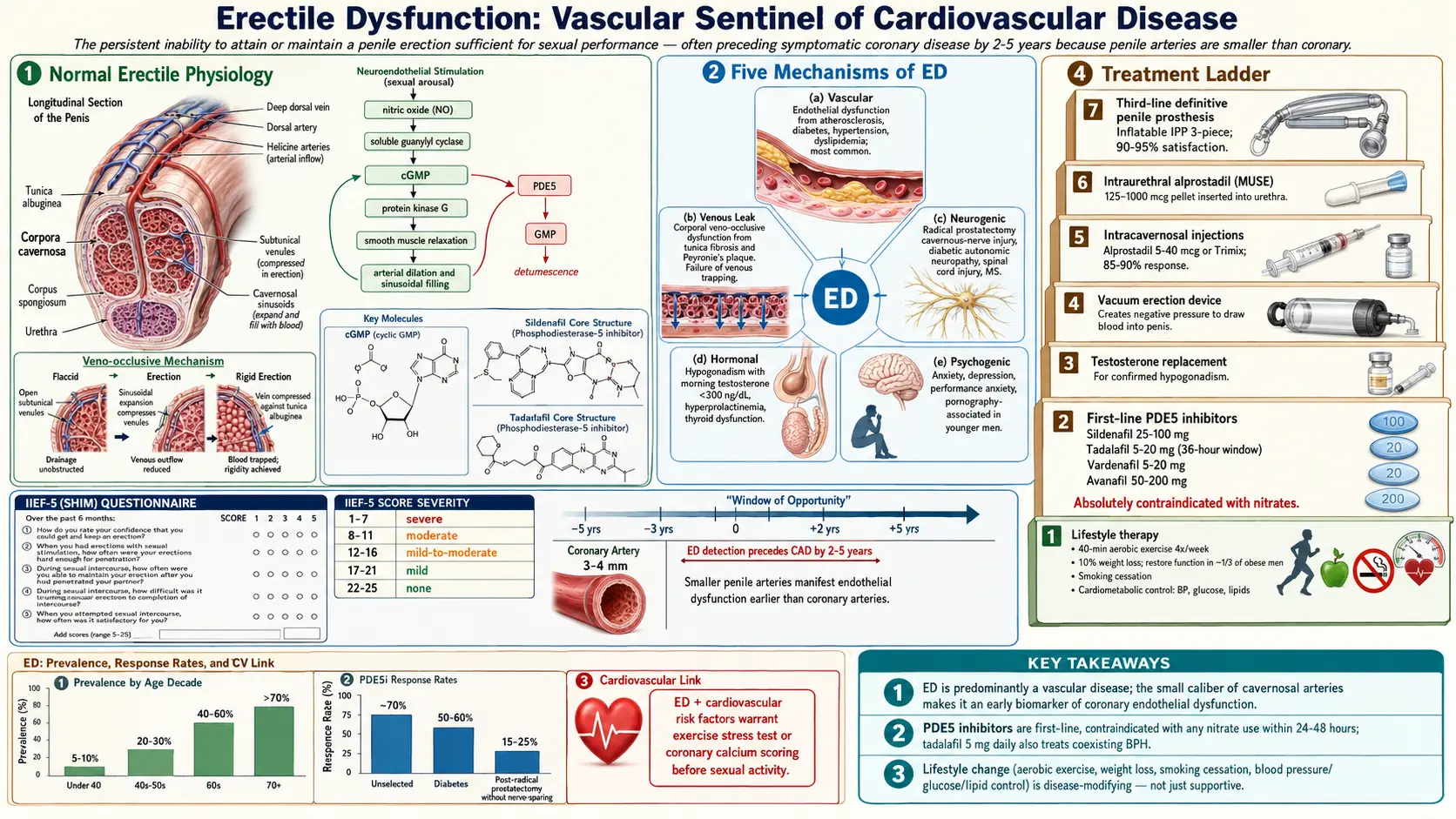
1. Overview
Erectile Dysfunction (ED) is defined as the persistent or recurrent inability to attain and/or maintain a penile erection sufficient for satisfactory sexual performance. The condition must be present for at least six months to distinguish it from transient situational difficulty. ED is among the most prevalent male sexual disorders worldwide and has profound implications for self-esteem, relationship quality, and overall psychological well-being.
Beyond its direct impact on sexual health, ED carries important clinical significance as a harbinger of systemic cardiovascular disease. Because the penile arteries (cavernosal diameter 1–2 mm) are smaller than coronary arteries (3–4 mm) and affected by endothelial dysfunction at earlier stages, ED often precedes symptomatic coronary artery disease by 2–5 years, providing a critical window for cardiovascular risk reduction.
2. Epidemiology
The Massachusetts Male Aging Study (MMAS) — a landmark community-based cohort — reported that approximately 52% of men aged 40–70 years experience some degree of ED, with complete ED in 10%, moderate in 25%, and minimal in 17%. Global prevalence estimates suggest that over 300 million men worldwide will be affected by 2025, reflecting aging populations and rising rates of metabolic risk factors.
Prevalence increases sharply with age: approximately 5–10% in men under 40, 20–30% in men aged 40–59, 40–60% in men aged 60–69, and over 70% in men aged 70 and above. Importantly, younger men (under 40) appear to represent a growing proportion of clinical presentations, with psychogenic and lifestyle factors predominating. Men with diabetes, cardiovascular disease, hypogonadism, or depression carry severalfold higher prevalence than age-matched controls.
3. Pathophysiology
Penile erection is a neurovascular event modulated by psychological, hormonal, and local vascular factors.
Normal Erectile Physiology
Sexual stimulation activates parasympathetic (S2–S4) and non-adrenergic non-cholinergic (NANC) nerve fibers, releasing nitric oxide (NO) from cavernous nerve terminals and vascular endothelium. NO activates soluble guanylyl cyclase, increasing cyclic guanosine monophosphate (cGMP), which activates protein kinase G. PKG phosphorylates myosin light chain phosphatase and potassium channels, reducing intracellular calcium and causing smooth muscle relaxation in the corpora cavernosa and helicine arteries. Increased arterial inflow, engorgement of the sinusoidal spaces, and compression of emissary veins against the tunica albuginea produce the veno-occlusive mechanism responsible for sustained erection.
Detumescence is mediated by sympathetic activation (norepinephrine-mediated smooth muscle contraction) and phosphodiesterase type 5 (PDE5), which hydrolyzes cGMP, restoring basal vascular tone.
Mechanisms of ED
- Vascular (endothelial) dysfunction: The most common mechanism in organic ED. Reduced endothelial NO bioavailability — due to oxidative stress, dyslipidemia, hyperglycemia, or hypertension — impairs smooth muscle relaxation. Atherosclerotic plaques in the pudendal, iliac, or cavernosal arteries reduce arterial inflow.
- Venous leak (corporal veno-occlusive dysfunction): Structural changes in the tunica albuginea (fibrosis, reduced elastin) or cavernosal smooth muscle (fibrosis from ischemia, Peyronie's disease) prevent adequate veno-occlusion.
- Neurogenic mechanisms: Damage to cavernous nerve fibers (radical prostatectomy, pelvic radiation, diabetes-associated autonomic neuropathy, multiple sclerosis, spinal cord injury) abolishes NO release at the cavernosal nerve terminal.
- Hormonal mechanisms: Testosterone modulates expression of NOS isoforms, PDE5, and maintains cavernosal smooth muscle architecture. Hypogonadism reduces NO synthesis and increases corporal fibrosis. Hyperprolactinemia suppresses gonadotropin secretion and testosterone production.
- Psychogenic mechanisms: Anxiety, depression, relationship conflict, and performance anxiety activate the sympathetic nervous system, increasing adrenergic tone and inhibiting parasympathetic pro-erectile signaling. Central serotonergic and dopaminergic pathway dysregulation is also implicated.
4. Etiology and Risk Factors
Cardiovascular and Metabolic Risk Factors
- Atherosclerosis and hypertension: Endothelial dysfunction is the shared pathophysiologic link between ED and cardiovascular disease.
- Diabetes mellitus: Two to three times greater ED prevalence; combined endothelial, autonomic, and hormonal mechanisms; earlier onset by 10–15 years.
- Dyslipidemia: High LDL and low HDL reduce endothelial NO production via oxidized LDL-mediated eNOS inhibition.
- Obesity: Associated with hypogonadism (aromatization of androgens), insulin resistance, sleep apnea, and systemic endothelial dysfunction.
- Metabolic syndrome: Encompasses and amplifies multiple individual risk factors.
Hormonal Causes
- Hypogonadism: Primary (testicular) or secondary (pituitary/hypothalamic); testosterone deficiency impairs NO-mediated erectile mechanisms.
- Hyperprolactinemia: Pituitary adenoma (macro or micro) suppresses LH/FSH and testosterone.
- Thyroid dysfunction: Both hypothyroidism and hyperthyroidism are associated with ED through hormonal and vascular mechanisms.
Neurological Causes
- Radical prostatectomy (cavernous nerve injury): ED rates of 40–80% depending on surgical technique and nerve-sparing status.
- Pelvic radiation therapy
- Spinal cord injury (erections preserved with S2–S4 intact reflexogenic arc)
- Diabetic autonomic neuropathy
- Multiple sclerosis, Parkinson's disease, stroke
Medications
- Antihypertensives: beta-blockers (most), thiazide diuretics; calcium channel blockers and ACE inhibitors are relatively sparing
- Antidepressants: SSRIs, SNRIs, TCAs (reduced libido and orgasmic dysfunction predominate; direct ED less common)
- Antipsychotics: dopamine receptor blockade and hyperprolactinemia
- Antiandrogens (finasteride, dutasteride, spironolactone, GnRH agonists)
- Recreational drugs: alcohol, cocaine, heroin, cannabis (chronic use)
Psychogenic and Relationship Factors
- Performance anxiety, generalized anxiety disorder, depression
- Relationship conflict, partner sexual dysfunction
- Post-traumatic stress disorder (PTSD)
- Pornography-associated ED in younger men
Structural/Local Causes
- Peyronie's disease: fibrous plaque formation in the tunica albuginea causing penile curvature and pain
- Priapism-associated fibrosis
- Penile trauma
5. Clinical Presentation
The diagnosis of ED is primarily established by history. The chief complaint is the inability to achieve or maintain erections adequate for sexual intercourse. Clinicians should characterize:
- Onset (gradual versus sudden) and duration
- Presence or absence of morning/nocturnal erections (preserved spontaneous erections favor psychogenic etiology)
- Erection firmness, penetration ability, and maintenance during intercourse
- Libido (reduced libido suggests hormonal etiology)
- Ejaculatory and orgasmic function
- Relationship status and partner factors
International Index of Erectile Function (IIEF)
The IIEF-5 (Sexual Health Inventory for Men, SHIM) is the most widely used validated instrument for ED assessment. Five questions scored 1–5 yield a maximum of 25 points. Severity classification:
- Severe ED: 1–7
- Moderate ED: 8–11
- Mild-to-moderate ED: 12–16
- Mild ED: 17–21
- No ED: 22–25
The full 15-item IIEF assesses five domains: erectile function, orgasmic function, sexual desire, intercourse satisfaction, and overall satisfaction, providing a comprehensive sexual health profile.
6. Diagnosis
Laboratory Evaluation
Recommended initial laboratory assessment includes:
- Fasting glucose and HbA1c: Screen for diabetes mellitus
- Fasting lipid panel: Cardiovascular risk stratification
- Total testosterone (morning, 8–10 AM): Assesses hypogonadism; if low, confirm with repeat measurement and add free testosterone, LH, FSH, prolactin, and SHBG
- PSA: In age-appropriate men being considered for testosterone therapy
- Thyroid-stimulating hormone (TSH): If thyroid dysfunction suspected
- CBC, renal function, liver function: In selected patients with systemic disease history
Cardiovascular Risk Assessment
All men presenting with ED should be assessed for cardiovascular risk using the Princeton Consensus guidelines. Men with ED and intermediate cardiovascular risk (e.g., multiple risk factors but asymptomatic) may warrant exercise stress testing or coronary calcium scoring before initiating PDE5 inhibitor therapy or sexual activity.
Nocturnal Penile Tumescence and Rigidity (NPTR)
The RigiScan device measures penile tumescence and rigidity at the tip and base during sleep. Preserved nocturnal erections (three or more episodes per night, minimum 70% rigidity, duration 10+ minutes) differentiate psychogenic from organic ED with approximately 85% sensitivity. Primarily indicated in medicolegal cases or when the distinction significantly alters management.
Penile Duplex Doppler Ultrasound
Gold standard for assessing penile arterial inflow and venous leak. Performed after intracavernosal injection of a vasoactive agent (prostaglandin E1 10–20 mcg). Normal: peak systolic velocity (PSV) above 30 cm/s, end diastolic velocity (EDV) below 5 cm/s, resistive index (RI) above 0.9. Arterial insufficiency: PSV below 25 cm/s. Veno-occlusive dysfunction: EDV above 5 cm/s with RI below 0.75.
Arteriography and Dynamic Infusion Cavernosometry
Reserved for surgical candidates (young men with post-traumatic arterial insufficiency or veno-occlusive dysfunction) in whom revascularization or venous ligation is being considered.
7. Treatment
Lifestyle Modification and Risk Factor Management
As the most evidence-based intervention for ED related to cardiovascular risk factors:
- Exercise: Aerobic exercise 40 minutes four times weekly for 6 months restores erectile function in men with ED secondary to sedentary lifestyle, obesity, or metabolic syndrome (Esposito et al., JAMA 2004). Pelvic floor exercises (Kegel) improve venous occlusion and erection quality.
- Weight loss: A 10% weight reduction in obese men restores normal erectile function in approximately one-third of patients.
- Smoking cessation, alcohol reduction
- Optimization of systemic diseases: Glycemic control, blood pressure, and lipid management are first-line disease-modifying interventions.
Phosphodiesterase Type 5 Inhibitors (PDE5i)
PDE5 inhibitors are the first-line pharmacological treatment for ED of all etiologies except when contraindicated. They enhance endogenous NO/cGMP signaling by preventing cGMP hydrolysis, requiring sexual stimulation for effect.
- Sildenafil (Viagra): 25–100 mg on demand, 30–60 minutes before sexual activity; duration 4–6 hours; high-fat meals reduce absorption.
- Tadalafil (Cialis): 10–20 mg on demand (half-life 17–21 hours; window 36 hours) or 2.5–5 mg once daily (continuous dosing restores natural erectile spontaneity); also approved for BPH-LUTS.
- Vardenafil (Levitra): 5–20 mg on demand; pharmacokinetics similar to sildenafil.
- Avanafil (Stendra): 50–200 mg; fastest onset (15–30 minutes); fewer visual side effects due to minimal PDE6 inhibition.
Contraindications: concurrent organic nitrates or nicorandil (risk of severe hypotension); caution with alpha-blockers; absolute contraindication in men who have used nitrates within 24 hours (sildenafil/vardenafil) or 48 hours (tadalafil). Common side effects: headache, flushing, nasal congestion, dyspepsia, myalgia, and transient visual color changes (sildenafil — PDE6 cross-reactivity).
Response rates: approximately 70% of unselected ED patients; reduced in severe diabetes (50–60%), post-radical prostatectomy without nerve sparing (15–25%), and severe vascular disease.
Testosterone Replacement Therapy (TRT)
Indicated for men with confirmed hypogonadism (morning total testosterone persistently below 300 ng/dL with symptoms). TRT alone may restore erectile function in hypogonadal men; TRT combined with PDE5 inhibitors is synergistic. Available formulations include transdermal gels, intramuscular injections (testosterone cypionate/enanthate every 1–2 weeks or undecanoate every 10 weeks), subcutaneous pellets, and buccal systems.
Vacuum Erection Devices (VED)
Non-pharmacological, mechanical option creating negative pressure around the penis to draw blood into the corpora cavernosa; a constriction ring maintains erection. Particularly useful post-prostatectomy or for penile rehabilitation. Patient satisfaction approximately 70–90% with proper instruction; pivotal concerns include pivoting sensation, ejaculatory restriction, and ecchymosis.
Intracavernosal Injections (ICI)
Second-line therapy with response rates of 85–90%. Alprostadil (PGE1) 5–40 mcg, alone or in combination (Trimix: alprostadil + phentolamine + papaverine). Self-administered 10–20 minutes before intercourse. Side effects include penile pain (alprostadil), priapism (less than 1%), and corporal fibrosis with repeated use. Priapism (erection exceeding 4 hours) requires emergency aspiration and intracavernosal phenylephrine injection.
Intraurethral Alprostadil (MUSE)
Medicated urethral suppository delivering alprostadil (125–1000 mcg); absorbed through the urethral mucosa into the corpora. Less effective than ICI (response rate 40–50%) but less invasive. Urethral burning is common.
Penile Prosthesis (Implant)
Third-line, definitive surgical treatment offering highest satisfaction rates (patient and partner satisfaction approximately 90–95%). Indicated for ED refractory to or contraindicated for medical therapy.
- Inflatable penile prosthesis (IPP, 3-piece): Two corporal cylinders connected to a scrotal pump and abdominal reservoir. The AMS 700 and Coloplast Titan are FDA-approved devices. Provides on-demand erection with natural flaccidity when deflated.
- Malleable (semi-rigid) prosthesis: Bendable rods implanted bilaterally; simpler device; always semi-erect but can be positioned downward.
Infection is the most feared complication (1–3%); antibiotic coating (InhibiZone, Titan with hydrophilic coating) reduces infection rates to 0.5–1%. Mechanical revision rate approximately 5% at 5 years.
Psychological and Psychosexual Therapy
Cognitive behavioral sex therapy, mindfulness-based interventions, and couple therapy are indicated for psychogenic ED or when psychological factors complicate organic ED. Combination of PDE5i and psychotherapy is superior to either alone in men with significant performance anxiety.
Low-Intensity Extracorporeal Shockwave Therapy (Li-ESWT)
Acoustic shockwave energy stimulates neovascularization, endothelial cell proliferation, and cavernosal nerve regeneration, potentially modifying the underlying vascular disease rather than merely treating symptoms. Multiple randomized trials demonstrate IIEF score improvements of 4–6 points versus sham. Optimal protocol and patient selection (best results in mild-to-moderate vasculogenic ED) remain under investigation.
8. Complications
- Cardiovascular events: ED is an independent predictor of major adverse cardiovascular events (MACE), myocardial infarction, and all-cause mortality, with hazard ratios of 1.3–2.0 after confounders are controlled.
- Psychological impact: Untreated ED causes significant depression, anxiety, shame, and diminished masculine self-concept. Relationship breakdown and sexual avoidance are common.
- Priapism from treatment: Intracavernosal therapy-induced ischemic priapism causes permanent corporal fibrosis and worsened ED if not treated within 4–6 hours.
- Peyronie's disease: ED and Peyronie's coexist in 30–60% of men; corporal fibrosis from both conditions can become self-reinforcing.
- Partner sexual dysfunction: Female partners of men with ED have higher rates of sexual dysfunction, depression, and relationship dissatisfaction.
9. Prognosis
The prognosis for ED depends heavily on etiology, severity, and the presence and reversibility of underlying risk factors. Men with psychogenic ED in the absence of organic disease have excellent prognosis with psychotherapy and PDE5 inhibitors. Men who successfully modify cardiovascular risk factors (weight loss, exercise, smoking cessation, optimized glycemic control) experience clinically meaningful spontaneous improvement in erectile function in approximately 30–50% of cases.
Post-radical prostatectomy ED follows a predictable nerve-regeneration timeline; return of function typically occurs over 6–24 months and is strongly predicted by nerve-sparing technique, surgeon volume, and patient age and baseline erectile function. PDE5 inhibitor penile rehabilitation (daily low-dose tadalafil) may accelerate and maximize recovery by maintaining oxygenation of cavernosal smooth muscle during the nerve recovery period.
10. Prevention
- Cardiovascular health maintenance: Aggressive management of hypertension, diabetes, dyslipidemia, and obesity.
- Regular aerobic and pelvic floor exercise
- Smoking cessation
- Moderate alcohol intake
- Nerve-sparing radical prostatectomy and prostate-sparing radiation techniques
- PDE5 inhibitor penile rehabilitation after radical prostatectomy or pelvic radiation
- Medication review: Substituting ED-causing antihypertensives (beta-blockers, thiazides) with alternatives (ARBs, nebivolol) when clinically appropriate
- Mental health treatment: Early intervention for depression and anxiety reduces risk of secondary psychogenic ED
11. Recent Research and Advances
Stem cell therapy: Intracavernosal injection of adipose-derived stem cells (ADSCs), bone marrow-derived mesenchymal stem cells, and platelet-rich plasma (PRP) is under clinical investigation for neurogenic and vasculogenic ED. Phase I/II trials show safety and preliminary efficacy signals for cavernous nerve injury models.
Gene therapy: Adeno-associated virus (AAV)-mediated delivery of hSlo (human BKCa potassium channel gene) demonstrated sustained (3-year) ED improvement in a Phase I trial (Christ et al., 2009). VEGF gene therapy targeting cavernosal angiogenesis is in early trials.
Melanocortin agonists: Bremelanotide (PT-141), a melanocortin receptor agonist acting centrally, is approved for hypoactive sexual desire disorder in women and shows promise for psychogenic and mixed ED through non-NO-dependent central pro-erectile mechanisms.
Topical alprostadil (Vitaros): Cream formulation applied to the urethral meatus; approved in Europe and Canada; avoids injection while providing local vasodilation.
Pulsed electromagnetic field therapy (PEMF) and low-intensity laser therapy: Emerging non-invasive modalities targeting cavernosal endothelial regeneration with phase II trial data suggesting additive benefit to PDE5 inhibitors.
Gut microbiome and ED: Reduced bacterial diversity and altered short-chain fatty acid production are linked to endothelial dysfunction and testosterone metabolism, opening microbiome-targeted interventions as a future direction.
12. References
- Feldman HA, Goldstein I, Hatzichristou DG, Krane RJ, McKinlay JB. Impotence and its medical and psychosocial correlates: results of the Massachusetts Male Aging Study. J Urol. 1994;151(1):54–61.
- Burnett AL, Nehra A, Breau RH, et al. Erectile dysfunction: AUA guideline. J Urol. 2018;200(3):633–641.
- Montorsi P, Ravagnani PM, Galli S, et al. Association between erectile dysfunction and coronary artery disease: matching the right target with the right test in the right patient. Eur Urol. 2006;50(4):721–731.
- Esposito K, Giugliano F, Di Palo C, et al. Effect of lifestyle changes on erectile dysfunction in obese men: a randomized controlled trial. JAMA. 2004;291(24):2978–2984.
- Rosen RC, Riley A, Wagner G, Osterloh IH, Kirkpatrick J, Mishra A. The international index of erectile function (IIEF): a multidimensional scale for assessment of erectile dysfunction. Urology. 1997;49(6):822–830.
- Buvat J, Maggi M, Guay A, Torres LO. Testosterone deficiency in men: systematic review and standard operating procedures for diagnosis and treatment. J Sex Med. 2013;10(1):245–284.
- Hatzimouratidis K, Amar E, Eardley I, et al. Guidelines on male sexual dysfunction: erectile dysfunction and premature ejaculation. Eur Urol. 2010;57(5):804–814.
- Carson CC, Rajfer J, Eardley I, et al. The efficacy and safety of tadalafil: an update. BJU Int. 2004;93(9):1276–1281.
- Mulhall JP, Brock G, Oger S, Elion-Mboussa A, Kissel JD, Viktrup L. An open-label extension study of tadalafil once daily for the treatment of erectile dysfunction. J Sex Med. 2015;12(6):1407–1416.
- Montague DK, Jarow JP, Broderick GA, et al. Chapter 1: the management of erectile dysfunction: an AUA update. J Urol. 2005;174(1):230–239.
- Fode M, Ohl DA, Ralph D, Sonksen J. Penile rehabilitation after radical prostatectomy: what the evidence really says. BJU Int. 2013;112(7):998–1008.
- Fojecki GL, Tiessen S, Osther PJ. Effect of low-energy linear shockwave therapy on erectile dysfunction: a double-blinded, sham-controlled, randomized clinical trial. J Sex Med. 2017;14(1):106–112.
- Yafi FA, Jenkins L, Albersen M, et al. Erectile dysfunction. Nat Rev Dis Primers. 2016;2:16003.
- Araujo AB, Travison TG, Ganz P, et al. Erectile dysfunction and mortality. J Sex Med. 2009;6(9):2445–2454.
- Shamloul R, Ghanem H. Erectile dysfunction. Lancet. 2013;381(9861):153–165.
- Mulhall JP, Goldstein I, Bushmakin AG, Cappelleri JC, Hvidsten K. Validation of the erection hardness score. J Sex Med. 2007;4(6):1626–1634.
Research Papers
- Clinical trials on erectile dysfunction treatment — PubMed search
- PDE5 inhibitors (sildenafil, tadalafil) — PubMed search
- ED as a marker of cardiovascular disease — PubMed search
- Low-intensity shockwave therapy for ED — PubMed search
- Testosterone replacement and erectile function — PubMed search
- Penile prosthesis for refractory ED — PubMed search
- Lifestyle and exercise interventions for ED — PubMed search
- L-arginine and nitric oxide pathway in erectile function — PubMed search
Connections
- Hypertension
- Arginine
- Benign Prostatic Hyperplasia
- Low Testosterone / TRT
- Zinc
- Ginseng
- Vitamin D3
- Testosterone Test
- Diabetes
- Depression
- Anxiety
- Obesity
- Cardiovascular Disease
- Metabolic Syndrome
- Atherosclerosis
- Fasting
- Peripheral Artery Disease
- Prostate Conditions