Secondary Osteoporosis Causes and Screening
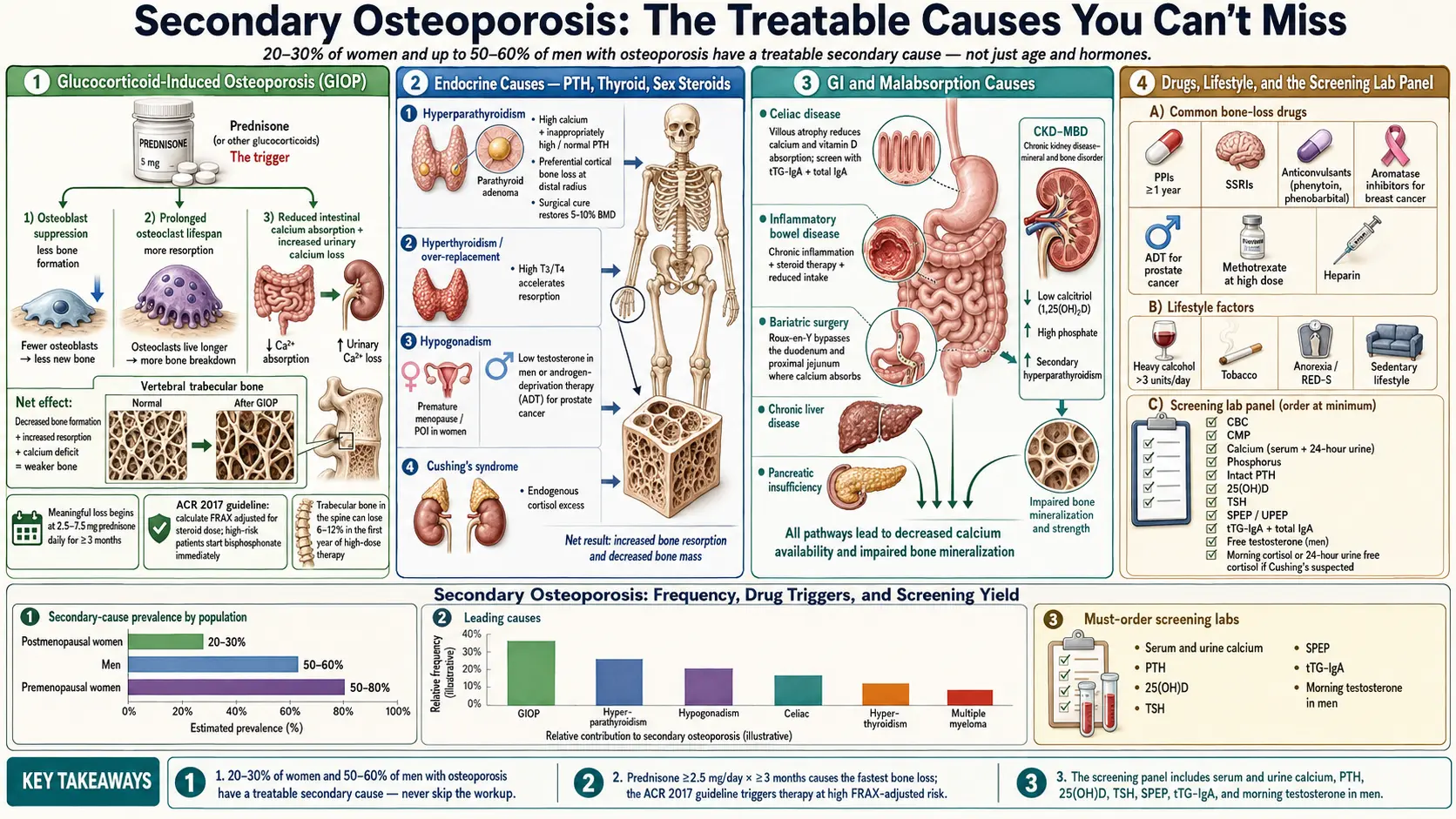
Table of Contents
- What Secondary Osteoporosis Means
- Who Should Be Screened for Secondary Causes
- Glucocorticoid-Induced Osteoporosis (GIOP)
- Hyperparathyroidism — Primary and CKD-MBD
- Hyperthyroidism and Over-Replacement
- Hypogonadism in Men — Including ADT
- Hypogonadism in Women — Amenorrhea, POI, Turner
- Celiac Disease and IBD
- Post-Gastric-Bypass and Bariatric Bone Loss
- Anorexia Nervosa and the Female Athlete Triad / RED-S
- Multiple Myeloma and Other Malignancies
- Chronic Kidney Disease and Renal Osteodystrophy
- The Diabetes Paradox — T1D and T2D
- Aromatase Inhibitors and ADT for Cancer
- SSRIs, PPIs, and Other Medications
- Alcohol and Tobacco
- The Screening Lab Panel
- Treating the Cause, Not Just the Bone
- Key Research Papers
- Connections
- Featured Videos
What Secondary Osteoporosis Means
"Primary" osteoporosis is what happens to most postmenopausal women and older men: estrogen or testosterone declines, bone turnover shifts toward resorption, and the skeleton slowly thins. "Secondary osteoporosis" means a specific, identifiable disease or medication is driving the bone loss — not just age and hormones.
This distinction matters because secondary causes are treatable, often with dramatic bone recovery once the underlying problem is addressed. A 32-year-old with vertebral fractures is not going through early menopause — she might have untreated celiac disease, an eating disorder, or a pituitary tumor driving cortisol excess. A 55-year-old man with a hip fracture after a minor fall is not just "unlucky" — he may have low testosterone, undiagnosed multiple myeloma, or years of proton-pump-inhibitor use.
Studies of osteoporosis referral clinics consistently find that 20–30% of postmenopausal women and up to 50–60% of men diagnosed with osteoporosis have at least one secondary cause. In premenopausal women and younger men, the proportion is even higher. If your doctor only prescribed a bisphosphonate and never ordered a secondary workup, the workup is missing — push for it.
Who Should Be Screened for Secondary Causes
The Endocrine Society, American College of Rheumatology, and American Association of Clinical Endocrinologists broadly agree: screen for secondary causes in anyone with osteoporosis whose presentation does not fit a simple age-plus-menopause story. Red flags include:
- Premenopausal women and men under 50 with osteoporosis of any severity.
- Z-score below −2.0 at any age (Z-score compares you to age-matched peers; see the DEXA explainer).
- Fragility fractures — a fracture from a fall at standing height or less — especially vertebral compression fractures.
- Rapidly falling bone density on repeat DEXA, greater than the scanner's least significant change.
- Poor response to first-line therapy — continued bone loss on bisphosphonate or denosumab.
- Any clinical clue: unexplained weight loss, chronic diarrhea, kidney stones, amenorrhea, hypogonadal symptoms, long-term steroids, or a family history of endocrine tumors.
Glucocorticoid-Induced Osteoporosis (GIOP)
Prednisone, prednisolone, methylprednisolone, dexamethasone, and their inhaled or injected cousins are the single most common cause of drug-induced osteoporosis. GIOP is also the fastest — trabecular bone (the spongy bone in vertebrae and the hip neck) can lose 6–12% in the first year of high-dose therapy, most of it in the first three to six months.
The threshold is lower than patients expect. Meaningful bone loss begins at 2.5–7.5 mg of prednisone daily continued for three months or more. "Low-dose" maintenance prednisone for rheumatoid arthritis, polymyalgia rheumatica, asthma, or IBD is enough to push the skeleton into negative balance. Even intermittent high-dose bursts (pulse steroids for lupus flares, repeated epidural injections) accumulate risk.
Glucocorticoids cause bone loss by three mechanisms at once: they suppress osteoblast bone-formation activity, prolong osteoclast bone-resorption lifespan, and reduce intestinal calcium absorption while increasing urinary calcium loss. The result is a skeleton that both builds less and tears down more, with a calcium deficit on top.
ACR 2017 GIOP guideline thresholds. The American College of Rheumatology recommends fracture-risk stratification for anyone starting prednisone ≥ 2.5 mg/day for an expected duration of three months or more. Risk is calculated with FRAX adjusted upward for steroid dose (see the FRAX article). High-risk adults — prior fragility fracture, T-score ≤ −2.5, or FRAX 10-year major osteoporotic fracture risk ≥ 20% / hip risk ≥ 3% — should start pharmacologic therapy immediately, not wait for a follow-up DEXA.
First-line treatment options:
- Oral bisphosphonates — alendronate 70 mg weekly or risedronate 35 mg weekly — are first-line for most GIOP patients because of decades of evidence, low cost, and oral convenience.
- IV zoledronate 5 mg yearly is preferred when swallowing, adherence, or GI side effects are problems — common in IBD, post-transplant, or polypharmacy patients.
- Denosumab 60 mg subcutaneous every 6 months is a strong alternative, particularly with renal impairment (GFR below 35) where bisphosphonates are contraindicated. Warning: stopping denosumab causes a rebound wave of vertebral fractures — never stop without a bisphosphonate bridge.
- Teriparatide (daily injection, 20 mcg) or abaloparatide is preferred for very high-risk GIOP patients — those with prior vertebral fractures or T-score below −3.5 — because it actually builds new bone rather than slowing loss. See the anabolic agents article.
- Calcium 1000–1200 mg/day and vitamin D 800–2000 IU/day are non-negotiable adjuncts for every GIOP patient.
Whenever possible, the best GIOP treatment is dose reduction or replacement of the steroid. Steroid-sparing biologics in RA, IBD, and asthma have made this much more achievable than it was twenty years ago.
Hyperparathyroidism — Primary and CKD-MBD
The parathyroid glands — four pea-sized glands behind the thyroid — pump out parathyroid hormone (PTH) whenever blood calcium drops. PTH pulls calcium out of bone to restore serum levels. Chronic PTH excess chronically strip-mines the skeleton, with a special preference for cortical bone (the dense outer shell of long bones, like the radius at the wrist).
Primary hyperparathyroidism is usually caused by a single benign adenoma in one of the four glands, more common in women over 50. The classic blood picture is high calcium with inappropriately high or normal PTH. Many patients are asymptomatic and picked up on routine labs; others develop kidney stones, fatigue, constipation, depression, or osteoporosis. Surgical removal of the overactive gland (parathyroidectomy) reverses bone loss — patients often gain 5–10% BMD over the first two years after surgery.
CKD-MBD (chronic kidney disease – mineral and bone disorder) is secondary hyperparathyroidism driven by failing kidneys: low calcitriol, high phosphate, and low calcium trigger chronic PTH elevation. This is a separate beast with its own treatment ladder — phosphate binders, active vitamin D analogues (calcitriol, paricalcitol), and calcimimetics (cinacalcet). Standard bisphosphonate dosing is usually contraindicated when GFR is below 30. See the Kidney Disease page for the broader CKD-MBD picture.
Hyperthyroidism and Over-Replacement
Thyroid hormone directly accelerates bone turnover. When levels run high — whether from Graves' disease, toxic multinodular goiter, or over-replacement with levothyroxine in someone being treated for hypothyroidism — resorption outpaces formation and bone thins quickly.
The risk is not limited to overt disease. Subclinical hyperthyroidism (suppressed TSH with normal free T4) carries measurable fracture risk, particularly in postmenopausal women. If you are on levothyroxine and your TSH is consistently below 0.3–0.5 mIU/L, you are almost certainly on too much. Ask your prescriber to reduce the dose unless you have a documented reason for deliberate TSH suppression (some thyroid-cancer survivors are kept low-TSH on purpose — but that population needs extra bone protection).
For context on thyroid diseases in general, see Graves' disease and Hashimoto's thyroiditis (Hashimoto's patients become osteoporosis-relevant only when they are over-replaced).
Hypogonadism in Men — Including ADT
Testosterone supports bone in two ways: directly, through androgen receptors on osteoblasts, and indirectly, by being aromatized to estradiol — and estradiol turns out to be the dominant skeletal hormone in men too. Men with low testosterone lose bone steadily.
Common causes of male hypogonadism include: primary testicular failure (Klinefelter syndrome, prior mumps orchitis, testicular trauma, chemotherapy), pituitary dysfunction (prolactinomas, non-functioning adenomas, hemochromatosis), chronic opioid use, heavy long-term alcohol use, obesity-related secondary hypogonadism, and — most dramatically — androgen deprivation therapy (ADT) for prostate cancer.
ADT and bone. Leuprolide, goserelin, degarelix, and the newer androgen-receptor blockers (enzalutamide, abiraterone) produce near-total testosterone suppression. Men on ADT lose bone at 4–7% in the first year, with fracture risk roughly doubling over a few years. Every prostate-cancer patient starting ADT should get a baseline DEXA, adequate calcium/vitamin D, and — if the planned duration is beyond six to twelve months — consideration of denosumab 60 mg every 6 months or zoledronate. Denosumab at 60 mg has a specific FDA indication for this population.
For non-cancer hypogonadism, treatment of the underlying cause (testosterone replacement when appropriate, opioid weaning, weight loss in obesity-related cases) often reverses bone loss. Confirm low testosterone with at least two morning fasting total-testosterone measurements before assigning the diagnosis — single random levels swing too much to trust.
Hypogonadism in Women — Amenorrhea, POI, Turner
Estrogen is the master regulator of female bone mass. Any state that removes it for months to years produces measurable bone loss, and the younger the patient, the bigger the eventual deficit — because peak bone mass (usually reached by the late 20s) is itself incomplete.
- Primary ovarian insufficiency (POI) — menopause before age 40, whether spontaneous, autoimmune, post-chemotherapy, or genetic. These patients need hormone therapy through at least the natural age of menopause (~51) not because of vasomotor symptoms but specifically to protect their skeleton and cardiovascular system.
- Turner syndrome (45,X) — includes ovarian dysgenesis from childhood. Estrogen replacement must start in adolescence and continue through the natural menopausal age.
- Hypothalamic amenorrhea — from stress, extreme exercise, or low energy availability (see the RED-S section below).
- Hyperprolactinemia from prolactinomas or certain antipsychotics, which suppresses GnRH and causes amenorrhea plus bone loss.
- Long-term GnRH agonist therapy for endometriosis or fibroids (leuprolide) — the same drug used for ADT in men.
A woman in her 20s or 30s who has not menstruated for six months or more and is not pregnant or on hormonal contraception needs a workup (FSH, LH, prolactin, TSH, estradiol, pregnancy test) and, depending on findings, estrogen restoration — whether combined oral contraceptive, transdermal estradiol plus progesterone, or causal treatment of the underlying driver.
Celiac Disease and IBD
Gut disease wrecks bones through several simultaneous routes: impaired absorption of calcium and vitamin D, chronic inflammatory cytokines (TNF-α, IL-6) that directly activate osteoclasts, low body weight, and — in IBD — repeated courses of glucocorticoids.
Celiac disease. Up to 40–70% of untreated celiac patients have osteopenia or osteoporosis at diagnosis, and the figure is striking even in patients whose GI symptoms are mild or silent. Many adults present with fragility fractures or unexplained osteoporosis and only later discover their celiac disease. Every premenopausal woman and every man under 50 with unexplained osteoporosis should have celiac serology — tissue transglutaminase IgA (tTG-IgA) plus total IgA. A strict gluten-free diet restores most of the lost bone over two to five years. See the celiac disease page for the full diagnostic and dietary picture.
Inflammatory bowel disease. Both Crohn's disease and ulcerative colitis cause bone loss, but Crohn's is worse because of small-bowel involvement, higher cumulative steroid exposure, and more frequent surgical resections that impair calcium, vitamin D, and magnesium absorption. Between 30% and 60% of IBD patients have low bone density. Nutrient deficiencies, chronic inflammation, undernutrition, and steroid bursts all contribute — see the IBD overview and the IBD nutrient deficiency deep-dive. Shifting from steroid-dependent regimens to biologic therapy (infliximab, adalimumab, ustekinumab, vedolizumab) reduces both inflammation and steroid exposure — and both protect bone.
Post-Gastric-Bypass and Bariatric Bone Loss
Roux-en-Y gastric bypass (RYGB) and biliopancreatic diversion bypass the duodenum and proximal jejunum — the main sites of calcium, iron, and fat-soluble vitamin absorption — and rapidly produce secondary hyperparathyroidism, vitamin D deficiency, and bone loss. Hip-neck BMD drops 8–14% in the first two years after RYGB, and fracture risk rises over the subsequent decade.
Sleeve gastrectomy is gentler but not neutral. Any bariatric patient should take lifelong calcium citrate 1200–1500 mg/day (split doses; citrate is better absorbed than carbonate with low stomach acid), vitamin D 3000 IU/day or more titrated to a 25-OH-D above 30 ng/mL, and a bariatric-specific multivitamin. Annual labs and a baseline-plus-follow-up DEXA are standard of care.
Anorexia Nervosa and the Female Athlete Triad / RED-S
Anorexia nervosa causes some of the most severe osteoporosis seen in young people. The mechanism is multi-hit: low body weight, hypothalamic amenorrhea (estrogen withdrawal), low IGF-1, elevated cortisol, and frank calcium/vitamin D deficiency. Over 50% of adult women with anorexia have osteoporosis; fracture risk is two to seven times higher than age-matched controls.
The female athlete triad — low energy availability, menstrual dysfunction, low bone density — and its broader relabeling as RED-S (Relative Energy Deficiency in Sport) describe the same physiology in athletes: under-fueling relative to training demands shuts down the hypothalamic-pituitary-ovarian axis. Distance runners, gymnasts, ballet dancers, and lightweight rowers are classically at risk, but the pattern appears in any sport where leanness is rewarded.
Treatment is weight restoration and resumption of menses, not a bisphosphonate. Oral contraceptives do not reliably protect bone in this population because they do not restore the metabolic milieu (IGF-1, leptin). Transdermal estradiol plus cyclic progesterone has modest evidence of benefit during recovery, and teriparatide has been used in severe adult cases with ongoing fractures. The definitive treatment is eating enough.
Multiple Myeloma and Other Malignancies
Multiple myeloma is a plasma-cell malignancy that produces osteoclast-activating factors and is classically heralded by unexplained osteoporosis, vertebral compression fractures, bone pain, or lytic lesions on imaging. Every patient over 40 with unexplained osteoporosis — especially if paired with anemia, elevated total protein, hypercalcemia, or kidney dysfunction — deserves serum and urine protein electrophoresis (SPEP/UPEP) with immunofixation plus serum free light chains.
Other malignancies that drive bone loss: metastatic breast and prostate cancer (both through direct metastasis and through the hormone therapies discussed below), mastocytosis (mast cells secrete heparin and other osteoclast activators), and a smaller list of leukemias/lymphomas. Any atypical osteoporosis pattern — focal lesions, rapid progression, associated systemic symptoms — warrants oncologic workup.
Chronic Kidney Disease and Renal Osteodystrophy
As GFR falls below 60 mL/min/1.73m2, the kidneys lose the ability to activate vitamin D (convert 25-OH-D to 1,25-diOH-D) and to excrete phosphate. The result is secondary hyperparathyroidism, abnormal mineralization, and a specific renal osteodystrophy pattern that is not simply "osteoporosis."
Practical implications: standard DEXA T-scores behave unreliably below GFR 30; bone biopsy is sometimes needed; most bisphosphonates are contraindicated below GFR 30–35; denosumab works at any GFR but carries an increased risk of profound hypocalcemia that requires careful calcium and calcitriol replacement. CKD bone disease is a nephrologist's game — see the Kidney Disease overview.
The Diabetes Paradox — T1D and T2D
Diabetes and bone have a confusing relationship that has taken decades to unravel.
Type 1 diabetes clearly reduces bone density. Insulin itself is an anabolic hormone for bone, and the long years of insufficient insulin action in T1D — plus frequent childhood onset before peak bone mass is established — lead to lower BMD and roughly 6-fold higher hip fracture risk than non-diabetic peers.
Type 2 diabetes is the paradox. BMD on DEXA is often normal or even slightly increased in T2D, yet fracture risk is 20–60% higher at any given BMD. The bone looks okay on paper but breaks anyway. Why? T2D bone has impaired collagen cross-linking from advanced glycation end-products (AGEs), reduced bone turnover, and microarchitectural defects that DEXA cannot see. FRAX underestimates risk in T2D — treat these patients as one T-score category worse than the scanner reports, or use the trabecular bone score (TBS) when available.
Certain diabetes drugs matter: thiazolidinediones (pioglitazone, rosiglitazone) increase fracture risk and should be avoided in anyone with bone concerns. SGLT2 inhibitors have had mixed signals — canagliflozin showed an early fracture signal in one major trial, though later data are reassuring. Metformin, DPP-4 inhibitors, and GLP-1 agonists appear neutral or bone-protective.
Aromatase Inhibitors and ADT for Cancer
Aromatase inhibitors (anastrozole, letrozole, exemestane) are the standard adjuvant hormonal therapy for postmenopausal hormone-receptor-positive breast cancer. They block the peripheral conversion of androgens to estrogen, producing near-zero circulating estradiol. The bone consequence is the same as surgical menopause, on top of whatever postmenopausal loss has already occurred. Expected BMD loss is 2–5% per year, and fracture risk rises proportionately.
Every patient starting an AI should get a baseline DEXA, calcium/vitamin D, and — if FRAX or the treating oncologist identifies elevated risk — either denosumab 60 mg every 6 months or zoledronate 4 mg IV every 6 months (the oncology dosing, separate from the osteoporosis 5 mg yearly). ADT in prostate cancer follows the same framework (see the men's hypogonadism section above).
SSRIs, PPIs, and Other Medications
Several common medications have modest but real effects on bone:
- Proton pump inhibitors (PPIs) — omeprazole, esomeprazole, pantoprazole. Long-term use reduces calcium absorption (calcium carbonate specifically needs gastric acid) and is associated with a 20–40% increase in hip fracture risk over many years. Switch to calcium citrate, which absorbs without acid, and re-evaluate whether the PPI is still needed — many patients are on them out of inertia.
- SSRIs — serotonin transporters exist on osteoblasts, and chronic SSRI use is associated with a ~25% higher fracture risk independent of fall risk. Do not stop a needed antidepressant for this reason alone, but it should factor into overall risk calculation.
- Anticonvulsants — phenytoin, phenobarbital, carbamazepine induce hepatic vitamin D metabolism. Long-term users need higher vitamin D doses.
- Heparin — long-term use during pregnancy can cause osteoporosis; low-molecular-weight heparin is safer.
- Medroxyprogesterone acetate (depot Provera) — suppresses estrogen and carries a black-box warning for bone loss with prolonged use.
- Long-term opioids — cause hypogonadism that drives bone loss.
- Loop diuretics (furosemide, bumetanide) increase urinary calcium. Thiazide diuretics do the opposite and are modestly bone-sparing.
Alcohol and Tobacco
Alcohol. Intakes above three standard drinks per day raise fracture risk substantially, both by direct osteoblast toxicity and through falls, malnutrition, and liver disease. Below two drinks per day the effect on bone is small. Binge patterns are worse than steady low-level drinking.
Tobacco. Current smokers have roughly 25–30% higher hip fracture risk. Mechanisms include direct osteoblast toxicity, reduced estrogen bioavailability, lower body weight, poor nutrition, and impaired fracture healing. Risk starts dropping within a year of quitting and approaches never-smoker levels after about a decade. Both alcohol and smoking are line items inside FRAX — they shift your 10-year fracture probability upward automatically.
The Screening Lab Panel
A reasonable screening panel for secondary causes in any newly diagnosed osteoporosis patient (yield varies but is clinically meaningful in most cohorts):
- CBC — anemia can point to myeloma, celiac, IBD.
- Comprehensive metabolic panel (CMP) — calcium, creatinine (for GFR), albumin, liver enzymes, phosphate if available.
- Intact PTH — paired with calcium, the key test for primary and secondary hyperparathyroidism.
- 25-hydroxyvitamin D — target above 30 ng/mL.
- TSH — check for overt or subclinical hyperthyroidism and for over-replacement in known hypothyroid patients.
- Serum and urine protein electrophoresis (SPEP/UPEP) with immunofixation, plus serum free light chains, in patients over 40 or with any atypical features — myeloma screening.
- 24-hour urine calcium — high values suggest idiopathic hypercalciuria (a treatable cause with thiazides) or hyperparathyroidism; low values suggest malabsorption or inadequate intake.
- Celiac serology — tTG-IgA plus total IgA, in every premenopausal woman, every man under 50, and anyone with GI clues.
- Morning total testosterone (with SHBG or free testosterone) in men — repeat to confirm.
- FSH, LH, estradiol, prolactin in premenopausal women with amenorrhea or irregular cycles.
- 24-hour urinary free cortisol or overnight dexamethasone suppression if Cushing's syndrome features are present — central obesity, purple striae, facial plethora, proximal muscle weakness.
- Tryptase if systemic mastocytosis is suspected (flushing, unexplained anaphylaxis, unusual fracture pattern).
Not every test is needed for every patient; the history steers the panel. But the first six bullets (CBC, CMP, PTH, 25-OH-D, TSH, SPEP/UPEP) are defensible as a baseline for nearly everyone.
Treating the Cause, Not Just the Bone
The payoff of secondary-cause screening is that treating the underlying condition often restores bone density without any osteoporosis-specific medication:
- Parathyroid surgery → 5–10% BMD gain over two years.
- Gluten-free diet in celiac → near-complete BMD recovery over two to five years.
- Testosterone replacement in genuinely hypogonadal men → meaningful BMD increase over one to two years.
- Controlling hyperthyroidism, reducing over-replacement, or tapering glucocorticoids → loss halts and often partially reverses.
- Weight restoration in anorexia → the single most important intervention; pharmacologic bone drugs have limited effect without it.
Pharmacologic therapy (bisphosphonate, denosumab, teriparatide, romosozumab) is still frequently needed when the damage is already severe or the cause cannot be fully reversed. But skipping the cause hunt means fighting the fire without turning off the gas.
Key Research Papers
Society guidelines and pivotal studies covering the dominant secondary causes of low bone mass: glucocorticoid-induced osteoporosis (ACR), male osteoporosis, primary hyperparathyroidism, ADT and aromatase-inhibitor bone loss, type-2 diabetes fragility, celiac disease, anorexia nervosa, and proton pump inhibitor exposure.
- Buckley L, Guyatt G, Fink HA, et al. 2017 American College of Rheumatology Guideline for the Prevention and Treatment of Glucocorticoid-Induced Osteoporosis. Arthritis & Rheumatology. 2017;69(8):1521–1537.
- Watts NB, Adler RA, Bilezikian JP, et al. Osteoporosis in Men: An Endocrine Society Clinical Practice Guideline. Journal of Clinical Endocrinology & Metabolism. 2012;97(6):1802–1822.
- Bilezikian JP, Brandi ML, Eastell R, et al. Guidelines for the Management of Asymptomatic Primary Hyperparathyroidism: Summary Statement from the Fourth International Workshop. Journal of Clinical Endocrinology & Metabolism. 2014;99(10):3561–3569.
- Khosla S, Amin S, Orwoll E. Osteoporosis in Men. Endocrine Reviews. 2008;29(4):441–464.
- Smith MR, Egerdie B, Hernández Toriz N, et al. Denosumab in Men Receiving Androgen-Deprivation Therapy for Prostate Cancer. New England Journal of Medicine. 2009;361(8):745–755.
- Eastell R, Adams JE, Coleman RE, et al. Effect of Anastrozole on Bone Mineral Density: 5-Year Results from the Anastrozole, Tamoxifen, Alone or in Combination Trial 18233230. Journal of Clinical Oncology. 2008;26(7):1051–1057.
- Napoli N, Chandran M, Pierroz DD, Abrahamsen B, Schwartz AV, Ferrari SL. Mechanisms of Diabetes Mellitus-Induced Bone Fragility. Nature Reviews Endocrinology. 2017;13(4):208–219.
- Schwartz AV, Vittinghoff E, Bauer DC, et al. Association of BMD and FRAX Score with Risk of Fracture in Older Adults with Type 2 Diabetes. JAMA. 2011;305(21):2184–2192.
- Mosekilde L, Beck-Nielsen H, Rud B, Stilgren L, Sorensen OH. Celiac Disease and Bone. Nutrition. 2005;21(11–12):1149–1153.
- Misra M, Klibanski A. Anorexia Nervosa and Bone. Journal of Endocrinology. 2014;221(3):R163–R176.
- Yu EW, Bauer SR, Bain PA, Bauer DC. Proton Pump Inhibitors and Risk of Fractures: A Meta-Analysis of 11 International Studies. American Journal of Medicine. 2011;124(6):519–526.
- Mountjoy M, Sundgot-Borgen J, Burke L, et al. The IOC Consensus Statement: Beyond the Female Athlete Triad — Relative Energy Deficiency in Sport (RED-S). British Journal of Sports Medicine. 2014;48(7):491–497.
- Shoback D, Rosen CJ, Black DM, Cheung AM, Murad MH, Eastell R. Pharmacological Management of Osteoporosis in Postmenopausal Women: An Endocrine Society Guideline Update. Journal of Clinical Endocrinology & Metabolism. 2020;105(3):587–594.
Live PubMed Searches
Live PubMed queries that update as new papers are indexed.
- PubMed: glucocorticoid-induced osteoporosis
- PubMed: primary hyperparathyroidism BMD
- PubMed: celiac disease osteoporosis
- PubMed: ADT and bone loss
- PubMed: aromatase inhibitor bone loss
- PubMed: T2D fracture paradox
- PubMed: bariatric surgery bone loss
- PubMed: anorexia nervosa BMD
- PubMed: PPI and fracture risk
- PubMed: myeloma as osteoporosis
Connections
- Osteoporosis
- Celiac Disease
- Kidney Disease
- Hashimoto's Thyroiditis
- Inflammatory Bowel Disease
- Denosumab, Romosozumab and Teriparatide
- DEXA Scan: T-Score and Z-Score
- FRAX Risk Assessment
- IBD: Fatigue, Anemia and Nutrient Deficiencies
- Graves' Disease
- Bisphosphonates
- Weight-Bearing and Resistance Exercise
- Fracture Prevention and Fall Risk
- Calcium, Vitamin D and Protein Intake
- Calcium
- Hyperparathyroidism
- Diabetes
- Multiple Myeloma