Postmenopausal Osteoporosis and Hormone Therapy
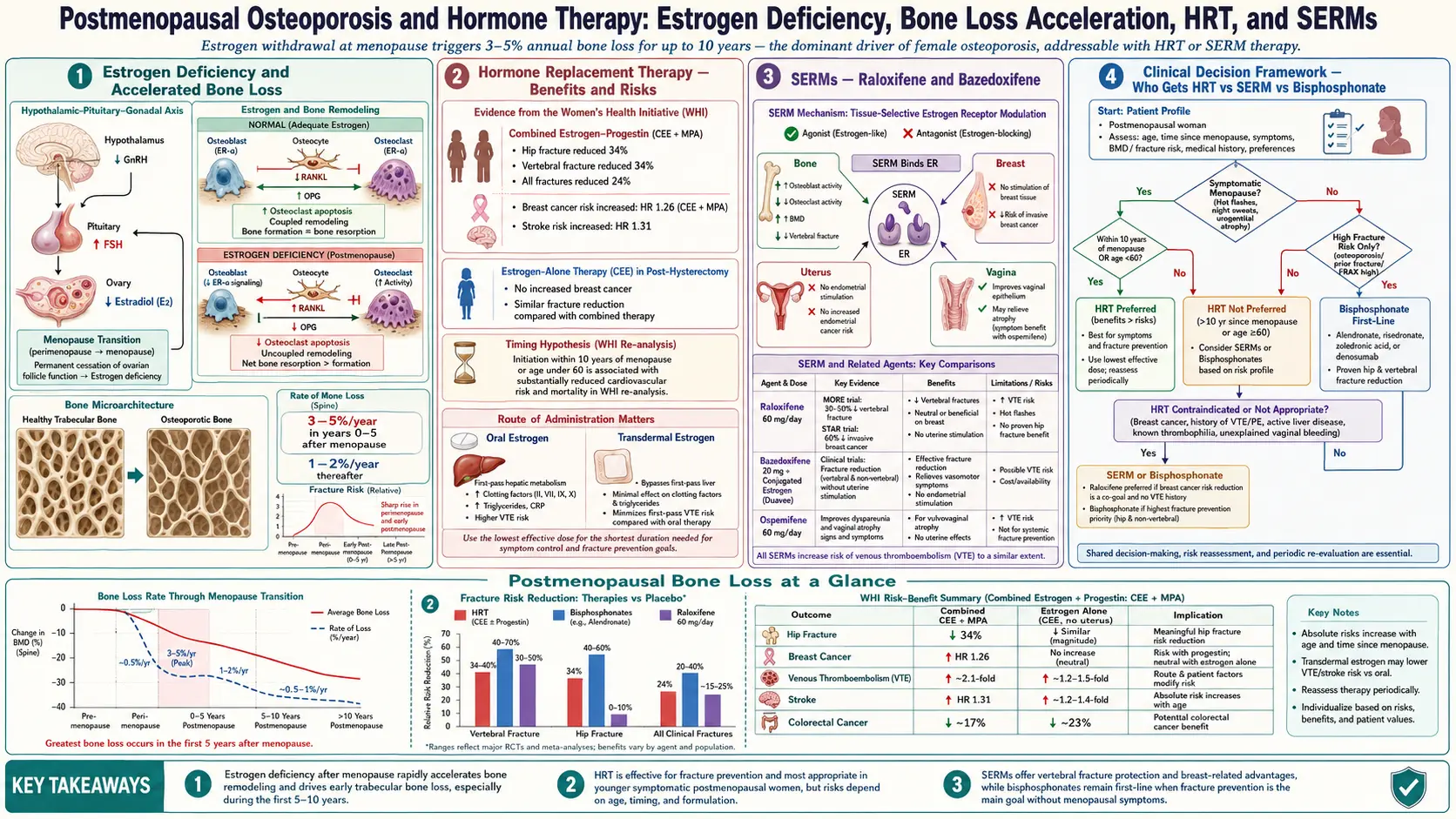
Table of Contents
- Why Menopause Hammers Your Skeleton
- The Bone Loss Pattern — Years 1 to 10
- Type I Primary Osteoporosis
- What Estrogen Actually Does to Bone
- The WHI Study — What It Really Showed
- The Timing Hypothesis — Age of Initiation Matters
- Hormone Therapy and Fracture Reduction
- Transdermal vs Oral Estrogen — The VTE Difference
- Why You Need Progesterone with an Intact Uterus
- SERMs — Raloxifene and the MORE Trial
- Bazedoxifene / Conjugated Estrogens (Duavee)
- Tibolone — The European Option
- When a Bisphosphonate Beats Hormone Therapy
- How Long to Stay on Hormone Therapy
- What Happens When HT Stops
- High-Risk Patients — Combining or Switching
- Non-Hormonal Options When HT Is Off the Table
- The Conversation to Have with Your Clinician
- Key Research Papers
- Connections
- Featured Videos
Why Menopause Hammers Your Skeleton
For most of your adult life, your skeleton is in a slow, steady remodeling loop. Cells called osteoclasts chew away small patches of old bone; cells called osteoblasts follow behind and lay down fresh bone in the same spot. For roughly thirty years — from peak bone mass in your late twenties until your mid-forties — the two teams stay nearly balanced, with maybe a 0.3–0.5% loss per year.
Then estrogen levels start falling. The ovaries wind down across perimenopause, and the final menstrual period marks a sharp cliff in circulating estradiol — from premenopausal levels of 50–400 pg/mL down to under 20 pg/mL within a year or two. Estrogen was quietly doing three things for your skeleton every single day: restraining the osteoclasts, prolonging the life of osteoblasts, and dampening inflammatory signals (especially RANKL) that recruit new bone-chewing cells. When estrogen drops, all three brakes release at once.
The result is the most rapid bone loss any healthy adult experiences outside of disease. A woman can lose more bone density in the five years after her final period than she lost in the previous twenty. This is not a gentle glide path. It is a free fall followed by a shallower but permanent descent.
The Bone Loss Pattern — Years 1 to 10
The numbers that matter to you, broken into phases:
- Perimenopause (roughly ages 45–51). Bone loss accelerates modestly — about 0.5–1% per year — even before periods stop.
- Early postmenopause (year 0 through year 3–5). The steepest phase. Trabecular (spongy) bone in the spine and inner hip loses 3–5% per year. Cortical (dense outer) bone loses roughly 1–2%. Over five years, that adds up to a 10–25% reduction in spinal bone density for an untreated woman.
- Late postmenopause (year 5 onward). The curve flattens to roughly 1–2% per year, a rate that continues for the rest of life. This slower loss is still substantial — another 20% over the next two decades.
Two women the same age can look entirely different on a DEXA scan depending on where they sit in this timeline. A 52-year-old who reached menopause at 48 is mid-avalanche. A 52-year-old whose periods stopped at 51 has barely started. Treatment decisions often hinge on which phase you are in — not just your T-score. See DEXA Scan, T-Score, and Z-Score Explained for the numbers that define osteopenia and osteoporosis.
Type I Primary Osteoporosis
Clinicians still use the old Riggs-Melton classification. Type I (postmenopausal) osteoporosis is the estrogen-deficiency form. It hits women six times more often than men, peaks in the 15 years after menopause, and preferentially damages trabecular bone — which is why the vertebrae and distal radius (wrist) are the early fracture sites. The classic presentation: a woman in her late fifties or sixties sustains a Colles fracture after a minor fall, or develops unexplained back pain that turns out to be a vertebral compression fracture.
Type II (senile) osteoporosis, by contrast, affects both sexes, peaks after age 70, and damages cortical bone more broadly. The hallmark fracture is the hip. In practice the two types blur — a 75-year-old woman usually has both processes stacked on top of each other — but the distinction matters because the early postmenopausal phase is the window where estrogen replacement is most mechanistically sensible.
What Estrogen Actually Does to Bone
At the cell level, estrogen works through the RANK / RANKL / OPG system. RANKL is a signaling molecule that osteoblasts (and immune cells) release to recruit and activate osteoclasts. OPG (osteoprotegerin) is a decoy receptor that mops up RANKL and blocks its signal. Estrogen suppresses RANKL production and boosts OPG. When estrogen falls, RANKL rises, OPG falls, and osteoclast activity surges.
Estrogen also lengthens osteoblast lifespan (osteoblasts die off faster without it) and shortens osteoclast lifespan (osteoclasts linger longer and chew more bone without it). The result of combining all three effects is what bone researchers call an uncoupled remodeling state — resorption running ahead of formation, with every remodeling cycle leaving a net bone deficit.
This is why hormone therapy is the single most physiologically direct osteoporosis treatment available. Bisphosphonates slow osteoclasts chemically; denosumab blocks RANKL directly; hormone therapy restores the upstream signal that controlled everything else.
The WHI Study — What It Really Showed
The Women's Health Initiative (WHI) is the single most influential — and most misread — study in the history of hormone therapy. Two randomized arms mattered: the estrogen-plus-progestin arm (n = 16,608, using conjugated equine estrogens 0.625 mg plus medroxyprogesterone acetate 2.5 mg daily) and the estrogen-alone arm (n = 10,739, conjugated equine estrogens in women with prior hysterectomy).
The combined arm was halted early in 2002 amid headlines about elevated breast cancer, stroke, coronary events, and venous thromboembolism (VTE). What the headlines buried:
- Hip fractures fell by 34%. Clinical vertebral fractures fell by 34%. Total fractures fell by 24%. This was the first randomized trial ever to prove hormone therapy cut hip fractures.
- The average participant age was 63, with many women 15+ years past menopause — the wrong population to enroll for a cardiovascular primary-prevention trial.
- The estrogen-alone arm (hysterectomized women) showed no increase in breast cancer — and later follow-up showed a significant decrease.
- The absolute risk increases were small — on the order of 8 additional breast cancers per 10,000 women per year in the combined arm.
A generation of women were taken off hormone therapy on the basis of relative-risk headlines that did not apply to women in their early fifties using modern formulations.
The Timing Hypothesis — Age of Initiation Matters
When researchers reanalyzed the WHI data by age and years-since-menopause, a cleaner picture emerged. Women who started hormone therapy within ten years of menopause or before age 60 had:
- Reduced all-cause mortality (hazard ratio roughly 0.70–0.75).
- No increase — and possibly a decrease — in coronary heart disease.
- Strong fracture protection.
- Modest, delayed breast-cancer risk (combined HT only).
Women who started HT 20+ years past menopause or after age 70 had the opposite: more cardiovascular events, more strokes, and no mortality benefit. The hypothesis: young postmenopausal arteries are still healthy and respond to estrogen with vasodilation and lipid benefits. Older arteries with established atherosclerotic plaque are destabilized by estrogen instead.
The clinical translation is now codified in North American Menopause Society (NAMS) 2022 and Endocrine Society guidelines: hormone therapy is reasonable for symptomatic women under 60 or within 10 years of menopause, and that window also provides the most favorable risk profile for bone protection.
Hormone Therapy and Fracture Reduction
Across the major randomized trials and meta-analyses, hormone therapy reduces fractures at every skeletal site studied:
- Hip: approximately 30–40% reduction.
- Vertebral (spine): approximately 30–35% reduction.
- Wrist: approximately 20–30% reduction.
- Total fractures: approximately 20–25% reduction.
Importantly, the WHI showed these reductions even in women with normal baseline bone density. Most osteoporosis drugs only work in women who already have osteoporosis; hormone therapy is one of the few proven interventions in the larger osteopenic population. That makes it a uniquely useful tool in the first decade after menopause, when T-scores have often drifted into osteopenia (−1.0 to −2.4) but not yet reached osteoporosis (≤−2.5).
Transdermal vs Oral Estrogen — The VTE Difference
Not all estrogen is delivered equally. Oral estrogen passes through the liver first, raising hepatic production of clotting factors and C-reactive protein. Transdermal estrogen (patch, gel, or spray) bypasses first-pass metabolism and enters the bloodstream directly.
The clinical consequence: observational cohort studies (notably the French ESTHER study) and subsequent meta-analyses show that oral estrogen roughly doubles the risk of venous thromboembolism (deep vein thrombosis and pulmonary embolism), while transdermal estrogen at standard doses carries no measurable increase. Stroke risk shows a similar pattern — elevated with oral, neutral with transdermal.
Current practice for any woman with baseline VTE risk factors (obesity, prior clot, Factor V Leiden, migraine with aura, heavy smoker, age over 60) is to default to transdermal estradiol. Typical starting doses: 0.025–0.05 mg/day patch (Climara, Vivelle-Dot, Minivelle) or a pump of 0.06% estradiol gel (Divigel, EstroGel). Bone-protective doses generally start at 0.05 mg/day patch.
Why You Need Progesterone with an Intact Uterus
Unopposed estrogen stimulates the uterine lining and substantially increases the risk of endometrial hyperplasia and endometrial cancer. Any woman who still has her uterus must combine estrogen with a progestogen to keep the lining in check. The options:
- Micronized progesterone (Prometrium, 100–200 mg nightly). Chemically identical to the body's own progesterone. Sedating — take at bedtime. Generally considered the most favorable profile for breast and cardiovascular risk.
- Medroxyprogesterone acetate (MPA). The progestin used in WHI. Widely available, cheap, but the synthetic progestin most strongly associated with the breast-cancer and cardiovascular signals.
- Levonorgestrel IUD (Mirena). Delivers progestin locally to the uterus with minimal systemic absorption. A reasonable option for women who tolerate oral progestogens poorly.
Women who have had a hysterectomy do not need a progestogen and can use estrogen alone — which simplifies everything and avoids the component most associated with increased breast-cancer risk.
SERMs — Raloxifene and the MORE Trial
Selective estrogen receptor modulators (SERMs) are drugs that act like estrogen in some tissues and against estrogen in others. Raloxifene (Evista, 60 mg daily) is the SERM with the largest osteoporosis evidence base.
The pivotal MORE trial (Multiple Outcomes of Raloxifene Evaluation, n = 7,705, postmenopausal women with osteoporosis, 3 years) and its four-year CORE extension showed:
- Vertebral fracture reduction of roughly 40–50%.
- No significant reduction in non-vertebral or hip fractures.
- A 72% reduction in invasive breast cancer (estrogen-receptor-positive tumors specifically).
- An approximate doubling of VTE risk — similar in magnitude to oral estrogen.
- Worsening of hot flashes — which makes raloxifene a poor choice for a woman still in the vasomotor-symptom phase of menopause.
Raloxifene earned FDA approval both for osteoporosis and for breast-cancer risk reduction. It is the right choice for a postmenopausal woman whose hot flashes have resolved, who has vertebral-dominant osteoporosis, who carries elevated breast-cancer risk, and who has no VTE history. It is a poor choice for symptomatic perimenopausal women or anyone with prior clots.
Bazedoxifene / Conjugated Estrogens (Duavee)
Duavee combines conjugated equine estrogens (0.45 mg) with the SERM bazedoxifene (20 mg). The concept is called a tissue-selective estrogen complex (TSEC): the estrogen delivers its bone and hot-flash benefits, while bazedoxifene blocks the estrogen effect specifically in breast and uterine tissue — so no separate progestogen is required to protect the endometrium.
The SMART trials showed Duavee improved lumbar spine bone density by roughly 1.5–2% more than placebo over two years, reduced hot flashes, protected the endometrium, and did not increase breast density on mammography (a surrogate for breast-cancer risk). VTE risk remained elevated, similar to oral estrogen.
Duavee fills a particular niche: a woman with an intact uterus who wants symptom relief and bone protection, can't or won't take a progestogen, and has no VTE history. It is not usually a first-line osteoporosis drug in its own right, but for the right candidate it is uniquely convenient.
Tibolone — The European Option
Tibolone (Livial, 2.5 mg daily) is a synthetic steroid that metabolizes into compounds with estrogenic, progestogenic, and weak androgenic activity. It is widely used in Europe and other countries but has never been approved by the US FDA.
The LIFT trial (Long-term Intervention on Fractures with Tibolone, n = 4,538, women aged 60–85 with osteoporosis) showed tibolone reduced vertebral fractures by 45%, non-vertebral fractures by 26%, and invasive breast cancer by 68%. But it increased the risk of stroke by 2.2-fold in this older cohort and was halted early for that reason.
If you are in a country where tibolone is available, it is a legitimate option for a younger postmenopausal woman (under 60) without cardiovascular risk factors who wants symptom relief, bone protection, and generally also reports improved libido (the androgenic component). In the US, you need to either go without or pursue compounding, which is not recommended.
When a Bisphosphonate Beats Hormone Therapy
Hormone therapy is not a first-line osteoporosis drug in most current US guidelines — not because it doesn't work, but because the average osteoporosis patient is 70+ years old, well outside the timing-hypothesis window, and a bisphosphonate delivers cleaner fracture reduction without cardiovascular concerns.
Choose a bisphosphonate over hormone therapy when:
- Age over 60 or more than 10 years past menopause.
- T-score already ≤−2.5 with established osteoporosis.
- Prior fragility fracture (especially hip or vertebral).
- Established cardiovascular disease, prior stroke, or clotting history.
- Personal or strong family history of breast cancer.
- Uterine cancer history.
- Active liver disease (bisphosphonate alendronate is actually fine; transdermal estrogen is also fine; oral estrogen is not).
Choose hormone therapy (often alongside a bisphosphonate conversation, not instead of) when:
- Early postmenopause (within 10 years, under age 60) with osteopenia not yet meeting osteoporosis thresholds.
- Disruptive hot flashes, night sweats, genitourinary symptoms, or sleep disruption alongside bone loss.
- Premature ovarian insufficiency (menopause before age 40) — HT through at least the average menopausal age of 51 is standard of care regardless of bone status.
- Intolerance to bisphosphonates (GI upset, jaw concerns, atypical femur concerns after prolonged use).
How Long to Stay on Hormone Therapy
There is no fixed expiration date. The old "five years and stop" rule was driven by WHI fallout and does not map cleanly onto modern formulations or the timing hypothesis. Current NAMS and Endocrine Society guidance is individualized:
- For vasomotor symptoms, continue as long as symptoms persist and benefits exceed risks — often 5–10 years, sometimes longer.
- For bone protection specifically, benefits last only as long as you take the drug. Stop, and bone loss resumes promptly.
- Reassess risk/benefit annually. Track blood pressure, breast exam and mammogram, any new cardiovascular symptoms, VTE risk factors.
- Women with premature ovarian insufficiency or early menopause (under 45) should continue through at least age 51.
"The lowest effective dose for the shortest needed duration" remains the boilerplate, but neither half of that phrase has a fixed number attached — it means the dose that controls your symptoms and the duration you continue to benefit.
What Happens When HT Stops
This is the single most under-discussed aspect of hormone therapy and osteoporosis. When you stop HT, bone density starts falling almost immediately, at a rate close to what it would have been in early postmenopause. Within two to three years, much of the bone density gained during HT is lost.
Fracture risk mirrors this: the WHI follow-up showed that within several years of stopping combined HT, hip-fracture incidence had drifted back up toward the placebo group rate. The bone benefit is not durable without the drug.
The practical implication: if you have used HT for symptom relief and are now in your early sixties considering stopping, have a plan. Either:
- Transition to a bisphosphonate (oral alendronate or risedronate weekly, or zoledronic acid IV yearly) as you taper off HT. This "locks in" the bone density gained and continues protection.
- Repeat DEXA 1–2 years after stopping HT and reassess then.
- Stay on HT at a lower maintenance dose if symptoms return and the risk-benefit still favors continuation.
High-Risk Patients — Combining or Switching
For women with severe osteoporosis, multiple vertebral fractures, or very low T-scores (≤−3.0), hormone therapy alone is rarely enough. These patients usually need an anabolic agent such as teriparatide, abaloparatide, or romosozumab for 1–2 years to rebuild bone, followed by a bisphosphonate or denosumab to maintain.
In these high-risk cases, if the woman is also early postmenopausal and symptomatic, transdermal estrogen can provide symptom relief alongside anabolic therapy — but the bone-rebuilding work is done by the anabolic agent, not the hormones. Think of HT in this context as adjunctive comfort and modest additional protection, not primary therapy.
Non-Hormonal Options When HT Is Off the Table
Some women cannot take hormones: prior estrogen-receptor-positive breast cancer, recent stroke, active VTE, active liver disease. For these patients the osteoporosis armamentarium still includes:
- Bisphosphonates — alendronate, risedronate, zoledronic acid. First-line for nearly everyone in this group.
- Denosumab (Prolia) — subcutaneous injection every 6 months. Particularly useful with kidney disease or poor oral bisphosphonate tolerance. Must not be stopped without a bisphosphonate bridge — rebound vertebral fractures are well documented.
- Raloxifene — if breast-cancer prevention is a priority and VTE risk is acceptable.
- Anabolic agents for severe disease.
- Calcium 1,000–1,200 mg/day, vitamin D to a level of 30–50 ng/mL, and protein 1.0–1.2 g/kg/day — foundational. See Calcium, Vitamin D, and Protein.
- Weight-bearing and resistance exercise 3–4 times per week. See the exercise protocol.
For vasomotor symptoms without hormones: low-dose paroxetine (Brisdelle, 7.5 mg) is FDA-approved for hot flashes. Venlafaxine, gabapentin, oxybutynin, and the neurokinin-3 antagonist fezolinetant (Veozah) are additional non-hormonal options. None of these protect bone.
The Conversation to Have with Your Clinician
Most primary-care offices are still working from 2003 WHI headlines. If you walk in asking about hormone therapy, you may get a knee-jerk "we don't do that anymore" reply that ignores two decades of reanalysis. To get a useful conversation, come in with:
- Your exact age and date of final menstrual period.
- A current DEXA result with T-scores at lumbar spine, total hip, and femoral neck.
- Your FRAX 10-year fracture risk.
- Personal and family history of breast cancer, cardiovascular disease, stroke, and clotting disorders.
- A list of your menopausal symptoms and how disruptive they are.
- A specific question: "Given my age, timing, and risk profile, am I a candidate for transdermal estradiol with micronized progesterone?"
If your clinician is uncomfortable, ask for a referral to a menopause specialist — the NAMS (now Menopause Society) website maintains a searchable list of certified practitioners. Women's health is an area where a second opinion from someone who reads the current literature often changes the plan.
Key Research Papers
The Women's Health Initiative primary trials and timing-hypothesis reanalyses, the ESTHER and other transdermal-route VTE studies, the SERM trials (MORE, RUTH), tibolone (LIFT), and current Menopause Society and Endocrine Society guidelines that frame hormone therapy as a bone treatment.
- Rossouw JE, Anderson GL, Prentice RL, et al. Risks and Benefits of Estrogen Plus Progestin in Healthy Postmenopausal Women: Principal Results From the Women's Health Initiative Randomized Controlled Trial. JAMA. 2002;288(3):321–333.
- Anderson GL, Limacher M, Assaf AR, et al. Effects of Conjugated Equine Estrogen in Postmenopausal Women with Hysterectomy: The Women's Health Initiative Randomized Controlled Trial. JAMA. 2004;291(14):1701–1712.
- Manson JE, Chlebowski RT, Stefanick ML, et al. Menopausal Hormone Therapy and Health Outcomes During the Intervention and Extended Poststopping Phases of the Women's Health Initiative Randomized Trials. JAMA. 2013;310(13):1353–1368.
- Rossouw JE, Prentice RL, Manson JE, et al. Postmenopausal Hormone Therapy and Risk of Cardiovascular Disease by Age and Years Since Menopause. JAMA. 2007;297(13):1465–1477.
- Cauley JA, Robbins J, Chen Z, et al. Effects of Estrogen Plus Progestin on Risk of Fracture and Bone Mineral Density: The Women's Health Initiative Randomized Trial. JAMA. 2003;290(13):1729–1738.
- Canonico M, Oger E, Plu-Bureau G, et al. Hormone Therapy and Venous Thromboembolism Among Postmenopausal Women: Impact of the Route of Estrogen Administration and Progestogens — The ESTHER Study. Circulation. 2007;115(7):840–845.
- Ettinger B, Black DM, Mitlak BH, et al. Reduction of Vertebral Fracture Risk in Postmenopausal Women with Osteoporosis Treated with Raloxifene: Results From a 3-Year Randomized Clinical Trial (MORE). JAMA. 1999;282(7):637–645.
- Barrett-Connor E, Mosca L, Collins P, et al. Effects of Raloxifene on Cardiovascular Events and Breast Cancer in Postmenopausal Women (RUTH). New England Journal of Medicine. 2006;355(2):125–137.
- Cummings SR, Ettinger B, Delmas PD, et al. The Effects of Tibolone in Older Postmenopausal Women (LIFT). New England Journal of Medicine. 2008;359(7):697–708.
- The 2022 Hormone Therapy Position Statement of The North American Menopause Society Advisory Panel. The 2022 Hormone Therapy Position Statement of The North American Menopause Society. Menopause. 2022;29(7):767–794.
- Eastell R, Rosen CJ, Black DM, Cheung AM, Murad MH, Shoback D. Pharmacological Management of Osteoporosis in Postmenopausal Women: An Endocrine Society Clinical Practice Guideline. Journal of Clinical Endocrinology & Metabolism. 2019;104(5):1595–1622.
- Greendale GA, Espeland M, Slone S, Marcus R, Barrett-Connor E. Bone Mass Response to Discontinuation of Long-Term Hormone Replacement Therapy: Results from the Postmenopausal Estrogen/Progestin Interventions (PEPI) Safety Follow-up Study. Archives of Internal Medicine. 2002;162(6):665–672.
Live PubMed Searches
Live PubMed queries that update as new papers are indexed.
- PubMed: postmenopausal osteoporosis hormone therapy
- PubMed: WHI timing hypothesis
- PubMed: transdermal estradiol VTE
- PubMed: raloxifene MORE trial
- PubMed: bazedoxifene Duavee
- PubMed: tibolone LIFT trial
- PubMed: estrogen RANKL OPG
- PubMed: bone loss after stopping HRT
Connections
- Osteoporosis
- Bisphosphonates
- DEXA Scan: T-Score and Z-Score
- Weight-Bearing and Resistance Exercise
- Denosumab, Romosozumab and Teriparatide
- Calcium, Vitamin D and Protein Intake
- FRAX Risk Assessment
- Secondary Osteoporosis Causes
- Fracture Prevention and Fall Risk
- Arthritis
- Pregnancy and Hashimoto's
- Cancer
- Stroke
- Calcium
- Cardiovascular Disease
- Perimenopause