Benign Prostatic Hyperplasia
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation and IPSS Scoring
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- References
- Research Papers
- Connections
1. Overview
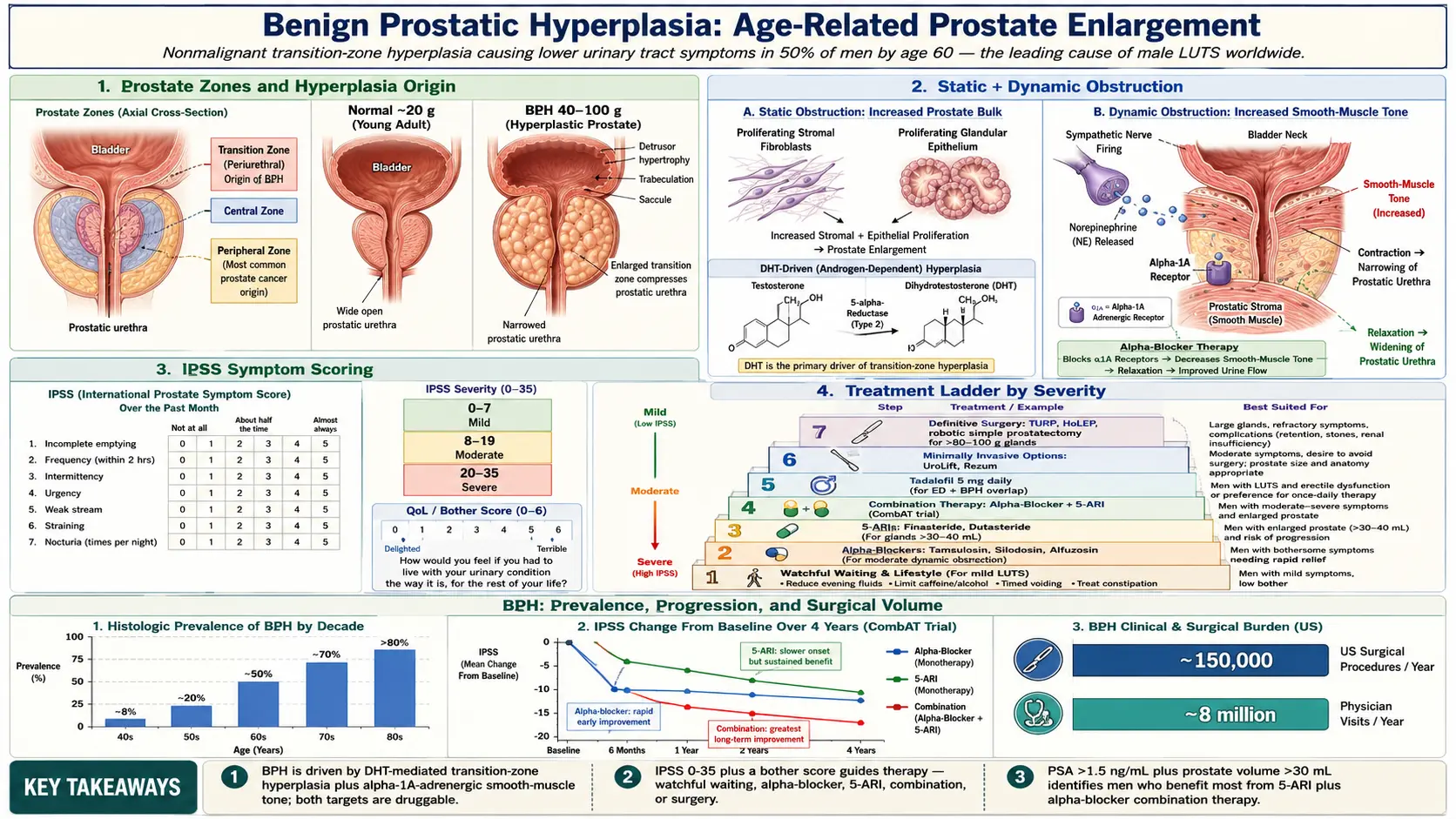
Benign Prostatic Hyperplasia (BPH) is a nonmalignant enlargement of the prostate gland resulting from the proliferation of both stromal and epithelial cells within the transition zone. It is among the most prevalent conditions affecting aging men and represents the leading cause of lower urinary tract symptoms (LUTS) in the male population. BPH-related LUTS significantly impair quality of life and impose a substantial economic burden on healthcare systems worldwide.
The prostate gland normally weighs approximately 20 grams in young adulthood. In BPH, this weight may increase to 40–100 grams or more, leading to partial or complete obstruction of the bladder outlet. The condition is distinct from prostate cancer, though both may coexist and share some overlapping symptomatology.
2. Epidemiology
BPH is strongly age-dependent. Histological evidence of hyperplasia is present in approximately 8% of men in their 40s, rising to over 80% by the ninth decade of life. Clinically significant BPH — defined by bothersome LUTS requiring intervention — affects roughly 50% of men by age 60 and up to 90% by age 85.
Global prevalence estimates suggest that over 200 million men worldwide are affected. In the United States alone, BPH accounts for more than 8 million physician office visits annually and approximately 150,000 surgical procedures. Age-standardized incidence rates are broadly similar across ethnic groups, though Black men may develop the condition earlier and with greater severity. The condition appears less prevalent in Asian populations, possibly reflecting dietary and hormonal differences.
3. Pathophysiology
The pathophysiology of BPH involves two principal components: static obstruction from glandular enlargement and dynamic obstruction mediated by smooth muscle tone.
Static component: Hyperplasia originates in the periurethral transition zone. Both stromal fibroblasts and smooth muscle cells proliferate abnormally, accompanied by glandular epithelial hyperplasia. The resulting nodular enlargement compresses the prostatic urethra, increasing outflow resistance. Dihydrotestosterone (DHT), derived from testosterone via 5-alpha-reductase (5-AR) types I and II, is the primary androgen driving prostatic growth. DHT binds androgen receptors with approximately fivefold greater affinity than testosterone and remains the key intraprostatic growth stimulus throughout life.
Dynamic component: Alpha-1 adrenergic receptors (predominantly the alpha-1A subtype) are densely expressed in prostatic stromal smooth muscle and the bladder neck. Sympathetic activation increases smooth muscle tone, further narrowing the urethral lumen independent of gland size. This dynamic element explains why alpha-blocker therapy produces rapid symptomatic relief even without reducing prostate volume.
Bladder changes: Chronic bladder outlet obstruction produces detrusor hypertrophy, trabeculation, and saccule or diverticulum formation. Over time, detrusor decompensation leads to impaired contractility, urinary retention, and upper tract changes including hydroureteronephrosis.
Inflammatory pathways: Emerging evidence implicates chronic intraprostatic inflammation — mediated by T-lymphocytes, macrophages, and pro-inflammatory cytokines (IL-6, IL-8, TNF-alpha) — in driving epithelial and stromal proliferation, providing a mechanistic link between metabolic syndrome, obesity, and BPH progression.
4. Etiology and Risk Factors
- Age: The single strongest risk factor; BPH is virtually absent before age 40 and near-universal among octogenarians.
- Androgens: Men castrated before puberty do not develop BPH, confirming the essential role of testicular androgens. However, absolute serum testosterone levels do not correlate linearly with BPH severity.
- Genetic predisposition: First-degree relatives of men with BPH requiring surgery have a fourfold increased risk. Candidate genes include those encoding 5-alpha-reductase, androgen receptor, and steroid hormone-binding globulin.
- Metabolic syndrome and obesity: Hyperinsulinemia stimulates prostatic growth via insulin-like growth factor (IGF-1) signaling. Adipose tissue aromatizes androgens to estrogens, potentially altering the androgen-to-estrogen ratio within the prostate.
- Cardiovascular disease: Shared inflammatory and metabolic pathways link BPH with hypertension, dyslipidemia, and atherosclerosis.
- Diet: High-fat, high-protein Western diets are associated with increased BPH risk. Lycopene (from tomatoes), soy isoflavones, zinc, and selenium may be protective.
- Physical inactivity: Sedentary lifestyle correlates with greater BPH symptom severity and faster progression.
- Ethnicity: Higher prevalence and earlier onset reported in Black men; lower rates in Asian men.
- Featured Videos
5. Clinical Presentation and IPSS Scoring
LUTS attributable to BPH are classified as obstructive (voiding) or irritative (storage) symptoms.
Obstructive (Voiding) Symptoms
- Weak or intermittent urinary stream
- Straining to initiate micturition
- Prolonged voiding time
- Sense of incomplete bladder emptying
- Terminal dribbling
- Urinary retention (acute or chronic)
Irritative (Storage) Symptoms
- Urinary urgency and urgency incontinence
- Increased frequency (more than 8 voids per 24 hours)
- Nocturia (two or more episodes per night)
- Dysuria (less common)
International Prostate Symptom Score (IPSS)
The IPSS (AUA Symptom Score) is the validated seven-question instrument used to quantify LUTS severity and monitor treatment response. Each question scores 0–5 for a maximum of 35 points. An eighth question assesses quality of life impact (0–6). Interpretation:
- Mild symptoms: IPSS 0–7
- Moderate symptoms: IPSS 8–19
- Severe symptoms: IPSS 20–35
A minimum clinically important difference of 3 points is required to constitute meaningful symptomatic improvement. The IPSS guides therapeutic decision-making: watchful waiting for mild scores, pharmacotherapy for moderate scores, and surgical intervention for severe scores or when complications are present.
6. Diagnosis
History and Physical Examination
A focused history elicits symptom duration, nocturia frequency, urgency episodes, prior urologic procedures, and medication use (particularly anticholinergics, decongestants, and diuretics that may worsen LUTS). Digital rectal examination (DRE) assesses prostate size, consistency, symmetry, and presence of nodules. DRE has poor correlation with actual prostate volume but remains important for excluding malignancy.
Urinalysis and Urine Culture
Urinalysis identifies hematuria, pyuria, glycosuria, and proteinuria, helping exclude infection, bladder malignancy, or diabetes mellitus as alternative diagnoses.
Prostate-Specific Antigen (PSA)
PSA measurement is recommended in men with life expectancy exceeding 10 years who are candidates for intervention. PSA correlates with prostate volume (approximately 0.3 ng/mL per gram of benign tissue) and predicts BPH progression risk. A PSA above 1.5 ng/mL in a man with moderate-to-severe LUTS identifies a higher-risk group warranting combination pharmacotherapy. PSA density, PSA velocity, and free/total PSA ratios assist in differentiating BPH from prostate cancer.
Uroflowmetry
Non-invasive measurement of maximum flow rate (Qmax) and voided volume. A Qmax below 10 mL/s strongly suggests bladder outlet obstruction, while Qmax above 15 mL/s is generally reassuring. Post-void residual (PVR) urine volume measured by ultrasound complements uroflowmetry; PVR greater than 300 mL increases the risk of urinary retention and renal impairment.
Transrectal Ultrasound (TRUS)
Provides accurate prostate volume measurement, critical for treatment planning. Prostate volume guides selection between alpha-1A-selective antagonists alone (any size) versus 5-ARI addition (volume above 30–40 mL). TRUS is also used to guide prostate biopsies when malignancy is suspected.
Pressure-Flow Urodynamics
The gold standard for diagnosing bladder outlet obstruction versus detrusor underactivity. Indicated when diagnosis is uncertain, prior to invasive intervention, or when initial therapy fails. The Bladder Outlet Obstruction Index (BOOI = PdetQmax − 2Qmax) classifies obstruction as obstructed (BOOI above 40), equivocal (BOOI 20–40), or unobstructed (BOOI below 20).
Cystoscopy
Reserved for cases with hematuria, suspected urethral stricture, or prior to minimally invasive surgical therapy. Visualizes the bladder neck configuration, urethral length, trabeculation, and diverticula.
7. Treatment
Watchful Waiting and Lifestyle Modification
Men with mild LUTS (IPSS 0–7) or moderate LUTS without bother may be managed expectantly. Behavioral measures include fluid restriction in the evening, double voiding, bladder retraining, and avoidance of caffeine, alcohol, and medications that worsen voiding. Regular reassessment every 6–12 months is recommended.
Alpha-1 Adrenergic Receptor Blockers
Alpha-blockers reduce dynamic obstruction by relaxing prostate and bladder-neck smooth muscle. They are the most commonly prescribed initial pharmacotherapy. Available agents include:
- Tamsulosin (0.4–0.8 mg once daily) — alpha-1A selective; minimal cardiovascular side effects
- Silodosin (8 mg once daily) — highly alpha-1A selective; high rate of retrograde ejaculation
- Alfuzosin (10 mg once daily) — uroselective; low orthostatic hypotension risk
- Doxazosin and terazosin — non-selective; require dose titration due to blood pressure effects
Clinical benefit appears within days to weeks. Symptom score improvements average 4–6 IPSS points. Alpha-blockers do not reduce prostate volume or alter disease progression.
5-Alpha-Reductase Inhibitors (5-ARIs)
5-ARIs block conversion of testosterone to DHT, inducing prostatic apoptosis and reducing gland volume by 20–30% over 6–12 months. Indicated for men with prostate volume above 30–40 mL or PSA above 1.5 ng/mL. Options include:
- Finasteride (5 mg daily) — inhibits 5-AR type II only
- Dutasteride (0.5 mg daily) — inhibits both 5-AR types I and II; greater DHT suppression (approximately 94% versus 70%)
Side effects include reduced libido, erectile dysfunction, ejaculatory disorders, and gynecomastia. 5-ARIs reduce PSA by approximately 50%, requiring PSA doubling for proper cancer screening interpretation after 6–12 months of therapy. Long-term use (MTOPS, CombAT trials) reduces acute urinary retention risk by approximately 60% and the need for surgical intervention.
Combination Therapy
The combination of an alpha-blocker plus a 5-ARI (e.g., dutasteride/tamsulosin, marketed as Jalyn) is superior to monotherapy for symptomatic relief and disease modification in men with enlarged prostates and moderate-to-severe LUTS. The CombAT trial demonstrated significantly greater IPSS reduction and reduced risk of clinical progression with combination therapy over 4 years.
Phosphodiesterase-5 Inhibitors
Tadalafil 5 mg once daily is approved for BPH-associated LUTS, either as monotherapy or combined with an alpha-blocker (with caution for hypotension). PDE5 inhibitor activity relaxes smooth muscle in the prostate, bladder neck, and urethra via the nitric oxide/cGMP pathway. It offers the additional benefit of treating co-existing erectile dysfunction.
Anticholinergic and Beta-3 Agonist Agents
When storage symptoms (urgency, frequency, nocturia) predominate despite adequate relief of outlet obstruction, an antimuscarinic agent (e.g., solifenacin, tolterodine) or a beta-3 agonist (mirabegron) may be added. Careful monitoring of PVR is required given the risk of urinary retention in men with significant outlet obstruction.
Minimally Invasive Surgical Therapies (MISTs)
- Prostatic Urethral Lift (UroLift): Implantable anchors retract obstructing lateral lobes without tissue removal. Preserves erectile and ejaculatory function. Indicated for prostate volumes 30–80 mL without prominent median lobe.
- Convective Water Vapor Ablation (Rezum): Steam energy induces coagulative necrosis of prostatic tissue. Effective for prostates 30–80 mL including median lobe. Retrograde ejaculation occurs in approximately 4% versus 90% with TURP.
- Transurethral Microwave Thermotherapy (TUMT): Microwave energy ablates periurethral tissue. Less durable than surgical options; use is declining.
- Prostatic Artery Embolization (PAE): Interventional radiology technique selectively embolizing prostatic arterial supply. Suitable for very large glands or surgical risk cases. Long-term durability data still accumulating.
Surgical Treatment
- Transurethral Resection of the Prostate (TURP): Historical gold standard. Monopolar or bipolar electrosurgical resection of obstructing tissue. Achieves approximately 85–90% symptomatic improvement. Retrograde ejaculation occurs in 65–90% of patients. Risk of TUR syndrome (dilutional hyponatremia) mitigated by use of isotonic irrigation with bipolar TURP.
- Laser Therapies:
- Holmium Laser Enucleation of the Prostate (HoLEP): Size-independent enucleation with outcomes equivalent to open prostatectomy and superior durability to TURP. Steep learning curve.
- Thulium Laser Enucleation (ThuLEP) and Diode Laser Vaporization (PVP): Alternative laser modalities with comparable efficacy.
- Open Simple Prostatectomy (Millin or Freyer): Reserved for glands greater than 80–100 grams. Increasingly replaced by robotic-assisted simple prostatectomy.
- Robotic-Assisted Simple Prostatectomy (RASP): Provides equivalent efficacy to open prostatectomy with reduced blood loss, shorter hospitalization, and faster recovery.
8. Complications
- Acute urinary retention (AUR): Annual incidence of approximately 1–2% in symptomatic men; 10-year cumulative risk approaches 25% in untreated men with IPSS above 7 and PSA above 1.4 ng/mL.
- Chronic urinary retention: High-pressure or low-pressure chronic retention may progress silently to bilateral hydronephrosis and obstructive nephropathy.
- Urinary tract infections and epididymo-orchitis: Stasis of residual urine promotes bacterial colonization.
- Bladder stones: Crystallization of urinary salts in stagnant residual urine.
- Bladder dysfunction: Detrusor overactivity, impaired compliance, and eventually detrusor failure (acontractile bladder).
- Hematuria: Varicose submucosal veins of the enlarged prostate may bleed spontaneously or after minor trauma; 5-ARI therapy reduces hematuria from BPH.
- Renal insufficiency: Obstructive nephropathy from long-standing high-pressure retention; reversible with timely relief of obstruction.
9. Prognosis
BPH is a slowly progressive condition in most men. Approximately 25% of men with moderate LUTS experience symptomatic improvement spontaneously over time, while approximately 50% remain stable. The remaining quarter progress. Key predictors of adverse progression include prostate volume above 30 mL, PSA above 1.5 ng/mL, maximum flow rate below 12 mL/s, and an elevated post-void residual.
With appropriate pharmacotherapy or surgical intervention, the vast majority of men achieve substantial and durable symptom relief. TURP and HoLEP produce the most durable long-term outcomes, with reoperation rates of less than 5% and less than 2% per decade, respectively. Quality of life is meaningfully restored in most patients.
BPH itself does not increase the risk of prostate cancer; however, concurrent prostate cancer requires independent evaluation and management.
10. Prevention
While the age-dependent nature of BPH limits complete prevention, modifiable risk factors offer meaningful opportunities for symptom attenuation and disease modification:
- Weight management: Obesity increases intraprostatic estrogen and IGF-1 signaling; weight reduction is associated with reduced LUTS severity.
- Regular physical activity: Exercise reduces sympathetic tone, insulin resistance, and systemic inflammation, all of which contribute to BPH pathogenesis. Men who exercise vigorously have approximately 25% lower risk of severe LUTS.
- Mediterranean diet: High consumption of vegetables, legumes, and whole grains, combined with reduced red meat and saturated fat intake, is associated with lower BPH risk.
- Lycopene: Dietary and supplemental lycopene may reduce intraprostatic oxidative stress and DHT levels.
- Reduction of alcohol and caffeine: Both increase diuresis, bladder irritability, and urgency symptoms.
- Statin use: Epidemiologic data suggest that HMG-CoA reductase inhibitors may reduce prostate inflammation and modestly lower BPH risk, though causality is unconfirmed.
11. Recent Research and Advances
Aquablation (AQUABEAM): Robotic waterjet ablation guided by real-time ultrasound imaging offers size-independent tissue resection with a favorable ejaculatory preservation profile (approximately 90% preservation rate). Multicenter randomized trials (WATER, WATER II) demonstrate non-inferiority to TURP in symptom relief with superior sexual function outcomes.
iTind (Temporary Implantable Nitinol Device): A retrievable nitinol device placed for 5–7 days that reshapes the prostatic urethra by mechanical dilation and controlled ischemia. Approved in 2021; pivotal trial data show IPSS improvement of approximately 9 points at 12 months with no retrograde ejaculation.
Novel pharmacological targets: Research into inhibitors of interleukin-6, the androgen receptor splice variant AR-V7, and Rho-kinase (ROCK) inhibitors is expanding the mechanistic understanding of BPH beyond the DHT/androgen axis.
Microbiome and BPH: Emerging data implicate gut and intraprostatic microbiome dysbiosis in BPH-associated inflammation. The role of Cutibacterium acnes and other microorganisms within prostatic tissue is under active investigation.
Genetic and biomarker studies: Genome-wide association studies have identified susceptibility loci including variants near GATA3, TERT, and steroid metabolism genes. Urinary biomarkers (e.g., exosomal miRNA signatures) are being evaluated as non-invasive predictors of BPH progression and treatment response.
12. References
- Roehrborn CG. Benign prostatic hyperplasia: an overview. Rev Urol. 2005;7(Suppl 9):S3–S14. PMID 16985902.
- McVary KT, Roehrborn CG, Avins AL, et al. Update on AUA guideline on the management of benign prostatic hyperplasia. J Urol. 2011;185(5):1793–1803.
- Foster HE, Barry MJ, Dahm P, et al. Surgical management of lower urinary tract symptoms attributed to benign prostatic hyperplasia: AUA guideline. J Urol. 2019;200(3):612–619.
- Roehrborn CG, Siami P, Barkin J, et al. The effects of combination therapy with dutasteride and tamsulosin on clinical outcomes in men with symptomatic benign prostatic hyperplasia: 4-year results from the CombAT study. Eur Urol. 2010;57(1):123–131.
- McConnell JD, Roehrborn CG, Bautista OM, et al. The long-term effect of doxazosin, finasteride, and combination therapy on the clinical progression of benign prostatic hyperplasia. N Engl J Med. 2003;349(25):2387–2398.
- Gilling P, Barber N, Bidair M, et al. WATER: a double-blind, randomized, controlled trial of aquablation vs transurethral resection of the prostate in benign prostatic hyperplasia. J Urol. 2018;199(5):1252–1261.
- Parsons JK, Messer K, White M, Barrett-Connor E, Bauer DC, Marshall LM. Obesity increases and physical activity decreases lower urinary tract symptom risk in older men. Eur Urol. 2011;60(6):1173–1180.
- Nickel JC, Saad F. The American Urological Association symptom index for benign prostatic hyperplasia. J Urol. 1992;148(5):1549–1557.
- Emberton M, Cornel EB, Bassi PF, et al. Benign prostatic hyperplasia as a progressive disease: a guide to the risk factors and options for medical management. Int J Clin Pract. 2008;62(7):1076–1086.
- Lerner LB, McVary KT, Barry MJ, et al. Management of lower urinary tract symptoms attributed to benign prostatic hyperplasia: AUA guideline 2021. J Urol. 2021;206(4):806–817.
- Stroup SP, Palazzi-Churas K, Kopp RP, Parsons JK. Trends in adverse events of benign prostatic hyperplasia (BPH) in the USA, 1998 to 2008. BJU Int. 2012;109(1):84–87.
- McNeal JE. The zonal anatomy of the prostate. Prostate. 1981;2(1):35–49.
- Kaplan SA, Roehrborn CG, Rovner ES, Carlsson M, Bavendam T, Guan Z. Tolterodine and tamsulosin for treatment of men with lower urinary tract symptoms and overactive bladder: a randomized controlled trial. JAMA. 2006;296(19):2319–2328.
- Cindolo L, Pirozzi L, Fanizza C, et al. Drug adherence and clinical outcomes for patients under pharmacological therapy for lower urinary tract symptoms related to benign prostatic hyperplasia. Eur Urol. 2015;68(3):418–425.
- Wein AJ, Kavoussi LR, Partin AW, Peters CA, eds. Campbell-Walsh-Wein Urology. 12th ed. Elsevier; 2020. Chapter 147: Benign Prostatic Hyperplasia.
- Osterberg EC, Osterberg N, Truesdale MD, Shindel AW. The role of phosphodiesterase inhibitors in BPH and LUTS. Curr Urol Rep. 2014;15(7):424.
Research Papers
- Clinical trials on benign prostatic hyperplasia treatment — PubMed search
- Tamsulosin for BPH (randomized controlled trials) — PubMed search
- Finasteride and dutasteride in prostate disease — PubMed search
- Saw palmetto for BPH — PubMed search
- Holmium laser enucleation of the prostate (HoLEP) — PubMed search
- Prostatic urethral lift (UroLift) clinical trials — PubMed search
- Rezum water vapor therapy for BPH — PubMed search
- Aquablation for BPH — PubMed search
Connections
- Erectile Dysfunction
- Urinary Tract Infections
- Zinc
- Saw Palmetto
- Prostate Conditions
- Low Testosterone / TRT
- Testosterone Test
- PSA Test
- Obesity
- Hypertension
- Diabetes
- Metabolic Syndrome
- Urinalysis
- Bladder Cancer
- Kidney Stones
- Interstitial Cystitis